Abstract
Spinal cord injury (SCI) is a traumatic injury of the central nervous system. Because neurons are damaged and difficult to regenerate after SCI, its repair remains challenging. However, recent research on stem cell therapy have favored its use after SCI. In this study, based on the establishment of a mouse SCI model, human menstrual blood–derived endometrial stem cells (MenSCs) were intrathecally injected to explore the role and molecular mechanism of MenSCs in SCI. MenSCs were transplanted following SCI in the animal model, and behavioral evaluations showed that MenSC transplantation improved functional recovery. Therefore, samples were collected after 7 days, and transcriptome sequencing was performed. Gene Ontology (GO) enrichment analysis revealed that SCI is closely related to immune system processes. After transplantation of MenSCs, the immune response was significantly activated. In the Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis, MenSC transplantation was found to be closely related to Th1, Th2, and Th17 cell differentiation pathways. Neuronal damage and glial cell proliferation and activation in the different groups were detected by fluorescence immunohistochemistry and Western blotting 7 days after SCI. Simultaneously, the activation of different types of microglia was detected and the expression of pro-inflammatory and anti-inflammatory factors was quantitatively analyzed. The results showed that MenSC transplantation and sonic hedgehog (Shh)–induced MenSCs accelerated neuronal recovery at the injured site, inhibited the formation of glial cells and microglial activation at the injured site, inhibited the expression of inflammatory factors, and improved the inflammatory microenvironment to achieve functional recovery of SCI. This study provides an experimental basis for the study of the role and molecular mechanism of MenSCs in SCI repair, and a reference for the role of Shh-induced MenSCs in SCI repair.
Introduction
Spinal cord injury (SCI) is a disease with high disability and mortality rates. It is mainly caused by traumatic factors and accounts for several traumatic diseases. It can lead to spinal cord neuron death, glial scar formation, and neurological symptoms, including neuralgia, abnormal motor or sensory function, and a high disability rate. Approximately, 180,000 people suffer from SCI annually worldwide 1 . After SCI, pathological changes such as congestion, liquefaction, necrosis, and nerve scar will occur in the lesion. SCI is divided into primary injury and secondary injury according to the time of injury and pathophysiologic changes 2 . Primary injury is caused by mechanical damage to the spinal cord directly caused by external forces, which is currently considered irreversible. Secondary injury is a series of pathophysiological changes of spinal cord neurons on the basis of primary injury. It is an active regulatory process at the cellular and molecular levels and has reversibility and feasibility. At the subacute phase of SCI [4–14 days post-injury (d.p.i.) in the mouse], the phenotypic changes of astrocyte proliferation play a crucial role in SCI pathology. In this process, naive astrocytes first as reactive astrocytes and then as scar-forming astrocytes 3 . Therefore, the treatment of SCI focuses on effective control of the secondary injury. In the repair treatment of SCI, promoting the formation of new neurons and the establishment of neural circuits at the injured site, inhibiting the formation of glial cells/macrophages, inhibiting the expression of pro-inflammatory factors, and promoting the expression of anti-inflammatory factors have become the focus of SCI repair research 4 . Existing treatments, including surgery and drug treatment after SCI, remain challenging. With the deepening of basic research on stem cells, there has been an increasing number of studies on stem cell treatment for SCI, which has good prospects for clinical applications.
With progress in stem cell research, there have been reports on the treatment of SCI with embryonic stem cells, neural stem cells (NSCs), and some mesenchymal stem cells (MSCs), all of which have achieved a certain degree of therapeutic effect5–8. Studies 9 have shown that transplantation of NSCs can significantly improve motor and sensory functions below the level of SCI patients. With the rapid development of biomedicine, stem cells have been successfully extracted from various tissues in recent years, such as bone marrow, umbilical cord blood, amniotic membrane, umbilical cord, and fat, and can be expanded in vitro. These cells have good stability and multidirectional differentiation potential. After appropriate induction in vitro, they can differentiate into various target cells, such as epidermal, fat, muscle, cartilage, neurons, and other tissue cells10–14. MSCs have been successfully used to treat bone and joint, cardiovascular, and immune system diseases, and have been found to have good histocompatibility and therapeutic effects15–17. The study found that, in an established experimental animal model of SCI, human umbilical cord–derived mesenchymal stem cells (hUC-MSCs) administered by intravenous injection or local transplantation can improve the motor function of SCI animals and change the expression of related factors in injured spinal cord tissue18–21.
Human menstrual blood–derived endometrial stem cells (MenSCs), an important member of the MSC family, have attracted extensive attention because of their rich sources, noninvasive collection and separation methods, high proliferative activity, and differentiation potential 22 . MenSCs have satisfactory results in the treatment of osteogenesis, cardiac, liver, lung, endometrial, stroke, and other diseases22–27. Owing to their excellent biological characteristics, MenSCs have received extensive attention as seed cells for the treatment of refractory diseases. Through animal models, it has been found that they have shown good improvement effects in the treatment of myocardial infarction, Duchenne muscular dystrophy, type I diabetes, colitis, endometrial injury repair, and other diseases28–32.
MenSC transplantation has beneficial therapeutic effects in the rehabilitation of a rat spinal cord hemisection model, mainly by enhancing the expression of brain-derived neurotrophic factor 33 . In our previous research, we studied adipogenesis, osteogenesis, and tumorigenesis of MenSCs. The results showed that MenSCs had good adipogenic and osteogenic characteristics but did not cause tumorigenicity in vivo, which also provided a guarantee for the clinical application of MenSCs 22 . In this study, we based on the establishment of a free-fall impact SCI model, and transplanted MenSCs and sonic hedgehog (Shh)–induced MenSCs at the injury site. At 1, 3, 5, 7, 14, 21, and 28 days after cell transplantation, the Basso Mouse Scale (BMS) scoring method was used to compare the treatment effects. Tissue material was collected at 7, 14, and 28 days, and RNAseq analysis was performed on tissues at 7 days. It has been reported that after SCI in mice, in the subsequent phase of SCI (4–14 d.p.i. in the mouse), it can effectively improve the functional recovery. But limited functional recovery in the chronic phase of SCI3,34 (after 14 d.p.i. in the mouse). Therefore, in this study, fluorescence immunohistochemistry and Western blotting (WB) experiments were focused on tissues at 7 days. The results of other stages are only for reference. Analyze the efficacy of SCI after cell transplantation and analyze potential therapeutic mechanisms. Transcriptome sequencing and experimental results showed that MenSCs promoted functional recovery by improving the inflammatory microenvironment in a mouse free-fall impact spinal cord model.
