Abstract
Non-small-cell lung cancer takes up the majority of lung carcinoma-caused deaths. It is reported that targeting PD-1/PD-L1, a well-known immune evasion checkpoint, can eradicate tumor. Checkpoint inhibitors, such as monoclonal antibodies, are actively employed in cancer treatment. Thus, this review aimed to assess the therapeutic and toxic effects of PD-1/PD-L1 inhibitors in treatment of NSCLC. So far, 6 monoclonal antibodies blocking PD-1/PD-L1 interaction are identified and used in clinical trials and randomized controlled trials for NSCLC therapy. These antibody-based therapies for NSCLC were collected by using search engine PubMed, and articles about the assessment of adverse events were collected by using Google search. Route of administration and dosage are critical parameters for efficient immunotherapy. Although antibodies can improve overall survival and are expected to be target-specific, they can cause systemic adverse effects in the host. Targeting certain biomarkers can limit the toxicity of adverse effects of the antibody-mediated therapy. Clinical experts with knowledge of adverse effects (AEs) of checkpoint inhibitors can help manage and reduce mortalities associated with antibody-based therapy of NSCLC.
Introduction
Lung cancer is the foremost reason for deaths related to cancer in the United States. According to statistics of the year 2019, the death toll related to cancers of respiratory system will exceed one-tenth of a million. There are several new treatment regimens endorsed by NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) for the management of non-small-cell lung cancer (NSCLC) 1 . Cancer cells are identified by the immune system, especially T-cells for control or elimination depending on receptors that strengthen the antitumor efficacy 2 . Such cells take part in multiple mechanisms to prevent this immune attack. PD-1 pathway is one mechanism for immune escape in several mouse tumor models. Manipulating such mechanism can allow the immune system to eliminate tumor 3 .
Over the past two decades, monoclonal antibodies (mAbs) have emerged as successful therapeutic agents for cancer in a multibillion-dollar market 4,5 . Recently, Relatlimab, Omburtamab, Etigilimab, Enoblituzumab and Tiragolumab are actively employed mAbs as immune checkpoint inhibitors (ICIs) at clinical level 6 . Programmed cell death protein (PD-1), a 288 amino acid, is a surface molecule often designated as a membrane protein that is expressed on immune cells including dendritic cells. Programmed death ligand 1 (PD-L1), a major PD-1 ligand and 40-kDa type 1 transmembrane protein, is often expressed in several types of malignant tumors and is related to survival and tumor progression 7,8 . PD-1 can interact with PD-L1 and result in the release of signals that can regulate T-cell mediated immunity 9 . This interaction ensures that immune system is activated at the proper timing and thereby the chances of chronic inflammation are reduced 10 . Cancer cells express proteins including PD-1 on their surface to trick the immune cells and evade their detection, and this action can stop a cytolytic activity 11 . An increase in PD-1 expression is considered as a hallmark for exhaustion of T-cells 12 . Notably, according to a meta-analysis based on randomized controlled trials, higher PD-L1 level in NSCLC cells is related to better efficacy of treatment by PD-1/PD-L1 inhibitors 13 .
Blockade of PD-1/PD-L1 pathway can cause limited toxicity compared to CTLA-4 blockade 14 . Blockade using monoclonal antibodies is a recently well-studied immune checkpoint inhibition and is used as a therapy for a variety of diseases including cancers of multiple origins 15 . This blockade of PD-1 using antibodies is now applied to treat patients with recurrence of cancer 16 . Although therapeutic targeting of PD-1/PD-L1 axis using ICIs can be more efficient than conventional chemotherapy, it can cause immune system related adverse effects (irAEs) predominantly in the digestive system, and these effects can be systemic. Drug resistance emerged with the blockade by ICIs is considered as a consequence of the progress of tumor neoantigens and the increase in immune checkpoint proteins that are not related to PD-1/PD-L1 axis 17 –19 .
Therefore, present review focuses on therapeutic roles and toxicity of ICIs targeting the PD-1/PD-L1 axis for clinical management of NSCLC. Clinical trials and randomized controlled trials of past decade were searched from PubMed for therapy and toxicity of ICIs in management of NSCLC.
