Abstract
Rheumatoid arthritis (RA) is an inflammatory disease of the joints, which causes severe pain and excessive systemic circulation of harmful inflammatory cytokines. Current treatments are limited, with some patients not responding well, and some experiencing severe and detrimental side effects. Mesenchymal stem cells (MSC) are cell-based therapeutics being evaluated as potent immunomodulators in RA and may provide relief to patients not responding well to drug-based treatments. We evaluated the safety and efficacy of BX-U001 human umbilical cord tissue–derived mesenchymal stem cells (hUC-MSC) to treat RA, in support of a successful investigational new drug application. A collagen-induced arthritis (CIA) mouse model of RA was established in DBA/1 J mice. Mice from the treatment assessment group were given a tail vein infusion of hUC-MSC 24 days after primary RA induction, while control assessment (CA) group mice were given cell-free carrier solution. All animals were evaluated daily for RA symptoms via clinical scoring, blood was taken periodically for cytokine analysis, and mice were dissected at end point for histological analysis. A linear mixed model was used to compare the rate of change among groups. The clinical scores of TA group were significantly reduced compared with CA group (P < 0.01), indicating therapeutic effects. The histological scores of the joints in TA group were significantly lower than those in the CA group (P < 0.05), but had no significant difference compared with Healthy groups (P > 0.05). The concentration of (interleukin) IL-6 in TA group was significantly reduced by 80.0% (P < 0.0001) 2 days after treatment and by 93.4% at the experimental endpoint compared with levels prior to hUC-MSC injection. A single intravenous infusion of hUC-MSC (2 × 106 cells/mouse), to CIA-induced DBA/1 J mice, resulted in significant alleviation of RA symptoms and may provide significant therapeutic benefits in humans.
Keywords
Introduction
Arthritis affects over 54 million Americans and can lead to rheumatoid arthritis (RA) 1,2 . RA is an inflammatory disease that affects between 1.5 and 3 million Americans and is characterized by inflammation of the synovial and cartilage tissue, causing damage to the articular cartilage and bone 3 –8 . RA can lead to reduced quality of life and serious damage to organ systems, caused by the high level of inflammatory cytokines circulating in the blood 9,10 . The causes of RA are still being elucidated, but there is a clear inflammatory cascade that involves the production of tumor necrosis factor alpha (TNF-α) and interleukin 6 (IL-6), following the production of autoantibodies including “Rheumatoid Factor” and anti-citrullinated protein autoantibodies 5,6,11 . The pro-inflammatory cytokines and autoantibodies activate macrophages to infiltrate and damage the tissues of the joint, while simultaneously producing more inflammation 12 –15 . There are also autoantibody-negative cases of RA, in which disease is characterized by inflammation and fibrosis, developed from the infiltration of fibroblast-like synoviocytes and increased thickening of synovial lining 16,17 . While autoantibody-positive RA is associated with worse prognosis, both types of disease result from macrophage-dependent inflammation 12 –18 .
One of the problems with treating autoimmune diseases, such as RA, is that the immune system is complex, and pharmaceutical drugs and biologics tend to target only one or a small subset of the system 19 . The current standard treatment regime varies by country, but is generally reliant on methotrexate and biologics (for instance, antibodies against TNF-α), but nonsteroidal anti-inflammatory drugs and corticosteroids are still common treatments 20 . These treatments are not at all targeted toward the specific causes of RA, cause immunosuppression, and have significant side effects 21,22 . There is great need for more effective therapeutics, and treatments that do not produce systemic immunosuppression 23 .