Materials and Methods
Animal Breeding
All animal experiments in this study used 7- to 8-week-old female C57BL/6 mice [purchased from Beijing Huafukang Biotechnology, License: SCXK (Jing) 2019–0008], which were raised in the animal room of the Henan Key Laboratory of Medical Tissue Regeneration, Xinxiang Medical College. The light–dark cycle was 12 h (light time 8:00–20:00), the temperature was maintained at 25°C, and the relative air humidity was 50% to 60%. The animal experiments involved in this study followed the guidelines of the Ministry of Science and Technology of the People’s Republic of China and were approved by the Animal Welfare Committee of Xinxiang Medical College.
Establishment of SCI Model in Mice
The mice were fasted for 6 to 8 h before the operation. The operation steps for spinal cord contusion in mice were as follows: the mice were anesthetized with isoflurane and fixed on the operating table in the prone position, keeping the spinal column straight. An incision approximately 2 cm long was made in the tenth segment of the thoracic vertebra (T10) of the mouse to expose the spinal cord, which was impacted with a spinal cord impactor. The area where the vertebral bone was open is 2.5 mm × 5.0 mm. The specification of the impactor used is 1.2 × 5.0 mm, impactor diameter is 1.2 mm, length is 5 mm, and the area of the spinal cord impacted by the impactor is 0.6 × 0.6π mm2. The weight and impact height of the spinal cord hammer were adjusted to 20 g and 20 cm, allowing the impactor to fall freely and impact the site of the T10 lamina accurately. Local edema and hemorrhage of the spinal cord were also observed after the impact. In the sham operation group, only the spinal cord was exposed without spinal cord impact. After the mice woke up, they were reared in separate cages, and their postoperative reactions were observed.
MenSCs Culture
P3 generation MenSCs were obtained from the Stem Cell Bank established by our research group (Zhongyuan Stem Cell Research Institute), and the cell line was established and identified in our laboratory 20 . MenSCs were cultured in DMEM (Dulbecco’s modified eagle medium) high-glucose medium [4.5 mg/ml glucose, 1% penicillin, 1% streptomycin, and 10% fetal bovine serum (FBS)] (Zhongqiao Xinzhou; China; ZP-101). MenSCs were cultured in a saturated humidity incubator at 37°C and 5% CO2, and the solution was changed every 2 to 3 days. When the cells grew to 80% to 90% confluence, trypsin (GENVIEW; USA; 204070101200) was used for digestion and passaging.
MenSCs Transplantation
First, P4 or Shh-induced MenSCs were expanded and cultured to exponential growth period. Now, take out the cultured MenSCs from the incubator, discard the culture medium of the culture bottle in the super-clean worktable, add 1 ml trypsin solution, digest for 5 min, and then add 5 ml culture medium to stop the effect of enzyme. Transfer the cell suspension into a 15 ml centrifuge tube, place it in the centrifuge, centrifuge at 1,200 rpm for 5 min, take the centrifuge tube back to the super-clean worktable after centrifugation, discard the supernatant, add 1 ml phosphate-buffered saline (PBS) solution, re-suspend the cells, place the tube in the centrifuge again, centrifuge at 1,200 rpm for 5 min, discard the supernatant after centrifugation, add PBS according to the cell density to prepare 1 × 105 cell/μl MenSCs suspension for cell transplantation. The prepared cell suspension was temporarily stored in a 4°C refrigerator to prepare an SCI model for transplantation. The prepared cells were injected within 6 h. After establishing the SCI model, use a glass tube capillary needle, aspirate 2 μl (2 × 105 cell) of the prepared cell suspension, and then, inject into the injury site with an injection depth of 0.5 mm and an injection duration of 10 s. After the injection, the muscle layer on the back of the injured site was sutured, and then the skin was sutured, and the operation was completed. Here, it should be noted that the prepared MenSCs or Shh recombinant protein (R-Shh)–induced MenSCs cell suspension was infused at the site of SCI using a capillary needle. After suspension infusion, the capillary needle still needs to remain in the damaged part for a period of time to prevent the cell suspension from flowing out. In addition, the concentration of the cell suspension should not be too high, or it would block the capillary needle; and it should not be too low, or the amount of cell transplantation would be too low.
Behavioral Experiments
Motor function recovery in SCI mice at 0, 1, 3, 5, 7, 14, 21, and 28 days after SCI was evaluated using the 9-point BMS scoring method 35 . SCI mice were placed in a self-made scoring device and three individuals were observed for 5 min. The BMS scores of the mice were recorded, and the average score was obtained. Animals that underwent non-model damage operators independently completed the scoring procedure.
RNA-Seq and Data Analysis
Spinal cord segments centered at the crush from mice with different treatments were collected for RNA-Seq, each group three samples at 7 days. The samples were sent to Personal Bio (Personal Gene Technology, Nanjing, China) for total RNA extraction for library construction and high-throughput sequencing (NCBI Accession: PRJNA858927 ID: 858927, https://www.ncbi.nlm.nih.gov/sra/PRJNA858927). Differential gene expression analysis, heatmap, Gene Ontology (GO) enrichment analysis, and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis were performed using the personal biogenes cloud platform gadget Complete. Transcripts with |log2FC| > 1 and adjusted P value < 0.01 were considered as differentially expressed genes (DEGs). DEGs were identified based on three contrasts: sham versus +MenSCs, SCI versus +MenSCs, and sham versus SCI on day 7.
Western Blotting
The extracted cell or tissue proteins were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), followed by wet membrane transfer. The proteins were transferred to a polyvinylidene fluoride membrane and then sealed with 5% skimmed milk powder solution at room temperature for 1 h. Finally, rabbit anti-Ki67 (Abcam; UK; ab15580; 1:500), mouse anti-c-Myc (Abcam; UK; ab56; 1:500), mouse anti-Shh (Santa Cruz; USA; sc-365112; 1:100), rabbit anti-Gli1 (Abcam; UK; ab49314; 1:200), rabbit anti-inducible nitric oxide synthase (anti-iNOS) (Abcam; UK; ab178945; 1:1,000), mouse anti-CD68 (Santa Cruz; USA; sc-70761; 1:100), mouse anti-CD206 (Santa Cruz; USA; sc-70585; 1:100), rabbit anti-tumor necrosis factor-α (anti-TNF-α) (Abcam; UK; ab215188; 1:1,000), rabbit anti-IL-1 β (Abcam; UK; ab254360; 1:1,000), mouse anti-IL-6 (Santa Cruz; USA; sc-32296; 1:100), rabbit anti-glial fibrillary acidic protein (anti-GFAP) (Abcam; UK; ab33922; 1:2,000), rabbit anti-neuronal nuclei (anti-NeuN) (Abcam; UK; ab177487; 1:2,000), rabbit anti-ionized calcium binding adapter molecule 1 (anti-Iba1) (Abcam; UK; ab153696; 1:1,000), mouse anti-IL-10 (Santa Cruz; USA; sc-365858; 1:100), and rabbit anti-transforming growth factor-β1 (anti-TGF-β1) (Abcam; UK; ab215715; 1:1,000) primary antibodies were incubated overnight at 4°C. The next day, the membranes were washed with tris buffered saline+tween (TBST) three times for 10 min each, horseradish peroxidase (HRP)–labeled goat anti-rabbit (ZSGB-BIO; China; ZB-2301; 1:10,000) and goat anti-mouse (ZSGB-BIO; China; ZB-2305; 1:10,000) secondary antibodies were incubated at room temperature for 2 h, followed by washing three times with TBST, and protein expression was detected using a gel imaging system.
