Abstract
Thin endometrium (TE) is a significant factor contributing to fertility challenges, and addressing this condition remains a central challenge in reproductive medicine. Menstrual blood–derived mesenchymal stem cells (MenSCs) play a crucial role in tissue repair and regeneration, including that of TE. The Wnt signaling pathway, which is highly conserved and prevalent in eukaryotes, is essential for cell proliferation, tissue development, and reproductive functions. MALAT1 is implicated in various transcriptional and molecular functions, including cell proliferation and metastasis. However, the combined effects of the Wnt signaling pathway and the long non-coding RNA (lncRNA) MALAT1 on the regulation of MenSCs’ regenerative capabilities in tissue engineering have not yet been explored. To elucidate the regulatory mechanism of MALAT1 in TE, we analyzed its expression levels in normal endometrium and TE tissues, finding that low expression of MALAT1 was associated with poor clinical prognosis. In addition, we conducted both
Keywords
Introduction
The growth and shedding phases of the menstrual cycle are essential for maintaining women’s reproductive health and ensuring successful pregnancies 1 . However, various factors such as invasive procedures, infections, certain medications, and underlying conditions can lead to thin endometrium (TE) and damage to the basal layer of the uterine cavity 2 . Such damage or excessive thinning of the endometrium impedes the essential regeneration and repair processes crucial for optimal fertility. TE not only hampers embryo implantation but also heightens the risk of female infertility and recurrent miscarriages3,4. While mild to moderate endometrial injuries can be addressed through conventional clinical treatments like hysteroscopic intrauterine adhesion (IUA) separation and high-dose estrogen therapy 5 , effective strategies for severe endometrial damage remain scarce. Consequently, repairing damaged endometrium and enhancing fertility in women with TE continue to be significant challenges in reproductive medicine.
The use of stem cells for endometrial repair has attracted considerable attention in recent years due to their inherent self-renewal capacity, which effectively supports the regeneration and proliferation of endometrial cells6,7. A notable source of these stem cells is menstrual blood–derived mesenchymal stem cells (MenSCs), naturally released during a woman’s menstrual cycle when the endometrium is shed 3 . MenSCs have shown substantial promise in various studies, especially regarding their robust proliferative ability. These cells can undergo multiple rounds of continuous culture without exhibiting chromosomal abnormalities, highlighting their stability and applicability in regenerative medicine 8 . When transplanted, MenSCs have yielded encouraging outcomes in enhancing endometrial hyperplasia, angiogenesis, and morphological repair. These outcomes are coupled with reductions in uterine collagen fibrosis and inflammation8 –10. In addition, MenSCs can promote functional recovery by improving the inflammatory microenvironment, as demonstrated in a mouse spinal cord injury model 11 . However, despite these significant discoveries concerning MenSCs’ therapeutic potential in endometrial injury repair, significant challenges remain in the application of these stem cells for disease therapy 12 , and a significant knowledge gap persists regarding their molecular mechanisms and the signaling pathways involved in these processes9,13.
The proliferation of stem cells and the pathophysiology of the endometrium are governed by a complex array of signaling pathways and cytokines 14 . Within this array, the Wnt signaling pathway is pivotal in various processes, including stem cell proliferation, tissue regeneration, and embryonic implantation development15,16. It regulates stem cell proliferation in the endometrium, which is crucial for successful implantation and pregnancy. Activation occurs through the binding of Wnt ligands to the Frizzled receptor and LRP5/6, preventing the degradation of β-catenin. This interaction initiates the activation of target genes essential for cell proliferation and differentiation, crucial for successful implantation 17 .
However, dysfunctions in this pathway can negatively affect endometrial receptivity 18 . Recent studies have highlighted the role of lncRNAs in critical regulatory functions, encompassing epigenetic, transcriptional, and post-transcriptional regulation 19 . Among these, MALAT1, a highly conserved lncRNA located at chromosome 11 q13.1, is one of the first lncRNAs linked to both pathological and physiological processes in humans 20 . MALAT1 functions as an endogenous miRNA decoy, modulating specific miRNA expression and influencing mRNA targeting. It plays a crucial role in various molecular biological processes, including proliferation and metastasis21,22. Numerous studies have highlighted MALAT1’s direct or indirect engagement in related signaling pathways via miRNA regulation23,24. Yet, its exploration in MenSCs remains limited.
In this research, we delineated the disparity in lncRNA MALAT1 expression between normal and thin endometrial tissues, marking the first identification of this relationship, which unveils a novel mechanism. This discovery is highly relevant for elucidating the mechanism by which MenSCs facilitate endometrial fertility restoration by the Wnt signaling pathway. Further analysis showed that MALAT1 inhibition curtailed cellular proliferation, as evidenced by MALAT1 knockdown, the CCK8 proliferation assay, and the cell scratch assay. Subsequent dual-luciferase assays determined its target genes, confirming its influence on endometrial repair and fertility enhancement in TE animal models. These findings suggest that targeting the MALAT1/miR-7-5p/TCF4 axis represents a promising therapeutic strategy for TE treatment, promoting endometrial tissue repair, regeneration, and fertility regulation.
Materials and Methods
Samples Collection
Human samples for this study were obtained from the Reproductive and Genetic Branch of the First Affiliated Hospital of the University of Science and Technology of China. Participants, aged between 25 and 35 years, had a history of healthy reproduction, no adverse pregnancy outcomes, and regular menstrual cycles. All tissue donation procedures adhered to the ethical guidelines approved by the University of Science and Technology of China, and each donor provided informed consent.
Normal endometrial tissue was harvested from patients undergoing hysterectomy for non-endometrial conditions, including benign gynecological diseases or early cervical cancer, and tubal dredging via hysteroscopy during the proliferative phase. Exclusion criteria included patients with endometriosis, adenomyosis, endometrial cancer, polycystic ovary syndrome (PCOS), and abnormal uterine bleeding (
Menstrual blood was collected from volunteer participants meeting the inclusion criteria of a history of healthy childbirth and moderate to heavy menstrual flow. Exclusion criteria were the use of intrauterine devices, the presence of endometrial lesions, endometriosis, adenomyosis, PCOS, and abnormal uterine bleeding (
Isolation and Culture of MenSCs
On the second day of menstruation, Diva Cups were used for collecting human menstrual blood. Intermediate tunica albuginea cells were isolated utilizing a lymphocyte isolation solution (Solarbio, 1.077 g/ml) and centrifuged at 1200 ×
MenSCs Identification
The third generation of MenSCs was cultured, with approximately 5 × 105 MenSCs collected and resuspended in a fluorescence-activated cell sorting buffer. The multipotent differentiation potential of MenSCs into various mesenchymal cell lineages was assessed using flow cytometry analysis with the Human MSC Analysis Kit (BD Biosciences). In addition, a human MSC functional identification kit (R&D Systems) was utilized in accordance with the manufacturer’s protocol to evaluate differentiation outcomes through immunofluorescence staining using osteocalcin as an osteocyte marker, FABP-4 as an adipocyte marker, and Aggrecan as a chondrocyte marker.
Histological Staining (H&E) and Masson Staining
After 14 days post-modeling, rats were euthanized, and the treated endometrium was fixed in 4% paraformaldehyde for 24 h and then dehydrated and embedded in paraffin. Paraffin-embedded samples were sectioned into 10 μm slices for staining. H&E and Masson’s trichrome staining were performed according to standard protocols. The number of glands, endometrial thickness, and fibrosis area ratio were calculated individually, followed by the calculation of mean values. Data analysis was conducted using ImageJ software.