PD-1/PD-L1 Axis As A Target for Therapy of NSCLC
The blockade of PD-1 and PD-L1 ligation can relieve the dysfunction, exhaustion, and tolerance of T-cells, so it has been proved to be a successful way to fight against cancer 20 . Yet, the antibodies may or may not have similarity in their binding sites on PD-1 21 . Relapse in disease can occur in patients with primary or acquired resistance to the antibody-based monotherapy and new approaches are employed recently to avoid or overcome the resistance to such therapy 22 . Genetic polymorphisms in PD-1 loci can increase the chances of developing several autoimmune disorders. PD-1/PD-L1 expression may vary with specific types of tumor so PD-1/PD-L1 can act as prognostic or diagnostic markers in several cancers. Therefore, several biomarkers such as elevated lymphocytes and eosinophils, low levels of circulating tumor DNA, and lactate dehydrogenase can help predict the responses to ICIs. Tumor mutational burden related to genes such as Polybromo-1 can also serve the purpose 23,24 .
Emerging Roles of Monoclonal Antibodies at the Nanoscale
As is known, mAbs are roughly 10 nm in size and possess unique properties. Dynamic light-scattering is used to analyze antibody formulations at the nanoscale. To serve therapeutic purposes, their affinity should be 1 nM or less for a specific antigen. They are absorbed via lymphatic system depending on convection and diffusion. These are plausible mechanisms for the uptake of mAbs 25 –27 . Glass and silica microparticles can adsorb mAbs via surface layering of 4-nm thickness 28 . Nanoparticles conjugated with antibodies are effectively delivered to their targeted site because of the size around 50 nm 29 . Administration of antibody-coated nanoparticles through subcutaneous route at fixed and optimized doses can improve targeted delivery and prevent irAEs 30,31 . Supportive of the fore-mentioned information, PD-L1 antibodies are conjugated onto copolymer nanoparticular surface and loaded with chemotherapeutic drugs to achieve targeted therapy and inhibition of PD-L1 expression in cancer cells 32 . Lipids, polymers and metal-based nanoparticles with an ideal size of 200 nm have been used in specific-targeting of lung cancer cells 33 .
Role of Biomarker Validation
Biomarkers can improve the specific targeting of tumor associated antigens and decrease systemic toxicity 34 . Tumor mutational burden, tissue polypeptide-specific antigen, and immunohistochemistry assays are valuable tools in biomarker validation for early detection of cancer 35 –38 . PD-L1 testing based on immunohistochemical platforms such as Dako and Ventana are used with four FDA approved antibodies for binding with precise epitope of PD-L1. Antibodies such as 22C3, 28–8, SP263 and SP142 are used to identify patients who can respond to immunotherapy for PD-L1 positive NSCLC 39,40 . Elevated expressions of PD-L1 is optimum for treatment using ICIs 13 . At a safe dose, combinatorial therapy of mAbs with other drugs can increase the OS of patients with NSCLC. Statuses of other oncogenes such as EGFR, ALK, KRAS, MET, ROS1, BRAF, and NTRK are critical in identification of specific ICIs for NSCLC 41,42 . Host microbiomes are tested and used as alternative biomarkers for NSCLC 43 . Also, several biomarkers are discovered to predict or influence the toxicity of PD-1/PD-L1 inhibitor-based therapy. For example, cytokines such as IL1Rα, IL-1α/β, IL-2, IL12p70, IL-13, GM-CSF, G-CSF, fractalkine, and IFN-α2 are proved as biomarkers highly expressed in patients having severe irAEs during ICI treatment 44 . TIM3 is proved to hinder anti-cancer immunity and regulate resistance to PD-1 and PD-L1 inhibitors 45 . The TLR3-specific adjuvant alleviates resistance to therapy using PD-L1 antibody without toxicity 46 . Targeting gut microbiota is reported to enhance efficiency and decrease toxicity of current therapy depending on various agents including anti-PD-L1 47 .
