Abstract
Although it is well known that liver allografts are often accepted by recipients, leading to donor-specific tolerance of further organ transplants, the underlying mechanisms remain unclear. We had previously used an in vitro model and showed that mouse liver sinusoidal endothelial cells (LSECs) selectively suppress allospecific T-cells across major histocompatibility complex (MHC) barriers. In the present study, we established an in vivo model for evaluating the immunomodulatory effects of allogeneic LSECs on corresponding T-cells. Allogeneic BALB/cA LSECs were injected intraportally into recombination activating gene 2 γ-chain double-knockout (RAG2/gc-KO, H-2b) mice lacking T, B, and natural killer (NK) cells. In order to facilitate LSEC engraftment, the RAG2/gc-KO mice were injected intraperitoneally with monocrotaline 2 days before the adoptive transfer of LSECs; this impaired the host LSECs, conferring a proliferative advantage to the transplanted LSECs. After orthotopic allogeneic LSEC engraftment, the RAG2/gc-KO mice were immune reconstituted intravenously with C57BL/6 splenocytes. After immune reconstitution, mixed lymphocyte reaction (MLR) assay using splenocytes from the recipients revealed that specific inhibition of host CD4+ and CD8+ T-cell proliferation was greater in response to allostimulation with irradiated BALB/cA splenocytes rather than to stimulation with irradiated third party SJL/jorllco splenocytes. This inhibitory effect was attenuated by administering anti-programmed death ligand 1 (PD-L1) monoclonal antibody during immune reconstitution in the above-mentioned mice, but not in RAG2/gc-KO mice engrafted with Fas ligand (FasL)-deficient BALB/cA LSECs. Furthermore, engraftment of allogeneic BALB/cA LSECs significantly prolonged the survival of subsequently grafted cognate allogeneic BALB/cA hearts in RAG2/gc-KO mice immune reconstituted with bone marrow transplantation from C57BL/6 mice. In conclusion, murine LSECs have been proven capable of suppressing T-cells with cognate specificity for LSECs in an in vivo model. The programmed death 1/PD-L1 pathway is likely involved in these suppressive effects.
Keywords
Introduction
Liver allografts are well accepted across major histocompatibility complex (MHC) barriers without immunosuppression in some species (5,6,10). The presence of a liver allograft can suppress the rejection of other solid tissue grafts (e.g., heart and skin) from the same donor (1); hence, the liver appears to favor immune tolerance rather than immunity. The mechanisms underlying such immune tolerance may be mediated by innate and adaptive immune regulators (26). We recently demonstrated a novel relevant mechanism of this liver allograft tolerance: naive allogeneic liver sinusoidal endothelial cells (LSECs) selectively inhibit cluster of differentiation 4 positive (CD4+) and CD8+ T-cells with direct allospecificity in mice in which liver allografts are normally accepted without recipient immune suppression across MHC barriers (22,23). Naive LSECs in mice constitutively express all molecules necessary for antigen presentation; that is, freshly isolated LSECs express MHC class II, CD40, CD80, and CD86 but do not induce allogeneic T-cell proliferation (22). In an allogeneic mixed hepatic constituent cell-lymphocyte reaction assay. Marked proliferation of reactive CD4+ and CD8+ T-cells was observed when LSECs were depleted from the hepatic constituent cell stimulators (23). The sinusoidal architecture, in which circulating leukocytes are forced into frequent contact with LSECs due to the small sinusoid diameter (7–12 μm), most likely promotes the immunomodulatory activity of LSECs against alloreactive T-cells. Such an interpretation, which is based on in vitro results, raised the question of whether recipients of isolated allogeneic LSECs, which may have a pivotal tolerogenic property, allospecifically suppress T-cells.
It was recently reported that inoculation with LSECs leads to host liver sinusoidal endothelium repopulation; that is, syngeneic LSECs transplanted into the liver via portal vein injection survived only in the liver without redistribution to other organs, including the lungs, heart, spleen, kidney, and intestines (2). By using a similar in vivo model system in the present study, we investigated the suppressive effects of exogenously inoculated allogeneic LSECs on allospecific immune responses. To exclude the possibility that the inoculated LSECs were eliminated by either innate or acquired immunity before achieving engraftment in the liver sinusoidal endothelium, we employed recombination activating gene 2 γ-chain double-knockout (RAG2/gc-KO) mice, which lack natural killer (NK), natural killer T (NKT), T-, and B-cells, as allogeneic LSEC recipients. The allogeneic LSEC chimeric mice were then reconstituted with host-type splenocytes (SPLs) or bone marrow cells (BMCs) to investigate the immune responses of allospecific T-cells.
Materials and Methods
Mice
Eight- to 12-week-old female C57BL/6 (B6, H-2b), BALB/cA (BALB/c, H-2d), and SJL/jorllco (SJL/j, H-2s) mice were purchased from Clea Japan (Tokyo, Japan). Female Cpt.C3-Tnfsf6gld mice (BALB/c-gld, H-2d, BALB/cA background) that were homozygous for the Fas ligand (FasL)gld mutation were purchased from The Jackson Laboratory (Bar Harbor, ME, USA). Age-matched (8- to 12-week-old) mice were used for the experiments. RAG2/gc-KO (H-2b) mice were purchased from Taconic Farms (Hudson, NY, USA). All animals were maintained under pathogen-free conditions and in compliance with national and institutional guidelines. Animal experiments were approved by the Institutional Review Board of Hiroshima University and were conducted in accordance with the National Institutes of Health guidelines.