Stem cell therapies have the potential to treat and manage autoimmune disorders in ways that conventional drugs cannot, because stem cells can harness the complex mechanisms that the body naturally uses to modulate and restore the imbalanced immunity with minimal toxicity 21 . In particular, mesenchymal stem cells (MSC) have been shown to have immunomodulatory effects on many types of immune cells and have limited side effects in past preclinical and clinical studies 22 . MSC are multipotent cells that can be readily isolated from many different adult tissues including fat, bone marrow, blood, and umbilical cord 21 . These cells can then be expanded in vitro to serve as “off-the-shelf” therapeutics. Human umbilical cord tissue–derived MSC (hUC-MSC) have been shown to have strong immunomodulatory effects and may have higher therapeutic potential for RA than bone marrow–derived MSC 22 . Due to MSC’s immune-evasive and immunomodulatory properties, they do not have the major graft-vs-host issues that other cell types do, making them amenable to allogenic transplantation. MSC have been shown to affect CD4+/CD8+ T cells, T regulatory cells (T-reg), B cell, monocytes, natural killer cells, and macrophages. The immunomodulatory mechanism of action is by direct contact inducing apoptosis of effector lymphocytes, and secretion of soluble mediators such as indoleamine 2,3-dioxygenase, prostaglandin E2, IL-6, and HLA-G5, which are known to dampen immune response 24 . This multipronged immunomodulatory power makes these cells capable of treating diseases caused by many different cell types. Previous preclinical animal studies have consistently shown reduction of RA symptoms upon treatment with MSC, regardless of the animal model used 22 . This preclinical study evaluates the potential of a new MSC product derived from human umbilical cord tissue (BX-U001) to treat a collagen-induced arthritis (CIA) mouse model of RA, and led to the successful filing of an investigational new drug (IND) application with the US Food and Drug Administration to initiate a clinical trial.
Materials and Methods
Isolation and Culture of hUC-MSC
The hUC-MSC used in this study were isolated and cultured from umbilical cord tissue by a standard tissue explant method
25
. Cells were cultured in hUC-MSC growth medium (Baylx, USA) at 37
hUC-MSC Differentiation Assays
For osteogenic and adipogenic differentiations, hUC-MSC were seeded onto 24-well tissue culture plates with 6 × 104 cells/well and incubated at 37°C in a humidified atmosphere containing 5% CO2. The differentiation induction medium from the Mesenchymal Osteogenesis Differentiation Kit and Mesenchymal Adipogenesis Differentiation Kit (EMD Millipore, Germany) were added after the confluence reached 90%. The medium was changed every 2 to 3 days based on the manufacturer’s protocol and adipogenic differentiation was confirmed by the deposition of lipid droplets in the cytoplasm using Oil Red O staining (EMD Millipore, Germany). The osteogenic differentiation was confirmed by positive staining of the extracellular calcium matrix using Alizarin Red S staining (EMD Millipore, Germany). Images of the stained samples were collected using a Nikon Ti-E microscope (Nikon, Japan).
Flow Cytometry
The phenotype of hUC-MSC was analyzed by flow cytometry after trypsinization. MSC were stained with fluorescein isothiocyanate-conjugated antibodies against CD34 and HLA-DR, or phycoerythrin-conjugated antibodies against CD45, CD90, CD105, and CD73. Mouse isotypic antibodies served as the control. Cells were stained in single label according to the manufacturer’s instructions and then analyzed by flow cytometry on a BD Accuri C6 flow cytometer (BD Biosciences, USA). Flow cytometry data were processed using FlowJo (FlowJo, USA).
hUC-MSC Potency Assay
hUC-MSC (1 × 104 cells/well) were co-cultured with anti-CD3/anti-CD28/IL-2 treated human PBMC (1 × 105 cells/well) in 96-well tissue culture plates. Activated PBMC cultured alone were used as positive control (control non-MSC, secretion inhibition efficiency = 0%). The TNF-α expression was then measured with a LEGEND MAX Human TNF-α ELISA Kit (Biolegend, USA) after 3 days’ co-culture.