Tissue Collection
Mice were anesthetized on the seventh day after SCI. First, the hearts were perfused with normal saline for 5 min, and whole-body blood was completely replaced. After perfusion with 4% paraformaldehyde (PFA) (Servicebio; Wuhan, China; G1101) for 15 min, the spinal cord was removed and fixed overnight with 4% PFA, followed by dehydration in 18% and 30% sucrose. After embedding the tissue with optimal cutting temperature compound (SAKURA; USA; 4583), frozen sections with a thickness of 20 μm were prepared.
Tissue Immunofluorescence
Tissue sections were fixed with 4% PFA solution for 15 min, incubated with 0.3% TritonX-100 (Sangon Biotech; China; 9002-93-1) for 5 min, and blocked with immunostaining blocking solution (Beyotime; China; P0260) at room temperature for 1 h. Finally, 300 μl of primary antibodies against NeuN (Abcam; UK; ab177487; 1:200), GFAP (Abcam; UK; ab33922; 1:500), Iba1 (Abcam; UK; ab153696; 1:100), iNOS (Abcam; UK; ab178945; 1:500), CD68 (Santa Cruz; USA; sc-70761; 1:50), and CD206 (Santa Cruz; USA; sc-70585; 1:50) were added and incubated overnight at 4°C. The next day, the samples were washed with tris-buffered saline (TBS) three times for 10 min each. Thereafter, CY™ 3 affinity goat anti-rabbit IgG (Jackson ImmunoResearch, USA; 111-165-003; 1:500) and CY™ 3 affinity goat anti-mouse IgG (Jackson ImmunoResearch, USA; 115-165-003; 1:500) secondary antibodies were incubated at 4°C in the dark for 4 to 6 h, and then washed with TBS three times for 10 min each time. 4′,6-diamidino-2-phenylindole (DAPI) (BIOSHARP; China; BS097; 1:1,000) was stained, and 10 μl anti-fluorescence quenching agent (vector labs; USA; H-1200) was added. Then, microscopic examination was performed.
Statistical Analysis
The results of the cellular and tissue immunofluorescence analyses were analyzed. Photoshop software was used to process images. The results of WB were quantified using ImageJ software. All analytical data are presented as the mean ± standard deviation (SD) of at least three independent experiments, and the results were analyzed using SPSS software (IBM, USA). After ensuring that the data had a normal distribution and variance equality (Levene’s test), analysis of variance (ANOVA) followed by least significant difference (LSD) were used to estimate the significant differences among the different groups, where a P value < 0.05 was considered significant.
Results
MenSCs Transplantation Significantly Improves the Locomotor Function of Mice
C57BL/6 mice–7–8 weeks were used to establish the free-fall spinal cord crush model (Fig. 1A). According to the BMS score, the SCI group was regarded as meeting the experimental standard if the score was zero after the crush. In the MenSC-treated group, MenSCs were injected intrathecally after crushing (Fig. 1B). In the sham operation group, only the spinal canal was dissected, without crushing. BMS scores were obtained on days 0, 1, 3, 5, 7, 14, 21, and 28 after the experiment in each group (Fig. 1B). From the statistical results of the BMS score, it can be seen that the MenSCs group had a significantly higher score than the SCI group from the first day, indicating that MenSCs significantly improved locomotor function in SCI mice (Fig. 1C).

SCI model establishment and transcriptome sequencing analysis. (A) SCI model establishment. (B) Experimental scheme. (C) BMS score statistical analysis. (D) Spinal cord tissue sample. (E) Number of differentially expressed genes in transcriptome sequencing (P < 0.05). (F) Selected gene heat map. (G) Sham vs SCI KEGG pathway. (H) Sham vs MenSCs KEGG pathway. (I) SCI vs MenSCs KEGG pathway. SCI = spinal cord injury; BMS = Basso Mouse Scale; KEGG = Kyoto Encyclopedia of Genes and Genomes; MenSCs = human menstrual blood–derived endometrial stem cells.
MenSC Treatment Alters the Expression of Genes Functionally Associated With Inflammation
We further explored the molecular mechanism of MenSC transplantation in the treatment of SCI and determined the possible direct and indirect target molecules of MenSCs in the injured spinal cord. We collected the spinal cord tissues of the sham operation, SCI, and MenSC treatment groups at 7 days and performed transcriptome sequencing analysis (Fig. 1D). Multiple pairwise comparisons were performed, and 7,822 DEGs (with |log2FC|>1 and adjusted P < 0.01) were identified based on three contrasts: sham versus SCI, sham versus MenSCs, and SCI versus MenSCs at 7 days (Fig. 1E). DEGs in different groups were compared. The results showed that, compared with the sham operation group, 1,793 genes were upregulated and 705 genes were downregulated after SCI, whereas 2,733 genes were upregulated and 1,954 genes were downregulated after MenSCs transplantation. In the SCI and SCI-transplanted MenSCs groups, 587 genes were upregulated, and 50 genes were downregulated (Fig. 1E).
Cluster analysis was performed based on transcriptome sequencing results. The results showed that three samples from the sham group were clustered together and then clustered together with one sample from the SCI group, although the other two samples from the SCI group were clustered together, and two samples from the MenSCs group were clustered together, clustered together with the SCI group, and finally clustered together with the sham (Fig. S1A). The clustering results showed that one sample in the SCI group did not meet the SCI standard, and one sample in the MenSCs group had a poor treatment effect. Other samples met the experimental requirements and reflected real data. Cluster analysis also showed significant differences between the MenSCs and SCI groups after transplantation; however, the expression of these genes was closer to that in the SCI group than in the sham group.
