Abstract
Transplantation of autologous skeletal myoblasts (SMBs) is a potential therapeutic approach for myocardial infarction. However, their clinical efficacy and safety is still controversial. Electrical coupling through gap junction between SMBs and host myocardium is essential for synchronized contraction and electrical stability. Here, we investigated the effect of heart beat-simulating environment, oscillating pressure, on the expression of connexin43 in two types of SMBs from rat and mouse. We found that connexin43 is markedly decreased under ischemia-mimicking conditions such as serum starvation and hypoxia (1% O2) in rat primary cultured SMBs and mouse C2C12 SMB cell line. Interestingly, the decrease of connexin43 expression under serum starvation was attenuated by oscillating pressure. Oscillating pressure treatment increased the expression of connexin43 twofold through AP-1 stimulation, which was blocked by PD98059, ERK inhibitor. In coculture of cardiomyocytes and C2C12, pressure-treated C2C12 and cardiomyocytes were able to form functional gap junction, which was demonstrated by both calcein-AM dye transfer assay and measurement of simultaneous contraction. In rat myocardial infarction model, transplantation of SMBs pretreated with oscillating pressure resulted in lesser ventricular dilatation and better systolic function than transplantation of untreated SMBs and control group. These results suggested that application of oscillating pressure on SMBs before transplantation may be useful to promote therapeutic efficacy for myocardial infarction by enhancing gap junction formation between transplanted and host cells.
Introduction
Cell transplantation is a promising therapeutic option for repairing the infarcted heart (3,11,15). Experimental studies for cell transplantation are performed using several cell types, including embryonic stem cells, fetal or neonatal cardiomyocytes, skeletal myoblasts (SMBs), and hematopoietic stem cells (5,15). Among these candidate cells, myoblasts have clinically attractive characteristics because of their ability to naturally generate a contractile phenotype, autologous origin, tolerance to ischemia, and their growth potential (8,20). Although skeletal myoblasts have been expected to be a good cell source for autologous transplantation, the safety and efficacy of their transplantation are still controversial. Transplantation of SMBs improved heart function in rabbit cryoinfarct model (30) as well as in human myocardial infarction (9). In recent clinical trials, however, SMBs injection after myocardial infarct failed to improve cardiac systolic function (17) and showed increased arrhythmia and sudden death (18). The mechanisms of ventricular arrhythmias associated with SMB transplantation are not fully understood yet.
In previous studies, SMBs could form functional gap junctions with cardiomyocytes in not only in an in vitro coculture system (24) but also in an in vivo model (29). However, newly formed gap junction between cardiomyocytes and SMBs seems to be maintained only transiently and eventually transplanted SMBs are electrically isolated from host cardiomyocytes. Several reports demonstrate that there is no functional communication between these cells in vivo (16,18). Because the heart functions by contraction and relaxation regulated through electromechanical coupling between cardiomyocytes (13), gap junction coupling between SMBs and cardiomyocytes is important for myocardial function and electrical stability. Thus, in order to improve safety and efficacy of SMB transplantation in myocardial infarction, strategies to enhance the formation of functioning gap junction would be critical.
The gap junction channel composed of connexins is important for electromechanical coupling of muscle cells. Three connexin isoforms are expressed by cardiac myocytes, connexins 43, 40, and 45 (31). Connexin43 is the dominant isoform in cardiac tissue and skeletal myoblasts. Connexin43 is essential for normal electrical conduction in the heart; mice with cardiac-restricted inactivation of connexin43 exhibit slower ventricular conduction and experience lethal arrhythmias (7). Overexpression of connexin43 in SMBs directly enhanced intercellular communication with adult cardiomyocytes, and engraftment of connexin43-expressing myocytes has the potential to reduce postinfarct arrhythmia (26,27). Overexpressing connexin43 in SMBs leads to enhanced cell-to-cell communication with cardiomyocytes and could improve cell integration in the myocardium after cell transplantation. Therefore, the exploration of the physiological and cellular mechanisms of the connexin43 expression in SMBs might be important for improving therapeutic efficacy of SMB transplantation for infarcted heart.
In this study, we evaluated effects of biological and physical environment of the infarcted myocardium on connexin43 expression on SMBs and its mechanisms. We tried to simulate the pathophysiological environment of the infarcted heart in vitro by exposing SMBs to hypoxia and serum deprivation as well as oscillating pressure, which are unique components experienced in the ischemic or infarcted myocardial environment. And we evaluated new strategy of pretreatment of SMBs with oscillating pressure to enhance the therapeutic efficacy for myocardial infarction.
Materials and Methods
Animal Care
All animal experiments were performed under approval from the Institutional Animal Care and Use Committee of Seoul National University Hospital. Animals were housed in Laboratory Animal Care Animal Facility of the Clinical Research Institute of Seoul National University Hospital.