Animal Studies
A total of 60 male DBA/1 J mice (Jackson Laboratories, USA) were randomly labeled and used for 2 identical experiments. Of those 60, 53 animals were selected randomly for primary induction (26 mice in the first study [Batch 1] and 27 mice in the second [Batch 2]). The remaining 7 mice were assigned to Healthy groups. On the day of hUC-MSC infusion, 41 mice met the disease-onset criteria (i.e., at least 1 leg reached the clinical score ≥2 on the day of infusion) and mice were regrouped randomly to ensure no significant differences of clinical scores existed between TA and CA groups. There were 21 mice in TA group (11 in Batch 1 and 10 in Batch 2), 20 in CA group (10 in Batch 1 and 10 in Batch 2), and 7 in Healthy group (4 in Batch 1 and 3 in Batch 2) (Table 1). Body weight (17 to 23 g for Batch 1; 16 to 24 g for Batch 2) of animals was measured every 3 days from the start of baseline blood collection (2 to 3 days before primary induction), to 18 days after the primary induction (Day 18), and was measured every other day thereafter (Fig. 1). The mice were monitored daily and assessed every other day, in a double-blind fashion, for signs of arthritis onset (based on clinical scores) by two independent investigators. Clinical arthritis was scored on a scale of 0 to 4, where 0 =no swelling, 1 =erythema and mild swelling limited to tarsals or ankle joint, 2 =erythema and mild swelling extending from ankle to tarsal joints, 3 =erythema and moderate swelling extending from ankle to metatarsal joints, and 4 =erythema and severe swelling surrounding the ankle, foot, and digits, or ankylosis of the limb. Each limb was graded and the grades from four limbs were summed to yield the arthritis score for each animal. The maximal score was 16. Paw thickness (exploratory outcome) was also assessed with a 0 to 10 mm Thickness Gage (Mitutoyo, Japan). All animals were sacrificed 42 days after primary induction (Day 42, end point) with CO2 (or isoflurane) overdose and cervical dislocation. Legs were harvested for further processing, followed by mouse dissection. Notes were taken and a histopathological exam was conducted if any abnormality in the volume, color, texture, and so on of organs (brain, heart, liver, spleen, lung, kidney, and injection-site tissue) was observed. All animal experiments and procedures were performed under permission from the UCI Institution of Animal Care and Use Committee (IACUC protocol number AUP-19-082) and conducted per the Animal Welfare Assurance (#A3416.01).
Animal Grouping.

CA: control assessment; hUC-MSC: human umbilical cord tissue–derived mesenchymal stem cells; TA: treatment assessment.

Timeline of using human umbilical cord tissue–derived mesenchymal stem cells to treat collagen-induced rheumatoid arthritis.
Arthritis Induction
For CIA, DBA/1 J mice were immunized with 100 µg of bovine type II collagen (Chondrex, USA), which was emulsified with Freund’s complete adjuvant containing 200 µg of Mycobacterium tuberculosis H37Ra (Sigma-Aldrich, Germany), via an intradermal injection at the base of the tail; and a booster emulsion of CII in Freund’s incomplete adjuvant was administered at 21 days (Day 21) after the primary immunization (Fig. 1) 26 . Disease onset was defined as at least one leg reaching the clinical score ≥2.
MSC Administration
At 24 days (Day 24) after primary induction of CIA (after disease onset), a single infusion of 200 µl carrier solution (Baylx, USA) or 2 million hUC-MSC suspended in 200 µl carrier solution (1 × 107 cells/ml) was intravenously infused via tail vein to each mouse from CA and TA groups. Intravenous infusion via tail vein was used to mimic the clinical administrative route and was done with 1 ml sterile syringes with 30 G × ½ disposable needles and an infusion speed of 8 to 20 μl/s. After drug administration, animals were closely monitored for 2 h. If any abnormality was observed (e.g., any profound paralysis that inhibits animals’ ability to eat or drink, any signs of pain including piloerected/unkempt fur, squinted eyes, ataxia, hunched posture, labored breathing, which is not relieved with analgesia), symptoms and their starting time, severity, duration, and so on were recorded in detail. Thereafter, monitoring was also conducted twice a day with notes taken accordingly.
Serum Cytokine Quantification
Murine blood was collected into ethylenediamine tetraacetic acid (EDTA)-coated collection tubes (Sai-infusion, USA) at baseline (2 to 3 days before primary induction), before treatment (Day 22), after treatment (Day 26), and at end point (Day 42) (Fig. 1). Then the sera were harvested from blood samples after centrifugation. The pro-inflammatory cytokine levels (including IL-1β, TNF-α, interferon gamma [IFN-γ], and IL-6) were determined by a MILLIPLEX MAP Mouse TH17 Magnetic Bead Panel Luminex assay (EMD Millipore, Germany) following the manufacturer’s protocol.