Clinical and Randomized Controlled trials for NSCLC Therapy using ICIs Targeting the PD-1/PD-L1 Axis
Atezolizumab, avelumab, durvalumab, cemiplimab, nivolumab, and pembrolizumab are fully humanized IgG1 and IgG4 antibodies that target the PD-1/PD-L1 axis in combinatorial first-line treatment of NSCLC. They can elicit anti-tumor effects through mechanisms involving adaptive and innate immunity 48 –53 . The search was performed in PubMed using the filter of 2010 to 2020 for clinical trials and randomized controlled trials in treatment of NSCLC targeting the PD-1/PD-L1 axis. A total of 8 trials were identified as a result of the search. We summarized the characters, applications and toxicities of 5 antibodies from the 8 researches in Table 1. The results indicated that the median overall survival (OS) was improved in Phase 2 and 3 trials using atezolizumab by more than 4 months versus docetaxel; increasing OS was coupled with higher PD-L1 level in NSCLC patients, and the adverse event profile for atezolizumab was more favorable than docetaxel 54,55 . In an open-label Phase 3 trial, avelumab improved the median OS by one month in comparison with docetaxel, which was insignificant; high PD-L1 expression in NSCLC patients was related to longer OS; and the ratio of treatment-related adverse event in patients receiving avelumab treatment was generally lower than those receiving docetaxel 56 . There was no report for durvalumab and cemiplimab according to the search. Yet, trials at various phases are ongoing for the treatment of NSCLC using cemiplimab 57 . Nivolumab had an OS of more than 40% after one year in a Phase 2, single-arm trial in NSCLC patients; the treatment-related immune-mediated adverse events after nivolumab treatment were neither frequent nor severe and the toxicity of nivolumab was weaker than toxic chemotherapy 58 . In combination with other chemotherapeutic interventions at Phase I level, nivolumab treatment resulted in OS of 12.2 and 17.4 months; combined use of other drugs did not lead to higher risk of nivolumab-related immune-mediated adverse events and these adverse events are generally manageable 59,60 . Pembrolizumab in combination with ipilimumab improved the OS compared to standard chemotherapy 61 . The median OS was more than 10 months, but the combined use of pembrolizumab and ipilimumab may lead to a higher risk of adverse events 62 .