Isolation of Rat Skeletal Myoblasts
SMBs were isolated from limbs of Fischer-344 rats as described previously with some modification (23). In brief, muscle tissues were digested for a total of 60–90 min with 0.2% collagenase type IV (Sigma) at 37°C. Digested tissues were gently minced using Pasteur pipette and transferred to new digestive solution. Tissues were digested again in 100 U/ml of collagenase type IV and 0.5 U/ml of dispase (Gibco) for 30 min at 37°C. After enzymatic digestion, the cells were filtered through a 40-μm cell strainer (BD Falcon), collected in the pellet, and washed twice with PBS. The cells were resuspended in F-10 media (Invitrogen Gibco) supplemented with 15% horse serum (Sigma) and 6 ng/ml bFGF (Invitrogen Gibco) and attached on the 1.5% of gelatin-coated dish for 2 h to avoid fibroblast contamination. Suspended cells were transferred and grown on the new 1.5% of gelatin-coated dish. For skeletal myotube formation, SMBs were cultured in differentiation induction media, F-10 media supplemented with 1.5% horse serum, 6 mg/ml insulin, 0.9 mM CaCl2. Characterization of isolated SMBs was processed through antimyosin heavy chain immunostaining. Over 90% cells of total cells were myosin heavy chain-positive myoblasts; the other cells were presumably fibroblasts.
Isolation of Neonatal Rat Cardiomyocytes
Neonatal rat cardiomyocytes were prepared as previously described (32). Briefly, hearts of 2-day-old Fischer-344 rats were digested using 0.1% trypsin in DMEM overnight at 4°C. Digested heart tissues were transferred and further digested in 0.1% collagenase type II at 37°C for 30 min. The dissociated cells were collected as pellet, resuspended, and preplated to remove the non-cardiomyocytes. The nonadherent cardiac myocytes were collected and cultured in DMEM/F12 containing 15 mM HEPES, 5% horse serum, insulin, transferrin, sodium selenite (ITS, Gibco), and 1% penicillin/ streptomycin (Gibco Invitrogen).
Cell Culture, Serum Starvation, Hypoxia, and Oscillating Pressure Chamber Condition
Mouse C2C12 myoblasts were grown in DMEM supplemented with 10% FBS (Gibco Invitrogen) and 1% antibiotic-antimycotics (Gibco Invitrogen). For serum starvation, rat SMBs and mouse C2C12 cells were cultures in F-10 media with 1.5% of horse serum and DMEM with 1% of FBS, respectively. For hypoxic condition, cells were incubated in a hypoxic chamber (Forma Scientific) that maintained a low oxygen tension (5% CO2 with 1% O2 balanced with N2). Oscillating pressure chamber was constructed as the model previously described with a little modification (4,19). Intrachamber pressure was regulated with check valve system and the pressure level was continuously monitored. The pressure cycle was at the trough pressure of 5–15 mmHg and the peak pressure of 120–150 mmHg and 150 cycles/min. Oscillating pressure 1 cycle is designed as 3-h pressing and 1-h resting (Supplementary Fig. S1). The pressure chamber was placed in a humidified 37°C cell incubator with 5% CO2.
For myocardial protein extraction, rat hearts subjected to myocardial infarction induction were obtained at postinfarction day 3, rinsed in PBS, and then frozen with liquid nitrogen. Frozen heart tissue was chopped into very small pieces, minced into powder, and placed in cell lysis buffer (0.075 M potassium acetate, 0.3 M NaCl, 0.1 M l-arginine basic salt, 0.01 M EDTA-HCl, 0.25% Triton-X100, pH 7.4). The mixture was centrifuged at 4°C, 1500 rpm for 20 min. The supernatant was dialyzed and isolated myocardial proteins were added to media at the concentration of 25 μg/ml.
RT-PCR Analysis
Total RNA from SMBs and C2C12 cells was prepared using TriZol (Gibco Invitrogen) according to the manufacturer's instruction. Genomic DNA was eliminated by RQ1 RNase-free DNase treatment (Promega) and cDNA was synthesized from 2 μg of total RNA with AMLV-RT (Promega). Oligonucleotide primers for PCR were designed as follows: connexin43, sense 5′-GACTTCAGCCTCCAAGGATTCCACC-3′ and antisense 5′-AGTTGGAGATGGTGCTTCCGGCC-3′; GAPDH, sense 5′-CCTCTGGAAAGCTGTGGCGT-3′ and antisense 5′-TTGGAGGCCATGTAGGCCAT-3′. The PCR products were separated on 1.2% agarose gels and visualized using ethidium bromide staining under UV transillumination.