Tissue Processing and Histology
Excessive fur and muscle were removed with scissors and tweezers from each mouse’s limbs before being fixed in 10% formalin for 2 days. The limbs were decalcified for 3 to 4 weeks in decalcification solution (14% EDTA, 0.2% of 4% paraformaldehyde in 1× phosphate-buffered saline, pH = 7.4); the decalcification solution was changed every 2 to 3 days. All limbs were processed in a TP1020-1-1 tissue processor (Leica, Germany) using an 18-h processing protocol. The 10-µm paraffin sections were stained using hematoxylin and eosin (H&E; EMD Millipore, Germany) to assess the difference in tissue morphology among TA, CA, and Healthy groups. Stained tissue was imaged on a Nikon Ti Eclipse (Nikon, Japan) using a 10× objective. Representative images of H&E-stained limbs from all three groups were independently scored by two blinded investigators on a scale from 0 to 4, where 0 =no cell infiltration, cartilage damage, or bone erosion; 1 =mild cell infiltration and synovial membrane hypertrophy, and little to no cartilage damage; 2 =obvious cell infiltration, mild cartilage damage, and little to no bone erosion; 3 =severe cell filtration, cartilage erosion, and bone erosion; and 4 =complete loss of joint integrity and ankylosis. Representative images are shown in Fig. 2 and average histology scores are shown in Table 2.
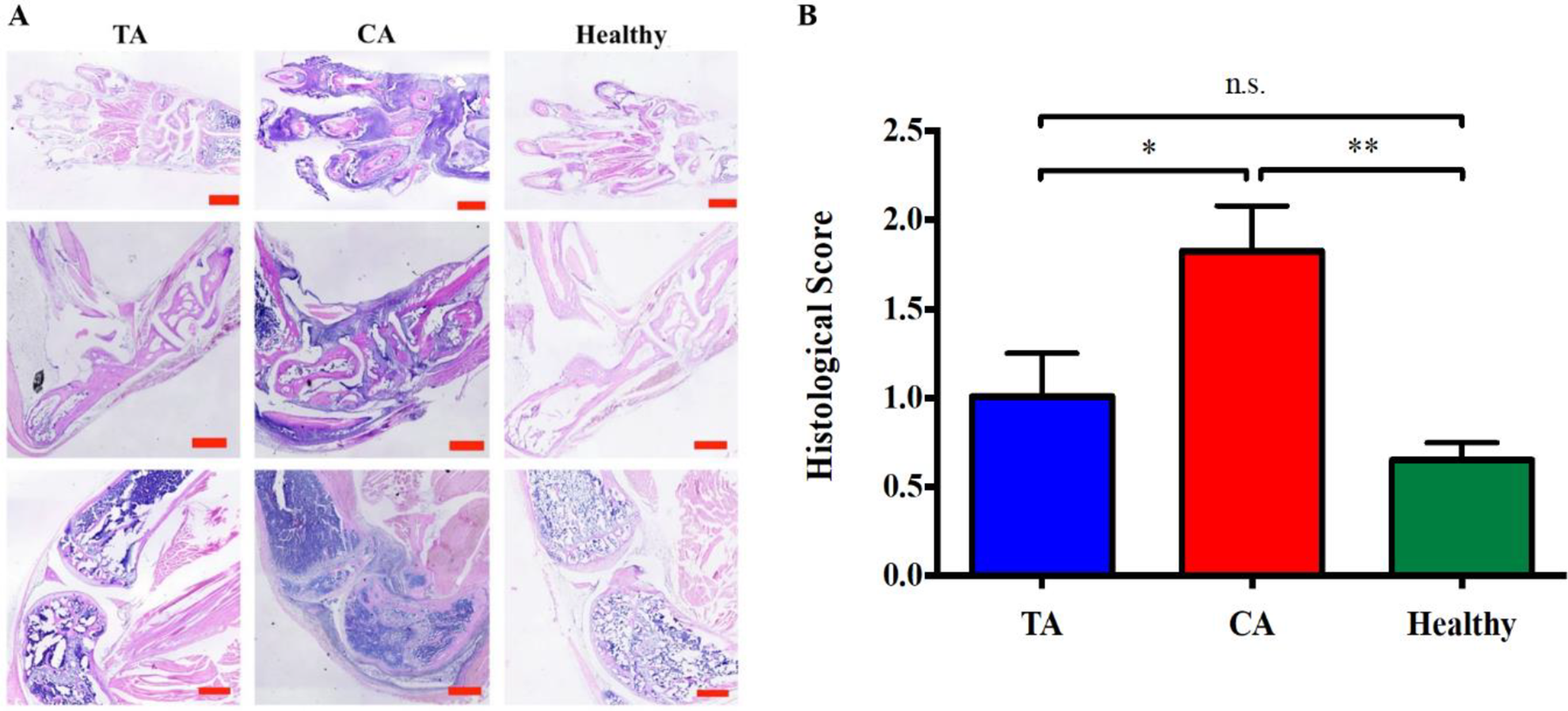
CA group mice had significantly better histological scores than those of TA group. (A) Representative pictures of histological sections. Scale bars = 500 µm. (B) Comparison of histological scores among three experimental groups at the end point (Day 42). Maximum score = 4. Mean ± standard error of the mean. TA group: n = 10, CA group: n = 9, Healthy group: n = 4. *P < 0.05, **P < 0.01 and n.s. = not significant.