GO enrichment analysis revealed that SCI activated the immune system process of spinal cord tissue, whereas MenSCs activated immune and defense responses (Fig. S1B, C, and D). Furthermore, based on the significance of gene expression differences in the different groups (P < 0.05), inflammation-related factors and glial cell markers were selected and a heat map was created (Fig. 1F). From the map, it can be seen that compared with the SCI group, MenSC transplantation significantly increased the expression of anti-inflammatory factors such as interleukin-10 (IL-10), while simultaneously inhibiting the expression of the glial cell marker GFAP (Fig. 1F). In this heat map, there were significant differences in Shh expression between the different groups (Fig. 1F). Compared with the sham group, the expression was significantly increased in the SCI group and significantly decreased in the MenSCs group (Fig. 1F). Shh is an important morphogenetic protein that is involved in spinal cord development. Furthermore, KEGG pathway analysis showed that, compared with the sham group, MenSC transplantation significantly activated the Th17, Th1, and Th2 cell differentiation pathways in the organic systems (Fig. 1G, H, I). These results indicate that the repair of SCI after transplantation of MenSCs occurs mainly through regulating the expression of the inflammatory microenvironment and related factors.
The Function of Shh in SCI
From the transcriptome sequencing results, it was found that the expression of Shh was low in the sham group but significantly increased in the SCI group. After MenSC transplantation, Shh expression was significantly reduced. Through fluorescence immunohistochemistry and WB experiments, we verified that the overall trend of Shh expression was consistent with the results of transcriptome sequencing (Fig. 2A–L). However, there were some differences. Transcriptome sequencing results showed that the expression of Shh in the SCI group was higher than that in the sham group, and higher than that in the stem cell transplantation group (Fig. 1F). In fluorescent immunohistochemistry, the expression of Shh in the SCI group was higher than that in the stem cell transplantation group and higher than that in the sham group (Fig. 2A–L). This also suggested that the amount of mRNA expression was not completely consistent with the amount of protein expression. However, the overall trend was that the expression of Shh in SCI increased, and the expression of Shh decreased after MenSCs transplantation. What is the function of Shh in SCI repair? Studies 36 have shown that Shh induces the differentiation of human adipose stem cells into neuron/motor neuron-like cells and has a significant repair effect on SCI. Therefore, we used the R-Shh to induce MenSCs in vitro and then performed cell transplantation to repair SCI. To explore the efficacy of Shh-induced MenSCs in SCI, MenSCs cultured in vitro were treated with R-Shh, an Shh inhibitor (HPI-1), and a Gli inhibitor (GANT-61) for 48 h. Compared with the control group, the cell morphology changed significantly. The cells in the R-Shh group became larger and polymorphic, and those in the HPI-1 and GANT-61 groups became fibrous, maintaining high stem cell characteristics (Fig. S2A–H). Furthermore, the expression of cell proliferation proteins Ki67 and c-Myc was detected. The results showed that R-Shh promoted the expression of Ki67 and c-Myc, whereas HPI-1 and GANT-61 inhibited their expression (Fig. S3A–E). These results indicated that R-Shh promotes the proliferation of MenSCs in vitro.
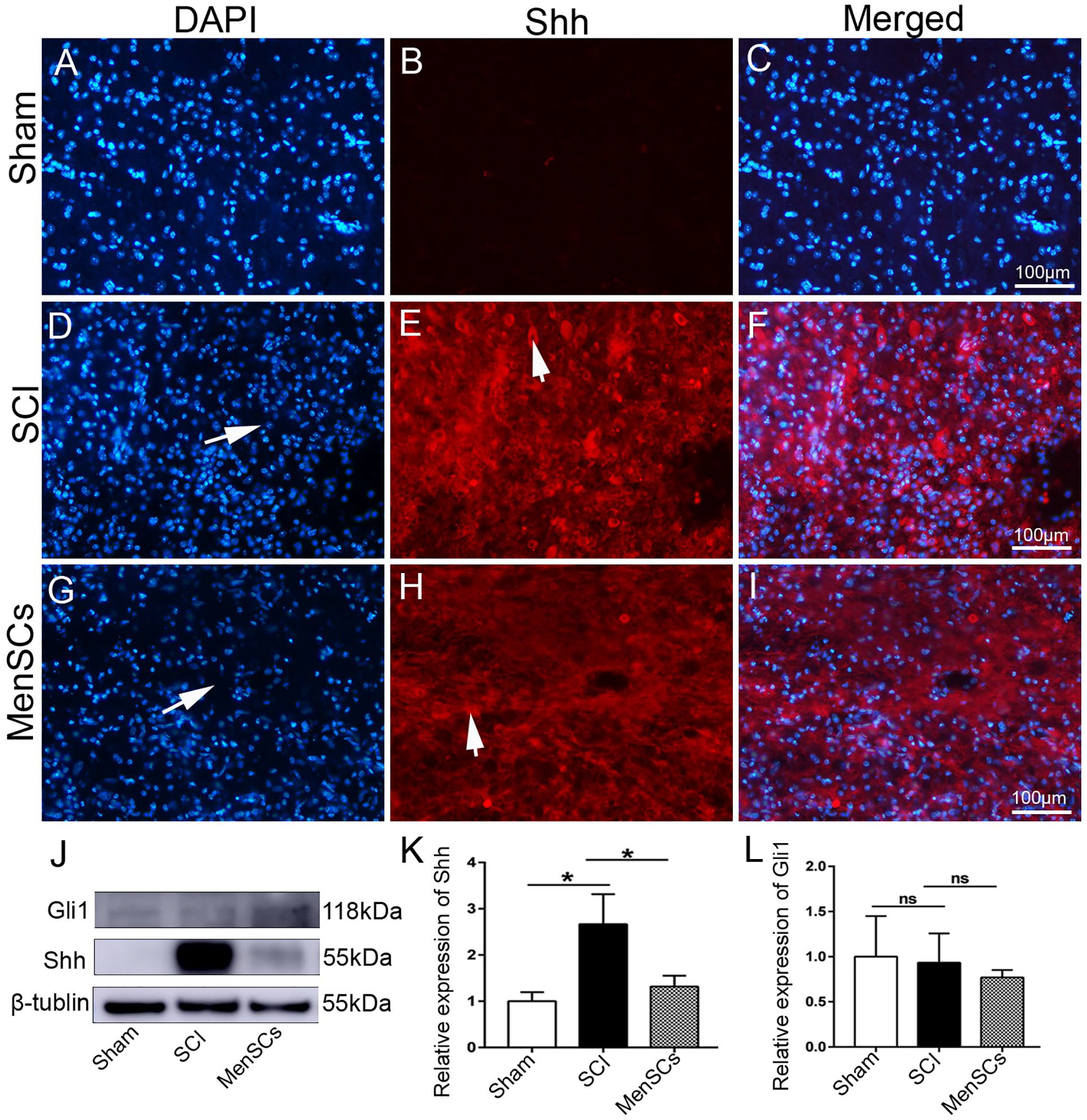
Shh expression in different groups. (A–I) Fluorescence immunohistochemistry. (A–C) Sham group. (D–F) SCI group. (G–I) MenSCs group. (A, D, G) DAPI staining nucleus (blue). (B, E, H) Shh expression (red). (C, F, I) Merge each group. (J–L) WB analysis. (J) Shh and Gli1 expression. (K) Relative expression of Shh. (L) Relative expression of Gli1. Arrows in (D) and (G) show the SCI area; arrows in (E) and (H) show Shh-positive cells; scale bar in (C), (F), and (I) is 100 μm for (A–C), (D–F), and (G–I). P < 0.05 were considered significant and are marked “*.” P > 0.05 were considered not significant and marked “ns.” DAPI = 4′,6-diamidino-2-phenylindole; Shh = sonic hedgehog; SCI = spinal cord injury; MenSCs = human menstrual blood–derived endometrial stem cells; WB = Western blotting.