Western Blot Analysis and Immunofluorescence Staining
Cells were lysed for 20 min in lysis buffer containing protease inhibitors (50 mM Tris, pH 7.2, 250 mM NaCl, 1% NP40, 0.05% SDS, 2 mM EDTA, 0.5% deoxycholic acid, 10 mM β-glycerol phosphate, 1 mM sodium orthovanadate, 100 mM NaF) and protease inhibitor cocktail (Roche), and centrifuged for 20 min at 14,000 rpm. Total protein (5–10 μg) were electrophoresed, transferred to PVDF membrane (Millipore), and the blot was immunostained with specific primary antibodies overnight at 4°C. Membranes were incubated with horseradish peroxidase-conjugated secondary antibodies (Amersham Biosciences) for 1 h at room temperature and immunoreactions were detected using the ECL kit (Amersham Bioscience). JNK inhibitor, SP600125 (A.G. Scientific, Inc.), and MAPK inhibitor, PD98059 (A.G. Scientific, Inc.) were used at 10 μM. These drugs were added to the cells 30 min before exposing the cells to oscillating pressure. Connexin43 (Santa Cruz); pc-Jun (Santa Cruz); c-Jun (Santa Cruz); c-Fos (Calbiochem); p-JNK (BD Biosciences); p-ERK (Cell Signaling); α-tubulin (Calbiochem); JNK (Santa Cruz); ERK (Cell Signaling).
For immunofluorescence staining, cells were washed twice with PBS, fixed with 100% methanol for 20 min at −20°C, and washed a further three times with PBS. After blocking with 1% of BSA/PBS for 1 h, cells were labeled with anti-connexin43 (Santa Cruz) and anti-cardiac TnT (Santa Cruz) overnight at 4°C. Cells were rinsed three times with 0.1 % Tween 20/PBS then incubated in secondary antibody solutions, donkey anti-rabbit Alexa Fluor 488 and donkey anti-goat Cy3. The nuclei were stained with DAPI (1 μg/ml in PBS) for 10 min, washed, and mounted using fluorescent mounting medium (DAKO Cytomation). Examinations were done using a fluorescence microscope (Olympus IX71, Japan) and confocal microscopy (NIKON PE2000, Japan).
Coculture of Cardiomyocytes with Myoblasts and Dye Transfer
For qualitative assessment of the gap junction communication between cardiomyocytes and myoblasts, dye transfer analysis was performed (4,6). Neonatal cardiomyocytes were cultured with C2C12 myoblasts cells or oscillating pressured C2C12 cells in DMEM/F12 containing 15 mM HEPES, 5% horse serum, insulin, transferrin, sodium selenite (ITS, Gibco Invtrogen), and 1% penicillin/streptomycin (Gibco Invitrogen). C2C12 cells were labeled with 2 μg/ml of DiI (Molecular Probes Inc.) and 5 μg/ml of calcein-AM (calcein acetoxymethyl-ester, Molecular Probes Inc.) and cultured with cardiomyocytes on 60 μ-Dish for Live Cell Analysis (ibidi, DE). After 48-h coculture, dye transfer was evaluated under fluorescence microscope (Olympus 1X71, Japan).
Rat Myocardial Infarction Model and SMB Transplantation
Ten-week-old Fisher-344 female rats were supplied from Central Lab. Animal Inc. Rats underwent surgery to induce myocardial infarction by ligation of the left coronary arteries or sham operation as described previously (4). One week after MI induction, rats were randomized into three groups: rats receiving control media only (control group), untreated SMBs (SMB group), and both pressure and myocardial protein-treated SMBs (pretreated SMB group). Pretreated SMB was cultured under oscillating pressure and myocardial protein contained culture media for 2 days before transplantation. Under anesthesia, each rat received four injections (total of 2 × 106 SMBs) into the peri-infarct area. Left ventricular systolic function was assessed by echocardiography (Sequoia, Acuson Corp., Mountain View, CA) at baseline, before SMB transplantation, and 8 week after SMBs transplantation.
Data Analysis and Statistics
Quantification of band intensity was analyzed using TINA 2.0 (RayTest) and normalized to the intensity of GAPDH or α-tubulin. All data are presented as median (range). Kruskal-Wallis test was used for comparison among three groups and a Mann-Whitney U-test for comparison between two groups. Bonferroni correction was performed in case of multiple comparisons. A probability value of p < 0.05 was considered significant.
Supplemental Materials
Supplementary material consists of two figures(Supplementary Fig. S1: http://hottopics.snuh.org/upload/Kim_HS_Supplementary_FiguresS1.pdf and Supplementary Fig. S2: http://hottopics.snuh.org/upload/Kim_HS_Supplementary_FiguresS2.pdf) and three videos (Movie M2a: http://www.liveto.com/m2comm/20090421s/CT-S2a.asx;MovieM2b: http://www.liveto.com/m2comm/20090421s/CT-S2b.asx;MovieM2c: http://www.liveto.com/m2comm/20090421s/CT-S2c.asx) as evidence for electrical coupling of cardiomyocytes and skeletal myoblasts.
Results
Connexin43 Expression on SMBs Was Reduced Under Serum Starvation and Hypoxic Condition
For success of SMB transplantation and synchronized contraction, electrical coupling through gap junction channel between SMBs and host myocardium is essential (28). We investigated influences of ischemic environment on connexin43 expression in SMBs (Fig. 1A, B). Rat primary cultured SMB and mouse C2C12 SMB cell line were evaluated in hypoxic and serum-deprived settings, which mimic ischemic myocardial environment. Primary rat SMBs and mouse C2C12 myoblasts expressed connexin43, yet connexin43 was markedly downregulated soon after incubation in serum starvation condition (Fig. 1A). Indeed, the expression of connexin43 showed quick reduction under hypoxic (1% O2) condition (Fig. 1B).