LSEC Isolation
Since we previously demonstrated that LSECs exclusively express CD105 at higher levels than the endothelium of the central veins or other vessels in the liver (23,27), CD105+ cells were positively selected for the isolation of LSECs from nonparenchymal cell fractions of the liver. Briefly, the livers of BALB/c or BALB/c-gld mice were perfused with portal injection using the two-step collagenase perfusion method (23,25,28). Disaggregated liver cells were centrifuged at 50 × g for 1 min for hepatocyte removal. The supernatant was subsequently centrifuged at 150 × g for 5 min, and hepatic nonparenchymal cells (HNPCs) were obtained and stained with biotin- conjugated anti-CD105 (MJ7/18, eBioscience, San Diego, CA, USA). The cells were then counterstained with streptavidin micro-beads (Miltenyi Biotec, Bergisch Gladbach, Germany) and magnetically sorted using an automated magnetic cell sorter (autoMACS, Miltenyi Biotec). This sorting technique yielded 4.8 ± 0.5 × 106 cells/body in the positive fraction, with 97.9 ± 0.3% purity (n = 4), indicating that marginal contamination with cells other than LSECs could not be ruled out.
Surface Marker Analyses of Freshly Isolated LSECs
The CD105+ LSECs were freshly isolated from BALB/c mice. The expression levels of H-2Kd/H-2Dd (MHC-class I), I-A/I-E (MHC-class II), CD80, CD146, FasL, and CD274 [programmed death ligand 1 (PD-L1)] were measured by flow cytometry (FCM; see later).
Orthotopic LSEC Engraftment
Isolated LSECs were prepared as a single-cell suspension. The LSECs (4 × 106 cells) in 0.5 ml of Medium 199 (Sigma-Aldrich, Saint Louis, MO, USA) containing 1% N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (HEPES) buffer (GIBCO; Invitrogen, Carlsbad, CA, USA) were injected into each recipient RAG2/gc-KO mouse through the portal vein using a 30-gauge needle. Medium 199 containing 1% HEPES buffer without LSECs was injected into control recipient RAG2/gc-KO mice. In some experiments, we administered monocrotaline (MCT; Sigma-Aldrich), a genotoxic pyrrolizidine alkaloid that impairs the host LSECs and confers a proliferative advantage on the transplanted LSECs. It was previously discovered that MCT injection to recipients prior to LSEC inoculation markedly accelerated the engraftment of inoculated LSECs, but that MCT-induced endothelial injury in additional vascular beds did not promote inoculated LSEC survival in extrahepatic locations (7). Two days before the adaptive transfer of LSECs, a 200–300 mg/kg dose of MCT was intraperitoneally injected into recipient RAG2/gc-KO mice to facilitate transplanted LSEC engraftment. The RAG2/gc-KO mice that received the same dose of MCT alone were used as controls. To evaluate orthotopic engraftment of transplanted allogeneic BALB/c LSECs in the recipient liver, the livers were immunostained using fluorescein isothiocyanate (FITC)-conjugated H-2Kd/H-2Dd monoclonal antibody (mAb) (34-1-2S; Beckman Coulter, Fullerton, CA, USA), phycoerythrin (PE)-conjugated anti-mouse CD146 (Abcam, Cambridge, UK), and anti-CD274-PE (MIH5; BD Pharmingen, San Diego, CA, USA) mAbs and examined with a fluorescence microscope (BZ-8000; KEYENCE, Osaka, Japan). To quantify LSEC chimerism, HNPCs were reisolated from the livers of the recipients of BALB/c LSECs. For the FCM analyses, the LSECs were selected by gating on CD105+ cells, and the proportion of cells expressing H-2Kd/H-2Dd (MHC-class I) among all of the LSECs was determined.
Immune Reconstitution of Recipient RAG2/gc-KO Mice
Immune reconstitution of the immunodeficient mice was performed by use of the method described previously (32). In brief, B6 SPLs (2×107) in 0.5 ml of Medium 199 containing 1% HEPES buffer were injected into recipient RAG2/gc-KO mice through the tail vein for immune reconstitution after engraftment of LSECs from either BALB/c or BALB/c-gld mice. In some in vivo experiments, to block PD-L1, recipient mice were pretreated with 500 μg of anti-PD-L1 blocking mAb (MIH5; eBioscience) or rat IgG isotype control antibody (Ab) (Beckman Coulter) just before immune reconstitution, and these antibodies were readministered 3 days later. One week after SPL inoculation, the recipient mice were sacrificed, and SPLs were obtained for subsequent mixed lymphocyte reaction (MLR) assay. In some experiments, bone marrow transplantation (BMT) was performed from B6 mice to recipient RAG2/gc-KO mice after LSEC engraftment. Briefly, recipient RAG2/gc-KO mice were irradiated (3 Gy) and injected with BMCs (1×107 cells/mouse) from B6 mice through the tail vein. After SPL inoculation or BMT, peripheral blood mononuclear cells (PBMCs) were obtained from recipient mice, and the CD4+ T, CD8+ T, NK, and NKT cell immune reconstitution levels were evaluated by FCM. Immune reconstituted recipient RAG2/gc-KO mice were used at least 6 weeks after BMT for the subsequent heart transplant experiment. Mice that did not receive LSECs but were immune reconstituted with either SPLs or BMCs were used as controls for subsequent studies.
