Abstract
Introduction
The development of programmed cell death protein 1 (PD-1) and programmed cell death ligand 1 (PD-L1) monoclonal antibodies, immune checkpoint inhibitors (ICIs) had been rough and challenging. With the continuous breakthrough of basic medical disciplines in this field, cancer immunotherapy has shown excellent efficiency in the treatment of a variety of malignancies, including but not limited to malignant melanoma, non-small cell lung cancer and lymphoma. Clinical trials and applications for other types of cancer are booming. With the increasing clinical applications of ICIs, immune-related adverse events (irAEs) have gradually attracted the attention of clinicians.
irAEs are characterized by excessively activated immune system leading to the off-target toxicities. Currently, both rare and common irAEs could be observed with the increasing treatment indications in different kinds of tumors, and the irAEs could further influence other organs. 1 Based on previous reports that any organs or systems could be affected by irAEs, such as endocrine, skin, ocular, hematological, neurological, and cardiovascular systems. The following factors could influence the frequency of irAEs, such as patient’s intrinsic risk factors, administration dose, and exposure time. 2 It was reported that irAEs influenced 17.1% of tumor patients receiving anti-tumor treatment including anti-PD-L1, anti-PD-1, and anti-CTLA-4.3–5 In this study, adverse events (AEs) of PD-1/PD-L1 inhibitors during the treatment of cancers was analyzed.
Methods and materials
Materials
Study subjects
Two hundred and forty patients (160 males and 80 females, age ranged from 19 to 86 years old) received PD-1/PD-L1 inhibitor therapy for different cancers at the Department of Oncology, Fujian Provincial Hospital from January 2020 to August 2021 were included in this study. There were 133 cases of lung cancer, 50 cases of urothelial carcinoma, 15 cases of gastric cancer, 14 cases of intestinal cancer, 10 cases of breast cancer, and 18 cases of other cancers. All patients received 547 cycles, with an average of 2.28 cycle per patient. The study has been approved by the Institutional Review Board of Shengli Clinical Medical College of Fujian Medical University, Fujian Provincial Hospital (Ethical batch number:K2021-07–041), and all experiments were performed in accordance with the guidelines. Written informed consent was obtained from all subjects before the study. The inclusion criteria were listed below as follows : patients diagnosed with tumor by pathological method and received PD-1/PD-L1 inhibitor therapy. The exclusion criteria were listed below: patients with disturbance of consciousness and mental illness.
Major inhibitors
Among the patients, there were 15 cases treated with nivolumab (3 mg/kg, every 2 weeks), 15 cases treated with pembrolizumab (2 mg/kg or 200 mg, every 3 weeks), 41 cases treated with sintilimab (200 mg, every 3 weeks), 40 cases treated with toripalimab (3 mg/kg, every 2 weeks), 72 cases treated with camrelizumab (200 mg, every 3 weeks), 44 cases treated with tislelizumab (200 mg, every 3 weeks), 5 cases treated with atezolizumab (1200 mg, every 3 weeks), and 8 cases treated with durvalumab (1500 mg, every 4 weeks, or 10 mg/kg, every 2 weeks).
Methods
Treatment plan All the 240 patients had advanced cancers
The patients and their family members were informed of the advantages/disadvantages and possible risks of using PD-1/PD-L1 inhibitors
Observation and evaluation of the AEs
The AEs related to PD-1/PD-L1 during the treatment were observed and evaluated based on the regulations in Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, US National Cancer Institute. The following aspects were evaluated: the categories of AEs, grades, onset timepoint and duration, management plan, prognosis, etc. We established an immune-related toxicity team of oncology and medical subspecialists to support multidisciplinary irAE diagnosis and management, and this team was composed of doctors from corresponding departments. The median duration of follow-up was 23 months. All patients received combination treatment of ICI and chemotherapy.
Statistic analysis
SPSS 25.0 software was used to perform the statistical analysis of relevant data. Enumeration data collected in the study were analyzed by chi-square test. p < 0.05 indicated that the difference was statistically significant.
