Abstract
Colorectal anastomotic leakage is one of the most feared and fatal complications of colorectal surgery. To date, no external coating material that can prevent anastomotic leakage has been developed. As myoblasts possess anti-inflammatory capacity and improve wound healing, we developed a multilayered human skeletal muscle myoblast (HSMM) sheet by periodic exposure to supraphysiological hydrostatic pressure during repeated cell seeding. We assessed whether the application of an HSMM sheet can promote the healing process after colonic anastomosis. Partial colectomy and insufficient suturing were employed to create a high-risk colo-colonic anastomosis model in 60 nude rats. Rats were divided into a control group (n = 30) and an HSMM sheet group (n = 30). Macroscopic findings, anastomotic bursting pressure, and histology at the colonic anastomotic site were evaluated on postoperative day (POD) 3, 5, 7, 14, and 28. The application of an HSMM sheet significantly suppressed abscess formation at the anastomotic site compared to the control group on POD3 and 5. The anastomotic bursting pressure in the HSMM sheet group was higher than that in the control group on POD3 and 5. Inflammatory cell infiltration in the HSMM sheet group was significantly suppressed compared to that in the control group throughout the time course. Collagen deposition in the HSMM sheet group on POD3 was significantly abundant compared to that in the control group. Regeneration of the mucosa at the colonic anastomotic site was promoted in the HSMM sheet group compared to that in the control group on POD14 and 28. Immunohistochemical analysis demonstrated that surviving cells in the HSMM sheet gradually decreased with postoperative time and none were detected on POD14. These results suggest that the application of a multilayered HSMM sheet may prevent postoperative colonic anastomotic leakage.
Introduction
In the United States, colorectal cancer is one of the most common causes of cancer death 1 . Surgical resection is the primary form of treatment for colorectal cancer. The most serious complication of colorectal surgery is colorectal anastomotic leakage. The incidence of anastomotic leakage in colorectal surgery is reported to range from 3% to 28% 2,3 . Anastomotic leakage is represented as a transmural defect of the intestinal wall at the anastomotic site that causes intraperitoneal leakage of contaminated bowel contents, resulting in infection, peritonitis, or abscess formation. Symptomatic anastomotic leakage leads to re-operation, prolonged hospital stay, and a poor oncological outcome 4,5 . In addition, one-third of all post-operative deaths were caused by anastomotic leakage 3,6 . Risk factors of anastomotic leakage are multifactorial, including restricted anastomotic blood supply, wound contamination, and uncontrolled inflammation 2,7 . Despite many years of research to solve these problems, no significant reduction in the incidence of colorectal anastomotic leakage has been achieved.
To prevent anastomotic leakage, a diverting stoma is often constructed after colorectal surgery to divert contaminated bowel contents away from the anastomotic site. Previous reports showed that, although diverting stomas did not reduce the incidence of symptomatic colorectal anastomotic leakage, they reduced the incidence of reoperation for anastomotic leakage 7,8 . The induction of other surgical interventions, including the stapled colorectal anastomosis technique 9 or laparoscopic surgery 10 , also failed to reduce the incidence of anastomotic leakage. External coating to wrap the colonic anastomotic site with various materials has been investigated in animal models. The external coating of colonic anastomosis sealed off colonic anastomotic defects and prevented anastomotic leakage 11 . Although many researchers have reported on the sealing effects of fibrin glue at the colonic anastomotic site, few have demonstrated any positive effects on colonic anastomotic healing 12 . van der Ham et al. reported that, after sealing off the colonic anastomotic defect with fibrin glue, the implanted fibrin glue was heavily infiltrated and surrounded by inflammatory cells 13 . Similar results were found by wrapping the colonic anastomotic site with a resorbable polyglycolic acid mesh 14 . Thus, there is a need to develop a novel external coating material that does not exacerbate the inflammatory reaction during the colonic anastomotic healing process.
Cell sheet therapy using skeletal myoblasts, mesenchymal stem cells, or induced pluripotent stem cells has been successful in wound healing in various tissues 15 . Thus, it was assumed that cell sheet transplantation would be effective for anastomotic healing after colorectal surgery. Sukho et al. reported that cell sheets using human adipose tissue–derived stem cell (ASC sheets) prepared in a temperature-responsive culture dish were implanted on colonic anastomotic sites in high-risk anastomotic leakage model rats 16 . Although the ASC sheets facilitated an accumulation of anti-inflammatory cells in the anastomotic site on postoperative day (POD) 7, they did not decrease the occurrence or severity of abscess formation on POD3. In addition, the ASC sheets did not mechanically reinforce the colonic anastomotic site. These findings indicated that the low mechanical ability of the ASC sheet failed to suppress the anastomotic leakage in the early postoperative phase.
Previously, we discovered a method to fabricate multilayered cell sheets from cultured cells by periodic exposure to extremely high hydrostatic pressure during repeated cell seeding 17 . Supraphysiological periodic hydrostatic pressure promoted stress fiber formation and fibronectin fibrogenesis of cultured cells and increased the mechanical strength of cell sheets. In this study, we made multilayered cell sheets derived from human skeletal muscle myoblasts (HSMM sheets) by periodic exposure to supraphysiological hydrostatic pressure. Previous reports showed that skeletal muscle myoblasts ameliorate tissue inflammation and enhance neovascularization through the secretion of HGF, VEGF, and SDF-1 18 –20 . In addition, myoblast sheets made in a temperature-responsive culture dish were shown to improve the cardiac performance of infarcted hearts 18 and to prevent complications of pancreatic fistula and gastric perforation 21,22 . We therefore hypothesized that myoblast sheets reinforced by supraphysiological periodic hydrostatic pressure is useful for the prevention of colonic anastomotic leakage. In this study, we assessed whether HSMM sheets promote the healing process after colonic anastomosis in rats.
Materials and Methods
Isolation and Expansion of Human Skeletal Muscle Myoblasts
Human skeletal muscle myoblasts (HSMMs) were obtained from two different male donors (31 and 64 years old) as previously described 23 . HSMMs were cultured at 37°C in a humidified atmosphere with 5% CO2 in SkGM-2 medium (Lonza, Walkersville, MD, USA). Muscle biopsy from donors and HSMM extraction were approved by the Osaka University Clinical Research Review Committee (Approval number, 16236-4), and all related procedures were conducted in accordance with the ethical principles of the Declaration of Helsinki.
Fabrication of Multilayered Cell Sheets from Human Skeletal Muscle Myoblasts by Periodic Hydrostatic Pressurization
To fabricate the first layer of the HSMM sheet, cells (1132 cells/mm2) were seeded on a permeable atelocollagen membrane (MEN-01, KOKEN Co., Ltd., Tokyo, Japan) coated with fibronectin (0.2 mg/ml, Sigma, St. Louis, MO, USA) and cultured with SkGM-2 medium containing ascorbic acid (AA2G, 50 μmol/L, Hayashibara Co., Ltd., Okayama, Japan) under atmospheric pressure (101 kPa). Twenty-four hours after seeding, the cells were exposed to periodic hydrostatic pressure (110–130 kPa, 0.002 Hz) for 24 h using a periodic hydrostatic pressurization system (JH0133, Koganei Corporation, Tokyo, Japan) as previously described 17 . To fabricate the second layer, the same procedure was repeated. This cell seeding and periodic hydrostatic pressure was repeated 15 times to obtain a collagen-rich multilayered HSMM sheet (Fig. 1A).