Histological Scores (Day 42, Mean ± Standard Error of the Mean).
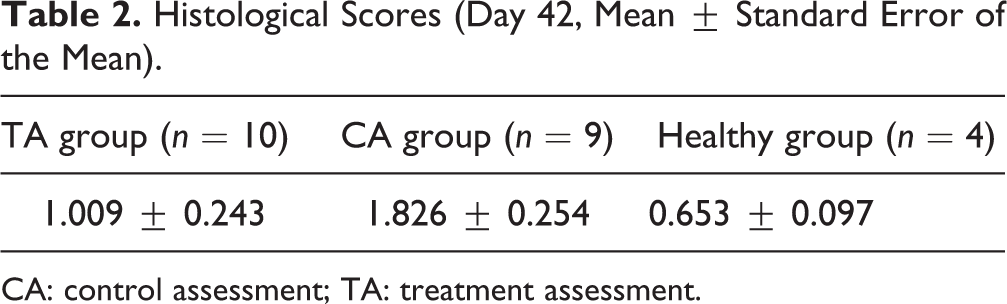
CA: control assessment; TA: treatment assessment.
Data Analysis and Statistics
Data were analyzed by Student’s two-tailed t test when comparing two groups. Data were expressed as the mean ± standard error of the mean, mean ± standard deviation of the mean, and 95% confidence interval (95% CI), and differences were considered significant at P <0.05. CIA mice that did not meet the disease-onset criteria for treatment (i.e., at least one leg has a clinical score ≥2) on the infusion day were removed from the study. Samples with failed readings or out of the upper limit of standard curve in Luminex assay were removed from the analysis. Luminex readings out of the lower limit of the standard curve were defined as “0”. See Supplementary Materials for detailed information for analysis on clinical scores and paw thickness.
Results
Generation and In Vitro Characterization of hUC-MSC
The hUC-MSC used in this study were generated from umbilical cord tissue and were characterized to have standard MSC phenotype according to the International Society for Cellular Therapy criteria (Fig. 3). We established a laboratory cell bank of hUC-MSC at passage 5. This cell bank was used for in vitro characterization and for the animal experiments in our IND-enabling studies. The hUC-MSC cell line displayed appropriate marker expression (Fig. 3A). The hUC-MSC showed high expression of the key MSC markers CD73, CD90, and CD105, while maintaining low expression of non-MSC markers including CD45, CD34, and HLA-DR. Multipotency was displayed via adipogenesis and osteogenesis (Fig. 3B). When co-cultured with activated PBMC, the hUC-MSC reduced TNF-α inflammatory cytokines by over 10-fold (Fig. 3C), indicating an anti-inflammatory functional potency.

In vitro characterization of hUC-MSC. (A) Flow cytometric analysis showed that hUC-MSC were positive for the surface markers CD90, CD105, and CD73, while negative for CD34, HLA-DR, and CD45. (B) Oil Red staining (left) showed adipogenic potential of hUC-MSC after adipogenesis differentiation; Alizarin red staining (right) showed mineralization potential of hUC-MSC after osteogenic differentiation. (C) hUC-MSC showed a superior TNF-α-inhibitive effect when co-cultured with PBMC. TNF-α expression level was analyzed in the presence of hUC-MSC and a non-MSC control following activation of PBMC with anti-CD3, anti-CD28, and IL-2. Data are presented as mean ± standard deviation.
hUC-MSC Improved Arthritis Assessment Outcomes
CIA was established in DBA/1 J mice to mimic human RA. CIA animals were grouped randomly into either a treatment assessment (TA) group, which received a tail vein infusion of 2 million hUC-MSC or a control assessment (CA) group, which received only cell-free carrier solution.