Reduction of connexin43 expression under ischemia mimic conditions in skeletal myoblasts. Western blot analysis of connexin43 in rat skeletal myoblasts (rSMB) and mouse skeletal myoblast (mC2C12) under serum starvation (A) and low oxygen (B) condition. Each sample (5 μg of total soluble proteins were used in SMBs, and 10 μg of proteins were used in C2C12 cells) was loaded for gel electrophoresis on 10% PAGE. Connexin43 was expressed at high level under normal condition, which was markedly decreased under serum starvation (serum starv.) and low oxygen (1% O2) conditions. Two independent experiments were performed and α-tubulin was used as loading control.
These results suggest that ischemic myocardial environment can unfavorably influence gap junction formation between the transplanted SMBs and cardiomyocytes because of the strong and rapid reduction of connexin43 in SMBs.
Exposure of SMBs to Oscillating Pressure Upregulated Connexin43 Expression and Delayed Connexin43 Reduction Under Serum Starvation Condition
We investigated whether heart beat-simulating environment, oscillating-pressure, would enhance connexin43 expression in myoblasts (Fig. 2). Expression of mRNA and protein of connexin43 increased about twofold by oscillating pressure in both rat SMBs and C2C12 (Fig. 2A, B). Oscillating pressure increased connexin43 expression in different types of cells (primary culture vs. cell line) and species (rat vs. mouse). In addition, the expression of connexin43 was continuously increased by increment of oscillating pressure exposure (Supplementary Fig. S1).
Increase of connexin43 mRNA and protein expression by exposure to oscillating pressure in both SMBs and C2C12. (A) When rSMBs and mC2C12 were cultured in oscillating pressure chamber, connexin43 protein and mRNA were upregulated. Representative Western blots and RT-PCR results are shown (n = 4). (B) Additional treatment of protein extracts from infarcted rat heart on oscillating pressure, which is ex vivo simulation of “in vivo infarcted heart,” did not show any additive effect on connexin43 expression. In other words, oscillating pressure alone is enough. Prs, oscillating pressure; PE, protein extracts from infarcted rat heart (25 μg/ml), PP, pressure and infarcted myocardial protein. α-Tubulin and GAPDH served as the controls for same protein and RNA levels. The quantification of the connexin43 immunoblot from two independent experiments is shown on the bottom. (C) Immunofluorescence analysis showed that connexin43 (green) expression on SMBs increased after exposure to oscillating pressure. Three independent experiments were performed (scale bar: 5 μm). (D) Pretreatment of oscillating pressure attenuates the decline of connexin43 induced by serum starvation. SMBs were pretreated with oscillating pressure then incubated in serum starvation condition for indicated days (3, 6, or 10 days). Quantitative graph from two independent experiments is shown below.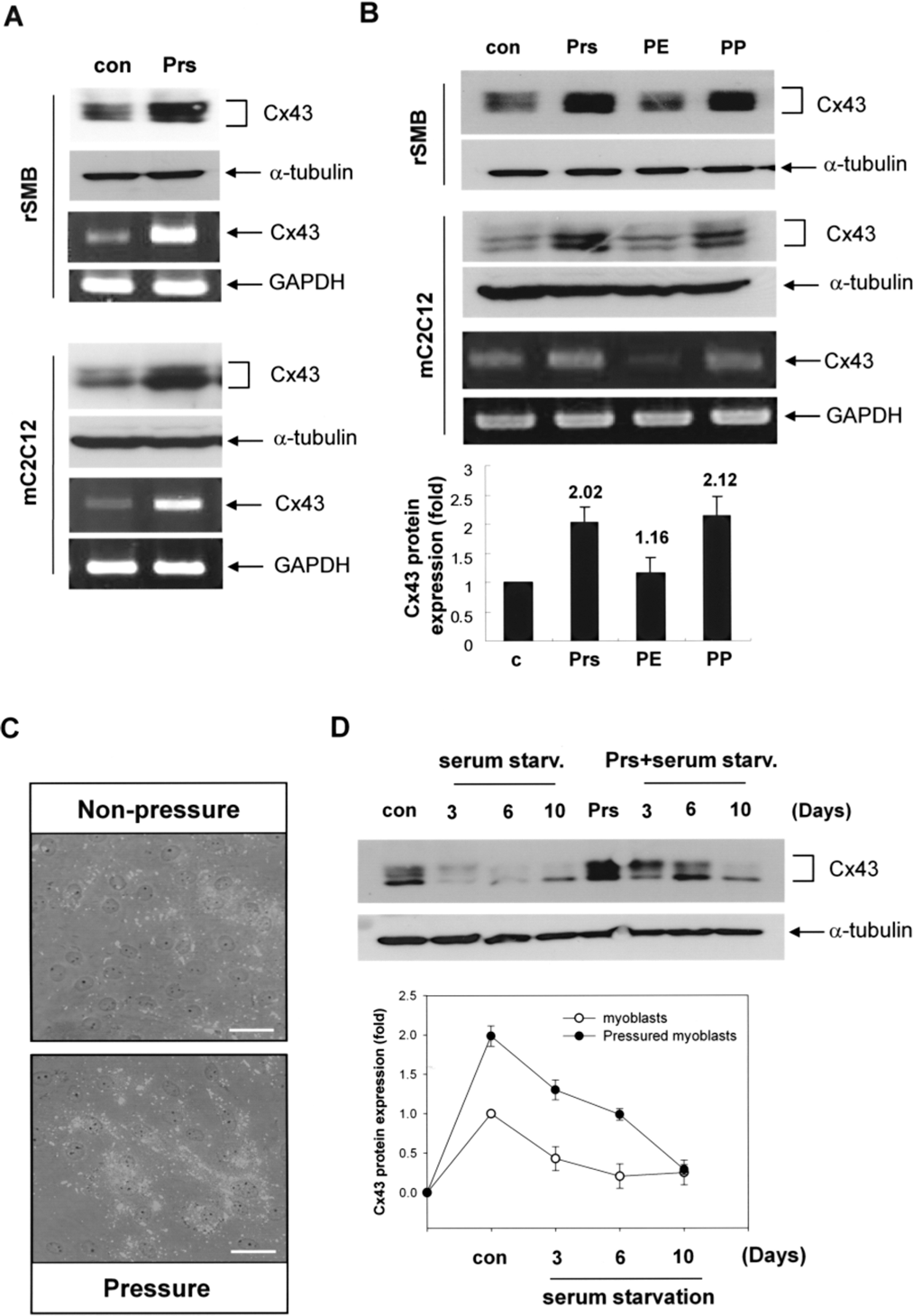