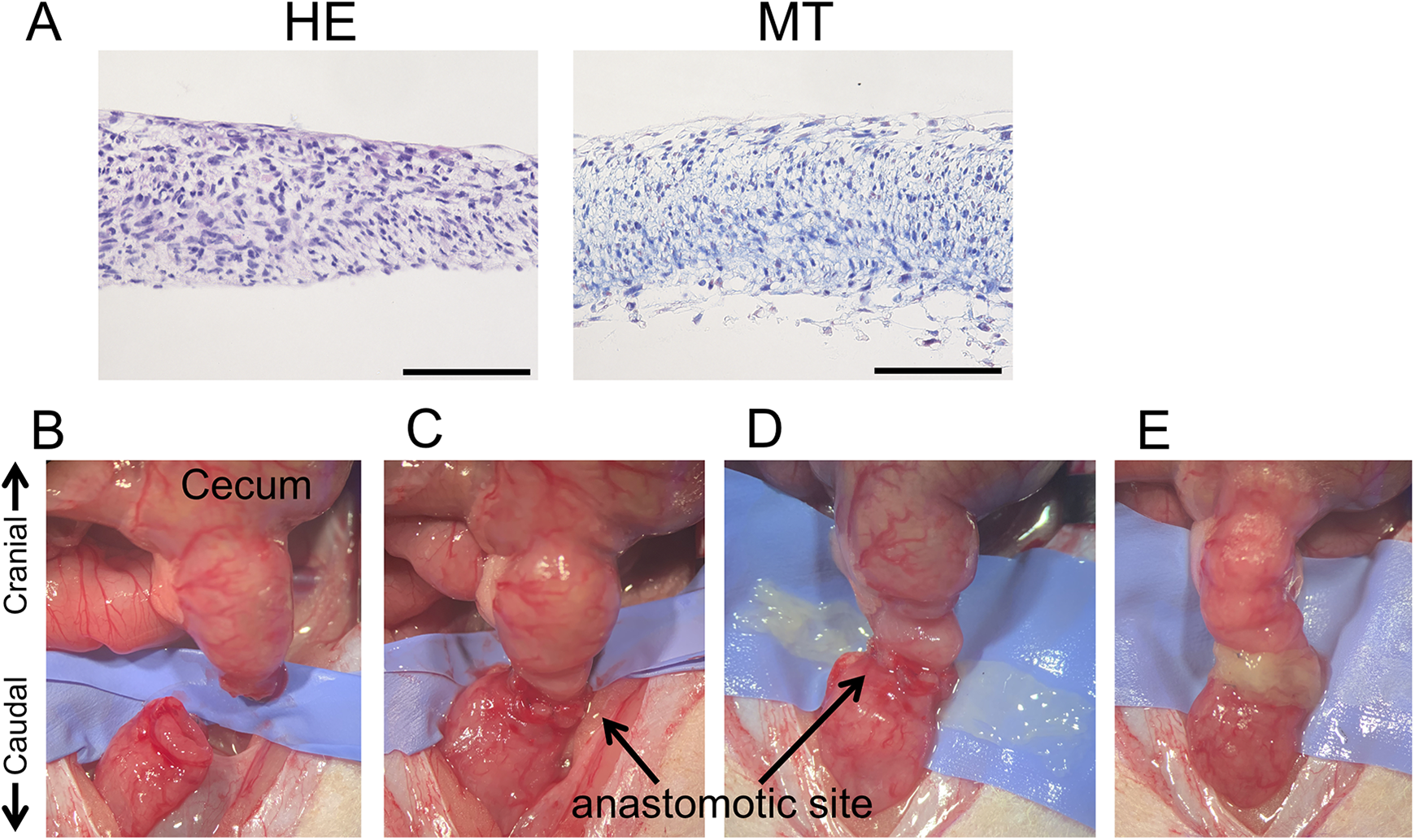
Transplantation of HSMM sheets. (A) Hematoxylin & eosin (HE; left) and Masson’s trichrome stains (MT; right) of an HSMM sheet. Scale bars, 100 μm. (B-E) HSMM sheet transplantation procedure. After resection of the colonic segment (B), a full-thickness end-to-end anastomosis was created (C). The anastomotic site was wrapped with an HSMM sheet (D-E).
Animals
Sixty adult male nude rats (Long-Evans-rnu/rnu, 250–350 g body weight) were obtained from Japan SLC, Inc. (Shizuoka, Japan). All rats had been housed in individual cages and were kept under standard laboratory conditions. All rats were given standard rat chow and water ad libitum. The animals were fasted 24 h prior to surgery with free access to water. All animal studies were approved by the Animal Care and Use Committee of Yokohama City University (Approval number, F-A-18-050).
Implantation
We implanted an HSMM sheet to the colonic anastomotic site of a previously described high-risk colo-colonic anastomotic leakage rat model 24 . Male nude rats were anesthetized in a closed chamber with 4% isoflurane. Anesthesia was maintained with 1.5% to 2.5% isoflurane by mask. A 5 cm midline laparotomy incision was made, and the right, middle, and left colic arteries were ligated with 6-0 nylon monofilament sutures (Natsume Seisakusho Co. Ltd., Tokyo, Japan). The colonic segment between the site 10 mm aborally to the cecum and 5 mm proximally the caudal mesenteric artery was resected (Fig. 1B). A full-thickness end-to-end anastomosis was performed by one-layer inverting suturing with 5 interrupted sutures (8-0 nylon monofilament suture, Bear Medic Co. Ltd., Tokyo, Japan) (Fig. 1C). Rats were randomized to control or HSMM sheet groups (n = 30 in each group). Immediately before implantation, the HSMM sheet was detached from an atelocollagen membrane using a cell scraper. In the HSMM sheet group, the dish side of the HSMM sheet was wrapped around the anastomotic site (Fig. 1D). The upper and lower side of the HSMM sheet was sutured to the serosal surface of the intestinal wall with 6 interrupted sutures (8-0 nylon monofilament suture) (Fig. 1E). The control group received the sutured end-to-end anastomosis with no other intervention. Lastly, the muscle layer and skin were closed with 5-0 nylon monofilament sutures (Akiyama Medical Mfg. Co., Ltd., Tokyo, Japan). Immediately after surgery, rats were warmed at 38°C and rehydrated with 0.9% saline solution. On POD3, 5, or 7 (n = 8 in each group), animals were anesthetized with isoflurane and re-laparotomy was performed for evaluation of short-term anastomotic healing. In addition, long-term anastomotic healing was evaluated on POD14 or 28 (n = 3 in each group). At the time of surgery and autopsy, the body weights of all animals were evaluated.
Macroscopic Observation of the Anastomotic Site
At the anastomotic site, macroscopic signs of anastomotic disruption, stricture, adhesion, and abscess formation were evaluated on POD3, 5, and 7. The number of abscesses at the anastomotic site was counted and abscess severity was scored according to the previously described abscess score: 0 = no abscess, 0.5 = one small abscess (<1 mm), 1 = several small abscesses, 2 = one medium abscess (1–3 mm), 3 = one large (3–5 mm), or several medium abscesses, or 4 = one very large (>5 mm) or several large abscesses 16 . The number of adhesions at the anastomotic site was counted and the adhesion score was evaluated through a modification of van der Ham et al.’s 13 adhesion score: 0 = no adhesions, 1 = minimal adhesions (i.e., mainly between the anastomotic site and the omentum), 2 = moderate adhesions (i.e., between the omentum and the anastomotic site and between the anastomotic site and a loop of the small intestine or cecum), or 3 = severe adhesions (i.e., between the omentum and the anastomotic site and between the anastomotic site and several small intestines or the small intestine and cecum). After macroscopic observation, the anastomotic bursting pressure was determined. In short, the anastomotic site was extracted and a catheter was placed at the distal end. The proximal and distal end of the colonic portion was ligated with 6-0 nylon monofilament sutures. A pressure transducer and a 30 ml syringe connected to an infusion pump were connected to the catheter. The pressure transducer was connected to a recorder. The anastomotic bursting pressure was measured by the air insufflation (2 ml/min), and the pressure at the time of the first air leak was recorded as the anastomotic bursting pressure. The site of the burst was also recorded. After the pressure measurement, histological analysis of the colonic anastomotic part was performed.
Histological Analysis
Colonic anastomotic samples were washed with phosphate-buffered saline (PBS) and opened longitudinally. Samples were fixed in 4% buffered formaldehyde, and embedded in paraffin. Samples were then sectioned into 4-μm-thick slices. Hematoxylin & eosin (HE) stain was performed for morphological analysis and measurement of the maximum distance between the scar margins at the anastomotic site. The measurement was performed using two or three representative images (20× magnification) per sample and then averaged. Masson’s trichrome (MT) stain (Sigma) was performed for the evaluation of collagen deposition with standard protocols. For immunohistochemistry, the sections were de-paraffinized, rehydrated, and incubated with primary antibodies for 24 h at 4°C. Inflammatory cell infiltration was evaluated using antibodies against myeloperoxidase (ab9535, Abcam, Cambridge, UK). Vascularization was evaluated using antibodies against CD34 (AF4117, R&D Systems, Minneapolis, MN, USA). Antibodies against α-smooth muscle actin (α-SMA) were purchased from Cell Signaling (#19245, Danvers, MA, USA). HSMM sheet survival was evaluated using antibodies against HLA (ab70328, Abcam) and human TRA-1-85 (MAB3195, R&D Systems). After washing with PBS, the sections were incubated for 30 min in biotinylated second antibodies (Vectastain Elite ABC IgG kit, Vector Labs, Burlingame, CA, USA) at room temperature. The targeted proteins were revealed with DAB (Dako Cytomation, Glostrup, Denmark). The sections were counterstained with Mayer’s hematoxylin. The negative control was confirmed by the omission of primary antibodies. The histological assessments on POD3, 5, and 7 were performed using seven representative images (200× magnification) per sample and then averaged. The histological sections on POD14 and 28 were assessed using three representative images (400× magnification) per sample and then averaged. The sections were photographed and analyzed using a BZ-X800 microscope (Keyence, Osaka, Japan).