MenSCs and Shh-Induced MenSCs (Shh + MenSCs) Promote the Recovery of Neurons at the Injured Site and Inhibit the Proliferation of Glial Cells
The direct result of SCI is neuronal injury, and the injured neurons cannot regenerate, which is also why SCI is difficult to repair completely. According to the results of fluorescence immunohistochemistry and WB detection in the different groups, compared with those in the sham group, NeuN-labeled positive cells in the injury site were significantly reduced in the SCI group, whereas in the MenSCs and Shh + MenSCs groups, there were some NeuN-positive neurons at the injury site (Fig. 3A–H). This also shows that MenSCs and Shh-induced MenSCs can promote the recovery of neuronal function at the site of injury. Compared with the Shh + MenSCs group, the MenSCs group had more NeuN-positive neurons (Fig. 3A–H), which also shows that these neurons are not differentiated by MenSCs. However, the neurons that have not completely died at the site of autologous SCI have recovered their functions under the improved microenvironment of MenSCs.

Expression in neurons and glial cells. (A–P) Fluorescence immunohistochemistry, (A–D) DAPI staining of nuclei (blue) in different groups, (E–H) NeuN expression (red), (I–L) GFAP expression (red), (M–P) Iba-1 expression (red). (R–T) WB analysis. (Q) NeuN, GFAP, and Iba-1 expression, (R) Relative expression of NeuN, (S) Relative expression of GFAP, (T) Relative expression of Iba-1. Arrows in (B, C, and D) show the SCI area; arrows in (E–H) show NeuN-positive cells; arrows in (I–L) show GFAP-positive cells; arrows in (M–P) show Iba-1 positive cells; the scale bar is 100 μm. P < 0.05 was considered significant and are marked “*.” P > 0.05 was considered not significant and marked “ns.” DAPI = 4′,6-diamidino-2-phenylindole; WB = Western blotting; SCI = spinal cord injury; MenSCs = human menstrual blood–derived endometrial stem cells.
Tissue injury can cause inflammatory reactions. In SCI, inflammatory reactions activate the proliferation of glial cells. From the results, it can be seen that, compared with those in the sham group, GFAP and Iba-1 labeled positive cells in the SCI group were significantly increased (Fig. 3I–J, M–N). However, compared with those in the SCI group (Fig. 3J, N), GFAP and Iba-1 labeled positive cells were significantly reduced in the MenSC group (Fig. 3K, O), and Shh + MenSCs (Fig. 3L, P), which also shows that transplantation of MenSCs can improve the inflammatory microenvironment and inhibit excessive proliferation of glial cells.
The results of the WB test showed that compared with the sham group, NeuN expression was the lowest in the SCI group and increased in the MenSCs and Shh + MenSCs groups (Fig. 3Q, R). GFAP and Iba-1 levels were lowest in the sham group, significantly increased in the SCI group, and decreased after transplantation of MenSCs, but abnormally increased in the Shh + MenSCs group, which may be related to the difference in samples or individuals (Fig. 3Q, S, T).
In differential gene expression analysis using transcriptome sequencing, it has been shown that MenSCs can inhibit the expression of GFAP after transplantation, and the protein expression results detected by WB are consistent with the results of the number of fluorescent immunohistochemically positive cells, which also validates the transcriptome sequencing results.
The tissues on the 14 and 28 days were sectioned and stained. It can be seen NeuN-labeled neurons in the sham group had uniformly marked positive nuclei on the 14 and 28 days (Fig. 4A: a–c; C: a–c). In the SCI group, there were no NeuN-positive neurons in the center of the injured site on the 14 and 28 days (Fig. 4A: d–f; C: d–f). However, in the cell transplantation group, there were a few NeuN-positive neurons at the injured site at 14 days (Fig. 4A: g–l), and the number of positive neurons at 28 days was more obvious (Fig. 4C: g–l) but compared with the sham group (Fig. 4A: a–c; C: a–c), there were fewer NeuN-positive neurons in the center of the injured site than in the surrounding tissues (Fig. 4A: g–l; C: g–l).

Formation of neurons, glial cells, and scars at the lesion. (A–D) Fluorescence immunohistochemistry. (A) NeuN-labeled positive neurons at the lesion site on day 14. (a, d, g, j) DAPI staining nucleus (blue). (b, e, h, k) NeuN expression (red). (c, f, i, l) merge. (B) shows GFAP-labeled positive neurons at the lesion site on day 14. (a, d, g, j) DAPI staining nucleus (blue); (b, e, h, k) GFAP expression (red); (c, f, i, l) merge. (C) shows NeuN-labeled positive neurons at the lesion site on day 28. (a, d, g, j) DAPI staining nucleus (blue); (b, e, h, k) NeuN expression (red); (c, f, i, l) merge. (D) shows GFAP-labeled positive neurons at the lesion site on day 28. (a, d, g, j) DAPI staining nucleus (blue); (b, e, h, k) GFAP expression (red); (c, f, i, l) merge. Scale bar is 100 μm. Arrows in A: a–c and C: a–c show NeuN-positive neurons, and A: d–l and C: d–l show the lesion. Arrows in B: a–c show GFAP-positive glial cells, and B: d–l and D: d–l show scars at the lesion. DAPI = 4′,6-diamidino-2-phenylindole; SCI = spinal cord injury; MenSCs = human menstrual blood–derived endometrial stem cells.
Among the GFAP-labeled glial cells, a few GFAP-positive cells were found in the sham group at 14 and 28 days (Fig. 4B: a–c; D: a–c). In the SCI group, there were a large number of GFAP-positive glial cells at the injured site and surrounding area at 14 and 28 days (Fig. 4B: d–f; D: d–f). However, in the cell transplantation group, on days 14 and 28, there were only a few GFAP-positive glial cells in the center and surrounding areas of the injury site, whereas some GFAP-positive cells were observed in the border area (Fig. 4B: g–l; D: g–l). The results show that the GFAP-labeled neurons are not regular glial cells but fibrous structures (Fig. 4B: d–f; D: d–f). At the same time, dense nuclei in the central area are surrounded by GFAP-labeled fibers, but this area is neither composed of NeuN-labeled neurons nor GFAP-labeled glial cells. Therefore, scars are formed in these areas (Fig. 4B: d–f; D: d–f, shown by arrows). However, no obvious scar formation was observed in the transplantation group (Fig. 4B: g–l; D: g–l).