Cell Transfection
Small interfering RNA (siRNA) oligonucleotides targeting MALAT1 (si-MALAT1) and a negative control siRNA (NC) were synthesized by Sangon Biotech. Chemically modified miR-7-5p mimics and inhibitors (AZENTA, China) were used to modulate miR-7-5p expression levels. Cells were transiently transfected using Lipofectamine 2000 (Invitrogen, Carlsbad, USA) following the manufacturer’s instructions. The sequences of the RNA of interest are presented in Supplemental Table S1.
Cell Proliferation
Each group of cells (4,000 cells/100 μl) was seeded into a 96-well plate. After treating the experimental group with si-MALAT1 for 0, 1, 2, 3, and 4 days, cell proliferation was measured in both groups using the CCK8 kit (biosharp, BS350B500T). Optical density (OD) was assessed at 450 nm using a multifunctional microplate reader, with six replicates for each cell group, and the experiment was replicated three times.
Wound-Healing Assay
Transfected cells in the logarithmic growth phase (2 × 105 cells/well) were seeded into six-well plates and grown to 50% confluence. In the experimental group, MALAT1 expression was specifically silenced. Upon reaching 90% confluence, a uniform scratch was made at the center of each well’s bottom using a 200 μl sterile pipettor tip. After performing three PBS washes to remove debris and detached cells, a fresh complete medium was added, and the cells were incubated for an additional 24 h under optimal conditions. Wound areas were monitored and measured at 0 and 24 h using inverted microscopy and Image J software, respectively. The cellular healing rate was calculated using the formula: (initial scratch width − final scratch width) / (initial scratch width) ×100%. This experiment was conducted three times independently, with average values reported. Cell migration was then monitored and quantified within the scratch region using ImageJ software.
Western Blot
Samples were incubated in Radio Immunoprecipitation Assay (RIPA) lysis buffer (Biosharp, BL504A) containing a protease inhibitor cocktail (Sangon Biotech, C600387) for 30 min on ice. The precipitated fraction was obtained through centrifugation at 12,000 ×
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
Total RNA was extracted using the RNA-Quick Purification Kit (ESscience, RN001) following the manufacturer’s instructions and quantified via quantitative polymerase chain reaction (PCR) analysis (qPCR) for further analysis. RNA concentration and purity were assessed with the NanoDrop One (Thermo Scientific, USA). Reverse transcription was performed using 500 ng of total RNA with the RT reagent kit (Vazyme, R-323-01).
For miRNA, the miRNA extraction kit (absin, abs60262) was used. miRNA reverse transcription employed the stem-loop-based miRNA 1st Strand cDNA Synthesis Kit (Vazyme, MR101-01), with normalization to U6 small nuclear RNA. cDNA samples were preserved at −20°C. qPCR analysis employed Cham Q Universal SYBR qPCR Master Mix (Vazyme, Q511-02) and miRNA Universal SYBR qPCR Master Mix (Vazyme, MQ101-01). mRNA and miRNA expression levels were analyzed using the 2−ΔΔCT method, normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and U6, respectively. Primer sequences for qPCR are listed in Supplemental Table S2.
Fluorescence In Situ Hybridization
To ascertain the subcellular localization of MALAT1 in MenSCs, fluorescence
Luciferase Reporter Assays
Sequences of MALAT1 or TCF4, containing either wild-type or mutant-type binding sites, were cloned into pmirGLO dual-luciferase vectors to create MALAT1/TCF4-wild-type (MALAT1-WT/TCF4-WT) and mutated-type (MALAT1-MT/TCF4-MT) vectors. These vectors were co-transfected into 293T cells with miR-7-5p mimics or negative control using Lipofectamine 2000 following the manufacturer’s guidelines. Luciferase activity was measured at 48 h post-transfection using a Multiscan Spectrum (SpectraMax iD3), with relative luciferase activity calculated as the ratio of firefly to Renilla luciferase activity. All vectors were sourced from Genecreate (Wuhan, China).
Bioinformatics Analysis
Target genes were predicted utilizing online DIANA Tools (https://diana.e-ce.uth.gr/lncbasev3/interactions) and TargetScan software (http://www.targetscan.org/vert_72/).
Rat Model of TE and Cell Implantation
Female Sprague-Dawley (SD) rats, aged 8–10 weeks old and weighing 160–200 g, were obtained from the Experimental Animal Center of Anhui Province, China. They were housed in a specific pathogen-free environment with a 12-h light/dark cycle and maintained at 22°C to 25°C, with
MenSCs were administered into the uterine lesion through the incision using a 20-μm pipette and secured at both lesion ends with arterial clamps to localize the cells to the injury site and prevent their dispersion. After several minutes, the clamps were released, and the incision was sutured with 7-0 absorbable thread. The rats were randomly allocated into five groups, each comprising three rats: a control group (no treatment), a sham group (saline injection), a TE model group (endometrial injury only), a TE-MenSCs group (TE model + MenSCs implantation), and TE-si-MALAT1-MenSCs group (TE model + si-MALAT1-MenSCs implantation). To control for inter-rat variability, both uterine horns of each rat served as self-controls: the right as the positive control and the left as treatment. In the TE-MenSCs group and TE-si-MALAT1-MenSCs groups, 1 × 106 MenSCs were injected into the injured site.
Fertility Assessment
Female rats of proven reproductive viability, including non-pregnant individuals, underwent fertility evaluation. Following the aforementioned procedures, rats were divided into groups, with three rats per group. Fourteen days post-modeling, mating trials were conducted, pairing female rats with males at a 3:1 ratio. Fourteen days post-mating, the females were humanely euthanized to inspect their uterine horns. Pregnancy success was determined by the presence of embryos in one or both uterine horns, and such cases were included in the fertility analysis. The gestational sac ratio was calculated by comparing the number of sacs in the left uterus to those in the right.
Endometrial Thickness Measurement
Endometrial thickness was measured using Caseviewer software on rat uterine sections, defined as the distance from the endometrium-myometrium junction to the uterine cavity. Four sections per layer were analyzed, and the average measurement was recorded as the layer’s endometrial thickness.