Statistical Analysis
Data are expressed as the mean ± standard error of the mean (SEM). Continuous and discontinuous variables were statistically analyzed using the unpaired t-test (Fig. 4–7 and Supplemental Fig. S1–S4) and the Mann–Whitney U test (Fig. 3), respectively. To compare the proportion between groups at each time, the two-tailed Chi-squared test was used (Table. 1). A value of P < 0.05 was considered statistically significant.
Post-Operative Macroscopic Observations at the Colonic Anastomotic Site.
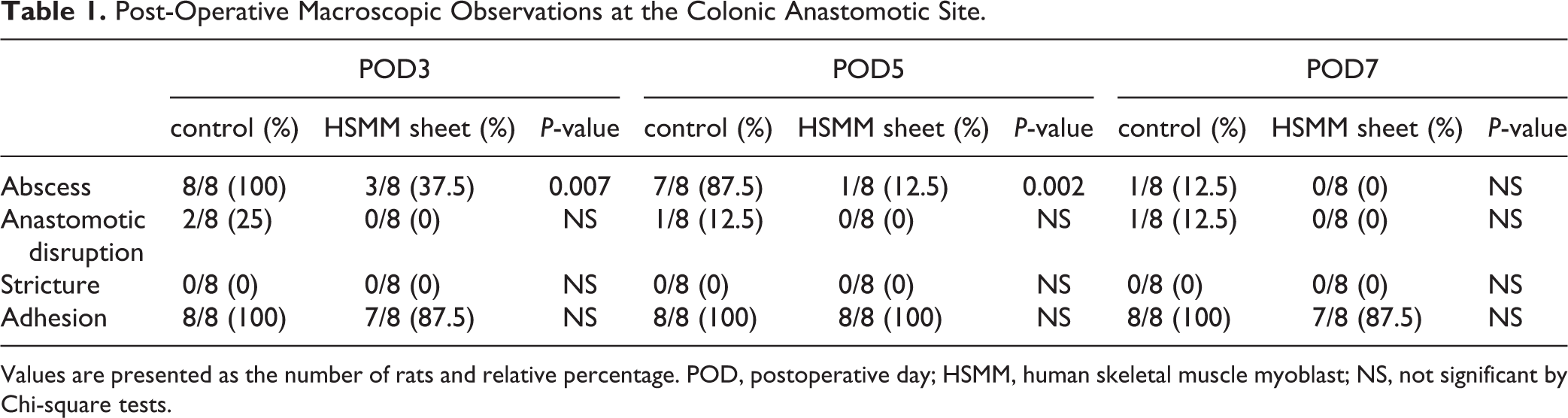
Values are presented as the number of rats and relative percentage. POD, postoperative day; HSMM, human skeletal muscle myoblast; NS, not significant by Chi-square tests.
Results
Overall Observations
Transplantations of the HSMM sheets were successful in all animals in the HSMM sheet group. Post-operatively, all animals in both groups survived and had diarrhea. The body weight and wellness did not differ between the control and HSMM sheet groups (Supplemental Fig. S1).
Postoperative Macroscopic Observations
To evaluate the preventive effect on anastomotic leakage by wrapping the HSMM sheet around the anastomotic site, we evaluated the macroscopic findings of the anastomotic site in the control and HSMM sheet groups. The HSMM sheets were visible around the anastomotic site and adhered firmly to the colonic serosal surface (Fig. 2A). Abscess formation at the anastomotic site in the control group was significantly more common than that in the HSMM sheet group on POD3 and 5 (P = 0.007 and 0.002, respectively) (Fig. 2B and Table 1). No significant difference in the prevalence of abscess formation was observed on POD7 between the two groups. There were no differences in the incidence of anastomotic disruption, stricture, and adhesions. The abscess number and score of the anastomotic site in the control group were significantly higher than that in the HSMM sheet group on POD3 and 5 (Fig. 3A, B). The abscess number and score between groups did not differ on POD7 (Fig. 3A, B). The adhesion number in the control group was significantly higher than that in the HSMM sheet group on POD3 and 7, although it did not reach statistical significance on POD5 (Fig. 3C). No significant difference in the adhesion score was observed between the two groups throughout the time-course (Fig. 3D).
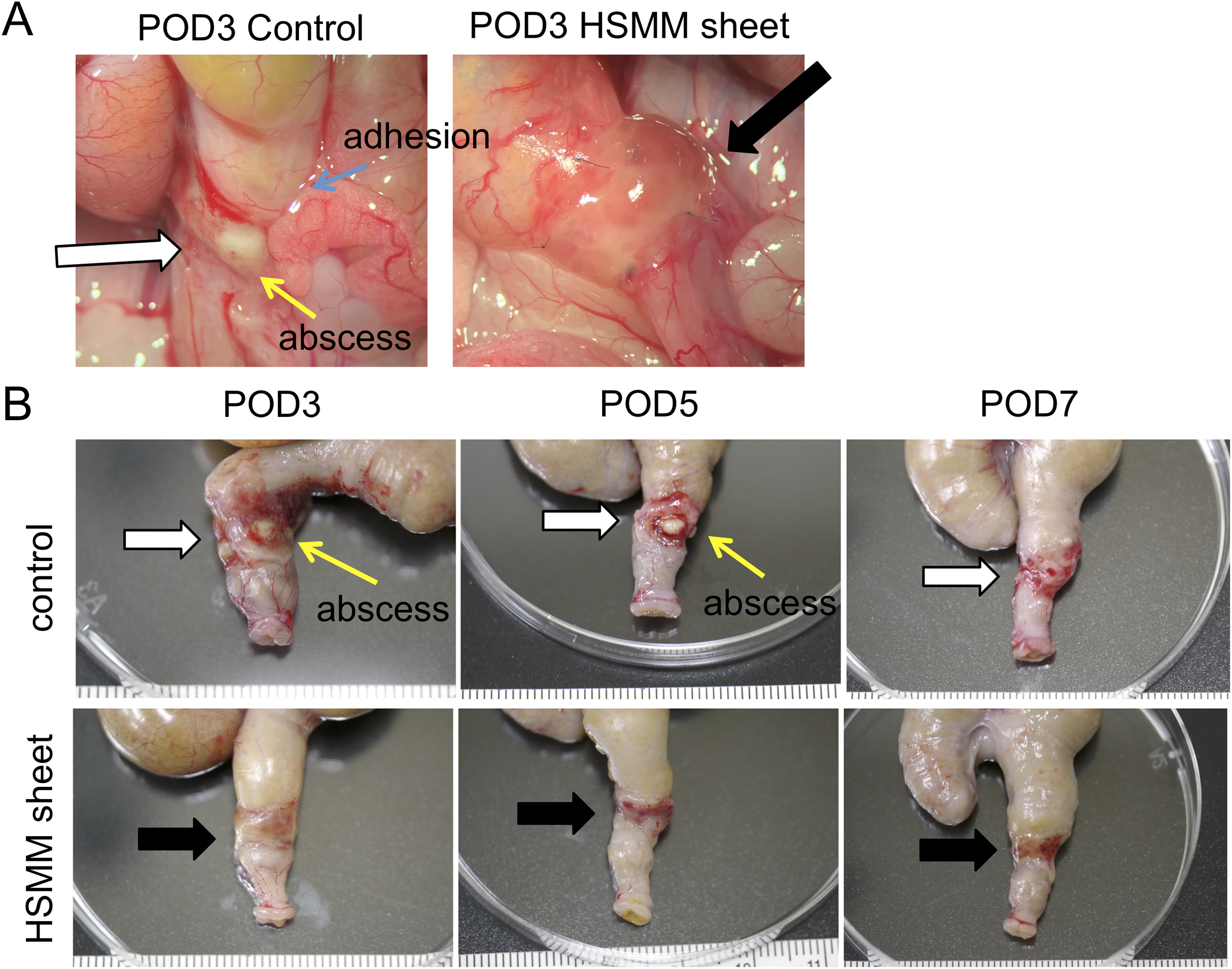
Representative images of macroscopic observation at the colonic anastomotic site. (A) Representative macroscopic images at the colonic anastomotic site after re-laparotomy on postoperative day (POD) 3 in the control group (left) and the HSMM sheet group (right). (B) Representative macroscopic images after resection of the anastomotic site on POD3 (left), 5 (middle), and 7 (right) in the control group (upper) and the HSMM sheet group (lower). White and black arrows indicate the anastomotic site and the transplanted HSMM sheet, respectively.