CFSE-MLR Assay
SPLs obtained from B6 (syngeneic stimulator), BALB/c (allogeneic stimulator), or SJL/j (third-party stimulator) mice were irradiated with 30 Gy and used as stimulator cells. For MLR using the carboxyfluorescein diacetate succinimidyl ester (CFSE) labeling technique (CFSE-MLR), SPLs from immune reconstituted recipient RAG2/gc-KO mice were labeled with 5 μM CFSE (Molecular Probes, Eugene, OR, USA) as described previously (21) and resuspended in culture medium as responder cells. The stimulator (2 × 106 cells) and responder (2 × 106 cells) cells were cocultured (1:1 ratio) in a total volume of 1 ml of medium in 48-well flat-bottom plates at 37°C in an incubator with an atmosphere of 5% CO2 in the dark for 4.5 days. In the resulting CFSE fluorescence histograms, CD4+ and CD8+ T-cells were selected by gating and analyzed for CFSE fluorescence. The numbers of division precursors and mitotic events of alloreactive T-cells were mathematically determined using logarithmic CFSE intensity on the basis of the peak of the undivided cells, while the mitotic index was calculated by dividing the total number of mitotic events by the total number of precursors (21). The T-cell stimulation index was calculated by dividing the mitotic index of T-cells responding to allogeneic or third-party SPLs by that of T-cells responding to the syngeneic SPLs.
FCM Analyses
The following reagents were used for surface staining: anti-CD3-FITC (145-2c11), anti-CD4-FITC (GK1.5), anti-CD8-FITC (53-6.7), anti-CD80-FITC (16-10A1), anti-I-A/I-E-FITC (2G9), anti-CD4-PE (GK1.5), anti-CD8-PE (53-6.7), anti-CD274-PE (MIH5), anti-mouse H-2Kd/H-2Dd-PE (SF1-1.1), anti-CD4-biotin (RM4-5), anti-CD8-biotin (53-6.7), anti-CD44-biotin (IM7), anti-CD45RB-biotin (16A), anti-CD62L-biotin (MEL-14), anti-CD105-biotin (MJ7/18), anti-NK1.1-allophycocyanin (PK136) mAbs, isotype-matched control Abs, streptavidin-allophycocyanin (BD Pharmingen), anti-CD146-FITC (ME-9F1; Miltenyi Biotec), FITC-conjugated mouse (C3H/HeJ) anti-mouse H-2Kd/H-2Dd (34-1-2S; Beckman Coulter), and anti-FasL-PE mAbs (MFL3; eBioscience). All analyses were performed using a FACSCalibur (BD Biosciences, San Jose, CA, USA). Nonspecific Fc-γ receptor binding of labeled mAbs was blocked by purified rat anti-mouse CD16/CD32 mAb (mouse BD Fc block) (2.4G2; BD Pharmingen). Dead cells were excluded from the analysis by forward scatter and propidium iodide (Sigma-Aldrich).
Heterotopic Heart Transplantation
Cervical heterotopic heart transplantation from BALB/c or SJL/j mice into the immune reconstituted recipient RAG2/gc-KO mice was performed using a modified cuff technique(20). Briefly, the right external jugular vein and the right common carotid artery were dissected free and fixed to the appropriate cuffs composed of polyethylene tubes (2.5F, Portex Ltd., London, UK). For anastomoses, the aorta and the main pulmonary artery of the obtained donor heart were drawn over the end of the common carotid artery and the external jugular vein, respectively. The graft ischemic time for the transplanted hearts was <30 min. Graft function was monitored by daily inspection and palpation. Rejection, defined as cessation of graft beating, was confirmed by histological analysis.
Statistical Analyses
The results were analyzed statistically by using Student's t test or the log-rank test for graft survival. Data are expressed as mean ± standard error of the mean (SEM). A value of p < 0.05 was considered statistically significant.
Results
Allogeneic LSEC Engraftment Was Successfully Achieved In Vivo
Previous studies have shown that allogeneic LSECs render allospecific T-cells hyporesponsive in vitro (22,23). In order to test the immunoregulatory effects of LSECs in vivo, we generated an LSEC engraftment model in which RAG2/gc-KO mice were orthotopically engrafted with allogeneic BALB/c LSECs (4 × 106 cells/mouse) and then immunologically reconstructed using SPLs (2 × 107 cells/mouse) from B6 mice 1 week after the LSEC inoculation. The inoculated LSECs were barely detected in the host liver sinusoid 2 weeks after inoculation in immunohistochemical studies using anti-H-2Kd/H-2Dd Ab (Fig. 1B). LSEC engraftment was successfully increased by MCT administration 2 days prior to the inoculation. A significant proportion of LSECs was stably detected even 7 weeks after LSEC inoculation in MCT-pretreated mice (Fig. 1C). By merging these immunofluorescence microscopic findings, we confirmed that the engrafted cells that were stained with anti-H-2Kd/H-2Dd Ab presented on the lining of the hepatic sinusoid face to the sinusoidal lumen and also were stained with anti-CD146 mAb, which is a LSEC-specific marker (Fig. 1D, E). The FCM analyses consistently revealed that MCT pretreatment enhanced LSEC engraftment; that is, the LSEC chimerism level increased (Fig. 2A, B). Immune reconstitution was confirmed by the presence of both CD4+ and CD8+ T-cells in the spleens of recipient RAG2/gc-KO mice by FCM 1 week after SPL inoculation. There was no statistically significant difference in the degree of CD4+ and CD8+ T-cell reconstitution between recipient mice with allogeneic LSECs and corresponding control mice without allogeneic LSECs (Fig. 3).