Glandular Counts
For each uterine section, four fields were photographed and analyzed at 20× magnification to count the uterine glands. The average number of glands per field was calculated to determine the glandular density.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 9.0, with results presented as the mean ± SEM (standard error of the mean). Differences between groups were assessed using two-way analysis of variance (ANOVA) and Student’s
Results
Isolation and Identification of MenSCs
MenSCs were isolated from female donors (
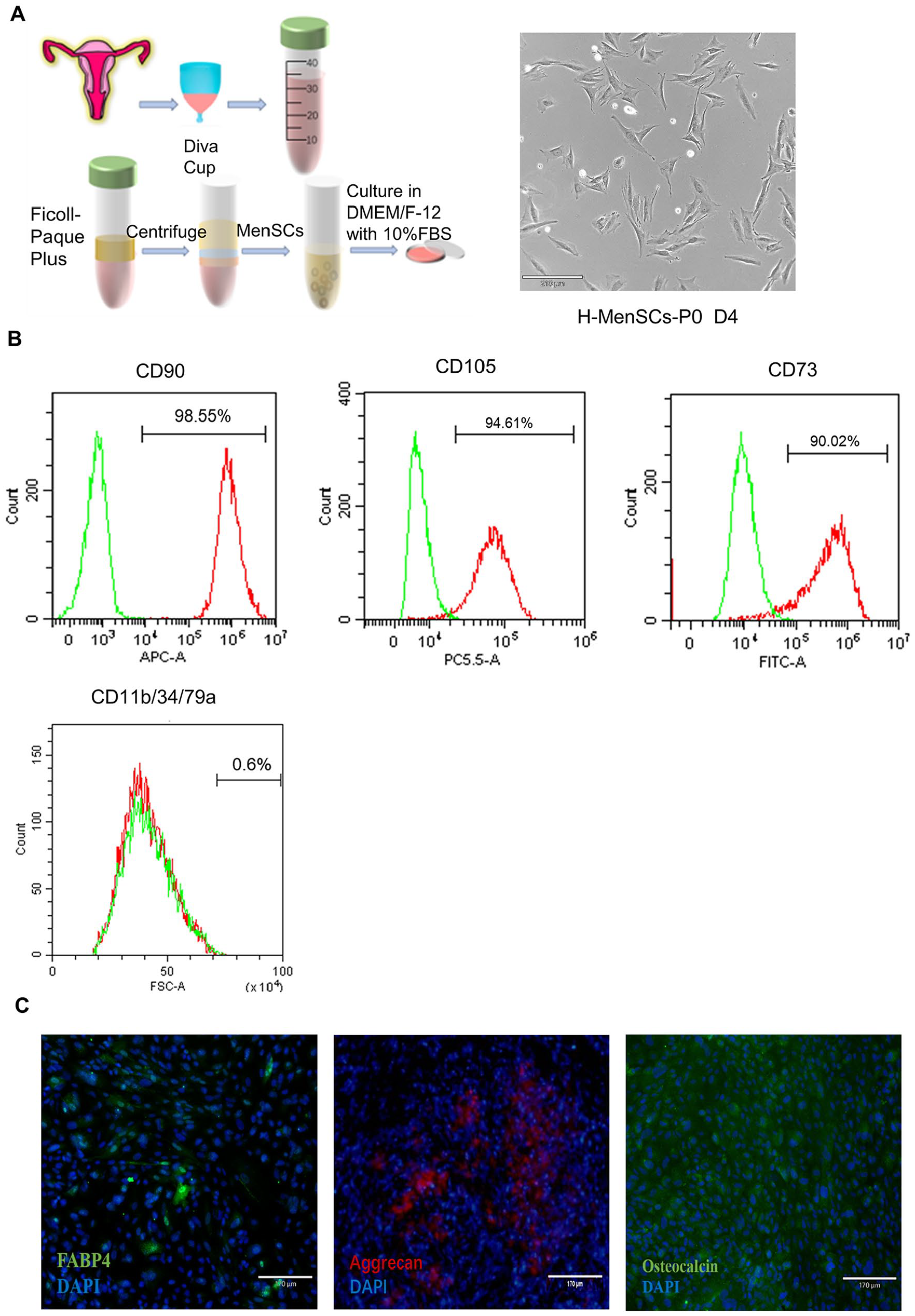
Isolation and identification of MenSCs. (A) Extraction process and brightfield morphology of primary MenSCs. Scale bar: 210 μm. (B) Flow cytometry analyses of MenSCs at passage 3 revealed the percentages of cells expressing CD73 (90.02%), CD90 (98.55%), CD105 (94.61%), and being negative for CD11b/CD34/CD79a (<1%). (C) Immunofluorescence staining demonstrated the differentiation potential of MenSCs into adipogenesis (FABP-4), osteogenesis (Osteocalcin), and chondrogenic (Aggrecan) lineages. Scale bar: 170 μm.
MALAT1 Promoted the Proliferation and Migration of MenSCs In Vitro
To elucidate the function of MALAT1 in endometrial tissues, RNA was extracted from tissue samples (
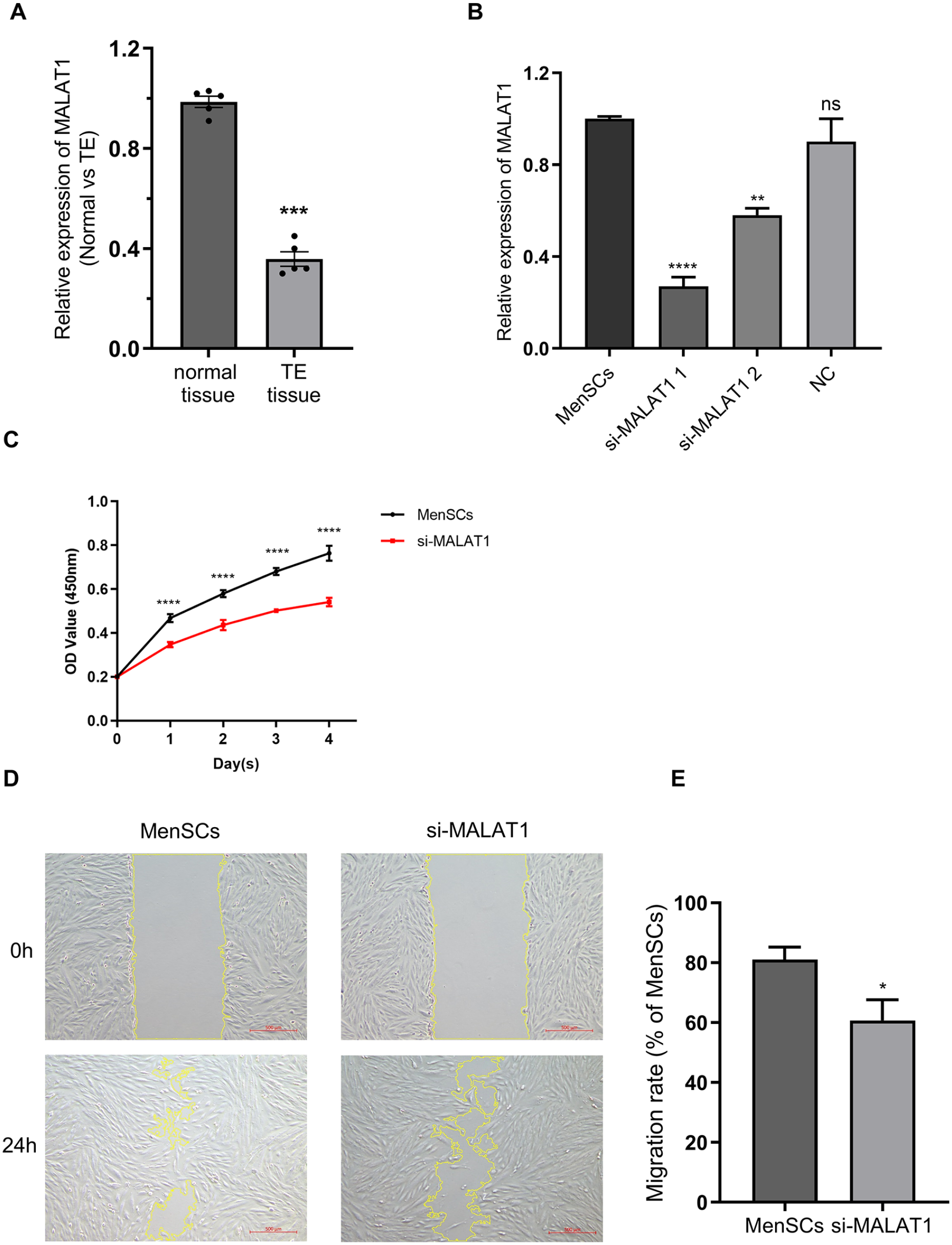
Investigation of the MALAT1 and its knockdown. (A) MALAT1 expression in normal and TE (
Further investigation into MALAT1’s role in MenSCs employed two specific siRNAs. Post-transfection, a marked decrease in MALAT1 expression was observed in comparison to the NC group, with si-MALAT1-1 showing more substantial inhibition than si-MALAT1-2 (Fig. 2B). Given its enhanced efficiency, si-MALAT1-1 was chosen for further experimentation. The knockdown’s efficacy was consistently monitored over a minimum of 5 days, confirming the siRNA’s potent and stable silencing of MALAT1 (Supplemental Fig. S1A). This finding underscores the potential utility of si-MALAT1 in subsequent MenSCs-related studies.