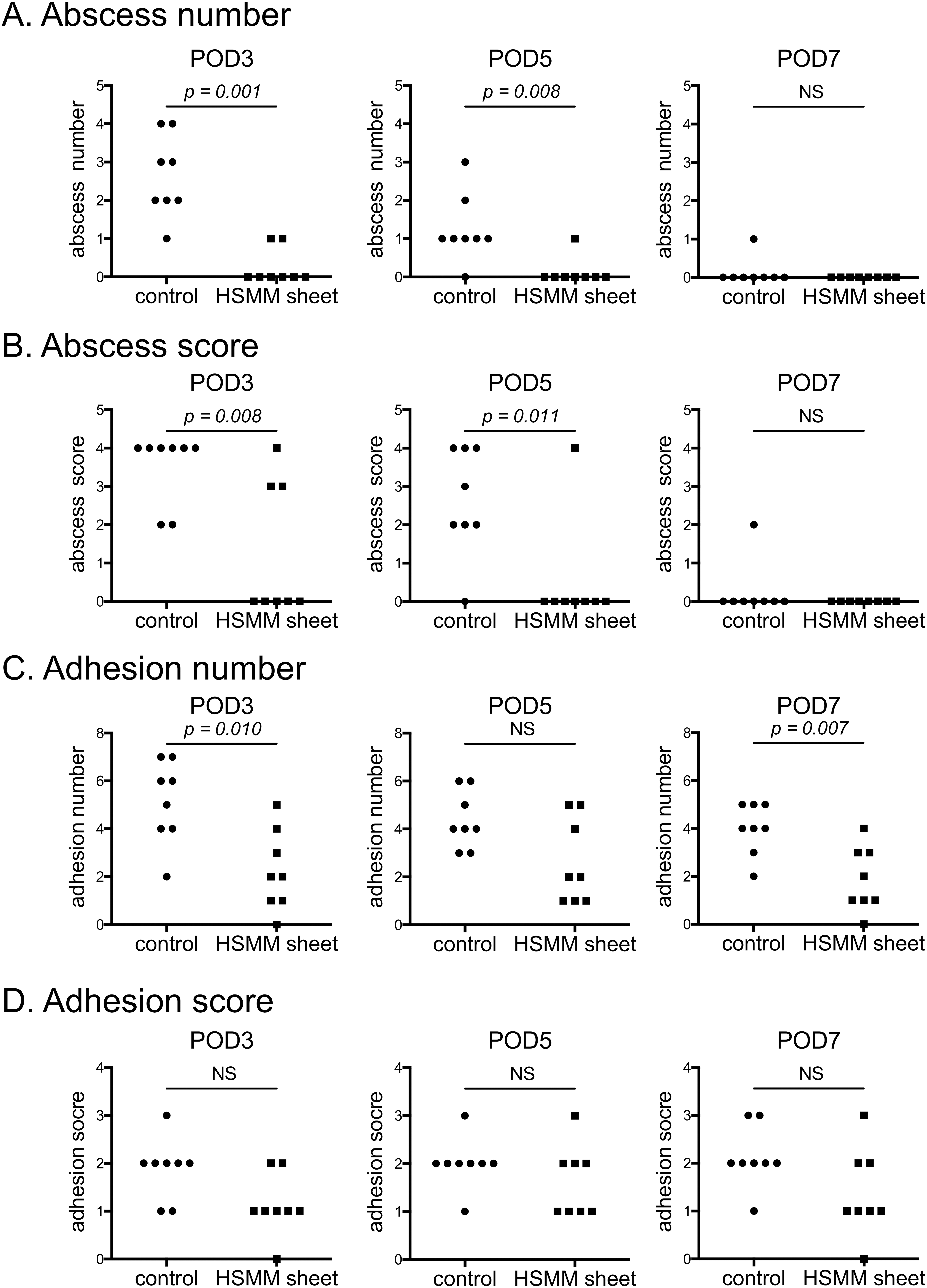
Macroscopic observations of the anastomotic site. (A) Abscess number, (B) abscess score, (C) adhesion number, and (D) adhesion score on postoperative day (POD) 3 (left), 5 (middle), and 7 (right). n = 8. Statistics were analyzed using Mann–Whitney U tests. NS, not significant.
Next, we evaluated the anastomotic bursting pressure, which is one of the parameters of anastomotic healing 25,26 . The anastomotic bursting pressure in the HSMM sheet group was significantly higher than that in the control group on POD3 and 5 (Fig. 4A, B, respectively). No significant difference between the two groups was evident on POD7 (Fig. 4C). Bursting occurred mainly at the anastomotic site in the control group (5/8; 62.5%) whereas no bursting at the anastomotic site occurred in the HSMM sheet group (0/8; 0%) on POD7 (P = 0.026). These findings indicated that the HSMM sheet reinforced the anastomotic site and promoted anastomotic healing in the early postoperative period.
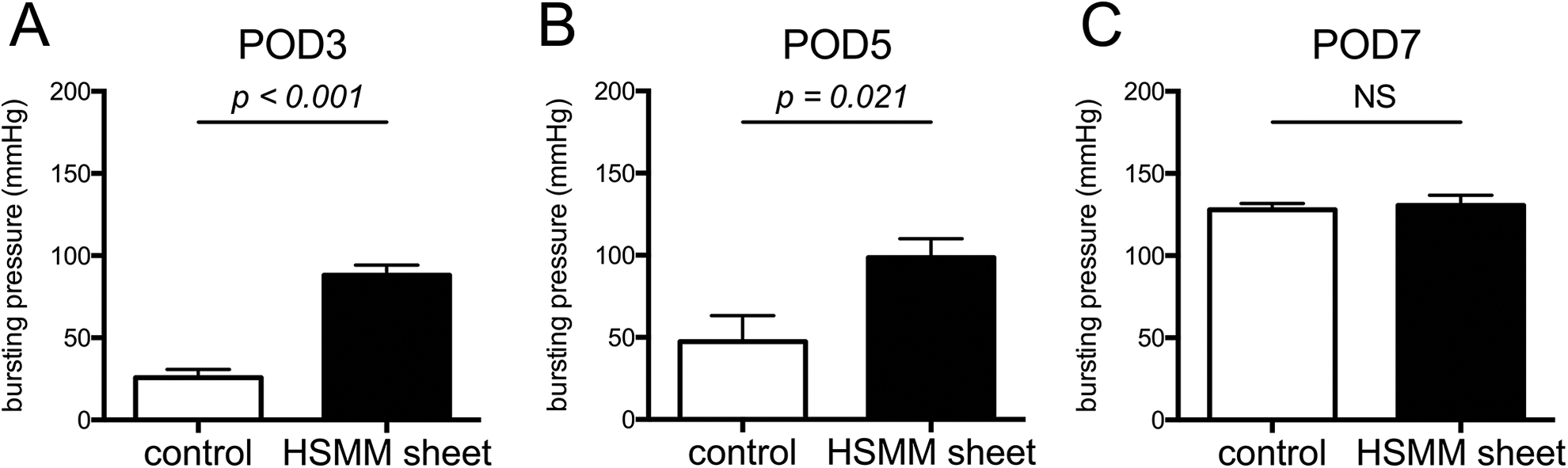
Anastomotic bursting pressure on postoperative day (POD) 3 (A), 5 (B), and 7 (C). Results are presented as the mean ± SEM. n = 8. Statistics were analyzed using the unpaired t-test. NS, not significant.
Histological Evaluation
To evaluate the effect of HSMM sheets on the colonic anastomotic healing process in the early postoperative period, histological and immunohistochemical analyses were performed. There was no difference in the maximum distance between the scar margins in the control and HSMM sheet groups at the anastomotic site on POD3, 5, and 7 (Supplemental Fig. S2). As shown in Fig. 5A and B, the infiltration of inflammatory cells (percentage area of myeloperoxidase-positive cells) at the anastomotic site was significantly suppressed in the HSMM sheet group compared with the control group on POD3, 5, and 7. The percentage area of collagens in the HSMM sheet group was greater than that in the control group on POD3 (Fig. 5C, D). There was, however, no difference in collagen deposition between groups on POD5 and 7 (Fig. 5C, D). As for vascularization, there were no obvious differences in CD34-positive capillary density between groups at any time point (Supplemental Fig. S3). The expression level of the contractility-related protein α-SMA was significantly higher in the HSMM sheet group than in the control group on POD3, and there were no differences between groups on POD5 and 7 (Fig. 6).