An in vivo liver sinusoidal endothelial cell (LSEC) engraftment model was established using allogeneic BALB/c LSECs that were transferred to RAG2/gc-KO mice through the portal vein. Recipient recombination activating gene 2 γ-chain double-knockout (RAG2/gc-KO) mouse livers were immunostained with fluorescein isothiocyanate (FITC)-conjugated anti-mouse major histocompatibility complex class I (H-2Kd/H-2Dd) (A–C) and phycoerythrin (PE)-conjugated anti-mouse cluster of differentiation 146 (CD146) monoclonal antibodies (mAbs) (D) for identification of transferred LSECs in the liver. (A) Livers from control RAG2/gc-KO mice that did not receive allogeneic BALB/c LSECs. (B) A few transferred LSECs were identified in the liver sinusoids 2 weeks after LSEC inoculation in recipient mice without monocrotaline (MCT). (C) Increased engraftment of the transferred LSECs was identified 7 weeks after LSEC inoculation in recipient mice pretreated with MCT. (D) Endothelial cells were immunostained with CD146 mAb. (E) Merged image of (C) and (D). The figures shown are representative of three independent experiments.

MCT pretreatment enhanced allogeneic LSEC engraftment. The RAG2/gc-KO mice were injected with BALB/c LSECs 2 days after the MCT pretreatment. Six weeks after the LSEC inoculation, hepatic nonparenchymal cells were reisolated from the recipient mice and stained for CD105 and H-2Kd/H-2Dd to analyze the donor-type MHC class I expression in the CD105+ LSECs by flow cytometry (FCM). (A) A representative figure in each group is shown. The thin and filled lines represent staining with isotype-matched control antibody (Ab) and anti-H-2Kd/H-2Dd mAb, respectively. The percent of Max is the number of cells in each bin of the histograms divided by the number of cells in the bin that contains the largest number of cells. The FCM profile from one experimental animal among four or five animals in each group is shown. (B) The percentages of H-2Kd/H-2Dd-positive cells among all of the CD105+ cells in the liver of the RAG2/gc-KO mouse recipients of BALB/c LSECs were determined by FCM analysis (MCT+, LSEC-: n = 4; MCT-, LSEC+: n = 5; MCT+, LSEC+: n = 5). The mean ± SEM values for the individual groups are shown. ***p < 0.001.

RAG2/gc-KO mice were immune reconstituted using B6 splenocytes (SPLs). BALB/c LSECs were inoculated in MCT-pretreated RAG2/gc-KO mice, and B6 SPLs were injected 6 weeks later. One week after SPL inoculation, the recipient mice were sacrificed, and SPLs were isolated for FCM analysis. Anti-CD4 and anti-CD8 mAbs were used for surface staining of these SPLs. In the FCM analyses, the percentages of CD4+ or CD8+ T-cells in the SPLs were measured in untreated control RAG2/gc-KO mice (n = 3) (left), immune reconstituted RAG2/gc-KO mice (MCT+, LSEC-: n = 4; MCT+, LSEC+: n = 4) (middle), and wild-type (WT) B6 control mice (n = 4) (right). The frequency of each gated population among total obtained cells is shown.
Orthotopic Engraftment of Allogeneic LSEC Led to Specific Inhibition of T-Cells Responding to Stimulation Cognate with LSECs
In order to address whether orthotopic engraftment of MHC-disparate allogeneic LSECs in the MCT-pretreated mice can induce specific inhibition of alloreactive T-cells after immune reconstitution with MHC-matched SPLs, we analyzed the responsiveness of reconstituted T-cells in MLR assays using the CFSE labeling technique. In syngeneic combinations using B6 SPLs as stimulators, the MLR assay revealed minimal T-cell proliferation in immune reconstituted recipient RAG2/gc-KO mice with and without allogeneic BALB/c LSEC engraftment (Fig. 4A, B). In fully allogeneic combinations using BALB/c SPLs, alloreactive T-cells of immune reconstituted recipient RAG2/gc-KO mice without allogeneic BALB/c LSEC engraftment proliferated markedly, as expected. In the recipient mice with allogeneic BALB/c LSEC engraftment, the response of the anti-BALB/c CD4+ and CD8+ T-cells was significantly less than that of the anti-third-party SJL/j CD4+ and CD8+ T-cells, whereas both responses were similar in the immune reconstituted RAG2/gc-KO control mice without allogeneic LSEC engraftment (Fig. 4C).

Allogeneic engraftment of the LSECs led to specific inhibition of T-cells responding to stimulation cognate with LSECs. RAG2/gc-KO mice were injected with BALB/c LSECs 2 days after MCT pretreatment. Six weeks after LSEC inoculation, B6 SPLs were injected into the recipient mice for immune reconstitution. One week after SPL inoculation, the recipient mice were sacrificed, and SPLs were obtained, carboxyfluorescein diacetate succinimidyl ester (CFSE) labeled, and cultured with stimulators. After 4.5 days of culture, the responder cells were analyzed by FCM. Using FCM analysis following mixed lymphocyte reaction (MLR), reactive T-cell proliferation can be visualized as the serial halving of CFSE intensity. (A, B) Representative FCM results of CD4+ and CD8+ T-cell division in the MLR using CFSE-labeled SPLs from the recipient RAG2/gc-KO mice as responders with irradiated SPLs from syngeneic B6 (left), allogeneic BALB/c (middle), or third-party SJL/j (right) mice as stimulators. The representative FCM profiles are shown (n = 4 in each group). A total of 10,000 cells are shown in the FCM profile figure. The percentage value in total cells is shown in each quadrant. (C) Stimulation indices of CD4+ and CD8+ T-cells are shown. In the FCM analysis, CD4+ and CD8+ T-cells were gated and analyzed for CFSE fluorescence, and their stimulation indices were calculated. The average values of four independent mice in each group are shown. *p < 0.05.