To evaluate disease severity and treatment efficacy, clinical assessments were taken by two independent investigators every other day for 18 days. The averaged clinical score on Day 24 (the day of hUC-MSC infusion) for TA group was 7.08 (95% CI: 6.30, 7.86), 7.51 (95% CI: 6.73, 28.30) for CA group, and 0.38 (95% CI: −0.57, 1.33) for the Healthy group, respectively. The Day 24 Healthy group clinical scores were not significantly greater than zero (P = 0.435) and there was no significant difference in clinical scores between TA and CA groups, prior to treatment (P = 0.126) (Supplemental Table S1). The Day 24 clinical scores of both TA and CA groups were significantly higher than that of the Healthy group (P < 0.0001).
After infusion, a significant difference in clinical scores between TA and CA groups was detected on Day 28, i.e., 4 days after the hUC-MSC infusion (−1.53, P = 0.003, 95% CI: −2.29, −0.77; with Bonferroni correlation for multiple comparison, Supplemental Table S1) which continued to increase until Day 42 (end point) (−4.49, P < 0.0001, 95% CI: −5.97, −3.01; Table 3, Fig. 4 and Supplemental Fig. S2). In Batch 1, a significant difference in clinical scores between TA and CA groups was observable by Day 32 (−2.82, P = 0.036, 95% CI: −4.52, −1.12) which continued to increase until Day 42 (end point) (−5.44, P < 0.0001, 95% CI: −7.40, −3.49) (Supplemental Table S2). In Batch 2, a significant difference in clinical scores between TA and CA groups started on Day 28 (−1.69, P = 0.015, 95% CI: −2.59, −0.79) and continued to increase until Day 38 (−3.95, P = 0.012, 95% CI: −6.02, −1.88) (Supplemental Table S3). All baseline clinical scores were zero.
Clinical Scores After hUC-MSC Infusion (Combined Batches; Mean and 95% Confidence Interval).

CA: control assessment; hUC-MSC: human umbilical cord tissue–derived mesenchymal stem cells; TA: treatment assessment.
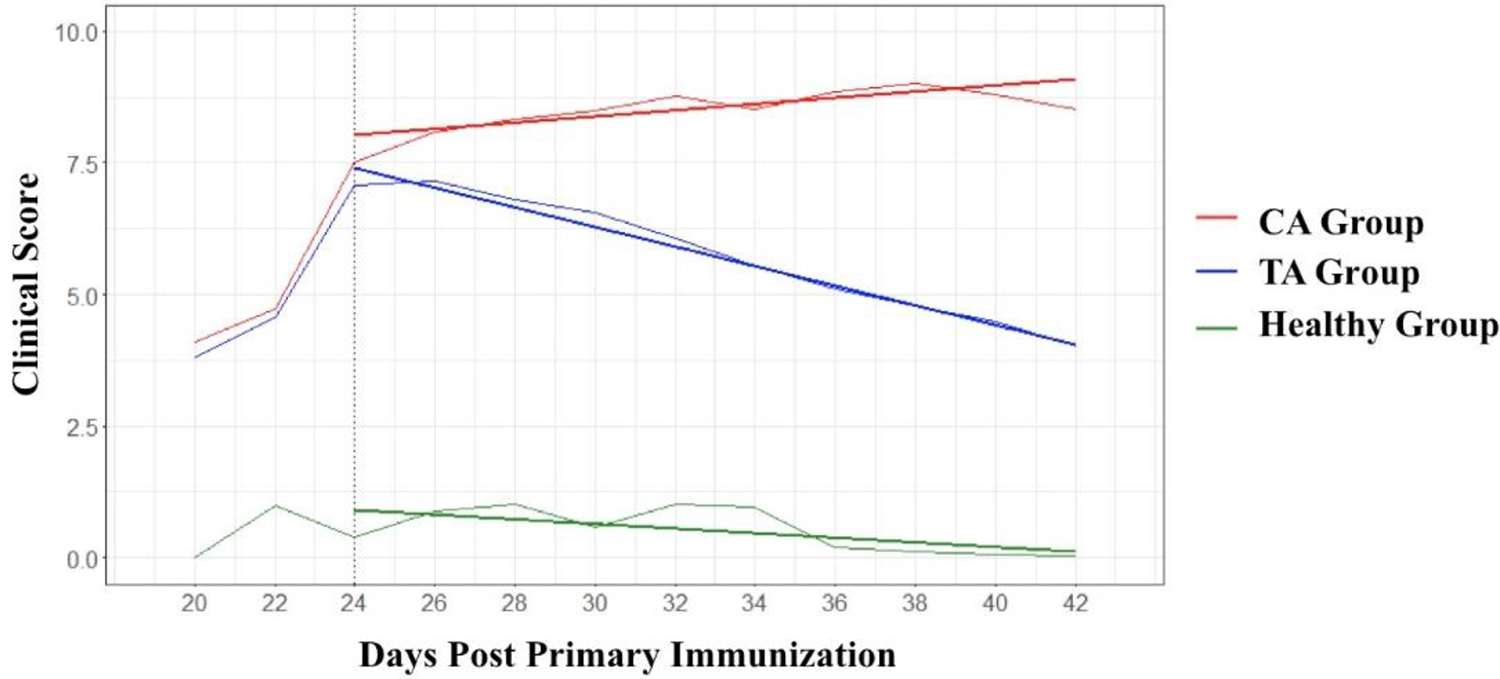
Average clinical score and model-based rate of change for each experimental group from Day 20 to Day 42 (two batches combined). TA group: n = 21, CA group: n = 20, Healthy group: n = 7. Human umbilical cord tissue–derived mesenchymal stem cells were infused on Day 24 (dash line). The three straight bold lines stand for the linear mixed model–fitted linear trends of clinical score in each group.