Characters, application and toxicities of antibodies against PD-1/PD-L1 summarized based on 8 clinical trials.

References: 54-61.
Systemic Toxicity of ICIs
The Immune-Related Adverse Events (irAEs) of ICIs increase at higher doses 63 . Inhibitors of PD-1/PD-L1 axis can cause systemic toxicities. PD-1 interacts with its alternate ligand PD-L2 when its expression is elevated in certain organs. This can result in resistance, survival or growth of tumor cells by T-cell inactivation, leading to an increase in toxicity (Fig. 1). The irAEs of ICIs include colitis, diarrhoea, hormonal imbalances, hypothyroidism, diabetes, acute injury to kidneys, pneumonitis, and myocarditis. Hepatic, pulmonary, and dermatological reactions such as pruritus and rashes are also evident 18,64,65,66 –70 .
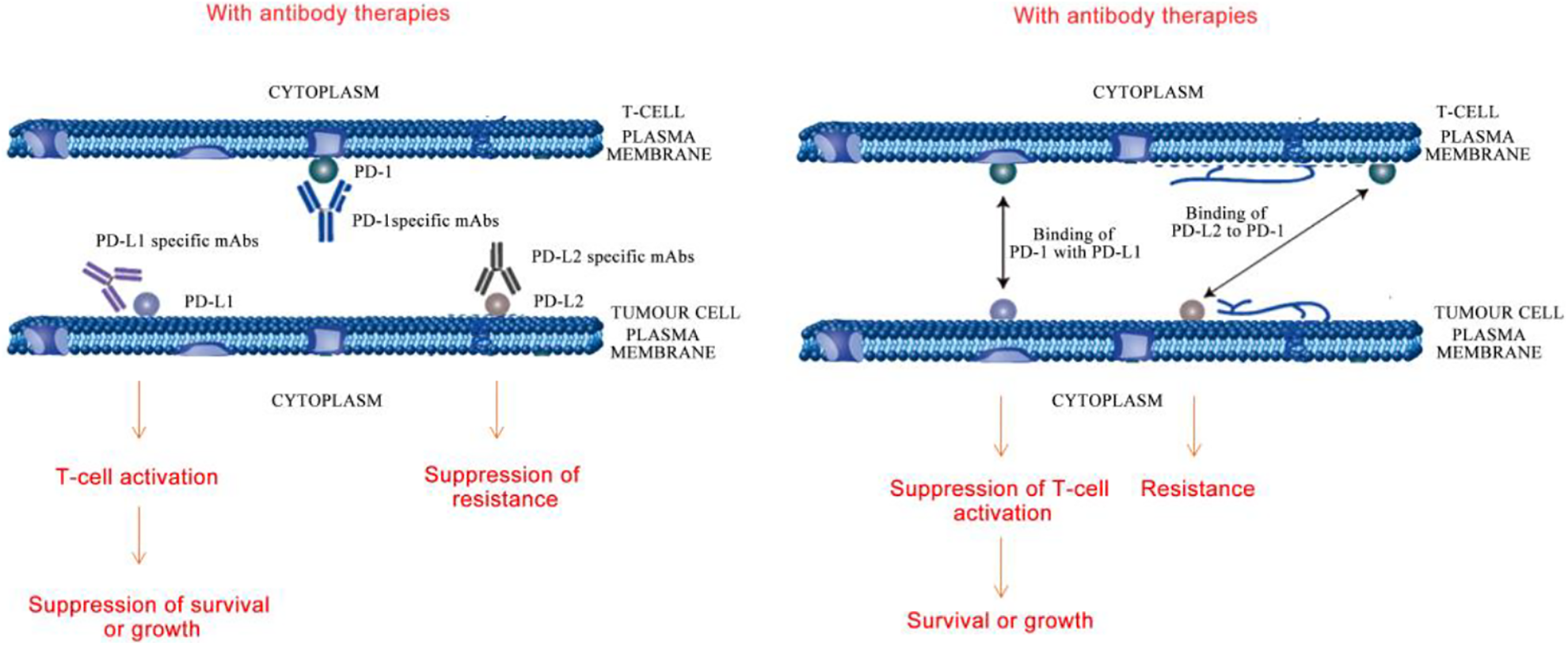
Pictorial representation of resistance, survival or growth among tumor cells as a result of T-cell inactivation leading to irAEs. Left: Blocking PD-1/PD-L1 by PD-1 or PD-L1 mAbs caused T-cell activation and suppressed tumor cell growth or survival; blocking PD-1/PD-L2 by PD-L2 mAbs repressed tumor cell drug resistance. Right: PD-1 on T-cell bound to PD-L1 on tumor cell to suppress T-cell activation and facilitate tumor cell growth or survival; PD-1 on T-cell bound to PD-L2 on tumor cell to facilitate tumor drug resistance.
Atezolizumab can induce systemic toxicities comparable to other PD-1 inhibitors such as avelumab and durvalumab by interacting with PD-L1, but is different from the irAEs of conventional chemotherapy. This can avoid interaction of PD-1 with PD-L1. 20 mg/kg is the tolerable dose in clinical trials involving atezolizumab and avelumab intended for NSCLC therapy at Phase I 50,71 –74 . Nivolumab at 3 mg/kg was the optimum dose in combination with ipilimumab (1 mg/kg) for treatment of NSCLC 75 . The 2-year OS for this antibody (5∼10 mg/kg) did range between 25∼62% based on the combinatorial agent used 76,77 . Pembrolizumab can cause thyroid dysfunction and even fatal pneumonitis in NSCLC patients 78,79 .