The PD-1/PD-L1 Pathway Was Involved in the Suppressive Effects of Engrafted Allogeneic LSECs on Alloreactive CD4+ T-Cells
The phenotypic analysis by FCM revealed that the freshly isolated BALB/c LSECs constitutively expressed FasL and PD-L1, which are death ligands, as well as MHC classes I and II and CD80, which are necessary for antigen-presenting cells (APCs), and CD146 (LSEC-specific marker) at the time of LSEC inoculation (Fig. 5). Hence, we explored the possibility that the Fas/FasL or programmed death 1 (PD-1)/PD-L1 pathway plays a role in the suppressive effect of LSECs on alloreactive T-cells in an in vivo model. FasL-deficient BALB/c (BALB/c-gld) LSECs were inoculated into RAG2/gc-KO mice after MCT treatment, and these mice were then immune reconstituted using B6 SPLs. The MLR assay utilizing SPLs from those mice revealed marked inhibition of alloreactive T-cell proliferation; that is, the stimulation indices of the anti-BALB/c CD4+ and CD8+ T-cells were still significantly less than those of third-party-reactive CD4+ and CD8+ T-cells in mice with BALB/c-gld LSECs (Fig. 6). In a separate experiment, LSECs from BALB/c mice were inoculated into RAG2/gc-KO mice, and then anti-PD-L1-blocking mAb was injected at 3-day intervals during immune reconstitution. Administration of anti-PD-L1-blocking mAb abrogated specific inhibition of the anti-BALB/c CD4+ T-cell response. Since PD-L1 expressed on host native LSECs also likely plays a significant role in the inhibition of self-reactive T-cells, the administration of anti-PD-L1 blocking mAb may interfere with control T-cell responses to syngeneic stimuli. In fact, the control anti-B6 T-cell responses in the anti-PD-L1 mAb-treated group were somewhat higher than those in the isotype-matched control Ab-treated group (data not shown). This result explains why anti-BALB/c responses were seemingly unaltered and anti-SJL T-cell responses were seemingly suppressed in the anti-PD-L1 mAb-treated group, compared to those in the isotype control Ab-treated group. Unexpectedly, administration of the isotype-matched control Ab did not result in any anti-BALB/c CD8+ T-cell inhibition, which suggested that the injected IgG interfered with these effects on CD8+ T-cells (Fig. 7A). CD8+ T-cells express Fc receptors for IgG (FcγR), but CD4+ T-cells do not, in response to certain stimuli (9); therefore, the Fc part of the injected IgG possibly binds FcγR on CD8+ T-cells, leading to interference with the LSEC-induced inhibitory effects on CD8+ T-cells. Thus, at least in alloreactive CD4+ T-cells, the PD-1/PD-L1 pathway could be involved in the suppressive effects of orthotopically engrafted allogeneic LSECs. Supporting this idea, immunohistochemical staining revealed that the engrafted allogeneic LSECs persistently expressed PD-L1 even 7 weeks after the inoculation (Fig. 7B).
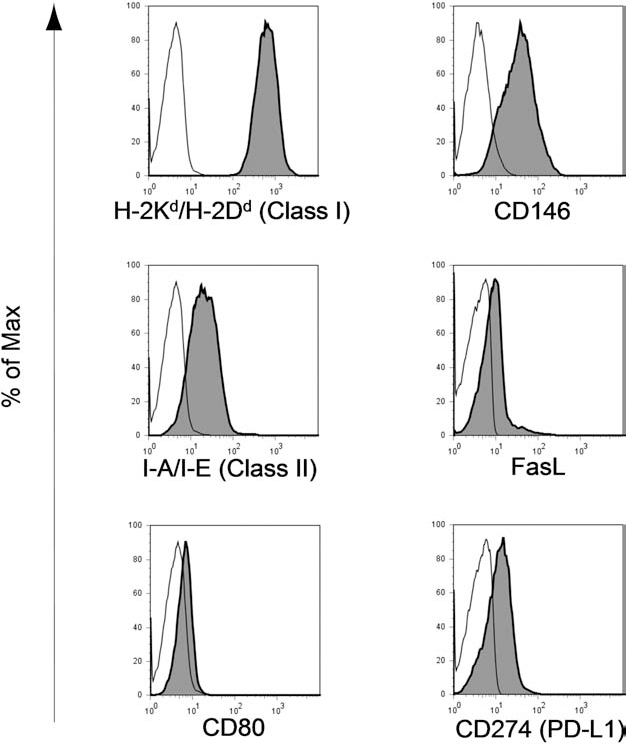
LSECs constitutively expressed Fas ligand (FasL) and programmed death ligand 1 (PD-L1), as well as molecules necessary for antigen presentation and CD146. The freshly isolated CD105+ LSECs from BALB/c mice were stained for H-2Kd/H-2Dd (MHC-class I), I-A/I-E (MHC-class II), CD80, CD146 (another LSEC specific marker), FasL, and CD274 (PD-L1). These cells were analyzed by FCM. Dead cells were excluded from the analysis by forward scatter and propidium iodide. Thin and filled lines represent staining with isotype-matched control Abs and mAbs for the indicated surface molecule, respectively. The percent of Max is the number of cells in each bin of the histograms divided by the number of cells in the bin that contains the largest number of cells.