CCK8 assays demonstrated that MALAT1 knockdown significantly reduced MenSCs proliferation (Fig. 2C). Furthermore, the Wound-Healing assay showed that si-MALAT1 considerably decreased the migratory ability of MenSCs compared with the control group (Fig. 2D, E), suggesting that MALAT1 facilitates MenSCs proliferation and migration
Silencing MALAT1 Inhibited the Wnt Signaling Pathway In Vitro
The Wnt signaling pathway is crucial for stem cell self-replication, self-renewal, and the maintenance of tissue homeostasis15,16. To explore the link between MALAT1 and the Wnt signaling pathway, qPCR (Fig. 3A) and Western blot (WB; Fig. 3B) analyses were performed. Results showed a significant reduction in
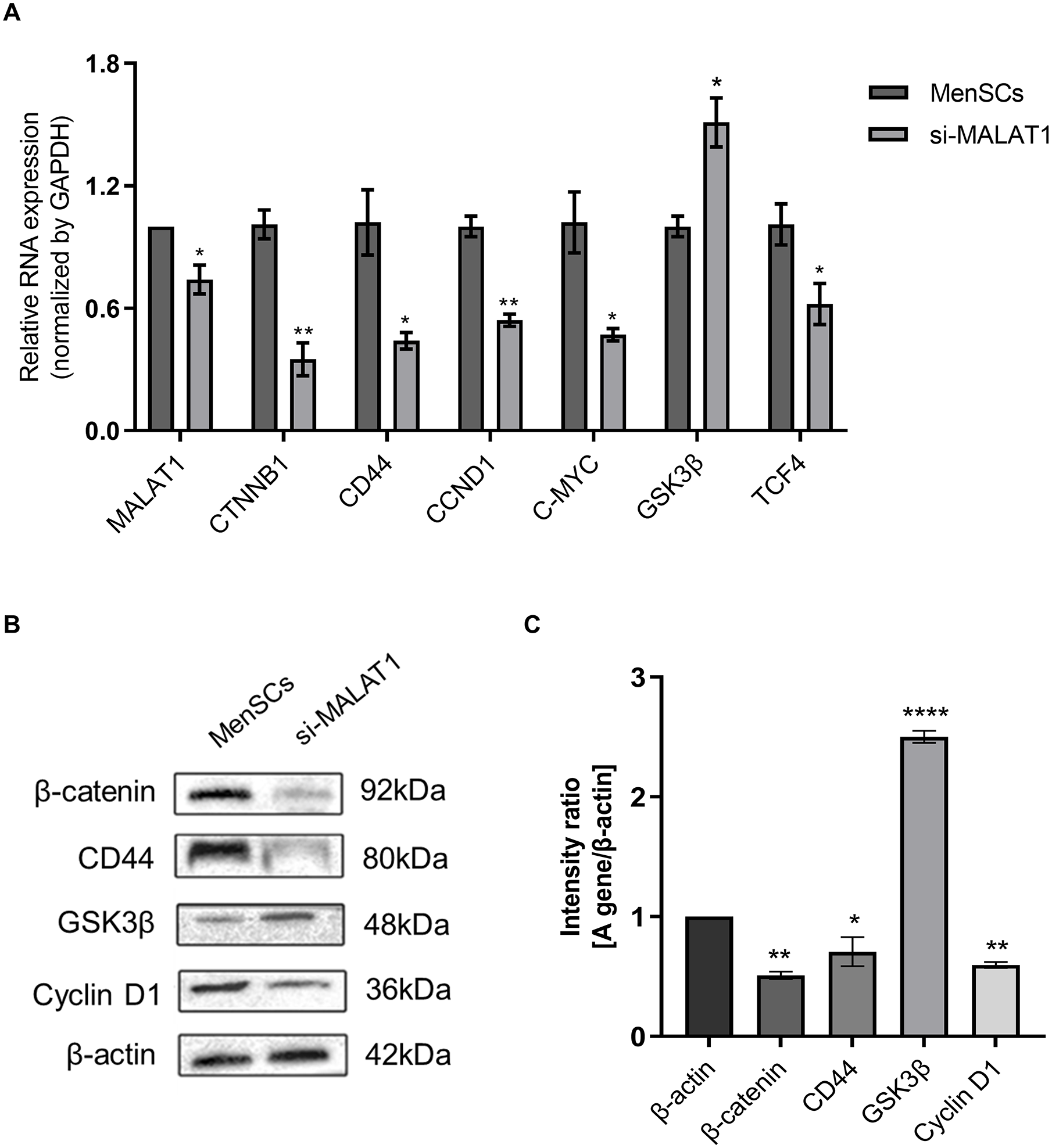
Relationship between MALAT1 and the Wnt signaling pathway. (A) Changes in RNA expression levels of
MALAT1 Acts as a Competing Endogenous RNA Against miR-7-5p
To advance our understanding of MALAT1’s mechanisms, its subcellular localization in MenSCs was determined. FISH assay findings indicated a primarily nuclear localization of MALAT1 (Fig. 4A). LncRNAs, such as MALAT1, can function as competing endogenous RNAs (ceRNAs), sequestering miRNAs 25 . Through bioinformatics analysis for target prediction, miR-7-5p emerged as a likely target of MALAT1 (Fig. 4B), suggesting MALAT1 serves as a ceRNA for miR-7-5p. Subsequent qPCR analysis showed that MALAT1 knockdown significantly increased miR-7-5p levels (Supplemental Fig. S1B), whereas miR-7-5p overexpression led to a marked decrease in MALAT1 expression (Supplemental Fig. S1C), supporting a reciprocal regulatory relationship between MALAT1 and miR-7-5p.