In order to test the in vitro biological and physical simulation for “the infarcted heart,” we evaluated effects of combination of oscillating pressure and the infarcted myocardial protein extracts on connexin43 expression in myoblasts (Fig. 2B). Although pressure increased connexin43 expression strongly, myoblasts cultured with the infarcted myocardial extracts showed minimal increase of connexin43 expression. Addition of the infarcted myocardial protein on oscillating pressure did not show additional upregulation of connexin43 in SMBs compared to oscillating pressure alone (Fig. 2B). Immunofluorescence staining against connexin43 also show that connexin43 increased after exposure to oscillating pressure in rat SMBs (Fig. 2C) and mouse C2C12 (data not shown). Thus, oscillating pressure might be a general enhancer for connexin43 expression in different skeletal myoblast cell types such as mouse/rat and cell line/primary cells.
We next examined the effect of oscillating pressure treatment on the SMBs under serum starvation (Fig. 2D). Connexin43 protein is markedly decreased under serum starvation. Interestingly, pretreatment SMBs with oscillating pressure showed delayed connexin43 reduction under serum starvation, suggesting that preconditioning of SMBs with oscillating pressure might give more chance to form electrical coupling with host cardiomyocytes after transplantation into myocardium.
Oscillating Pressure Upregulated Connexin43 via AP-1
Because connexin43 promoter activity is determined by the transcription factor AP-1 (31,33), we tested whether AP-1 is regulated by oscillating pressure (Fig. 3). Oscillating pressure markedly enhanced c-Fos and phosphorylated c-Jun, which are the components of AP-1 (Fig. 3A). Because the expression and activation of c-Fos and c-Jun are regulated by extracellular-regulated kinase (ERK) and c-Jun N-terminal kinase (JNK) activation (34), we examined whether oscillating pressure affects ERK and JNK activity (Fig. 3B). Pressure increased the phosphorylation of both ERK and JNK. But only PD98059, an inhibitor of ERK, reversed connexin43 expression, whereas JNK inhibitor, SP600125, did not. These results suggest that connexin43 expression by oscillating pressure is regulated through AP-1 activation via at least ERK pathway in myoblasts.
Mechanism for induction of connexin43 by oscillating pressure in SMBs. (A, B) C2C12 cells were cultured in pressure condition, and the activation of c-Jun, c-Fos, JNK, and ERK was determined by Western blotting. SP600125 (10 μM) or PD98059 (10 μM) was incubated for 30 min before exposure to pressure condition. The quantification of the intensity from three independent experiments is shown on the right. (A) Oscillating pressure markedly enhanced c-Fos and phosphorylated c-Jun, which are required for AP-1 transcription factor activity. (B) In the analysis of signal transduction pathway, pressure increased phosphorylation of ERK and JNK. But only PD98059, an inhibitor of ERK, reversed connexin43 expression, whereas JNK inhibitor, SP600125, did not.
Pretreatment of SMBs with Oscillating Pressure Enhanced Gap Junction Formation with Cardiomyocytes
We check whether pressure preconditioned myoblasts can form gap junction with adjacent cardiomyocytes in vitro. We first confirmed localization of connexin43 expression between myoblasts and cardiomyocytes in a co-culture system (Fig. 4A). C2C12 cells were pretreated with oscillating pressure before coculture with cardiomyocytes to facilitate gap junction formation by enhancing connexin43 expression. C2C12 (blue nucleus, arrows in Fig. 4A) lay near cardiomyocytes that stained with cardiac troponin T (cTNT) (red cytosol in Fig. 4A). Confocal microscopy revealed the expression of connexin43 at contact sites between C2C12 and cardiomyocytes (arrowheads in Fig. 4A).