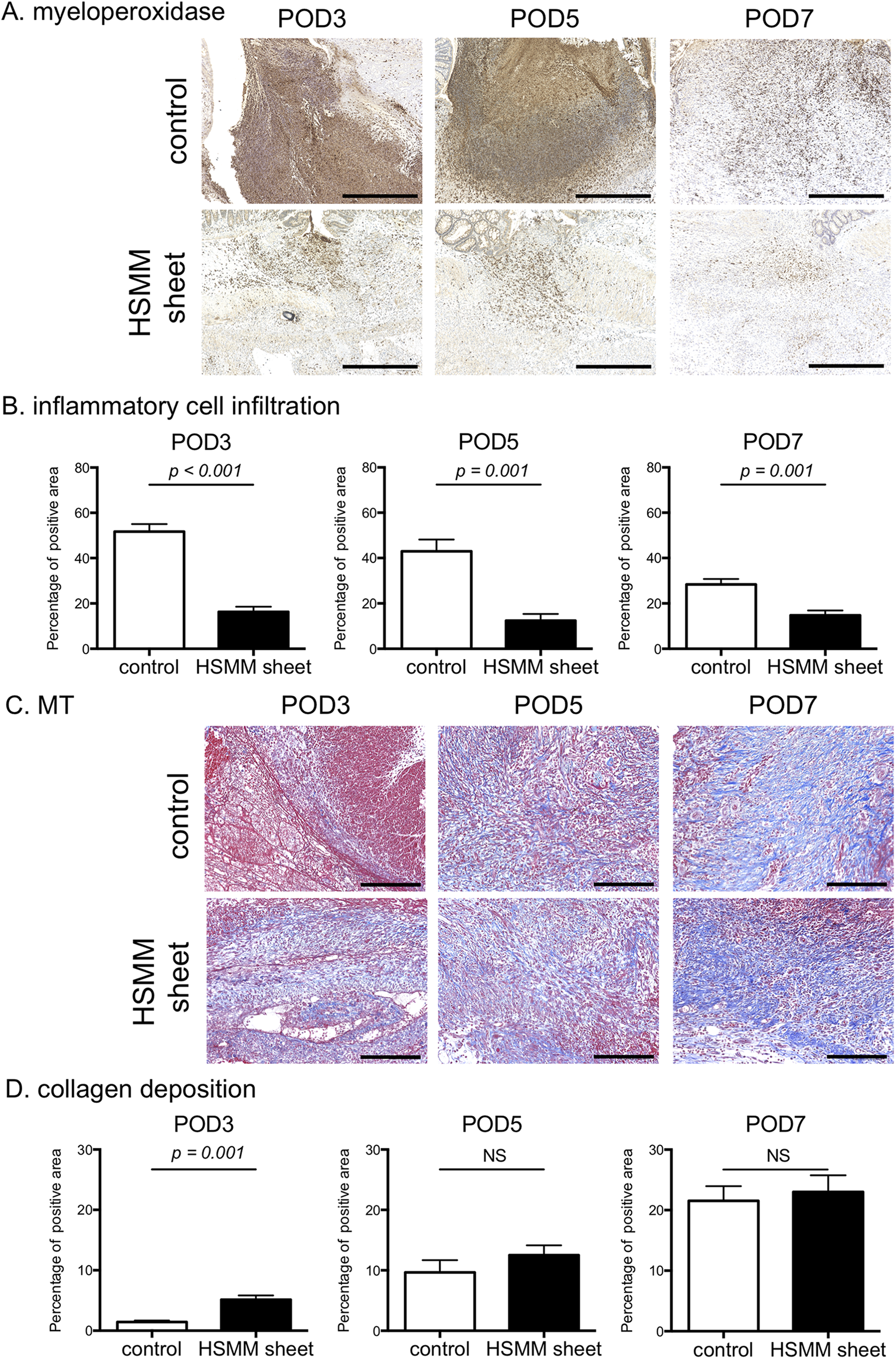
Comparison of inflammatory cell infiltration and collagen deposition between the control group and the HSMM sheet group on postoperative day (POD) 3, 5, and 7. (A) Representative images of immunohistochemical stain for myeloperoxidase of the anastomotic site in the control (upper) and the HSMM sheet (lower) group on POD3 (left), 5 (middle), and 7 (right). Scale bars, 500 μm. (B) Inflammatory cell infiltration assessed by area percentage of myeloperoxidase-positive cells on POD3 (left), 5 (middle), and 7 (right). (C) Representative images of Masson’s trichrome (MT) stain of the anastomotic site in the control (upper) and the HSMM sheet (lower) group on POD3 (left), 5 (middle), and 7 (right). Scale bars, 200 μm. (D) Collagen deposition assessed by area percentage of collagen stained by MT stain on POD3 (left), 5 (middle), and 7 (right). Results are presented as the mean ± SEM. n = 8. Statistics were analyzed using the unpaired t-test. NS, not significant.
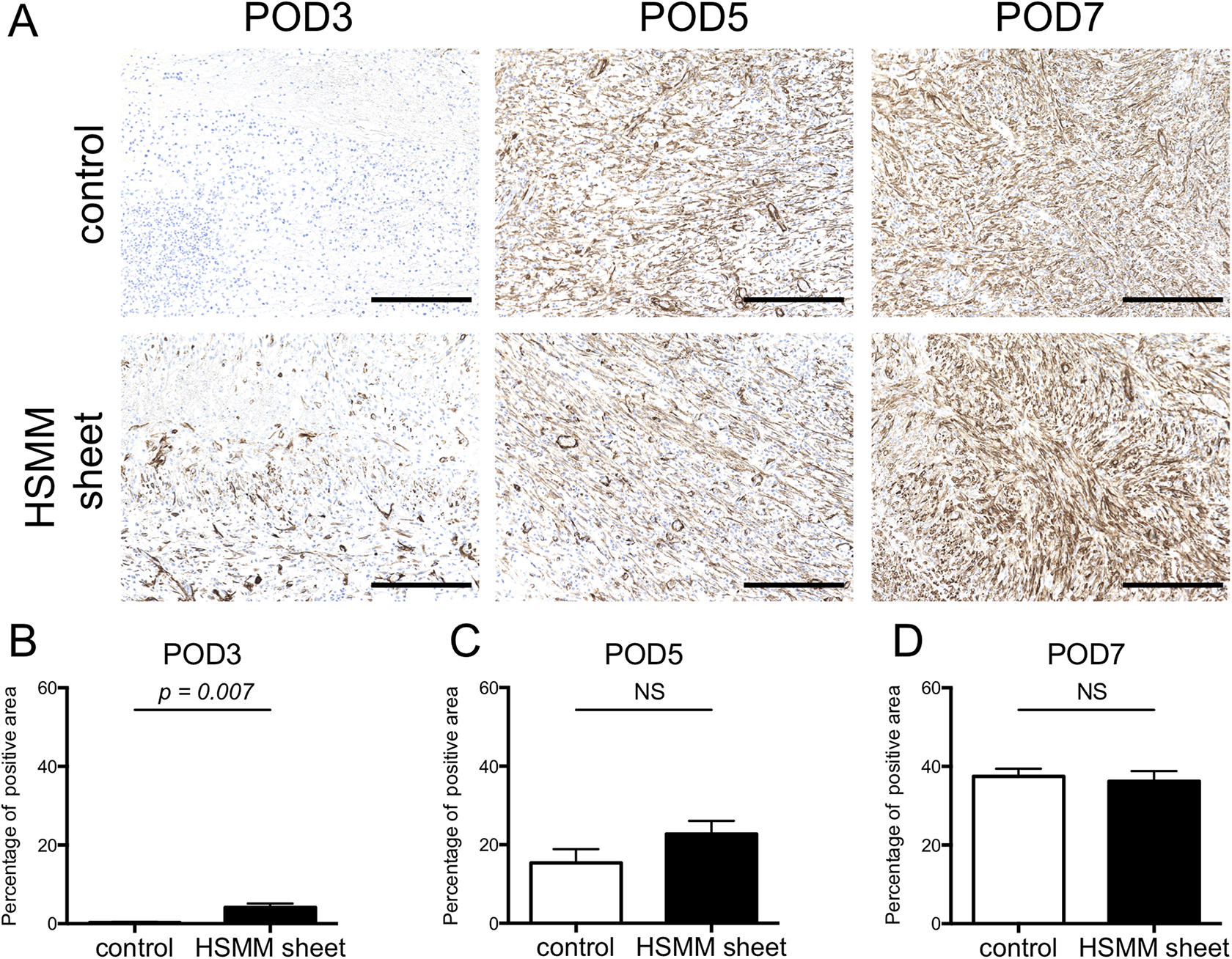
Expression level of α-SMA-positive cells at the anastomotic site on postoperative day (POD) 3, 5, and 7. (A) Representative images of immunohistochemical stain for α-SMA of the anastomotic site in the control (upper) and the HSMM sheet (lower) group on POD3 (left), 5 (middle), and 7 (right). Scale bars, 200 μm. (B-D) Comparison of area percentage of α-SMA-positive cells at the anastomotic site between the control and HSMM sheet group on POD 3 (B), 5 (C), and 7 (D). Results are presented as the mean ± SEM. n = 8. Statistics were analyzed using the unpaired t-test. NS, not significant.