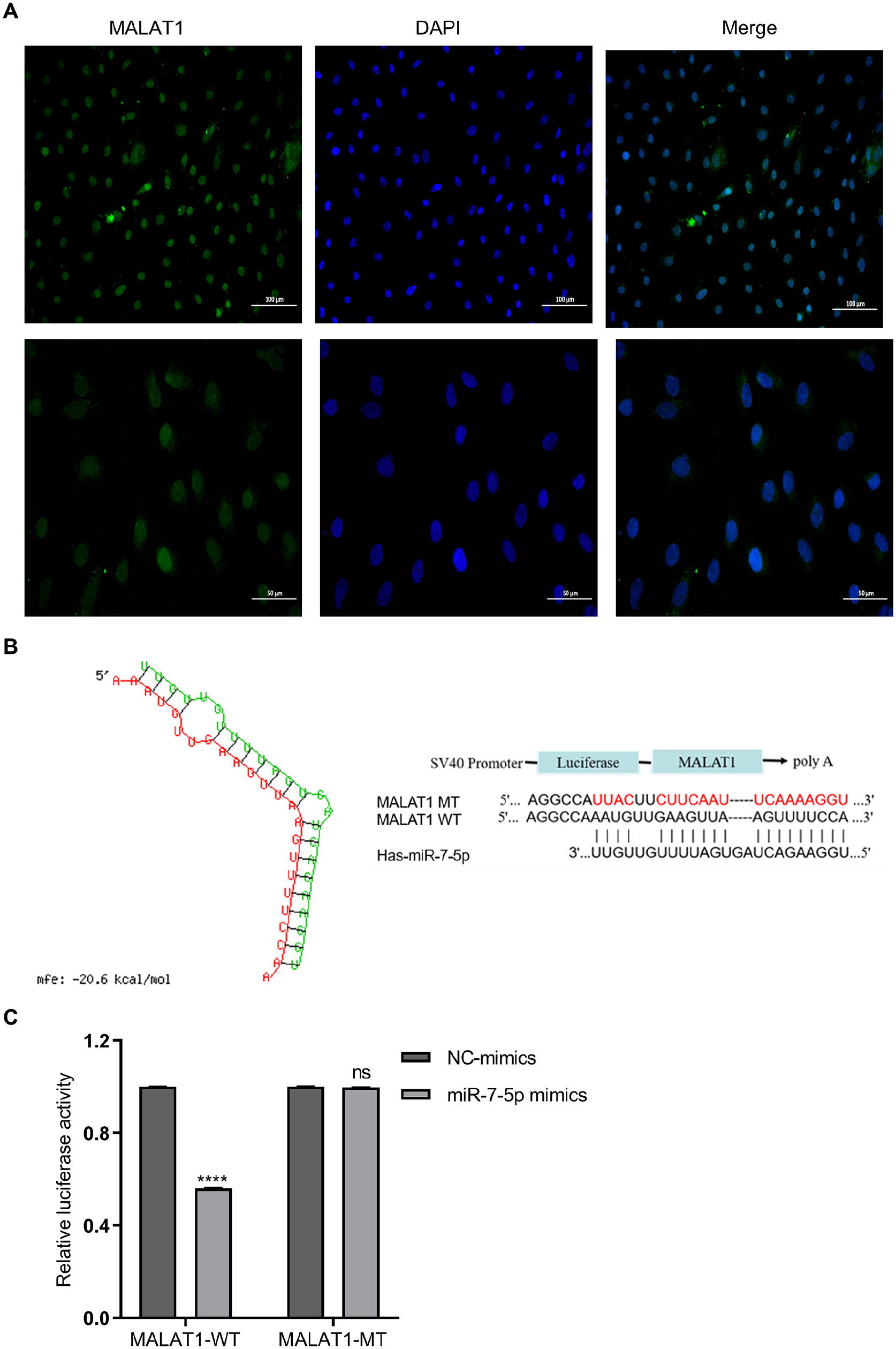
Localization of MALAT1 and its function as a sponge of miR-7-5p. (A) Localization of MALAT1 in MenSCs confirmed via FISH assay, with MALAT1 marked in green and the nuclear marker (DAPI) in blue. Scale bar: 100 μm (top) or 50 μm (bottom). (B) Comparison of wild-type (WT) and mutant-type (MT) complementary binding sequences between MALAT1 and miR-7-5p. (C) Variations in relative fluorescein activity between MALAT1-WT and MALAT1-MT upon altering miR-7-5p levels. Data are presented as mean ± SEM, ****
To verify the direct interaction between miR-7-5p and its target seed sequences in the 3′-UTR of MALAT1, we engineered MALAT1-WT and MALAT1-MT constructs containing the original and mutated miR-7-5p binding sites within MALAT1’s 3′-UTR, respectively. Supplemental Fig. S1D shows that miR-7-5p levels were significantly reduced in the miR-7-5p inhibitor group compared with the MenSCs and miR-NC groups, while an increase was observed in the miR-7-5p mimics group. The luciferase reporter assay results (Fig. 4C) demonstrated a notable reduction in luciferase activity in cells co-transfected with MALAT1-WT and miR-7-5p mimics, relative to the NC group (
MiR-7-5p Directly Regulates the Stability of TCF4 in MenSCs
miRNAs modulate gene expression by targeting specific mRNA sites to influence their translation. To elucidate how miR-7-5p affects MenSCs, particularly its role in the Wnt signaling pathway, we conducted a bioinformatics analysis using TargetScan Release 7.2 to identify miR-7-5p targets. This analysis highlighted a conserved binding site in the 3′-UTR of transcription factor 4 (TCF4) (Fig. 5A), a key transcription factor in the Wnt pathway, working in tandem with β-catenin to activate target gene transcription 26 .