Evidences for the formation of functional gap junctions between pressure-pretreated SMBs and cardiomyocytes in a coculture system. (A) Confocal microscopy of cardiomyocytes cocultured for 2 days with pressure-treated C2C12. Cells were fixed after coculture and stained for cTNT (red) and connexin43 (green). (a) Arrows indicates C2C12 cells (blue nucleus) near cardiomyocyte (red cytoplasm). Gap junction marked with green connexin43 (arrowheads) is formed between cardiomyocytes expressing cTNT (red) and C2C12 cells. (b) 3D confocal microscopic image shows that connexin43 is indeed detectable in the interface between cardiomyocytes and C2C12. Scale bars: 5 μm. (B) Dye transfer studies in coculture of cardiomyocytes and pressure-treated C2C12 (a–d) or naive C2C12 (e–h) skeletal myoblasts at day 2. Before coculture, myoblasts were stained with both DiI (red) and calcein-AM dye (green). (a–d) Green fluorescence dye was transferred from pressure-treated C2C12 to cardiomyocyte. (c) Beating cardiomyocyte uptaking green fluorescence from C2C12 cells is marked (**). (e–h) In contrast, dye transfer was not shown in coculture of naive C2C12 cells and cardiomyocytes. (g) Beating cardiomyocyte not uptaking green fluorescence from C2C12 cells is marked (*). Arrows indicate C2C12 myoblasts doubled stained with DiI and calcein (orange). Scale bars: 5 μm. Supplemental videos further depicting Figure 4 are available.
Secondly, to test whether the pressure-pretreated SMBs can form functional gap junction with cardiomyocytes, we performed calcein-AM dye transfer experiment (Fig. 4B). The unlabeled cardiomyocytes were cocultured with C2C12 labeled with a combination of DiI and calcein-AM; DiI is not transferable through gap junctions whereas calcein-AM can pass through gap junctions. Cardiomyocytes stained with green fluorescence (calcein-AM) without red fluorescence (DiI) represent cardiomyocytes that received calcein via functional gap junctions from the dual-labeled C2C12 myoblasts. We found that some beating cardiomyocytes were green fluorescence positive (asterisks in Fig. 4Bc, Bd) near the dual-labeled C2C12 (orange; arrows in Fig. 4Ba, Bb), suggesting that cardiomyocytes and C2C12 pretreated with oscillating pressure can form functional gap junction between them. There were also beating cardiomyocytes with green fluorescence negative (asterisk in Fig. 4Bg, Bh), indicating no gap junction formation between C2C12 skeletal myoblasts and cardiomyocytes. The beating cardiomyocytes with green fluorescence ere more frequent when cocultured with C2C12 preex-posed to oscillating pressure.
However, C2C12 did not show their own spontaneous contractions. They showed contraction-like movement in accordance with contraction of cocultured cardiomyocytes (Supplementary Fig. S2). In Figure S2, we were able to see DiI-labeled C2C12 (asterisk in Fig. S2) attaching to cardiomyocytes that were actively contracting (arrows in Fig. S2). Thus, SMBs pretreated with oscillating pressure can form functional electromechanical junctions and perform synchronous contractile movements with cardiomyocytes via gap junctions, suggesting that oscillating pressure on SMBs might be applicable for enhancing the therapeutic efficacy of SMBs transplantation for salvaging infarcted myocardium.
Pretreatment of SMBs with Oscillating Pressure Improved the Therapeutic Efficacy of Transplantation in Myocardial Infarction
We tested the effect of SMBs transplantation on myocardial repair in rat MI model (Fig. 5). We pretreated SMBs with oscillating pressure and infarcted myocardial protein extracts, then evaluated effects on improvement of cardiac function. Even though the combined treatment of infarcted myocardial protein extracts with oscillating pressure showed minimal additional effects on connexin43 expression in vitro, we treated both oscillating pressure and myocardial proteins extracts before transplantation to maximize connexin43 expression on SMBs. In rat MI model, change of left ventricular systolic function and remodeling showed significant difference among three groups (p = 0.012 for change of left ventricular ejection fraction and p = 0.003 for change of left ventricular end systolic dimension with Kruskal-Wallis test). Transplantation of untreated SMBs (naive SMBs) improved left ventricular remodeling compared to control group, which received media injection only [change of left ventricular end systolic dimension in the control vs. the untreated SMBs: +14.0mm (8.0, 16.0) vs. −1.5 mm (-6.0, 3.0), p = 0.032; change of left ventricular ejection fraction: −15.4% (-29.8, −10.5) vs. −0.5% (-15.2, 6.5), p = 0.126]. Transplantation of treated SMBs was significantly more effective in preventing left ventricular dilatation than naive SMBs and in improving systolic function than control [change of left ventricular systolic dimension in the untreated SMBs vs. treated SMBs: −1.5 mm (-6.0, 3.0) vs. −11.0 mm (-17.0, −8.0), p = 0.032; change of left ventricular ejection fraction in the control vs. treated SMBs: −15.4% (-29.8, −10.5) vs. +7.1% (-3.0, 19.4), p = 0.016]. Additionally, the area of viable grafted SMBs in the heart transplanted with the treated SMBs is significantly larger than that with untreated SMBs (p < 0.05, data not shown).