Engraftment with FasL-deficient LSECs did not attenuate the inhibitory effect of LSECs on alloreactive T-cells. RAG2/gc-KO mice were injected with BALB/c (n = 4) or BALB/c-gld (n = 5) LSECs 2 days after MCT pretreatment. Six weeks after LSEC inoculation, B6 SPLs were injected into the recipient mice for immune reconstitution. One week after SPL inoculation, the recipient mice were sacrificed, and the SPLs were obtained, CFSE labeled, and cultured with irradiated SPLs from syngeneic B6, allogeneic BALB/c, or third-party SJL/j mice as stimulators. After 4.5 days of culture, the responder cells were analyzed by FCM. Using FCM analysis following MLR, reactive T-cell proliferation can be visualized as the serial halving of CFSE intensity. In the FCM analysis, CD4+ and CD8+ T-cells were gated and analyzed for CFSE fluorescence, and their stimulation indices were calculated. The average values of four or five independent mice in each group are shown. *p < 0.05.

Administering anti-PD-L1 mAb during immune reconstitution attenuated the inhibitory effect of LSECs on alloreactive CD4+ T-cells. (A) RAG2/gc-KO mice were injected with BALB/c LSECs 2 days after MCT pretreatment. Six weeks after LSEC inoculation, B6 SPLs were injected into the recipient mice for immune reconstitution. To block PD-L1 signaling in vivo, the recipient mice were pretreated with 500 μg of anti-PD-L1 blocking mAb (n = 5) or isotype-matched Ab (n = 3) just before SPL inoculation. These antibodies were readministered 3 days later. One week after SPL inoculation, the recipient RAG2/gc-KO mice were sacrificed, and then the SPLs were isolated, CFSE-labeled, and cultured with irradiated SPLs from syngeneic B6, allogeneic BALB/c, or third-party SJL/j mice as stimulators. After 4.5 days of culture, the responder cells were analyzed by FCM. By using FCM analysis after the MLR, the reactive T-cell proliferation can be visualized as the serial halving of CFSE intensity. In the FCM analysis, CD4+ and CD8+ T-cells were gated and analyzed for CFSE fluorescence, and their stimulation indices were calculated. The average values of three or five independent mice in each group are shown. *p < 0.05. (B) The engrafted LSECs persistently expressed PD-L1. RAG2/gc-KO mouse livers were obtained 7 weeks after LSEC inoculation and immunostained with FITC-conjugated anti-mouse H-2Kd/H-2Dd (left) and anti-CD274-PE mAbs (middle) to estimate PD-L1 expression on the transferred LSECs. The right panel shows the merged image of the left and middle panels.
Engraftment of Allogeneic LSECs Prolonged Survival of Subsequently Grafted Cognate Allogeneic Hearts in RAG2/gc-KO Mice Immune Reconstituted by BMT
It has been shown that the presence of a liver allograft can suppress rejection of other organ grafts from the same donor (1). To address the possibility that allogeneic LSECs play a role in such immunoregulatory effects on subsequently transplanted organs, allogeneic hearts from BALB/c mice were transplanted into immune reconstituted recipient RAG2/gc-KO mice in which allogeneic LSECs from BALB/c mice had been engrafted after MCT pretreatment. When heart allografts from BALB/c mice were transplanted to the recipients 1 week after B6 SPL inoculation for immune reconstitution, LSEC engraftment did not result in prolonged heart allograft survival (data not shown).
One possible explanation for the failure to prevent allograft heart rejection in mice immune reconstituted with SPLs is that extensive T-cell proliferation occurred after the adoptive transfer of mature T-cells in immune-deficient mice; this process, termed homeostatic proliferation, might lead to insusceptibility of those T-cells to LSEC-induced suppression. Consistent with this hypothesis, the proportion of CD44high+ memory phenotype T-cells in the peripheral blood of mice that received B6 SPLs was much higher than that of wild-type (WT) B6 control mice during the observed period (Fig. 8A). In order to prevent homeostatic proliferation of mature T-cells, RAG2/gc-KO mice with BALB/c LSECs were immune reconstituted by BMT from B6 mice. RAG2/gc-KO mice that received B6 BMCs displayed similar levels of CD44high+ memory T-cells and CD45RBhigh+/CD62Lhigh+ naive T-cells to those in WT B6 control mice. The proportion of NK1.1+ CD3- NK cells in the mice immune reconstituted using B6 BMCs fully resembled that in the WT B6 control mice, whereas the level in the mice immune reconstituted using B6 SPLs was markedly lower. In contrast, the proportion of NK1.1+ CD3+ NKT-cells in the mice immune reconstituted using B6 BMCs did not markedly differ from that in the mice immune reconstituted using B6 SPLs (Fig. 8B).