Toxicity of ICIs on Different Systems of the Human Body
Fatigue of all grades is one of the common irAEs induced by ICIs 80 . Macules and papules are skin-related irAEs of grade 1 and 2. Vitiligo-like depigmentation is related to grade 3 or 4 symptoms 81 . With incidence lower than 1%, AEs of ICIs associated with cardiovascular system are myocarditis, pericarditis, cardiac fibrosis, cardiac arrest, arrhythmias, heart failure, large pericardial effusion, tamponade and Takotsubo syndrome. Electrocardiogram, troponin monitoring and diagnosis of myocarditis are baseline tools to detect abnormalities related to heart 82 –84 . Disorders of the coagulation-fibrinolysis system can occur 85 . Grade 1 to 4 diarrhoea and colitis are lower GI-based irAEs. ICIs can induce upper GI tract toxicity, resulting in decline of appetite and nausea 86,87 . Esophagitis, gastritis, duodenitis, and jejunitis are other upper GI tract-associated irAEs 88 . Thyroid dysfunction and hypophysitis are the major toxicities associated with the endocrine system, although the effects can be systemic 89 . Pneumonitis is a late grade irAE in NSCLC patients 90 . Other pulmonary events such as dyspnoea, hypoxia and lung opacities were also observed 91 . Polyneuropathy, myasthenia gravis, Bell’s palsy and encephalopathy are neurological irAEs 92 . Loss of skeletal muscle mass and proteinuria are other irAEs of ICIs for NSCLC (Fig. 2). These irAEs are enlisted in Table 2.
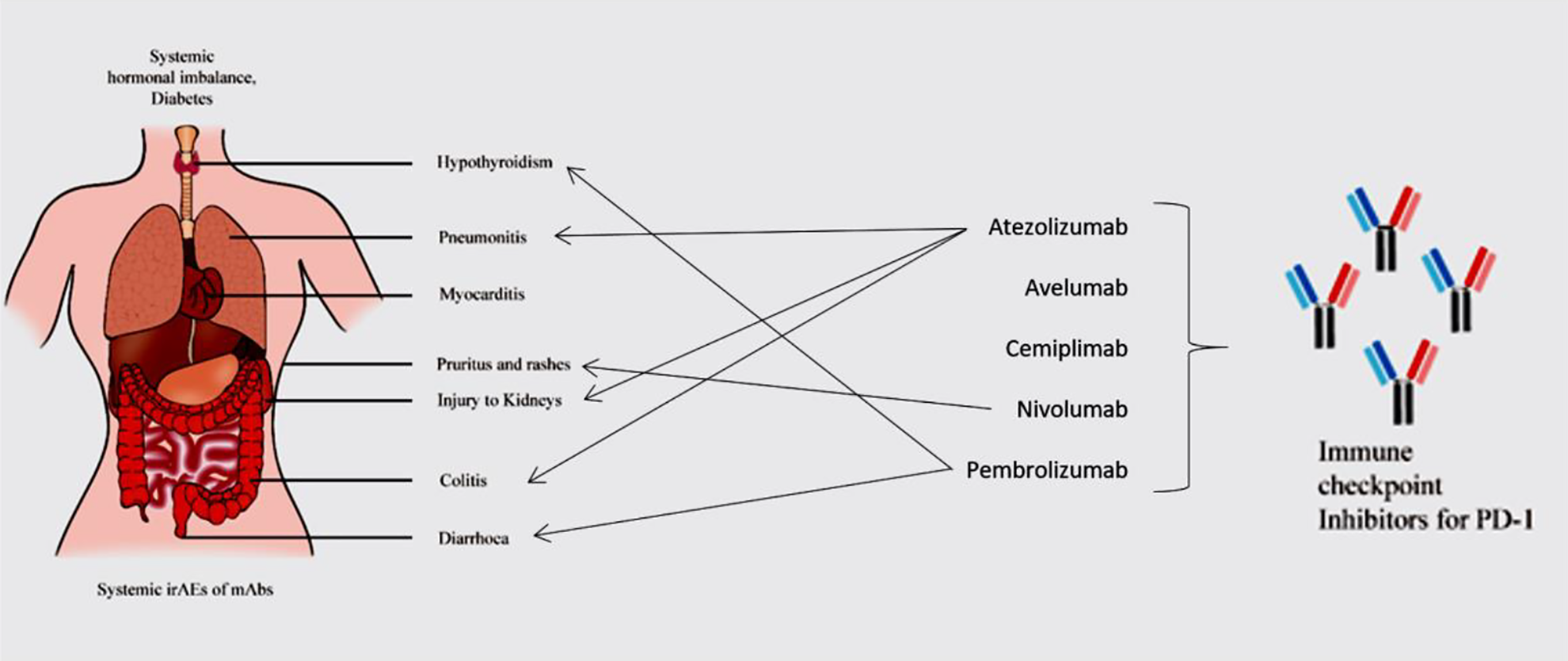
Systemic Toxicity of ICIs.