Pretreatment of SMBs improved the therapeutic efficacy to salvage the infarcted rat myocardium. (A) Schematic time table of rat MI surgery, cell transplantation, and echocardiographic examinations. (B, C) Postinfarction echocardiographic results before and 7 weeks after SMBs transplantation to the infarcted rat heart. Circles and bars indicate median value and range, respectively. Sham, sham operated group; PP, SMBs treated with oscillating pressure and myocardial protein extract; S, SMBs alone; Con, control with media injection. LVESD, left ventricle end-systolic dimension; EF, left ventricular ejection fraction. Transplantation of SMBs improved LV contractility and prevented LV dilatation in comparison with control group. Such therapeutic effects of cell transplantation were significantly potentiated by pretreatment of SMBs before transplantation.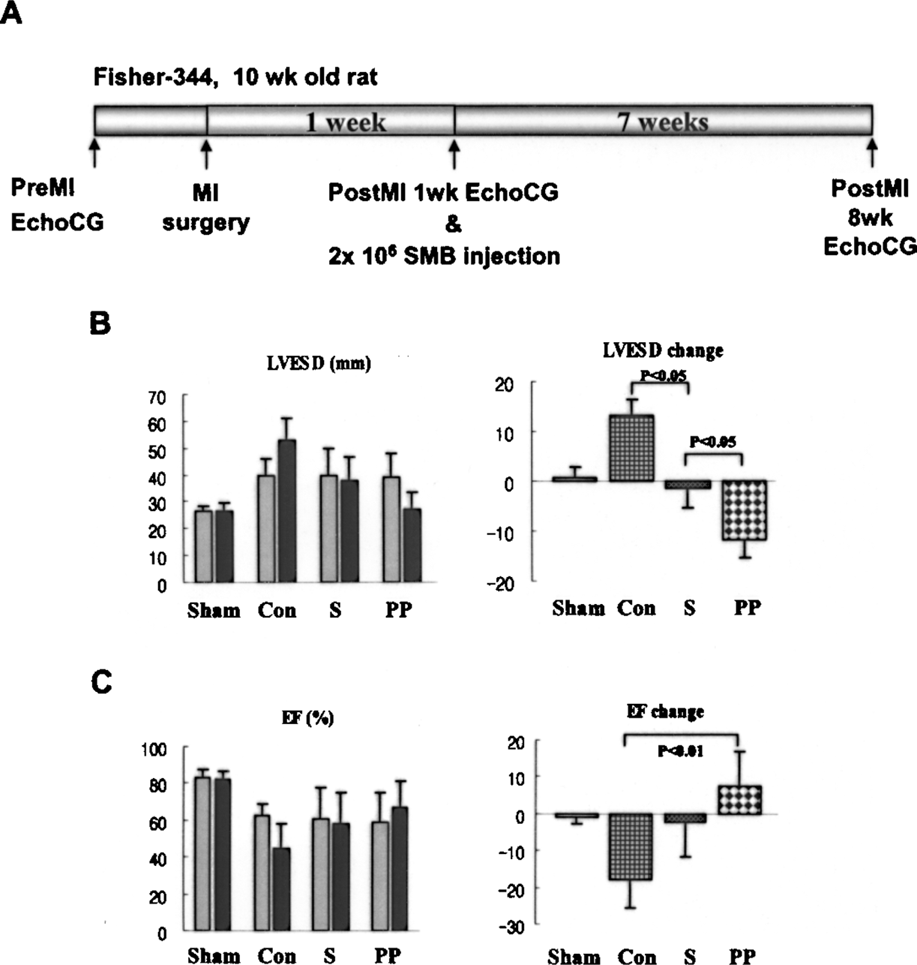
Discussion
The novel findings of this study are as follows. 1) Connexin43 expression on SMBs is decreased in serum-deprived or hypoxic condition that is an important pathobiologic component of ischemic myocardium. 2) Such reduction of connexin43 was relieved by pretreatment of SMBs with oscillating pressure, which was mediated by AP-1 and ERK-dependent pathways. 3) Transplantation of SMBs pretreated with oscillating pressure achieved a greater therapeutic efficacy than naive SMBs in improving heart function after myocardial infarction.
Pathophysiological Microenvironments of the Ischemic Heart Are Important Modulators of Gap Junction Formation and Integration Between Skeletal Myoblasts and Resident Cardiomyocytes
Although most of preclinical studies showed favorable effects of SMBs transplantation in myocardial infarction (9,18,30), recent clinical trial reported disappointing results in terms of recovery of cardiac systolic function (17). For successful SMB transplantation in myocardial infarct patients, the fundamental understanding of the mechanisms involved in the intercellular communication between SMBs and host cardiomyocytes may have crucial importance.