Next, we evaluated the long-term histological anastomotic healing after transplantation of the HSMM sheet. The inner layer of the anastomotic site was covered by regenerated mucosa in the HSMM sheet group on POD14 and 28 whereas discontinuity of the mucosa at the anastomotic site was observed in the control group on POD14 and 28 (Fig. 7A, B). The degree of inflammatory cell infiltration at the anastomotic site was less in the HSMM sheet group than in the control group on POD14 and 28 (Fig. 7C–E). There were no differences in collagen deposition, vascularization, and α-SMA expression between groups on POD14 and 28 (Supplemental Fig. S4). These findings indicated that the prevention of postoperative anastomotic leakage by wrapping the HSMM sheet around the anastomotic site suppresses the inflammatory reaction and affects the improvement of anastomotic healing in the late postoperative phase.
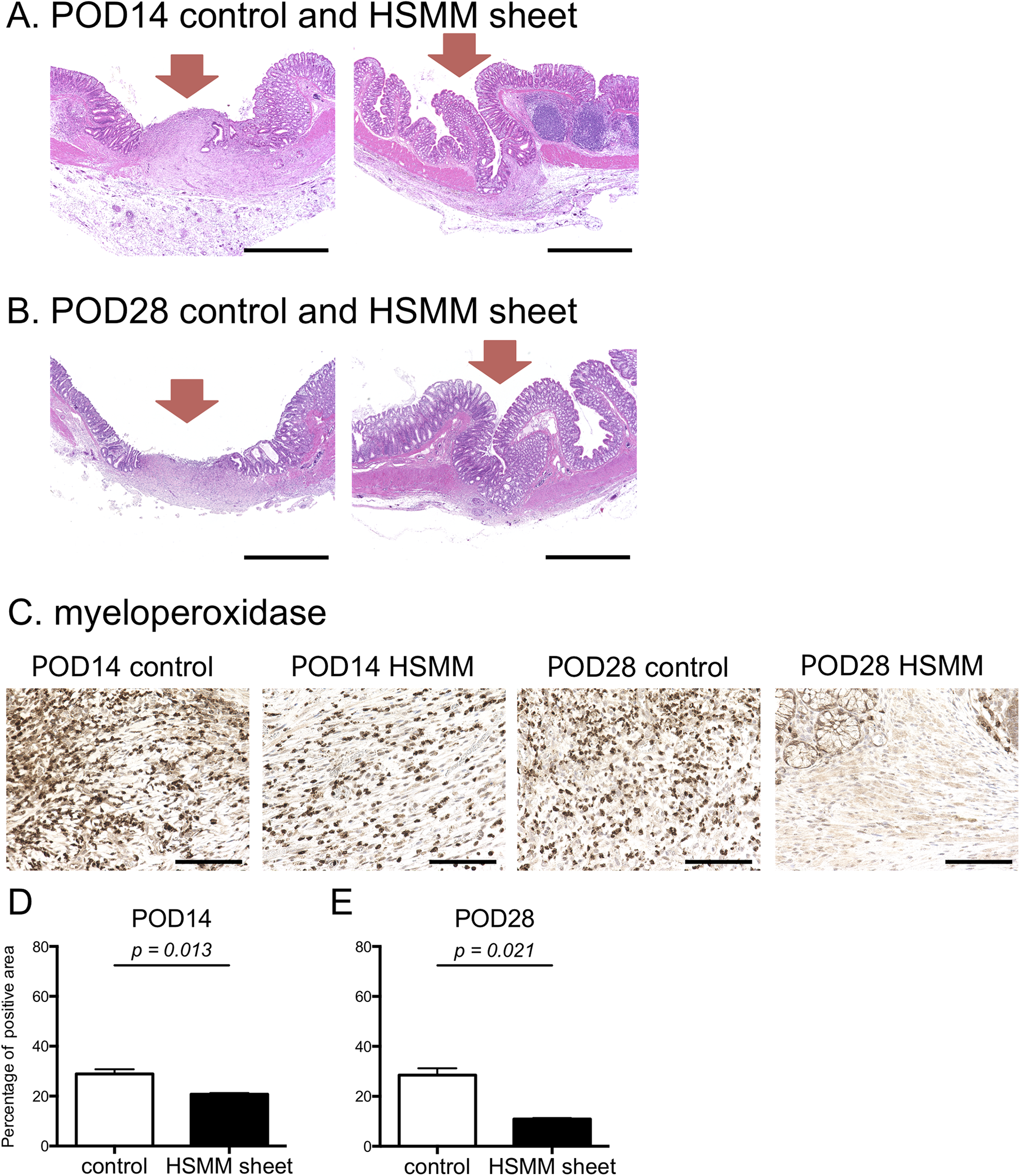
Histological evaluation at the anastomotic site 14 and 28 days after transplantation of the HSMM sheet. (A, B) Representative images of hematoxylin & eosin stain in the control (left) and the HSMM sheet (right) group on postoperative day (POD) 14 (A) and 28 (B). There was discontinuation of the mucous membrane in the control group on POD14 and 28. Recovery of the continuity of the mucous membrane in the HSMM sheet group is evident on POD14 and 28. Scale bars, 1 mm. Red arrows indicate the anastomotic site. (C) Representative images of immunohistochemical stain for myeloperoxidase of the anastomotic site in the control and the HSMM sheet group on POD14 and 28. Scale bars, 100 μm. (D–E) Comparison of inflammatory cell infiltration at the anastomotic site on POD14 (D) and 28 (E). Results are presented as the mean ± SEM. n = 3. Statistics were analyzed using the unpaired t-test.
Lastly, to evaluate how long implanted HSMMs remained, immunohistochemical stains for HLA and TRA-1-85 were performed. On POD3, there were abundant HLA-positive cells in the implanted HSMM sheet (Fig. 8A). HLA-positive cells at the anastomotic site gradually diminished on POD5 and 7, however (Fig. 8B, C), and were no longer detected on POD14 and 28 (Fig. 8D, E). Similar findings regarding immunohistochemical stain for TRA-1-85 were found (Supplemental Fig. S5). These findings indicated that the HSMM sheets were gradually replaced by host-derived cells after transplantation.