Results
General data
The study included 240 patients received PD-1/PD-L1 inhibitor therapy, which was consisted of 160 male patient (82.5%) and 80 female patients (17.5%). The age of patients ranged from 19 to 86 years old, with a median of 58 years old.
Occurrence of AEs
Prevalence of adverse effects (AEs) in 240 patients treated with PD-1/PD-L1 antibody inhibitors (cases, %).
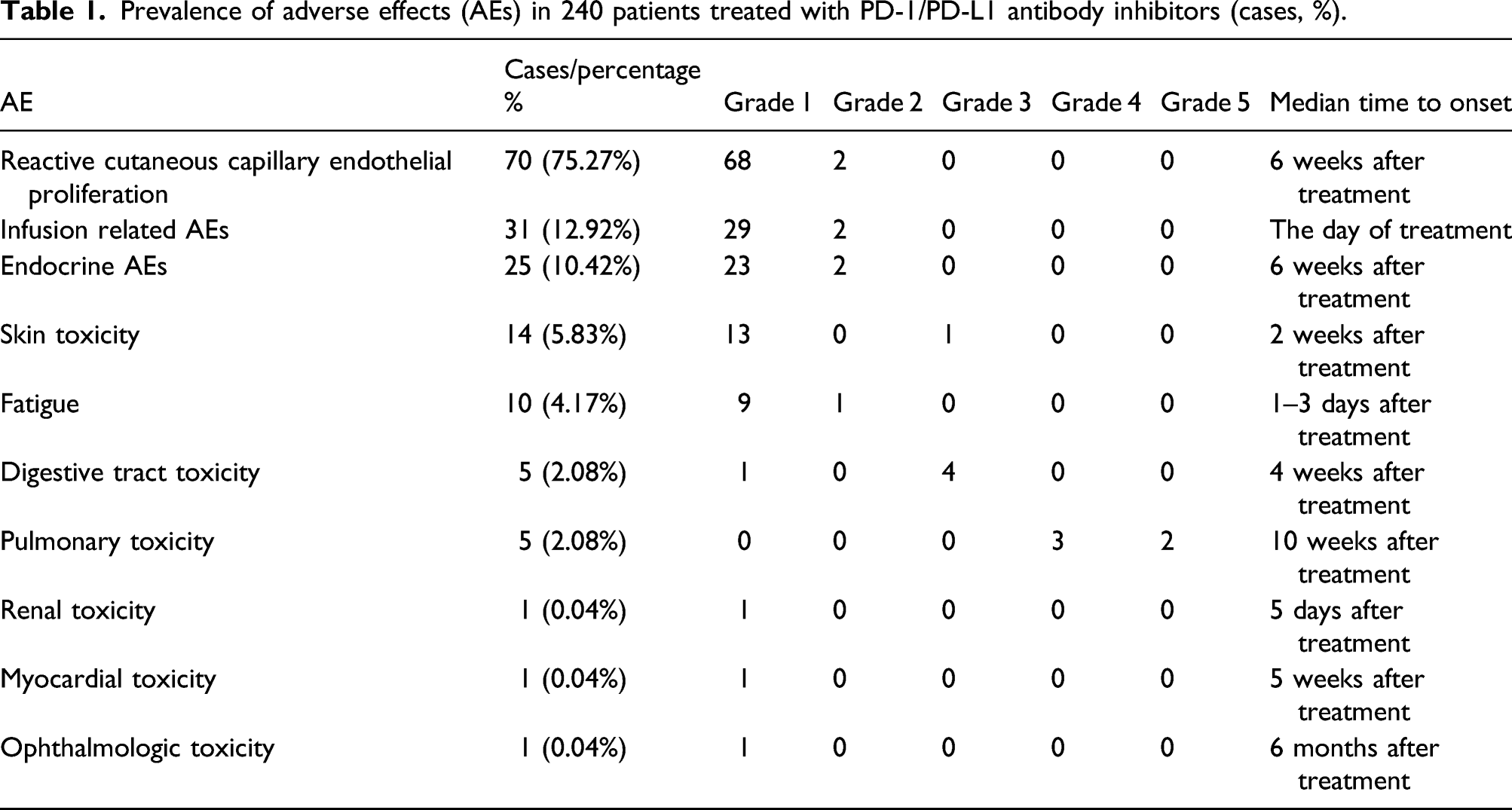
Serious adverse effects in patients treated with PD-1/PD-L1 antibody inhibitors.