References: 76-90
The toxicities associated with immunotherapy may be autoimmune (on target, off-tumor toxicity) or cytokine-associated. The mechanisms related to such toxicities may vary from the reactions similar to immune system-related allergies to the auto-immune reactions, such as reactions observed in chemotherapy or the entry of T-cells into central nervous system. There are several tests that individually identify the irAEs specific for each organ by detecting the specific antigens on toxicity-induced organs using specific antibodies. Steroids and other immunosuppressive agents are used for management of such irAEs 93,94 .
Challenges and Future Directions for Therapy of NSCLC using Checkpoint Inhibitors
Resistance of NSCLC cells to available drugs can limit the therapeutic potential of a drug and therefore requires proper management to control progression 95 . Immune escape and evasion in NSCLC patients play a critical role in cancer progression 96 . Amplification of tumor-infiltrating lymphocytes after collection from the patient, growth at laboratory scale and giving back can boost the immune system 97,98 . Clinicians are aware that the management of irAEs of ICIs can follow guidelines for cancer care and the challenges imposed 99 . Early detection is critical to the management of increase in systemic toxicity and mortality 100 .
Peptide antagonists for PD-1 and its ligand PD-L1 are emerging alternatives for cancer therapy and a few examples have been elucidated 101,102 . A novel peptide RK-10 identified from RCSB protein data bank has been known to detect PD-L1 in several circulating and tumor cells and tissues of patients suffering from various cancers 103 . T-cell therapy allowing the same cells to secrete certain peptide fragments may improve both safety and anti-tumor efficacy as such peptides can stay localized to tumor cells 104 . Non-blocking antibodies for PD-1 possess anti-tumor effect similar to that of anti-PD-1 monotherapy using blocking antibodies 105 . An interesting study suggests that on-target lifespan of anti-PD-1 antibodies is much shorter than usually expected on the tumor-infiltrating T-cells. According to the authors, second generation antibodies can be a fascinating approach for they have much more specific Fc regions that possess extended binding to such T-cells 106 .
Analyzing the composition of gut microbiota is a key point in determining the therapeutic efficacy of antibodies against PD-1/PD-L1. Fecal microbiome could improve sensitivity of anti-PD-L1 therapy which differs between melanoma patients who respond to therapy and who do not. Such patients had unique microbiome, and enhanced immunity was observed in mice transplanted with fecus of patients who responded to checkpoint blockade immunotherapy 107 .
Therefore, novel therapeutic regimens with specific targeting capacity are mandatory for prolonging the survival among NSCLC patients 108 .
Conclusions
NSCLC can be treated efficiently by targeting the PD-1/PD-L1 axis using checkpoint inhibitors such as monoclonal antibodies. Atezolizumab, avelumab, durvalumab, cemiplimab, nivolumab, and pembrolizumab are fully humanized antibodies used in clinical trials and randomized controlled trials for NSCLC therapy. Treatment modalities are critical parameters in targeted therapy. Identification of biomarkers and targeted therapy can help in limiting the toxicity of adverse effects of antibody-mediated therapy. Further research in identifying checkpoint inhibitors can help eradicate cancers of various origins.
Supplemental Material
Supplemental Material, sj-docx-1-cll-10.1177_09636897211041587 - Therapeutic and Systemic Adverse Events of Immune Checkpoint Inhibitors Targeting the PD-1/PD-L1 axis for Clinical Management of NSCLC
Supplemental Material, sj-docx-1-cll-10.1177_09636897211041587 for Therapeutic and Systemic Adverse Events of Immune Checkpoint Inhibitors Targeting the PD-1/PD-L1 axis for Clinical Management of NSCLC by Jing Chen, Yaser Alduais and Baoan Chen in Cell Transplantation
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial statement for research, authorship and/or publication of thiu article: The work was supported by National Natural Science Foundation of China (No.81903091), the Fundamental Research Funds for the Central Universities (No.2242020R20006) and the Jiangsu Planned Projects for Postdoctoral Research Funds.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