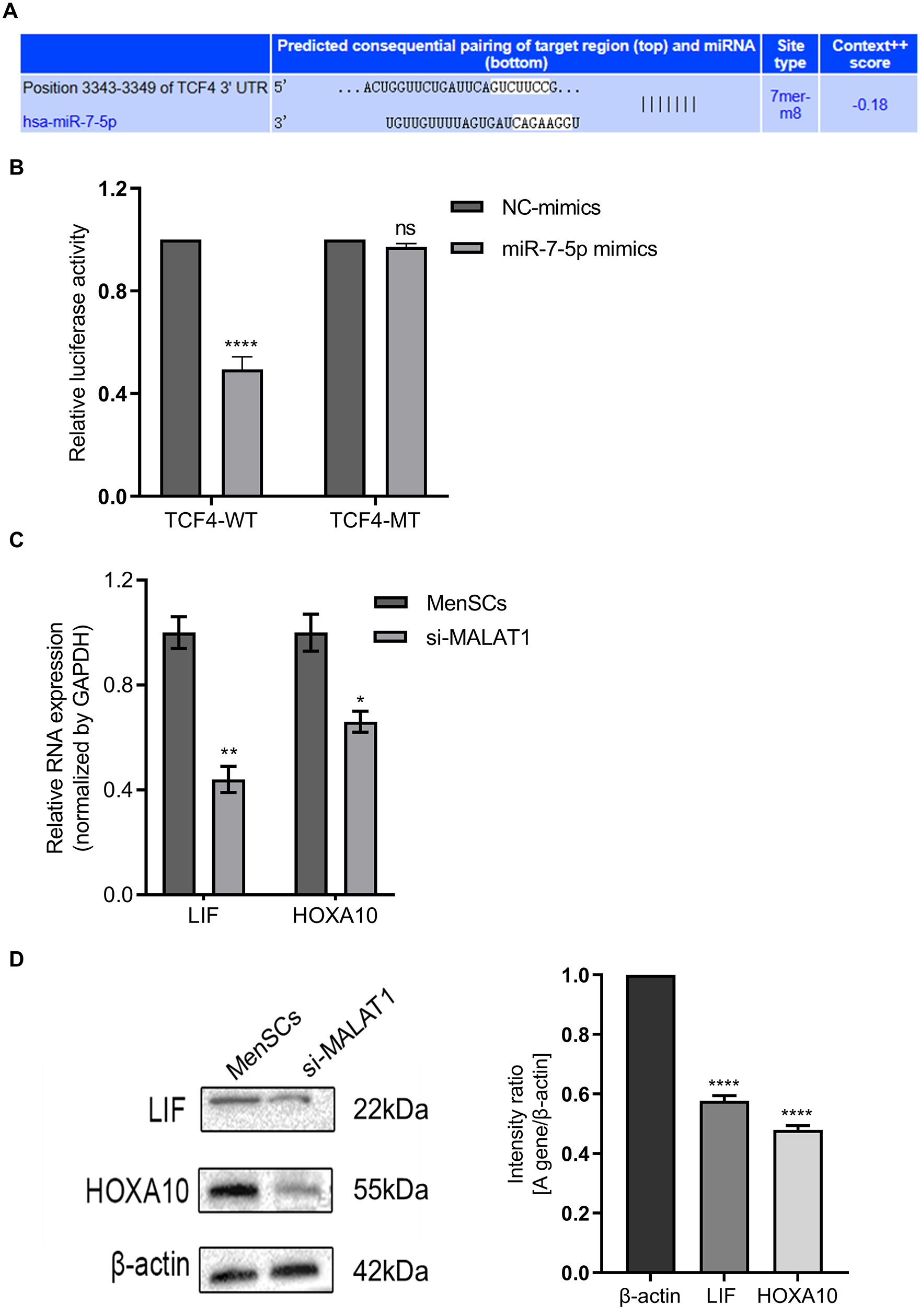
miR-7-5p directly regulates TCF4 stability and influences endometrial regeneration. (A) Analysis of the binding sites between miR-7-5p and TCF4. (B) Variations in relative fluorescein activity between TCF4-WT and TCF4-MT in response to miR-7-5p levels. (C and D) qPCR and Western blot (WB) analyses were used to assess the expression changes in endometrial receptivity-related genes LIF and HOXA10 following MALAT1 knockdown in MenSCs, along with relative quantitative assessments. Data are presented as mean ± SEM, ****
The TCF4-WT luciferase reporter, harboring miR-7-5p’s putative binding site, was constructed. Luciferase activity in this wild-type reporter significantly decreased following miR-7-5p overexpression. However, this inhibitory effect was absent in TCF4-MT, where miR-7-5p’s binding site was eliminated, within MenSCs lines (Fig. 5B). These experiments confirm TCF4 as a direct target of miR-7-5p in MenSCs.
Downregulation of MALAT1 Impairs Endometrial Regeneration
Endometrial regeneration is essential for fertility restoration and successful embryo implantation, depending critically on the precise modulation of the Wnt signaling pathway. Key genes such as
Knockdown of MALAT1 Inhibits Endometrial Regeneration and Gland Formation
Assessing endometrial regeneration is critical, with a focus on the endometrium’s thickness and structural integrity—key determinants of a successful pregnancy 28 . Research indicates that an endometrial thickness of less than 6 mm is associated with a significantly reduced embryo implantation rate of 29.43% in women undergoing assisted reproductive technologies 29 .
To investigate the impact of MALAT1 on endometrial functionality, we analyzed the endometrial morphology and thickness across different rat groups (Supplemental Fig. S2A). Measurements of endometrial thickness were taken from three sections per rat using paraffin-embedded tissues, averaging four perpendicular measurements within the same plane for each section. The TE model group exhibited a notably reduced endometrial thickness compared with the sham group (Fig. 6B), with an increased Masson positive ratio relative to the NC group (Supplemental Fig. S2B). Although there was no significant difference between the TE-MenSCs and sham groups, the TE-MenSCs group showed a markedly greater thickness than the TE model group. Conversely, the TE-si-MALAT1-MenSCs group exhibited a substantial reduction in endometrial thickness relative to the TE-MenSCs group, along with a significant enlargement of the uterine cavity area (Fig. 6B and Supplemental Fig. S2A).
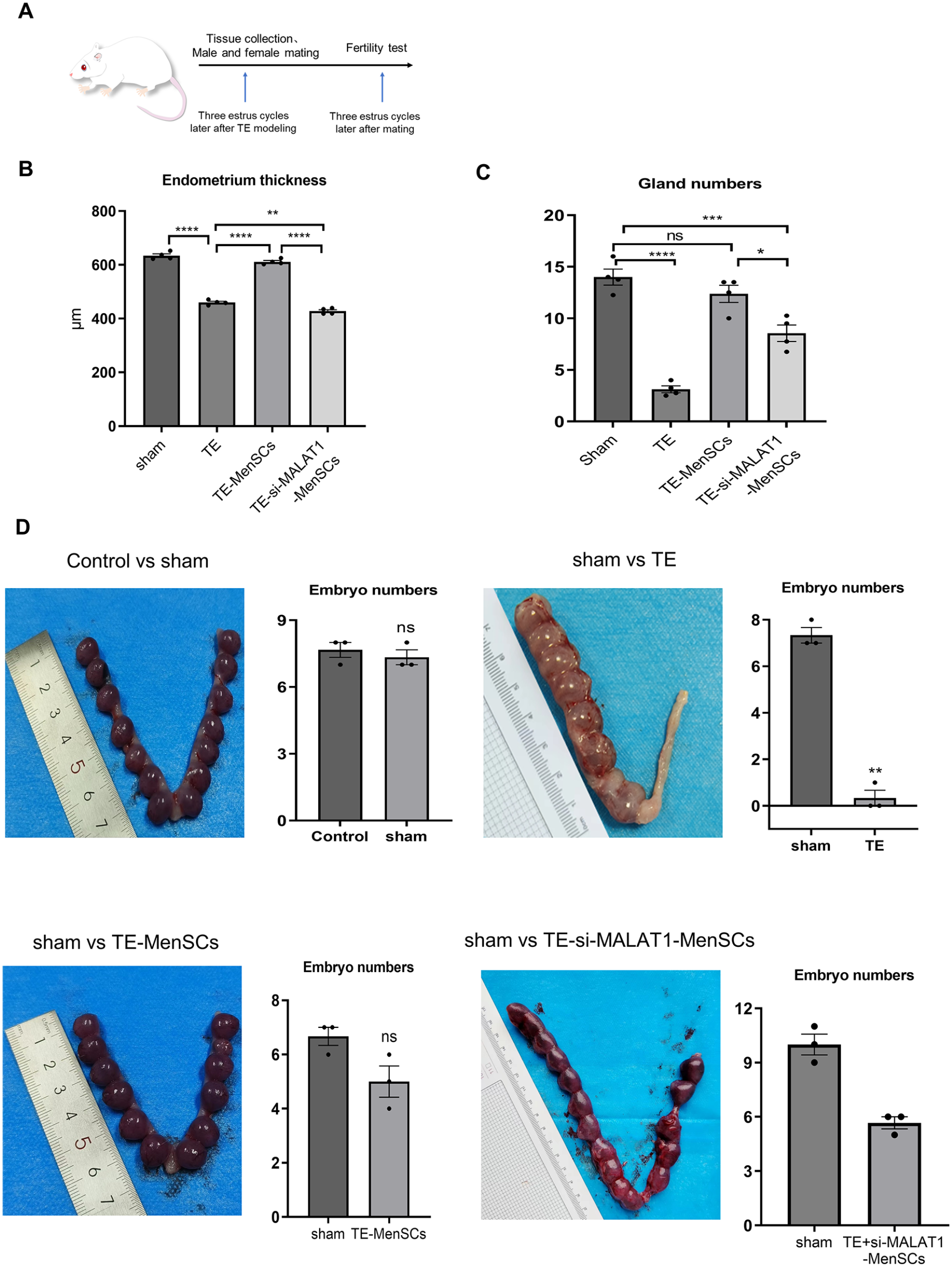
The effects of MALAT1 on endometrium and fertility were verified
Furthermore, the number of endometrial glands is also crucial for endometrial functions and can indicate outcomes of endometrial regeneration. As depicted in Fig. 6C, although the number of glands in the TE-si-MALAT1-MenSCs group was higher than that in the TE group, it was significantly lower than that in the TE-MenSCs group, suggesting that MALAT1 deletion may affect the number of endometrial glands.