MenSCs Transplantation Significantly Reduces Macrophages After SCI
After SCI, microglia are activated, and macrophages are formed. CD68 is a marker of macrophages, iNOS is a marker of M1 macrophages, and M1 macrophages mainly secrete pro-inflammatory factors. CD206 is a marker for M2 macrophages that secrete anti-inflammatory factors. Therefore, different macrophage types were detected in these tissues. Fluorescent immunohistochemical results showed that compared with the sham group, the number of CD68 and CD206 labeled cells in the SCI group increased significantly, whereas those in the MenSCs, and Shh + MenSCs groups decreased significantly (Fig. 5A–P). The expression of CD68, CD206, and iNOS proteins was detected by WB (Fig. 5Q). The results showed that, compared with the sham group, the SCI group showed a significant increase (Fig. 5R–T). Compared with the SCI group, the relative expression of MenSCs and Shh + MenSCs groups decreased, and there was a significant difference between the CD68 and iNOS groups (Fig. 5R–T). The above results showed that, compared with the SCI group, after MenSC transplantation, CD68, iNOS, and CD206 expression decreased in both fluorescent immunohistochemistry and WB results, indicating that the total number of macrophages and M1 and M2 macrophages decreased.
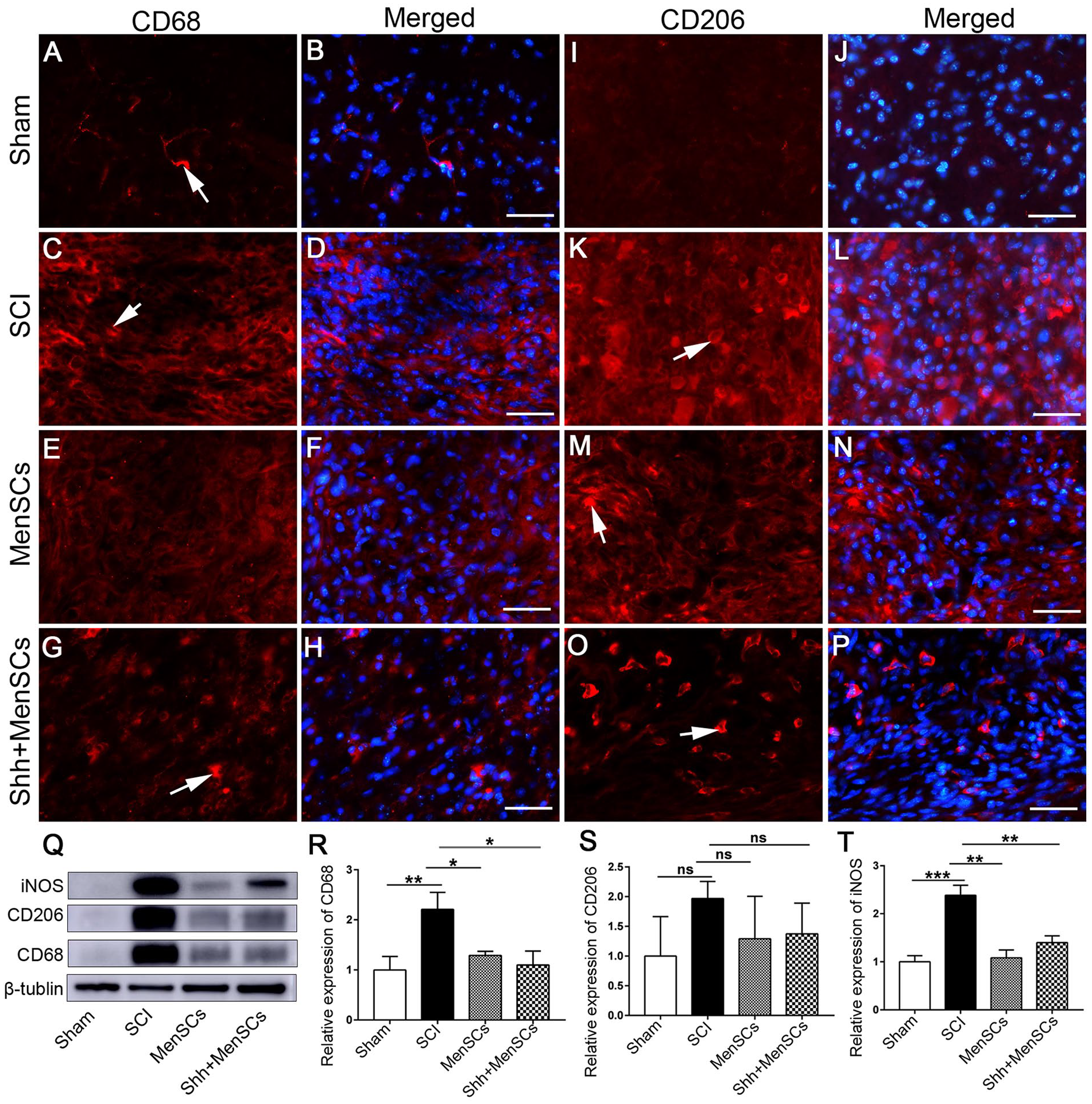
Expression in microglia. (A–P) Fluorescence immunohistochemistry. (A, C, E, G) CD68 expression (red). (B, D, F, H) Merge of (A, C, E, G) and DAPI staining nucleus, respectively. (I, K, M, O) CD206 expression (red). (J, L, N, P) Merge of (I, K, M, O) and DAPI staining nuclei, respectively. (Q–T) WB analysis. (Q) CD68, CD206, and iNOS expression. (R) Relative expression of CD68. (S) Relative expression of CD206. (T) Relative expression of iNOS. Arrows in (A, C, G) show CD68 positive cells; arrows in (K, M, O) show CD206 positive cells; the scale bar is 50 μm. P < 0.05 was considered significant and are marked “*.” P > 0.05 was considered not significant and marked “ns.” DAPI = 4′,6-diamidino-2-phenylindole; iNOS = inducible nitric oxide synthase; SCI = spinal cord injury; MenSCs = human menstrual blood–derived endometrial stem cells; WB = Western blotting.