AEs: adverse effects
Infusion related AEs
There were 31 cases of infusion related AEs with the prevalence rate of 12.92%. Among them, there were eight cases of fever, four cases of dizziness, four cases of arthralgia/myalgia, two cases of sweating, two cases of urticaria and pruritus, three cases of hypertension, three cases of shortness of breath, three cases of cough/wheezing, one case of angioedema, and one case of flushing/headache. Twenty-two patients experienced infusion related AEs after the first cycle with PD-1/PD-L1 monoclonal therapy, and in nine patients (29.03%) at the second cycle. All infusion related AE occurred on the day of treatment. After adjustment of infusion speed or withheld of infusion and symptomatic management, patients’ symptoms were spontaneously relieved. The infusion of PD-1/PD-L1 was continued later.
Endocrine AEs
There were 25 cases of endocrine AEs with the prevalence rate of 10.24%. Among them, there were 12 cases of hyperglycemia/diabetes mellitus, 11 cases of thyroid disease, and 2 cases of hypophysis. Among patients with hyperglycemic/diabetic AE, 58.3% (7/12) occurred after 3–7 days, 25% (3/12) after 1 month, and 16.7% (2/12) after 7–8 months of treatment. 66.7% (8/12) were transient hyperglycemia, which did not require special management, but occurred in multiple cycles, and 33.3% (4/12) were persistent hyperglycemia, which could be controlled within normal after oral hypoglycemic drug therapy. AE of thyroid disease occurred from 2 to 21 weeks, with an average of 6 weeks. Most patients have no symptoms associated with abnormal thyroid function. For patients with severe symptoms, relevant indicators can return to normal after proper drug therapies. Thyroid dysfunction: The most common is hyperthyroidism/hypothyroidism. The thyroid function test should be monitored. Related drugs therapy should be administered according to specific indicators when thyroid function is abnormal. Two cases developed hypophysis, which was improved after oral hydrocortisone treatment.
Skin toxicity
There were 14 cases of skin toxicities with the prevalence rate of 5.83%. Patients with rashes had a better response to drug. General rash refers to relatively mild papules and macules. If there is local rash and pruritis, glucocorticoid ointment or oral glucocorticoids were the proper choices. For patients with severe symptoms, referral to dermatologists were required. In this group, 5 cases of AEs occurred on the day of treatment, 5 cases after 2 weeks of treatment, and 4 cases after 6 weeks of treatment. Thirteen cases were grade 1 and 2 skin AEs with short duration and relieved within 1–3 days. One case of grade 3 skin AE was improved after once oral methylprednisolone 1 mg/kg. There was no grade 4 skin AE.
Fatigue
There were 10 cases of fatigues with the prevalence rate of 4.17%. The main manifestations were fatigue and weakness, which will relieve on their own in 3–5 days. If the fatigue persists or aggravated, medical attention was required. Fifty percentage (5/10) of the patients had repeated recurrence during following cycles. Fatigue was relieved without special treatment 1–3 days after infusion.