Our study showed that skeletal myotube can form gap junction with cardiomyocytes, and beating together in in vitro condition (data not shown). However, connexin43, which is an important component of gap junction formation for synchronized heart beating (7,31), dramatically decreased in condition such as serum starvation and hypoxia, which are biologic features of ischemic myocardium (Fig. 1A, B). Interestingly, connexin43 expression is markedly enhanced by heart beat-simulating pressure condition (Fig. 2). Thus, pretreatment of SMBs with oscillating pressure significantly retarded the reduction of connexin43 under serum starvation condition (Fig. 2D).
Several Potential Benefits From Induction of Connexin43 Expression on SMBs Before Transplantation to Infarcted Heart
In a previous study (12), it took over 2 weeks for grafting skeletal myoblasts to proliferate and differentiate into myotubes, and finally integrate into infarct heart tissue. In other words, new myotubes should have a period of time after transplantation to make new contacts with the resident cardiomyocytes via putative gap junctions that are composed of connexin43. It could explain that pressure-pretreated SMBs show better recovery of cardiac systolic function (Fig. 5B). Because SMBs treated with oscillating pressure were able to maintain connexin43 expression, which otherwise decreased under ischemic microenvironment, they can have time to form gap junction with neighboring cardiomyocytes. We suggest that commitment of SMBs with pressure before transplantation would help SMBs to incorporate more easily into host myocardium.
Secondly, the functioning gap junction between transplanted SMBs and cardiomyocytes has a potential to prevent apoptotic death of cardiomyocytes via transferring prosurvival factors from SMBs. In our previous study (10), transplanted mesenchymal stem cell can salvaged endangered cardiomyocytes by transferring prosurvival factors through gap junction in the situation of myocardial infarction.
Thirdly, when SMBs were transplanted, connexin43 protein decreased in transplanted SMBs, which can induce arrhythmia (7). To prevent the decline of connexin43 in SMBs, several experiments were tried to overexpress connexin43 by gene manipulation (21,25). It was reported that SMBs stably overexpressing connexin43 enhanced the formation of active electrical communication and the steady-state conductance between SMBs and cardiomyocytes (26,27). Connexin43 overexpression has antiarrhythmic effects in coculture (1). Our finding about connexin43 upregulation by pressure and prolonged existence of connexin43 protein in ischemic condition suggests that pretreatment of SMBs with oscillating pressure may have beneficial effects on prevention of arrhythmia after transplantation of SMBs to the infarcted heart.
Regulatory Mechanisms of Connexin43 Upregulation by Oscillating Pressure
There have been few studies to investigate the relationship between mechanical stresses and connexin43 expression. In cardiomyocytes, mechanical stretch increased the expression of connexin43 via c-Fos upregulation (33), and in endothelial cells stretch and shear stress induced the expression of connexin43 (14). In our study, oscillating pressure increased connexin43 in SMBs (Figs. 2 and 3). These results from different studies suggest that cells respond to mechanical stimuli with modulating connexin43 and gap junction formation.
Connexin43 promoter contains SP1, SP3, and AP-1 binding element (31). We observed that AP-1 was important in connexin43 expression in SMBs. Figure 3A shows that oscillating pressure upregulates and activates c-Jun and c-Fos, the molecular constituents of AP-1 heterodimers, leading to induction of connexin43 promoter activity through AP-1 binding site. Regarding the upstream signaling pathway of AP-1/connexin43, one study demonstrated that connexin43 transcription was upregulated by the Ras-Raf-MAPK pathway in mouse fibroblasts (2), whereas another showed that it was downregulated by JNK pathway in cardiomyocytes (22). We showed that pressure-induced connexin43 expression in SMBs was blocked by ERK inhibitor but not by JNK inhibitor (Fig. 3B), indicating that pressure signal for connexin43 upregulation is mediated at least by ERK pathway in SMBs. Taken together, the upstream signaling pathways to control AP-1/connexin43 expression vary depending on the class of stimulation and cell types.
In summary, the pathophysiologic microenvironment of ischemic heart (i.e., hypoxia and nutritional deprivation) may inhibit connexin43 expression of transplanted SMBs, leading to poor engraftment or survival of transplanted SMBs. But pretreatment of SMBs with oscillating pressure enhanced connexin43 expression through ERK/AP-1 pathways. Preconditioning SMBs with oscillating pressure potentiates the cellular coupling with cardiomyocytes, possibly leading to the improved survival of SMBs and electrical communication between two cell types. This could be one of the promising strategies to overcome the potential arrhythmogenecity of SMBs transplantation as well as to enhance its therapeutic efficacy for the patients with myocardial infarction.
Footnotes
Acknowledgments
This study was supported by a grant for Stem Cell Research Center (SC4210) and the Innovative Research Institute for Cell Therapy (A062260).