MenSCs and Shh + MenSCs Improve the SCI Microenvironment by Inhibiting the Expression of Inflammatory Factors
KEGG analysis of transcriptome sequencing revealed that, compared with other groups, MenSC transplantation significantly activated Th1, Th2, and Th17 cell differentiation pathways (Fig. 1H, I). Th1, Th2, and Th17 cells belong to different subsets involved in Th0 differentiation. Cytokines α-interferon (IFN-α), interleukin-12 (IL-12), and γ-interferon (IFN-γ) regulate Th1 cell differentiation; interleukin-4 (IL-4) and interleukin-13 (IL-13) regulate Th2 cell differentiation; and interleukin-6 (IL-6) and transforming growth factor-β (TGF-β) regulate the differentiation of Th17 cells.
From the above results, we found that the number of both M1 and M2 macrophages decreased after MenSC transplantation. M1 macrophages mainly secrete pro-inflammatory factors; however, they must be activated by factors secreted by helper cells Th1. M2 macrophages mainly secrete anti-inflammatory factors that are activated by the factors secreted by Th2 cells. Th1 cells secrete cytokines, such as IL-2 and IFN-γ, and TNF-α, and Th2 cells secrete cytokines, such as IL-4, IL-5, IL-6, IL-9, IL-10, and IL-13. We then tested whether there were differences in the expression of these inflammatory factors in the different groups using WB (Fig. 6A–G).

Expression analysis of inflammatory factors. (A–D) Expression analysis of pro-inflammatory factors: (A) TNF-α, IL-1β, and IL-6 expression. (B) Relative expression of TNF-α. (C) Relative expression of IL-1β. (D) Relative expression of IL-6. (E–G) Expression analysis of anti-inflammatory factors. (E) IL-10 and TGF-β expression. (F) Relative expression of IL-10. (G) Relative expression of TGF-β. P < 0.05 was considered significant and are marked “*.” P > 0.05 was considered not significant and marked “ns.” SCI = Spinal cord injury; MenSCs = human menstrual blood–derived endometrial stem cells; IL-1β = interleukin-1β; IL-10 = interleukin-10; IL-6 = interleukin-6; TGF-β = transforming growth factor-β; TNF-α = tumor necrosis factor-α.
The results showed no significant differences in TNF-α levels among the four groups (Fig. 6B). The IL-1β and IL-6 levels were significantly higher in the SCI group than in the sham group (Fig. 6C, D). Compared with the SCI group, the expression of IL-1β in the MenSCs and Shh + MenSCs groups decreased; however, the difference was not significant (Fig. 6C). IL-6 decreased significantly, which was statistically significant (Fig. 6D). In the detection of anti-inflammatory factors, compared with that in the sham group, the expression of IL-10 and TGF-β1 in the SCI, MenSCs, and Shh + MenSCs groups increased, whereas compared with that in the SCI group, the expression of IL-10 and TGF-β1 in the MenSCs and Shh + MenSCs groups decreased, but the difference was not significant (Fig. 6E–G).
Discussion
Several studies have been conducted on the treatment of SCI using stem cells. The ideal treatment strategy is to transplant homologous stem cells, such as NSCs, which can differentiate into functional neurons after transplantation to achieve SCI repair 37 . However, there are many problems associated with the role of NSCs in the treatment of SCI. The main reason for this is that most transplanted NSCs differentiate into astrocytes rather than neurons in the SCI microenvironment 38 . Other types of stem cells of human origin also have positive effects on SCI in experimental animals, such as human adipose–derived stem cells (hADSCs), hUC-MSCs, and human placenta–derived mesenchymal stem cells (hPMSCs)36,39,40. These results confirmed that human-derived stem cells can be effectively used in experimental animals.
In this study, the role and molecular biological mechanisms of MenSCs after transplantation were studied using a mouse model of crush SCI. The animal behavior experiment showed that the BMS score of mice transplanted with MenSCs was significantly higher than that of the SCI group after transplantation for 1 days, which showed that MenSC transplantation could significantly improve motor function recovery in SCI animals. Previous studies 33 have shown that in the rat spinal cord hemisection model, the Basso, Beattie, and Bresnahan (BBB) scores of rats were significantly higher than those of the untreated group of MenSCs transplantation after 7 days. These results indicate that MenSCs have good therapeutic effects in SCI. The stem cells used in this study were derived from human stem cells. What are the effects and molecular mechanisms of SCI repair? Transcriptome sequencing and GO enrichment analysis revealed that SCI was closely related to immune system processes. Through transplantation of MenSCs, immune response and immune defense are significantly activated. KEGG analysis revealed that MenSC transplantation was closely related to Th1, Th2, and Th17 cell differentiation pathways. These results provide important insights. We speculate that the improvement in SCI functional recovery by MenSCs may be closely related to the improvement of the inflammatory microenvironment. On the basis of this information, we conducted a series of experimental studies on inflammation.
In the present study, the results showed that in SCI group, neurons were damaged, and GFAP and Iba-1 labeled glial cells increased significantly, while in MenSCs group, neurons recovered significantly, and the number of GFAP and Iba-1 positive cells also decreased significantly. This is because SCI leads to a massive inflammatory response, and glial cells are activated 33 . Activated microglia form macrophages under inflammatory conditions. Macrophages in the central nervous system (CNS) are divided into M1 and M2 microglia. Microglia play different roles in diseases of the nervous system 41 . M1 microglia secrete pro-inflammatory factors with the help of Th1 cells, whereas M2 microglia secrete anti-inflammatory factors from Th2 cells. This is consistent with the results of transcriptome sequencing. Transcriptome sequencing revealed that MenSC transplantation was significantly related to Th1, Th2, and Th17 cell differentiation processes.
Th1 and Th2 cells play important roles in SCI. Th1 cells and M1 macrophages secrete pro-inflammatory factors such as cytokines (IFN-γ, TNF-α, β-β, and IL-1-β). Th2 cells, regulatory T cells, and M2 macrophages secrete anti-inflammatory cytokines, such as cytokines (IL-4, IL-10, and IL-13) 42 . Th17 cells can produce IL-17 and play an important role in the immune response, and IL-17 is a pro-inflammatory factor 43 . In this study, MenSCs regulated the differentiation and activation of Th2 and M2 macrophages, produced anti-inflammatory factors, reduced the inflammatory response at the injured site, and thus promoted the recovery of SCI function. In the present study, M1 and M2 microglia were detected using iNOS and CD206, respectively. The results showed that, compared with that in the SCI group, the expression of iNOS and CD206 in the MenSCs transplantation and Shh + MenSCs groups was significantly reduced. In contrast, WB quantitative analysis showed that the expression of CD206 in the MenSC transplantation and Shh + MenSCs groups was slightly higher than that in the sham group, whereas the expression of iNOS was not different from that in the sham group. Therefore, MenSCs improved the highly inflammatory microenvironment, reduced the overactivation of microglia in a highly inflammatory environment, and made M1 and M2 microglia reach an appropriate ratio. This regulation improves the SCI microenvironment and provides an environment that is conducive to neuronal recovery. We know that the formation and activation of M2 microglia can promote SCI repair, which is also supported by relevant experimental results 44 . However, SCI is a highly inflammatory environment, and the overexpression of inflammatory factors promotes microglial formation and activation in the CNS. Under these conditions, MenSC transplantation can inhibit this pathway, and inhibition of the formation and activation of M1 or M2 microglia is beneficial for neuronal recovery.