Gastrointestinal toxicity
There were 5 cases of gastrointestinal toxicities with the prevalence rate of 2.08%. Among them, there was 1 case of autoimmune colitis and 4 cases of autoimmune hepatotoxicity, and 60% (3/5) of the patients with hepatotoxicity were diagnosed with liver cancer or liver metastatic cancer. In 3 patients with grade 3 irAE hepatotoxicity, the PD-1/PD-L1 inhibitors were discontinued immediately. 3 days of liver protection therapy and methylprednisolone (2 mg/kg, IV) did not improve the liver function. Mycophenolate mofetil and gamma globulin (5g, every day) were added into the management. The liver function indicators improved after 15 days of treatment. Glucocorticoid dosage was reduced according to the standard. When the glucocorticoids were fully stopped, liver function indicators were tested and PD-1/PD-L1 inhibitors were terminated after it was confirmed that the dosage had returned to normal level. For the other patients with grade 1 irAEs, their liver functions were improved by routine liver protection therapy. The patient with autoimmune colitis presented with hemorrhagic colitis, which was characterized by multiple diffuse bleeding under colonoscopy and a large amount of T cell infiltration was observed pathologically. This case of irAE was determined to be grade 3. The bleeding stopped after the first day of intravenous methylprednisolone 2 mg/kg, and the patient was cured after 3 weeks of treatment.
Pulmonary toxicity
There were 5 cases of pulmonary toxicities with the prevalence rate of 2.08%. Autoimmune pneumonia: the most common symptoms were different levels of dyspnea, a new onset or worsening cough, sputum, chest pain, and infection caused by pathogens. The onset of pulmonary events was 3–21 weeks after the treatment. Four cases occurred within 10 weeks and one at the 21st weeks. For two patients had grade 5 irAEs, one of them was a lung cancer patient who developed grade 2 autoimmune pneumonia 2 months after the first cycle of durvalumab. This patient was treated with methylprednisolone 2 mg/kg intravenously per day for 7 days. The pneumonia was improved down to grade 1. During the process of dosage decreasing, the autoimmune pneumonia recurred and upgraded to a grade 3 irAE after re-evaluation. After 2 months of treatment (methylprednisolone, 2 mg/kg, immunoglobulin, stuzumab, infliximab, anti-infection, non-invasive ventilator assisted ventilation, etc.), the patient still died due to multiple organ failure caused by pneumonia. Another lung cancer patient developed grade 3 autoimmune pneumonia 2 months after the use of sintilimab due to delayed treatment and diagnosis. The patient died of respiratory failure after intensive treatment. The total mortality rate of pulmonary event was 0.83%.
Renal toxicity
There was one case of renal toxicity with the prevalence rate of 0.04%. One case had renal dysfunction during hospitalization, the patient’s renal function was improved after the administration of nephroprotective drugs to limit renal damage.
Myocardial toxicity
There was one case of myocardial toxicity with the prevalence rate of 0.04%.
Ocular toxicity
There was one case of ocular toxicity with the prevalence rate of 0.04%. The symptoms occurred 6 months after treatment and relieved with oral methylprednisolone, 0.5 mg/kg.
Reactive cutaneous capillary endothelial proliferation(RCCEP)
There were 70 cases of RCCEPs with the prevalence rate of 75.27%. All patients presented with RCCEP were treated with camrelizumab. RCCEP was induced from the side effects caused by the off-target effect unique to camrelizumab, which are generally manifested as skin capillary hyperplasia. All cases were evaluated as grade 1 and 2 AEs.
The relationship between baseline data and AEs
Factors of adverse effects.