Therapeutic Effects of MALAT1 on Fertility in TE Rats
The goal of endometrial restoration is to establish an environment conducive to embryo implantation, growth, and development 30 . Assessment of the estrous state in rats, via analysis of vaginal secretions, precedes the animal experiments. An increased presence of anucleated keratinized epithelial cells in vaginal secretions correlates with a thicker uterine lining, aiding in the establishment of the TE model 31 . To evaluate the therapeutic potential of MALAT1 in endometrial repair, female rats were sacrificed 14 days post-mating to examine their uteri. Figure 6D presents representative uterine images from each group, with pregnancy outcomes detailed in Table 1.
Reproductive Outcomes in Experimental Rats.
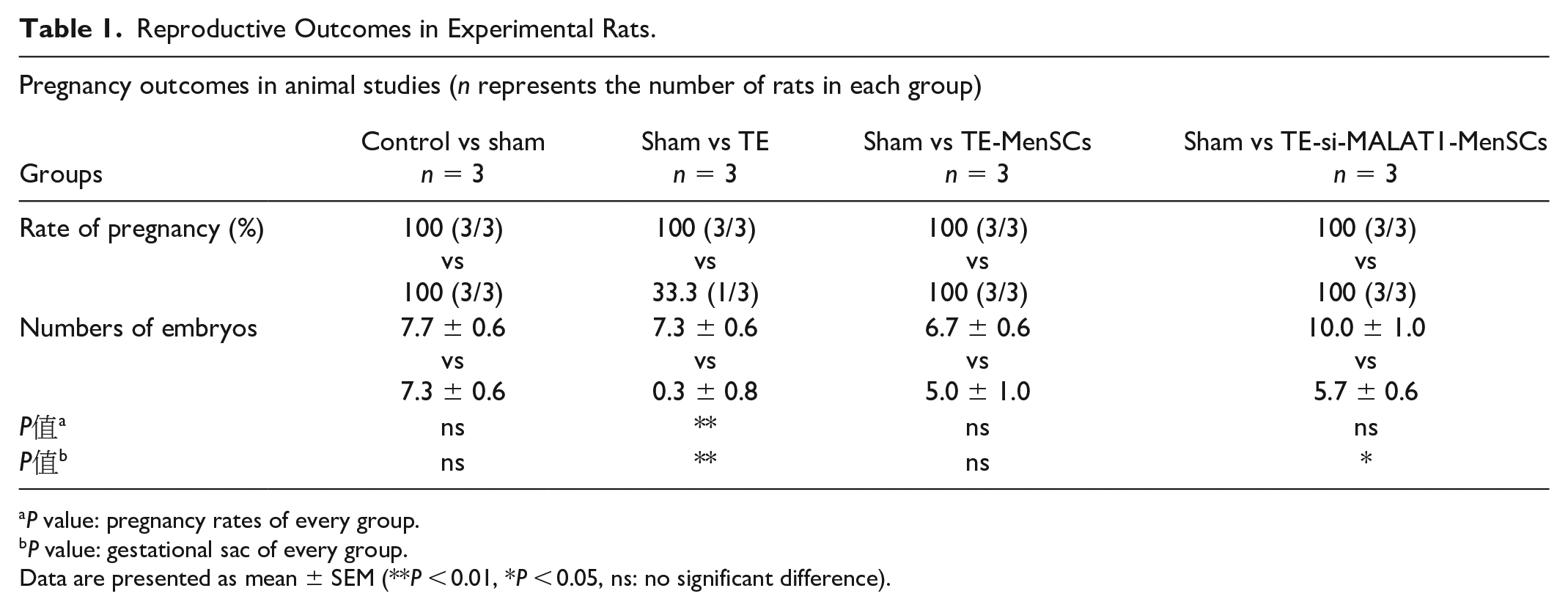
Data are presented as mean ± SEM (**
To minimize variability related to age and developmental stages among the rats, a self-control approach was employed in our experiments, using the right uterus as the control and the left uterus for experimental interventions. The pregnancy rate in the TE group was significantly reduced to 33.3% (
Discussion
In this study, we present initial evidence indicating that lncMALAT1 modulates miR-7-5p to influence the transcription factor TCF4 within the Wnt signaling pathway, aiding MenSCs-driven TE repair and fertility restoration.
The endometrium, a vital tissue lining the uterus, is essential for pregnancy and undergoes significant changes during the menstrual cycle to facilitate embryo implantation and growth 32 . Endometrial dysfunction can disrupt this critical balance, impairing conception and the ability to sustain pregnancy 3 . MenSCs, derived from menstrual blood, provide a non-invasive source of MenSCs with unique advantages for treating autologous endometrial injuries33,34. These cells have been successfully used in both animal models and human studies to repair endometrial damage, demonstrating benefits such as improved endometrial thickness and vascularization 35 .
Considering the properties of MenSCs and their potential in endometrial tissue remodeling, in alignment with the physiological patterns of the menstrual cycle, their application in endometrial regeneration has been explored. Researchers have assessed the biosafety of MenSCs in TE animal models, focusing on toxicity and tumorigenicity, and concluded that MenSCs transplantation in rat TE models is safe and viable 36 . Moreover, a meta-analysis has confirmed the safety and efficacy of MenSCs transplantation in treating TE, extending menstrual duration, and increasing endometrial thickness 37 . Clinical trials have shown that autologous MenSCs transplantation into patients with severe IUA enhances endometrial thickness and pregnancy rates, positioning this approach as a promising strategy for endometrial regeneration 38 . MenSCs transplantation promotes endometrial hyperplasia, angiogenesis, and morphological restoration, which in turn improves fertility and pregnancy outcomes. In addition, MenSCs help reduce endometrial collagen fibrosis and inflammation9,10. However, the precise mechanisms through which MenSCs facilitate endometrial repair remain to be fully elucidated. In our study, we collected menstrual blood samples from healthy women and isolated MenSCs using a lymphatic separation technique. These cells underwent flow cytometry to analyze surface marker expression, confirming their identity as MSCs. Furthermore, we conducted multilineage differentiation assays to verify the multilineage differentiation capacity of these stem cells.