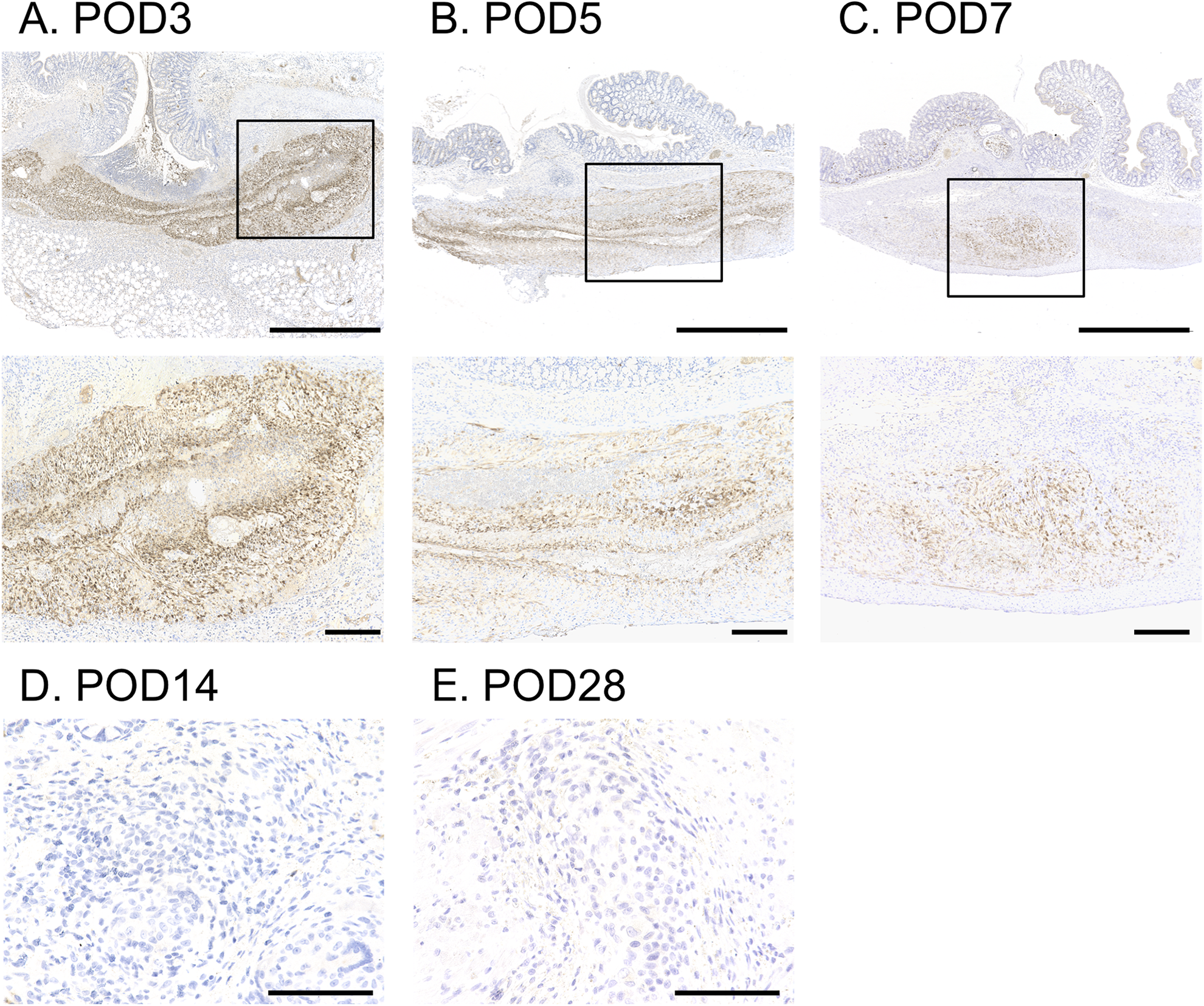
Remaining HSMM sheets at the anastomotic site. (A–C) Representative images of immunohistochemical stain for HLA on postoperative day (POD) 3 (A), 5 (B), and 7 (C). Upper images are low power field (scale bars, 1 mm). The selected area with the solid line is enlarged and illustrated below (scale bars, 200 μm). (D–E) Representative images of immunohistochemical stain for HLA of the anastomotic site on POD14 (D) and 28 (E). Scale bars, 100 μm. HLA-positive cells gradually diminished with POD, and were no longer detected on POD14 and 28.
Discussion
Cell sheet technology is currently advancing in regenerative medicine. Cell sheets enable the local delivery of cultured cells with high cell viability and preserved the extracellular matrix structure 27 . Cell sheets have succeeded in regenerating various tissues and promoting wound healing as a substitution for injured tissue or a sustainable carrier of cytokines 15 . One challenge, however, is that a long culture period (more than several months) is required to prepare cell sheets with high mechanical properties 28 . Previously, we demonstrated that supraphysiological periodic hydrostatic pressure promoted stress fiber formation and fibronectin fibrogenesis in cultured cells 17 . In addition, the combination of repeated cell seeding and periodic exposure to supraphysiological hydrostatic pressure enabled the fabrication of multilayered cell sheets from cultured cells. Using this technique, we made collagen-rich multilayered cell sheets derived from HSMMs within a month-long culture period. In this study, the application of an HSMM sheet to the colonic anastomotic site suppressed abscess formation and adhesion number, which are the most common complications of colorectal surgery 2,29 , in the early postoperative period. Importantly, HSMM sheets reinforced the anastomotic strength. In addition, it was clarified that these preventive effects of anastomotic leakage led to the promoted regeneration of intestinal mucosa at the anastomotic site.
In the clinical setting, partial colectomy can result in severe surgical trauma and an inflammatory response 30 , both of which are risk factors for colorectal anastomotic leakage after colorectal surgery 2 . Wu et al. created a partial colectomy model in rats 24 and reported that these model rats exhibited a higher incidence of anastomotic leakage than simple colon transection or short-length resection model rats 13,14 . Using this high-risk anastomotic leakage model, Sukho et al. evaluated the efficacy of ASC sheets to prevent anastomotic leakage 16 . Although the application of ASC sheets resulted in a reduced incidence of anastomotic disruption, they did not suppress abscess formation on POD3. In addition, the ASC sheets did not reinforce the mechanical anastomotic strength due to their lack of mechanical ability. Based on these findings, we speculated that the ASC sheets could not withstand the intestinal pressure at the anastomotic site and that small leakages occurred from cracks in the sheets. In our study employing the same animal model, the application of HSMM sheets prevented anastomotic abscess formation on POD3. In addition, histological inflammatory cell infiltration was also suppressed, and HSMM sheets reinforced the anastomotic strength on POD3. Previous reports showed that anastomotic strength on POD3 was at its lowest after colonic surgery, which suggests that this is the period of highest risk for colonic anastomotic leakage and requires mechanical support 31,32 . These findings indicated that cell sheets that reinforce anastomotic mechanical strength might be effective in the prevention of colonic anastomotic leakage in the early postoperative period.
In this study, the HSMM sheets contained abundant collagens, which provide mechanical properties to cell sheets 33 . In addition, collagen deposition in the HSMM sheet group on POD3 was marked compared to that in the control group. The collagenous component in the HSMM sheets appeared to seal off the defects of the bowel wall and firmly adhere to the anastomotic site. The existing collagen network in the anastomotic site is responsible for anastomotic strength 25 . Thus, collagen fibers in HSMM sheets may contribute to the reinforcement of the anastomotic strength. Because previously reported cell sheets made by a temperature-responsive culture lacked collagen fibers 34 , the collagen-rich property of the HSMM sheets is thought to be an advantageous feature for the prevention of anastomotic leakage.
In this study, the expression level of contractility-related proteins at the anastomotic site in the HSMM sheet group was remarkable compared to that in the control groups on POD3. α-SMA is expressed in myofibroblasts and smooth muscle cells, which are the key cells of colonic anastomotic healing, and provide mechanical properties for contraction of the anastomotic site 35 . Contraction of the anastomotic site lead to enhanced mechanical anastomotic strength 32,35 . In addition, because myoblasts have been reported to differentiate to myofibroblasts by stimulation with fibrogenic cytokines including TGF-beta 36 , transdifferentiation of skeletal myoblasts in the HSMM sheet may also increase the mechanical strength. Thus, the abundant expression of contractility-related protein after implantation of the HSMM sheet might contribute to the prevention of anastomotic leakage in the early postoperative period.
One of the positive effects of myoblast therapy in the treatment of heart failure is the induction of angiogenesis through the secretion of paracrine mediators, including VEGF, HGF, and SDF-1 18,34,37 . Due to the need for the induction of microcirculation for tissue regeneration, angiogenesis is an indispensable process for colorectal anastomotic healing 38,39 . Although the HSMM sheets were expected to promote anastomotic vascularization via paracrine effects, the present study showed no difference in anastomotic capillary density between the control and treated groups. The amino acid sequence of VEGF is highly conserved across species, and a previous report showed that human VEGF binds to its receptors of rat vascular endothelial cell in various tissues 40 . In addition, the local or systemic injection of recombinant human VEGF promoted wound healing through increased angiogenesis in an animal model with colonic anastomoses 41 or gastric mucosal injuries 42 . Based on this evidence, we speculated that human VEGF secreted by human skeletal myoblasts was likely to bind to rat VEGF receptor at colonic anastomotic sites. Although we do not know why the HSMM sheet did not promote anastomotic vascularization, we will need to modify the fabrication method of the HSMM sheet to promote vascularization in future studies. Recently, many researchers have studied the prefabrication of the endothelial cell network in engineered tissues in vitro 43,44 . Vascularization in cell sheets is crucial for cell survival and the persistence of the functional capacity of cell sheets after transplantation. By applying these techniques to our method, it may be possible to further accelerate the beneficial effects of the HSMM sheet on anastomotic healing.
Previous reports showed that myoblast or ASC sheet survival time after transplantation to an injured heart was 1 to 2 months 45 –48 . In this study, HSMMs were present for at least 7 days after transplantation. This result was equivalent to that reported by Sukho et al. for ASC survival 16 . Transplanted cell survival is largely influenced by the degree of inflammation after transplantation 48 . A severe inflammatory reaction after partial colectomy might cause a shorter survival time in transplanted cells around the anastomotic site 30 . Importantly, however, because clinical anastomotic leakage most often occurs within 7 days of surgery 2 , living HSMMs might contribute to the prevention of colonic anastomotic leakage in the clinical setting.