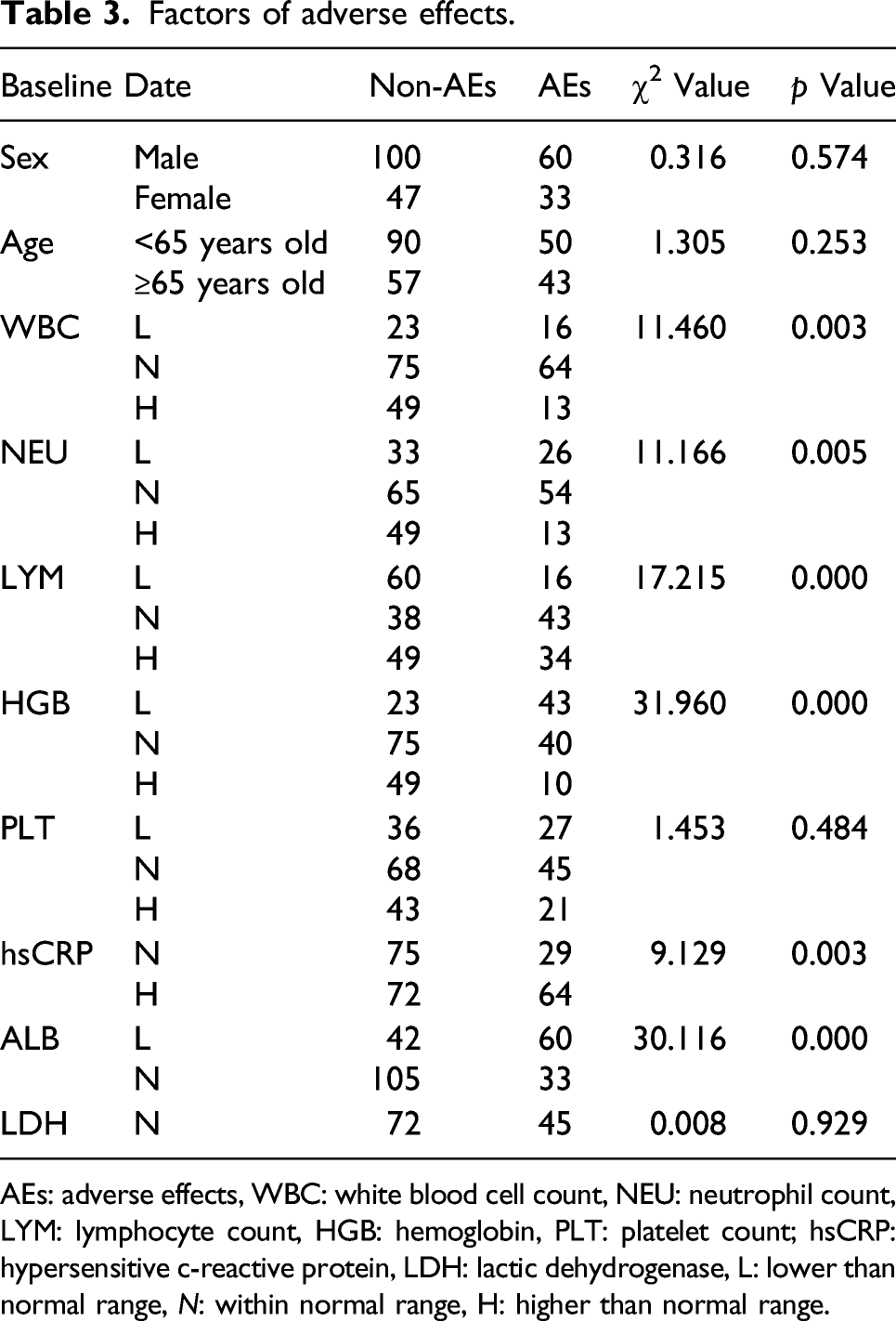
AEs: adverse effects, WBC: white blood cell count, NEU: neutrophil count, LYM: lymphocyte count, HGB: hemoglobin, PLT: platelet count; hsCRP: hypersensitive c-reactive protein, LDH: lactic dehydrogenase, L: lower than normal range, N: within normal range, H: higher than normal range.
Discussion
ICIs is a class of drugs that stand out in the treatment of malignancies. Different from traditional chemoradiotherapy, ICIs acts on the effector T cells of the immune system and breaks through immune tolerance and immune escape by restoring their specific recognition and specific killing function of tumor cells, compared with the direct killing of tumor. 6 With breakthroughs in ICIs and the update of immunotherapy in the guidelines as the first line treatment of various malignancies, the suitable patients for ICIs have grown rapidly. In the increasing clinical applications, the toxicity characteristics of immunotherapy have been paid more attention by oncologists. Different pharmacological mechanisms also bring different AEs. Therefore, clinicians need a profound understand of the toxicities of ICIs and manage the AEs of ICI as early as possible to achieve better therapeutic effects.