Thereafter, with the help of Th1 and Th2 cells, what happens to the expression of pro-inflammatory and anti-inflammatory factors secreted by M1 or M2 microglia? Our results also showed that pro-inflammatory and anti-inflammatory factors differed between the different groups. Stem cell transplantation can inhibit the expression of pro-inflammatory factors and promote the expression of anti-inflammatory factors33,37,45,46. In this study, after MenSC transplantation, the expression trends of the pro-inflammatory factors IL-1β and IL-6 were consistent with those reported in previous studies. MenSCs inhibited the expression of pro-inflammatory factors. This is consistent with a previous trend of decreasing numbers of M1 microglia. However, the protein expression levels of anti-inflammatory factors detected by WB and fluorescence immunohistochemistry are not completely consistent with existing research results. In our results, compared with those in SCI, the expression levels of IL-10 and TGF-β1 in the MenSCs transplantation group and Shh + MenSCs transplantation group were lower. However, in the transcriptome sequencing results, the expression of IL-10 in the MenSC transplantation group was significantly higher than that in the SCI group, which was consistent with the results of previous studies. We noticed that the transcriptome sequencing results are not completely consistent with the WB results, which attracted our attention.
In stem cells that inhibit Th1 and Th17 differentiation, MSCs suppress the proliferation and phenotype of mature Th1 and Th17 cells and do not generate Treg cells 47 . Direct studies 48 have also shown that MenSCs significantly inhibit Th17 differentiation. Seven-day differentiated allogeneic nonchondrogenic and chondrogenic MSC pellets co-cultured with memory T cells resulted in a significant increase in Th2 and a decrease in Th1 T cells 49 . These studies have shown that MSCs inhibit the differentiation of Th1 and Th17 cells and promote the differentiation of Th2 cells. This also confirmed our finding that MenSCs inhibited the expression of pro-inflammatory factors and promoted the expression of anti-inflammatory factors. What is the mechanism by which MenSCs regulate pro-inflammatory and anti-inflammatory factors? Exosomes play a major role in communication between MSCs and target tissues 50 . This study shows that exosomes may induce the conversion of Th1 cells into Th2 cells and reduce the potential of T cells to differentiate into interleukin 17-producing effector T cells (Th17) 50 . This study also supports the above research view that the mechanism of MenSCs pro-inflammatory and anti-inflammatory effect in SCI is mainly through exosomes or cytokines secreted by MenSCs.
In the present study, we added a group of MenSCs induced by Shh. Among the various detection indicators, we found no significant difference between the Shh + MenSCs and MenSCs groups, and some indicators were not as ideal as the MenSCs group. This also shows that the mechanism by which MenSCs promote the functional recovery of SCI may not be through the differentiation of MenSCs into neurons or glial cells but through the factors secreted by MenSCs to improve the SCI microenvironment and recover the function of SCI. This study shows that improving the inflammatory microenvironment promotes functional recovery in SCI, which needs to be further studied.
Conclusion
MenSC transplantation and Shh-induced MenSCs accelerated the recovery of neurons at the injured site, inhibited the formation of glial cells, inhibited microglial activation at the injured site, inhibited the expression of inflammatory factors, and improved the inflammatory microenvironment to achieve functional recovery of SCI. This study provides an experimental basis for studying the role and molecular mechanism of MenSCs in SCI repair, and a reference for the role of Shh-induced MenSCs in SCI repair.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231154579 – Supplemental material for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model
Supplemental material, sj-docx-1-cll-10.1177_09636897231154579 for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model by Yaping Shi, Yunfei Liu, Bichao Zhang, Xiaoying Li, Juntang Lin and Ciqing Yang in Cell Transplantation
Supplemental Material
sj-docx-2-cll-10.1177_09636897231154579 – Supplemental material for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model
Supplemental material, sj-docx-2-cll-10.1177_09636897231154579 for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model by Yaping Shi, Yunfei Liu, Bichao Zhang, Xiaoying Li, Juntang Lin and Ciqing Yang in Cell Transplantation
Supplemental Material
sj-jpg-3-cll-10.1177_09636897231154579 – Supplemental material for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model
Supplemental material, sj-jpg-3-cll-10.1177_09636897231154579 for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model by Yaping Shi, Yunfei Liu, Bichao Zhang, Xiaoying Li, Juntang Lin and Ciqing Yang in Cell Transplantation
Supplemental Material
sj-jpg-4-cll-10.1177_09636897231154579 – Supplemental material for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model
Supplemental material, sj-jpg-4-cll-10.1177_09636897231154579 for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model by Yaping Shi, Yunfei Liu, Bichao Zhang, Xiaoying Li, Juntang Lin and Ciqing Yang in Cell Transplantation
Supplemental Material
sj-jpg-5-cll-10.1177_09636897231154579 – Supplemental material for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model
Supplemental material, sj-jpg-5-cll-10.1177_09636897231154579 for Human Menstrual Blood–Derived Endometrial Stem Cells Promote Functional Recovery by Improving the Inflammatory Microenvironment in a Mouse Spinal Cord Injury Model by Yaping Shi, Yunfei Liu, Bichao Zhang, Xiaoying Li, Juntang Lin and Ciqing Yang in Cell Transplantation
Footnotes
Acknowledgements
We thank Dr. Yanli Liu, Yanan He, and Feng Yang for kindly providing us the MenSCs and purchase of laboratory animals and reagents.
Author Contributions
Ciqing Yang and Juntang Lin conceived and designed the experiments. Yaping Shi, Yunfei Liu, Bichao Zhang, and Xiaoying Li performed the experiments. Yaping Shi analyzed the data. Yaping Shi and Ciqing Yang wrote the paper.
Ethical Approval
This study was approved by the ethical review board of Xinxiang Medical University, Xinxiang, China.
Statement of Human and Animal Rights
All the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care and Use Committee guidelines of Xinxiang Medical University, Xinxiang, China.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Henan Province Science and Technology Project (212102310215), the Key Scientific Research Project of Henan Province Colleges and Universities (23A180005), Henan Key Laboratory of Neurorestoratology (HNSJXF-2021-010), Xinxiang Medical University Research Innovation Support Program (YJSCX202168Y), the PhD Research Startup Foundation (505090) of Xinxiang Medical University.
Data accessibility statement
All data generated or analyzed during this study are included in the main text and the supplementary information files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