RAG2/gc-KO mice immune reconstituted with bone marrow cells (BMCs) showed a lower proportion of memory T-cells but a higher proportion of natural killer (NK) cells in their peripheral blood mononuclear cells (PBMCs) compared with the mice that were immune reconstituted with SPLs. B6 BMCs or SPLs were injected into RAG2/gc-KO mice through the tail vein. PBMCs from those RAG2/gc-KO or WT B6 control mice were analyzed by FCM. Anti-CD3, anti-CD4, anti-CD8, anti-CD44, anti-CD45RB, anti-CD62L, and anti-NK1.1 mAbs were used for surface staining of the PBMCs. (A) The proportions of CD44high memory phenotype, CD45RBhigh, and CD62Lhigh naive phenotype cells among each CD4+ and CD8+ T-cell fraction were determined after bone marrow transplantation (BMT) (filled circles) or SPL inoculation (open circles). The reconstituted CD4+ T-cells began to be detectable 4 weeks after BMT. (B) The proportions of NK1.1+ CD3- NK and NK1.1+ CD3+ NKT-cells among the PBMCs were determined after BMT (filled circles) or SPL inoculation (open circles). The mean values ± SEM of four independent mice in each group are shown. WT B6 mice were used as controls. The thin line indicates the mean and the dotted line indicates the SEM (n = 4). *p < 0.05, **p < 0.01, ***p < 0.001.
Six weeks after BMT, there was no statistically significant difference in immune reconstitution levels (i.e., the proportion of CD4+ and CD8+ T-cells in the PBMCs) between recipient RAG2/gc-KO mice with allogeneic LSECs and corresponding control RAG2/gc-KO mice without allogeneic LSECs (Fig. 9A). When heart allografts from BALB/c mice were transplanted to recipients 6 weeks after BMT, heart survival in mice with allogeneic BALB/c LSEC engraftment was significantly prolonged compared with that in control mice without LSEC engraftment (Fig. 9B). On the other hand, survival of heart grafts from third-party SJL/j mice was not prolonged at all in mice with BALB/c LSEC engraftment (data not shown). These findings suggest that allogeneic LSECs can prolong the survival of subsequently grafted cognate allogeneic hearts.

Transfer of allogeneic LSECs through the portal vein leads to prolonged survival of subsequently transplanted donor-type heart allografts in recipient mice immune reconstituted by BMT. (A) RAG2/gc-KO mice were reconstituted by B6 BMT. The RAG2/gc-KO mice were injected with BALB/c LSECs 2 days after the MCT pretreatment. Two weeks after the LSEC inoculation, B6 BMCs were injected into the recipient mice for immune reconstitution. Six weeks after BMT, PBMCs were obtained from the recipient mice, and the immune reconstitution level was evaluated by FCM. Anti-CD4 and anti-CD8 mAbs were used for the PBMC surface staining. In the FCM analyses, the percentages of CD4+ or CD8+ T-cells among PBMCs were measured in immune reconstituted RAG2/gc-KO mice with (n = 8) or without (n = 6) allogeneic LSEC inoculation. The frequencies of each indicated population among total number of obtained cells are shown. (B) Heterotopic heart transplantation after immune reconstitution by BMT in recipient RAG2/gc-KO mice with or without allogeneic BALB/c LSECs transfer. Heart allografts from BALB/c mice were subsequently transplanted into the recipient mice 6 weeks after BMT. Heart graft survival curves are shown. There is a significant difference (**p < 0.01) in heart allograft survival between recipient RAG2/gc-KO mice with (solid line, n = 16) or without (dotted line, n = 8) allogeneic LSEC transfer.
Discussion
LSECs have been described as a new type of APC that induces immune tolerance in naive T-cells (4,11,12,16). Following priming by antigen-presenting LSECs, CD4+ T-cells fail to subsequently differentiate into the T helper 1 phenotype; instead, they differentiate into regulatory T-cells that express interleukin (IL)-4 and IL-10 upon restimulation (13,14). LSECs also have the capacity to present exogenous antigens on MHC class I molecules to CD8+ T-cells, a process termed cross-presentation (16). Initially, stimulation of naive CD8+ T-cells by LSECs results in T-cell proliferation and cytokine release. However, it ultimately leads to antigen-specific tolerance, as demonstrated by the simultaneous loss of cytokine expression and the failure of CD8+ T-cells to develop into cytotoxic effector T-cells.
We previously demonstrated similar immunoregulatory effects of LSECs on T-cells with direct allospecificity beyond MHC barriers using an in vitro mixed LSEC-lymphocyte coculture model (23). In that model, cell–cell contact was necessary to induce the inhibitory effects of LSECs on alloreactive T-cell proliferation. In vivo, the cumulative surface area of LSECs is very large, and hepatic microcirculatory parameters allow frequent contact between LSECs and passenger leukocytes. Considering the large volume of blood that passes through the liver daily, it is probable that LSECs are ideally positioned within the liver to regulate alloimmune responses. By using this anatomical advantage of LSECs, we investigated the immunoregulatory effects of LSECs on alloreactive T-cells in an in vivo model in which exogenously inoculated allogeneic LSECs were engrafted orthotopically on the liver sinusoidal endothelium. MCT, which induces persistent cell cycle arrest through the G2/M block in endothelial cells (24,29), was used in the present study to promote LSEC engraftment. MCT does not affect hepatocytes in this manner unless hepatic injury is redirected by additional drug toxicities or accumulation of toxic MCT intermediates through activation of cytochrome P450-induced metabolism MCT in hepatocytes (30). By this mechanism, MCT impairs only the host LSECs and confers a proliferative advantage to the transferred LSECs. Consistently, MCT administration significantly enhanced orthotopic engraftment of allogeneic LSECs (Fig. 1C). In this MCT-pretreated mouse model, allogeneic LSEC engraftment suppressed CD4+ and CD8+ T-cell proliferation in response to allostimulation by cells from mice cognate with LSEC donors.