ICIs achieve anti-tumor effects by adjustment of humoral immunity. The mechanism is to cut off the negative regulation of T cells and remove the immune suppression caused by tumor cells. However, the abnormally enhanced immune response may destroy the immune tolerance balance, and the affected normal tissues will have an autoimmune inflammatory response, known as irAEs.7–9 Different irAEs occurred in different ICIs signaling pathways. The prevalence of irAEs in PD-1 and PD-L1 inhibitors was about the same, 64% and 66%, respectively. 10 The prevalence and severity of irAEs in CTLA-4 inhibitors were significantly higher than those in PD-1/PD-L1 inhibitors. 11 Common irAEs for PD-1 inhibitors include pneumonia, myalgia, hypothyroidism, hypothyroidism, arthralgia, vitiligo, etc. ICIs therapies may cause a series of irAEs, which may affect eye, respiratory tract, cardiovascular system, digestive tract, liver, endocrine, reproduction, skin, nerve, hematology, skeletal muscle, and other systems. 12 Most irAEs are mild to moderate in severity, and occasionally severe, even life-threatening. 13
IrAEs occur at different times for different ICIs and most of them are reversible. Relatively common AEs involve skin (34–45%), endocrine (5–10%), liver (5%), gastrointestinal (<19%)), lung (5–53%), rheumatoid/skeletal muscle (15%), and infusion related AEs (10%). 13 Cardiovascular, hematologic, renal, nerve, and eye AEs may occur, but AEs of nerve (6.1%), hematologic, kidney (<5%), heart (<1%), and eye (<1%) are relatively rare. 13 Eight brands of PD-1/PD-L1 inhibitors used in this study showed that the overall prevalence of irAEs was 58–94.1%. The prevalence of grade 3 and above AEs was 7.0–30.6% (referring to instruction of various drugs). In this group, the prevalence of irAEs was 38.75%, 83 cases (34.58%) of grade 1 and 2 AEs, 10 cases (4.17%) of grade 3 and above AEs. The mortality rate of grade 5 AE was 0.83%. The low prevalence and severity of irAEs may be related to the insufficient observation of grade 1 and 2 AEs during early application of treatment. In this group of patients, the very common (≥10%) of AEs included RCCEP, with an prevalence of 75.27%, and all patients were treated with camrelizumab. RCCEP was induced from the side effects caused by the off-target effect. The prevalence rate of infusion related AE was 12.92%. The prevalence rate of endocrine toxicity was 10.42%. Common (1–10%) AEs include: skin toxicity (5.83%), fatigue (4.17%), gastrointestinal toxicity (2.08%), pulmonary toxicity (2.08%), and renal, myocardial, and ocular toxicity was 1 case each, with the prevalence of 0.04%. Neurotoxicity was not observed in this group. In this group of patients, the prevalence of infusion related reactions is high and can occur repeatedly. Although the reactions are slight, they still need to be paid attention and carefully observed. Hepatotoxicity was more common in gastrointestinal toxicity, because a considerable part of the patients in this group were liver cancer and liver metastatic cancer patients, which may be related to the patients’ poor liver function. It has been reported that in the study of PD-1/PD-L1 treatment for lung cancer, 5% (16/319) patients developed multi-system irAEs, 14 patients developed dual-system irAEs, and two patients developed triple-system irAEs. Further study indicated that patients with pulmonary inflammatory response, dermatitis and hypothyroidism were most likely to suffer from multi-system irAEs, which is of great significance for interdisciplinary patient management in the era of cancer immunotherapy. 14 IrAEs of immunotherapy have the characteristics of delayed onset and long-term lasting, which are not possessed by traditional chemoradiotherapy. Physicians should note during follow-up that irAEs of ICIs may occur within 1 year of the initiation of PD-1/PD-L1 inhibitor therapy, which is the delayed effect of ICIs. In previous studies, irAEs will appear in sequence, presenting a typical time distribution pattern. The occurrence time of irAEs varies in different studies. Eigentler et al. 15 showed that irAEs usually occurred at different times after administration, generally within 1–6 months. The onset of various irAEs is different and the time ranges, among which the recovery time of endocrine toxicity is the longest. Studies also show that most of irAEs are reversible. Weber et al.16,17 showed that skin AEs appeared first (in 3 weeks), followed by gastrointestinal AEs (in 5–10 weeks), and endocrine disease and hepatic related AEs occurred last (9–12 weeks). The onset of AEs in this group was basically consistent with the study above.