Recently, lncRNAs have emerged as significant players in biology, known for their regulatory roles in diverse biological processes 39 . MALAT1, a lncRNA prevalent across human tissues, has been extensively studied for its function. Research has revealed that MALAT1 significantly affects MSCs’ self-renewal and differentiation by modulating internal signaling pathways, gene expression, and protein synthesis 40 . Cooper et al. 41 discovered that exosomes from adipose-derived MSCs, containing MALAT1, enhance the migration of human skin fibroblasts and ischemic wound healing. In addition, MALAT1 activates the Wnt signaling pathway and interacts with the miR-124 and miR-378a/FGF2 axis to accelerate wound repair21,42. Furthermore, exosomes carrying the MALAT1-201 are found to promote trophoblast cell proliferation and migration while inhibiting apoptosis 43 .
In this experiment, utilizing CCK8 and cell scratch assays, we discovered that MALAT1 plays a crucial role in regulating MenSCs proliferation. Knockdown of MALAT1 led to a reduction in cell proliferation and inhibited cell migration and expansion. However, it is important to recognize that the findings of this study are specific to MenSCs and may not extend to other stem cell types or human organ systems. Future research should explore various stem cell types to elucidate MALAT1’s comprehensive role in biological processes. The nucleus, a pivotal cellular organelle, is responsible for DNA replication, transcription, and critical RNA processing activities. The nuclear enrichment of MALAT1 indicates its potential involvement in gene regulation and RNA metabolism. Although MALAT1’s functions in various physiological and pathological contexts have been documented, its specific roles and mechanisms in MenSCs are yet to be fully understood. This study suggests that MALAT1 might regulate gene expression or RNA metabolism in MenSCs, influencing their characteristics and capabilities. We initiated an in-depth investigation into the potential interaction between MALAT1 and miR-7-5p using extensive database searches. Through comprehensive analysis, we identified their binding sites and proposed regulatory mechanisms. Subsequent dual-luciferase reporter assays confirmed the interaction between MALAT1 and miR-7-5p, providing quantitative insights into their binding dynamics and functional implications.
The Wnt signaling pathway was originally identified for its crucial role in embryonic development, governing cell fate determination, tissue patterning, and organogenesis 44 . In adult organisms, this pathway remains active, influencing a broad array of physiological processes, and is also involved in tissue repair and regeneration post-injury or damage. However, the concurrent regulation of MenSCs by both lncRNA and the Wnt signaling pathway constitutes a rare phenomenon. MenSCs, mesenchymal stem cells originating from the female endometrium, hold significant promise in regenerative medicine. Thus, delving deeper into how lncRNAs modulate MenSCs’ functionality through the Wnt signaling pathway will yield critical insights into the underlying molecular mechanisms. In this study, we have identified MALAT1 in the endometrium using a qPCR assay.
As a lncRNA, MALAT1 predominantly operates in the nucleus, influencing a variety of biological processes. Through a dual luciferase assay, we further established that TCF4 is a downstream target gene of this axis. TCF4 is not merely a target of MALAT1 but also a crucial downstream component of the Wnt signaling pathway, playing a pivotal role in cell proliferation, differentiation, and stem cell self-renewal. In this research, we delineated the MALAT1/miR-7-5p/TCF4 signaling axis, elucidating lncRNA’s regulatory influence on the Wnt signaling pathway. This discovery underscores the significance of lncRNAs within a complex molecular interaction network and offers invaluable insights into treating endometrium-associated infertility.
To reinforce our findings, we utilized an animal model of TE. In TE rats, uterine damage led to reduced basal cell numbers and diminished regenerative capacity. Our experiments demonstrated that transplantation of MenSCs ameliorated these conditions, notably enhancing the damaged endometrium’s area and thickness, fostering gland development, reconstituting its structure, and enhancing the intrauterine environment. Given the inherent reduction of stem cells in TE, these effects are likely attributable to the paracrine actions of the transplanted MenSCs. In contrast, TE-si-MALAT1-MenSCs exhibited significantly lower endometrial thickness and gland numbers than those treated with MenSCs alone. Furthermore, the TE-si-MALAT1-MenSCs group showed a notable reduction in gestational sac numbers compared with both the sham group and TE-MenSCs group.
The results of this study are encouraging, yet to ascertain its safety and efficacy, further large-scale clinical trials and comprehensive analyses of its mechanisms are imperative. Concurrently, it is essential to intensify research on additional factors influencing the MenSCs’ mechanism in improving TE fertility, to gain a more holistic understanding of the process and devise more effective treatment strategies. Moreover, these findings underscore the importance of considering uterine microstructural changes in evaluating and forecasting patients’ postoperative recovery. Relying solely on a single parameter, such as thickness, does not adequately capture the myriad of complex factors at play in the restoration process. The findings demonstrate that lncMALAT1 expression affects TE rat endometrial regeneration and fertility. By modulating the MALAT1/miR-7-5p/TCF4 axis, one can effectively treat TE, enhance endometrial tissue repair and regeneration, and regulate fertility. This study lays a foundation for a deeper understanding of endometrial regeneration mechanisms and disease treatment, potentially guiding future endometrial improvement strategies and fertility enhancement by regulating lncMALAT1 expression, indicating its significant clinical potential.
Conclusions
Our findings reveal the critical role of the MALAT1/miR-7-5p/TCF4 axis in MenSCs’ improvement of TE, which could be used as novel therapeutic targets in the future.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241259552 – Supplemental material for MALAT1/miR-7-5p/TCF4 Axis Regulating Menstrual Blood Mesenchymal Stem Cells Improve Thin Endometrium Fertility by the Wnt Signaling Pathway
Supplemental material, sj-docx-1-cll-10.1177_09636897241259552 for MALAT1/miR-7-5p/TCF4 Axis Regulating Menstrual Blood Mesenchymal Stem Cells Improve Thin Endometrium Fertility by the Wnt Signaling Pathway by Huiru Wang, Kai Chen, Lu Zong, Xin Zhao, Jingxin Wang, Shiwei Fan, Bing Shen and Shengxia Zheng in Cell Transplantation
Footnotes
Author Contributions
H.W., K.C.: performed the experiments and drafted and revised the manuscript; L.Z., X.Z.: collected the human menstrual blood samples; J.W., S.F.: data analysis and interpretation; B.S., S.Z.: conception and design of the experiments; and S.Z.: final approval of the manuscript. All authors discussed the results and commented on the manuscript.
Availability of Data and Material
Relevant data can be found within the article and its supplementary information.
Ethical Approval
This study was approved by the Ethics Committee of the University of Science and Technology of China (NO. 2021KY015) on February 7, 2021.
Statement of Human and Animal Rights
The studies involving human subjects were conducted according to the principles outlined in the Declaration of Helsinki. The animal experiment was approved by the Animal Ethics Committee of the University of Science and Technology of China (USTC, code no. 2023-N (A)-92) on August 30, 2023.
Statement of Informed Consent
Written informed consent and anonymized human samples were obtained from all participants before their use.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Grant No. 81971339), the Natural Science Foundation of Anhui Province (Grant No. 1808085MH242) and FDCT grants from the Macao Science and Technology Development Fund (grant code 002/2023/ALC and 006/2023/SKL).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