Colorectal anastomotic leakages are commonly detected starting 3 days after colorectal surgery 2 . Based on epidemiological studies, previous reports using an animal model of anastomotic leakage analyzed the tissue healing process from 3 to 7 days after surgery 16,24 . Although clinical research revealed that some patients are diagnosed with anastomotic leakage within 2 days after colorectal surgery 49 , a previous report showed that the acute inflammatory reaction on POD1 was similar level to that on POD3 in a colon transection model 50 . Further study with a shorter follow-up time (e.g., POD1) will provide insight into the molecular mechanisms related to the acute anastomotic healing process after transplantation of the HSMM sheet.
In this study, the mucosal regeneration of the anastomotic site was promoted and the infiltration of inflammatory cells was suppressed in the HSMM sheet group compared to that in the control group on POD14 and 28. Clinically, half of the patients with anastomotic leakage were diagnosed after discharge from the hospital due to abdominal symptoms 49 . Most studies using animals evaluated the short-term histological anastomotic healing within 1 week of the operation. Based on our findings, anastomotic leakage in the early postoperative phase may be involved in delayed anastomotic healing. The prevention of early-phase anastomotic leakage may affect the reduced incidence of late-phase anastomotic leakage.
Our study has some limitations that should be noted. First, we could not clarify the mechanism of the preventive effect of anastomotic leakage by application of an HSMM sheet. Although reinforcement of the anastomotic site by wrapping with an HSMM sheet is a preventive effect factor, other mechanisms (including paracrine effects) may be involved. Similar to ASCs, myoblasts have been reported to release abundant angiogenic and anti-inflammatory factors 19,47 . These cytokines are also involved in cell proliferation and migration 18,34,37 . Thus, the paracrine effects of HSMMs may be involved in suppressing the inflammatory reaction and improving wound healing at the colonic anastomosis. Further studies are needed to verify if these paracrine effects of HSMMs are beneficial to colonic anastomotic healing.
Our current method for fabrication of multilayered cell sheet needs 28 days. The long culture period is related to the enhanced mechanical properties of cell sheets due to the formation and maturation of extracellular matrix 28 . This relatively long fabrication period made it possible to provide sufficient mechanical strength to the HSMM sheet to prevent anastomotic leakage, and fabricated HSMM sheets can be easily handled due to their stiffness and elasticity. Prolonging the culture period leads to high medical costs and delayed surgery schedules, however. Thus, further improvement of the current fabrication protocols to shorten the culture period is needed for clinical application.
Finally, our surgical procedure for the transplantation of cell sheets was more complicated than that using cell sheets prepared in a temperature-responsive culture dish 16 . A temperature-responsive culture dish was coated with poly (N-isopropyl acrylamide) 15 . This special polymer changes from hydrophobic to hydrophilic at 20°C, and cell sheets can be detached from these culture dishes without trypsinization or scraping. These cell sheets preserve adhesive proteins on the dish side of the cell sheets, and can adhere to the colonic serosal surface spontaneously and rapidly without suturing or glue. These cell sheets thus enable reduced transplantation time. Future research will examine the simplification of the surgical procedure by applying our technology to these special culture dishes.
In conclusion, this study demonstrated the prevention of colonic anastomotic leakage by application of a multilayered HSMM sheet. Application of the HSMM sheet reinforced anastomotic strength and enhanced anastomotic healing with a decreased incidence of abscess formation. Multilayered HSMM sheets may thus be useful in reducing the incidence of complications after colorectal surgery.
Supplemental Material
Supplemental Material, sj-doc-1-cll-10.1177_09636897211009559 - Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats
Supplemental Material, sj-doc-1-cll-10.1177_09636897211009559 for Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats by Takashi Nakamura, Utako Yokoyama, Tomomitsu Kanaya, Takayoshi Ueno, Takanori Yoda, Atsushi Ishibe, Yuko Hidaka, Masanari Umemura, Toshio Takayama, Makoto Kaneko, Shigeru Miyagawa, Yoshiki Sawa, Itaru Endo and Yoshihiro Ishikawa in Cell Transplantation
Supplemental Material
Supplemental Material, sj-tiff-1-cll-10.1177_09636897211009559 - Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats
Supplemental Material, sj-tiff-1-cll-10.1177_09636897211009559 for Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats by Takashi Nakamura, Utako Yokoyama, Tomomitsu Kanaya, Takayoshi Ueno, Takanori Yoda, Atsushi Ishibe, Yuko Hidaka, Masanari Umemura, Toshio Takayama, Makoto Kaneko, Shigeru Miyagawa, Yoshiki Sawa, Itaru Endo and Yoshihiro Ishikawa in Cell Transplantation
Supplemental Material
Supplemental Material, sj-tiff-2-cll-10.1177_09636897211009559 - Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats
Supplemental Material, sj-tiff-2-cll-10.1177_09636897211009559 for Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats by Takashi Nakamura, Utako Yokoyama, Tomomitsu Kanaya, Takayoshi Ueno, Takanori Yoda, Atsushi Ishibe, Yuko Hidaka, Masanari Umemura, Toshio Takayama, Makoto Kaneko, Shigeru Miyagawa, Yoshiki Sawa, Itaru Endo and Yoshihiro Ishikawa in Cell Transplantation
Supplemental Material
Supplemental Material, sj-tiff-3-cll-10.1177_09636897211009559 - Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats
Supplemental Material, sj-tiff-3-cll-10.1177_09636897211009559 for Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats by Takashi Nakamura, Utako Yokoyama, Tomomitsu Kanaya, Takayoshi Ueno, Takanori Yoda, Atsushi Ishibe, Yuko Hidaka, Masanari Umemura, Toshio Takayama, Makoto Kaneko, Shigeru Miyagawa, Yoshiki Sawa, Itaru Endo and Yoshihiro Ishikawa in Cell Transplantation
Supplemental Material
Supplemental Material, sj-tiff-4-cll-10.1177_09636897211009559 - Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats
Supplemental Material, sj-tiff-4-cll-10.1177_09636897211009559 for Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats by Takashi Nakamura, Utako Yokoyama, Tomomitsu Kanaya, Takayoshi Ueno, Takanori Yoda, Atsushi Ishibe, Yuko Hidaka, Masanari Umemura, Toshio Takayama, Makoto Kaneko, Shigeru Miyagawa, Yoshiki Sawa, Itaru Endo and Yoshihiro Ishikawa in Cell Transplantation
Supplemental Material
Supplemental Material, sj-tiff-5-cll-10.1177_09636897211009559 - Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats
Supplemental Material, sj-tiff-5-cll-10.1177_09636897211009559 for Multilayered Human Skeletal Muscle Myoblast Sheets Promote the Healing Process After Colonic Anastomosis in Rats by Takashi Nakamura, Utako Yokoyama, Tomomitsu Kanaya, Takayoshi Ueno, Takanori Yoda, Atsushi Ishibe, Yuko Hidaka, Masanari Umemura, Toshio Takayama, Makoto Kaneko, Shigeru Miyagawa, Yoshiki Sawa, Itaru Endo and Yoshihiro Ishikawa in Cell Transplantation
Footnotes
Acknowledgments
We are grateful to Fumiko Kato for her excellent technical assistance.
Author Contributions
UY, IE, and YI conceived of the study. TN, UY, AI, and YI were involved in the study design and data analysis. TN, YH, and TY performed experiments. TK, TU, SM, and YS provided human skeletal muscle myoblasts and supported the technical issues of cell culture. TT and MK aided the periodic hydrostatic pressurization system. TN wrote the draft. UT and YI made a critical revision of the manuscript. All authors read and approved the final manuscript.
Ethical Approval
This study was approved by the Animal Care and Use Committee of Yokohama City University (Approval number: F-A-18-050). The protocol for using human skeletal muscle myoblasts was approved by the Osaka University Clinical Research Review Committee (Approval number: 16236-4).
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the Institutional Animal Care Guidelines of Yokohama City University, Japan, and approved by the Animal Care and Use Committee of Yokohama City University.
Statement of Informed Consent
Written informed consent was obtained from all donors for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (TN, JP18K15013; UY, JP17K19403, JP18K08767, JP20K21638; YI, JP19H036567, JP18KT0073), Japan Agency for Medical Research and Development (AMED) (YI, JP19ek0109240, JP19ek0210117, JP19lm0203087), a grant for Strategic Research Promotion of Yokohama City University (YI), and Kitsuen Research Foundation (YI, 2071000014). These funders had no role in this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