The management principle of PD-1/PD-L1 AEs was stratified treatment. Grade 1 toxicity may be closely monitored with continued application of PD-1/PD-L1. Grade 2 requires withheld of PD-1/PD-L1 until symptoms and/or laboratory indicators return to grade 1 or lower, during which time the use of topical or systemic oral glucocorticoid therapy of 0.5–1 mg/kg/d may be considered as appropriate. Grade 3 requires the withheld of PD-1/PD-L1 and the initiation of systemic oral or intravenous methylperone 1–2 mg/kg/d. If AEs are not relieved 3–5 days after glucocorticoid therapy, the use of PD-1/PD-L1 should be discontinued under the guidance of oncologists. For patients with AE remission, the restart of immunotherapy should be discussed based on the patient-based risk/benefit ratio. Grade 4 requires permanent discontinuation of PD-1/PD-L1, intravenous methylprednisolone 1–2 mg/kg/d for three consecutive days, and gradually reduced to 1 mg/kg/d. If symptoms fail to alleviate after 3–5 days of glucocorticoid therapy, the oncologist is required to make further treatment plan according to the actual situation after intervention. The clinical symptoms of grade 1 and 2 AEs are mild, and most patients can return to normal after symptomatic treatment or oral glucocorticoid therapy. Grade 3 and above AEs had more severe clinical symptoms and higher risks. In this group of patients, there were 10 cases (4.17%) of AEs higher than grade 3, grade 3 AEs: 3 cases of hepatic toxicity, 1 case of colitis, 1 case of skin toxicity; grade 4 AEs: 3 cases of pneumonia; grade 5 AEs: 2 cases of pneumonia. One patient was a lung cancer patient who developed grade 2 autoimmune pneumonia 2 months after the first cycle of durvalumab. This patient was treated with methylprednisolone 2 mg/kg intravenously per day for 7 days. The pneumonia was improved down to grade 1. During the process of dosage decreasing, the autoimmune pneumonia recurred and upgraded to a grade 3 irAE after re-evaluation. After 2 months of treatment (methylprednisolone, 2 mg/kg, immunoglobulin, stuzumab, infliximab, anti-infection, non-invasive ventilator assisted ventilation, etc.), the patient still died due to multiple organ failure caused by pneumonia. Another lung cancer patient developed grade 3 autoimmune pneumonia 2 months after the use of sintilimab due to delayed treatment and diagnosis. The patient died of respiratory failure after intensive treatment. The total mortality rate of pulmonary event was 0.83%. Among the above 10 cases of grade 3 or above, 8 cases of AE were completely alleviated after glucocorticoid impulse therapy, maintenance therapy and symptomatic therapy, among which 6 cases continued to receive PD-1 monoclonal antibody inhibitor, and the rest 2 cases were discontinued. The two patients with grade 5 irAEs (autoimmune pneumonia) were lung cancer patients. One case was complicated with chronic obstructive pneumonia and had poor pulmonary function. The other case had a history of pulmonary radiotherapy before ICI therapy and died of systemic multi-organ failure due to delayed monitor of autoimmune pneumonia.