The possible mechanisms for LSEC-induced suppressive immune regulation specifically on allogeneic T-cells might be associated with the death-inducing molecules that are constitutively expressed on LSECs (e.g., FasL, PD-L1). Morita et al. recently demonstrated the critical role of the PD-1/PD-L1 pathway in establishing immunological spontaneous tolerance status in mouse liver allografts (18). They found that PD-L1 was highly expressed on donor-derived tissue cells and is associated with infiltrating T-cell apoptosis in the allografts, and that blockade of this pathway leads to severe acute rejection.
Although the cell type that contributes to PD-L1-induced negative regulation of T-cells in liver allografts remains to be identified, both their results and ours suggest that the signal between PD-1 on allogeneic T-cells and PD-L1 on LSECs likely inhibits corresponding T-cell function. The results of our previous in vitro study suggested that the Fas/FasL pathway also would be involved in LSEC- induced suppression of allogeneic T-cells (23). The lack of a significant role of FasL in LSECs could be explained by a limitation of allogeneic LSEC engraftment, since allogeneic LSECs have never replaced most native LSECs in an in vivo model. Alternatively, even when we used FasL KO mice as LSEC donors in the adoptive transfer to the allogeneic WT recipient mice, we could not rule out the possibility of bidirectional transfer of MHC class II molecules between the WT recipient LSECs and the FasL-mutant donor LSECs after their inoculation. On the basis of the previous demonstration that T-cells can be positive for both donor and recipient MHC class II by intercellular transfer of MHC molecules and play a significant role in the antigen presentation (3), it is possible that recipient LSECs expressing FasL acquire donor MHC molecules, which might complicate the interpretation of this study. In contrast, when we used antibodies blocking PD-L1, they were able to block PD-L1 on both the donor and recipient LSECs, even if MHC transfer occurred. From such a standpoint, instead of the use of FasL KO mice, the use of anti-FasL-blocking mAb with/without anti-PD-L1-blocking mAb in the LSEC-chimeric mouse model might provide additional valuable information. Thus, to address the precise role of FasL and PD-L1 on LSECs in their inhibitory effects on alloreactive T-cells, further studies are required.
In addition to the death-inducing molecules, regulatory T-cells (Tregs) might also play a role in LSEC-induced suppression of allogeneic T-cells. It has been demonstrated that LSECs prime CD4+ T-cells to a CD45RBlow memory phenotype lacking marker cytokine production for effector cells, and that those T-cells functionally belong to the CD25low FoxP3- Tregs (forkhead box P3 negative regulatory T-cell) family (15). Those LSEC-primed Tregs are thought to contribute to shifting antigen-dependent immune responses to tolerance toward exogenous antigens or endogenous self-antigens. A similar mechanism might be involved in the immunosuppressive effects of LSECs toward the alloreactive T-cells observed in the present study.
Engraftment of allogeneic LSECs prolonged the survival of subsequently grafted cognate allogeneic hearts in RAG2/gc-KO mice that were immune reconstituted by BMT (Fig. 9B) but not in RAG2/gc-KO mice that were immune reconstituted using SPLs. This difference could be explained by the homeostatic proliferation of inoculated splenic T-cells, which might lead to insusceptibility of those T-cells to LSEC-induced suppression. It has been reported that lymphopenia induces rapid expansion of T-cells, conversion from a naive to a memory-like phenotype, and development of effector functions (8,17). This phenomenon is thought to have an adverse effect on organ allograft tolerance induction in mouse models (19,31). An alternative explanation might be the insufficiency of either NK or NKT cells that have the capacity to release immunoregulatory cytokines, such as IL-10, in mice immune reconstituted with syngeneic SPLs. NK cells were supplied in the mice that were immune reconstituted by BMT (Fig. 8B) and might provide factors that induce the immunosuppressive features of the LSECs. It has been reported that host NK cells destroyed graft-derived APCs in a skin allograft model (33). In the absence of NK cells, donor APCs can survive and then migrate to host lymphoid and extralymphoid sites, where they directly stimulate the activation of alloreactive T-cells. T-cells activated in the absence of NK cells are possibly more resistant to LSEC-induced immunosuppression, and under such conditions, the prolongation of heart allograft survival might be difficult to achieve.
Nevertheless, only T-cells with direct allospecificity (i.e., those that recognize alloantigens via the direct pathway) would be suppressed by direct exposure to orthotopically engrafted allogeneic LSECs; T-cells with indirect allospecificity would theoretically not be influenced in the present in vivo model. This could be why most of the allogeneic hearts were eventually rejected even in LSEC-chimeric mice immune reconstituted by BMT, in contrast to the in vitro MLR assay results. Further studies are required to confirm this hypothesis.
In conclusion, allogeneic LSECs are capable of suppressing T-cells with specificity cognate to the LSECs in an in vivo murine model. The PD-1/PD-L1 pathway is likely involved in these suppressive effects.
Footnotes
Acknowledgments
This work was supported by Grants-in-Aid for Young Scientists (Start-up) (2189015500) from the Japan Society for the Promotion of Science. The authors declare no conflict of interest.