The overall response rate of PD-1/PD-L1 immunotherapy is low, and it has become a promising method for the treatment of malignant tumors. IrAEs of ICIs limit and affect clinical application, and PD-1 induced irAEs are general and complex. IrAEs occur at different time points, generally within 6 months. In addition to the long-term and chronic characteristics of AE in endocrine organs, most OF irAE has good tolerance and reversibility, which is tolerable and reversible. Although the prevalence of severe AE is relatively low and rare, the severity of AEs is significant with serious consequences. In severe AEs, pericarditis, pulmonary and neurological AEs are of particular concern. Therefore, clinicians should not only be aware of the anti-tumor efficacy, but also pay attention to the occurrence of AEs. For clinicians in different practice environments, especially for oncologists, the ability of diagnosing and grading all types of irAEs is required. Early control the progress of AEs is essential to complete the treatment of PD-1/PD-L1 inhibitors and ensure the safety of therapy, to achieve the treatment goal and prolong the overall survival of patients. Most irAEs can be reversed without interfering the therapeutic efficiency and can be controlled within a reasonable range by withheld and/or rational use of corticosteroids. Therefore, early diagnosis and treatment are the most important in irAEs management. At present, the research on irAEs predictors is not enough. Through statistical analysis, there was a relationship between irAEs and white blood cell count, neutrophil count, hemoglobin count, level of hypersensitive CRP, and level of LDH, which was in line with previous study. However, the irAEs were not correlated with the age, sex, and platelets count, which were not in line with previous studies. No confirmed index that related to irAEs was found by former clinical retrospective studies. More large-sample, multicenter, prospective clinical studies are needed to provide solid evidence. Genetic irAEs predictors, such as carrying PTEN gene, etc. are worth further study in the future.
Some reports indicated the potential negative effect of concomitant medications with ICIs on the final outcomes of patients. 18 It was reported that pre-treatment with antibiotics before ICIs therapy could exerted negative influences including shortening of over survival and progression-free survival due to the dysfunction of gut. 19 However, the co-medication with proton pump inhibitors suggested lower occurrence of irAEs. irAE occurred in 33% of patients receiving proton pump inhibitors, while in 50% of patients without receiving proton pump inhibitors. 20 In addition, in the present study, high level of body mass index was related with higher risk of irAEs.
There are some limitations in this study. In this study, only one hospital and 240 patients were enrolled, which might influence the accuracy of conclusion. In addition, a key limitation for optimal clinical characterization of irAEs is the use of CTCAE as these criteria can be difficult to apply and do not allow accurate reporting of the severity and effect of some irAEs, such as rheumatic or systemic irAE. Finally, we were unable to separate out the specific toxicity related to immunotherapy or related to chemotherapy (for example, digestive or hepatic). ICI may alter the presentation of AEs typically seen with traditional cancer therapies and may produce challenges in identifying the etiology. The strength of this study is that we investigated multi-system of irAEs.
Conclusion
In clinical scenarios, the irAEs of PD-1/PD-L1 inhibitors may involve multiple organ systems of patients. The onset time points and duration of different AEs varies. Most of the AEs can be controlled and even reversed by discontinuation of ICIs and administration of corticosteroids. Among patients with malignancies, lung cancer patients who use ICIs are at a higher risk of autoimmune pneumonia, especially those with a history of radiotherapy, chronic obstructive pneumonia, or impairment of pulmonary function. The occurrence of autoimmune pneumonia and other AEs should be closely monitored in during and after immune therapy. The risk of autoimmune hepatitis is also increased for patient with liver cancer and metastatic malignant tumor of the liver.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study has been approved by the Institutional Review Board of Shengli Clinical Medical College of Fujian Medical University, Fujian Provincial Hospital (Ethical batch number: K2021-07-041), and all experiments were performed in accordance with the guidelines.
Informed consent
Written informed consent was obtained from all subjects before the study.