When comparing the clinical scores between Day 24 and Day 42 (end point), a significant decrease of 3.12 (P < 0.0001, 95% CI: 2.04, 4.20) was observed in TA group, whereas the clinical score was significantly increased by an average of 1.13 (P = 0.047, 95% CI: 0.05, 2.21) 18 days after hUC-MSC infusion in CA group (Table 4). No significant change of clinical score was detected in Healthy group (P = 0.668, 95% CI: −2.18, 1.39; Table 5). Importantly, a significant difference in the 18-day clinical score trend was observed between TA and CA groups (−4.25, P < 0.0001, 95% CI: −5.78, −2.72; Table 5). Consistency was observed between both batches (Supplemental Tables S2 and S3).
Changes of Clinical Score Between Day 24 and Day 42 (Estimate = Day 42 − Day 24).

CA: control assessment; TA: treatment assessment.
Comparison (TA vs. CA) of Clinical Score Between Day 24 and Day 42 (Estimate = TA − CA).

CA: control assessment; TA: treatment assessment.
A comparison between TA and CA group clinical scores over the time was also performed (Fig. 4 and Table 4). The model-based rate of change in clinical score was 1.86 decrease (95% CI: 1.58, 2.14) per 10 days in TA group and 0.60 increase (95% CI: 0.32, 0.88) per 10 days in CA group. Overall hUC-MSC infusion displayed a significant therapeutic effect on reducing clinical scores in a CIA murine model (−0.25 per day, P < 0.0001, 95% CI: −0.29, −0.21) (Table 6). Similar results were also observed, showing consistency between batches. The trend detected in Healthy group was not significantly different compared to zero (−0.04 per day, P > 0.05, 95% CI: −0.09, 0.00). Representative pictures of paws are shown in Supplemental Fig. S3.
Comparison (TA vs. CA) on Clinical Score Over the Time (Estimate = TA − CA).

CA: control assessment; TA: treatment assessment.
Joint swelling was measured with thickness gages (front and back paws) every other day for 18 days. On the day of hUC-MSC infusion (Day 24), paw thickness between TA and CA groups had no significant difference (−0.01 mm, P = 0.874, 95% CI: −0.16, 0.14). When analyzed separately, the thickness of neither front paws (0.03 mm, P = 0.311, 95% CI: −0.03, 0.08) nor hind paws (−0.05 mm, P = 0.051, 95% CI: −0.11, 0.00) showed significant difference between TA and CA groups.
The mean and 95% CI of paw thickness measured every 2 days after Day 24 were calculated for front and hind legs separately (Supplemental Table S4) and combined (Supplemental Table S5). No significant difference was observed in overall, front, or hind paw thickness between TA and CA groups from Day 26 to Day 42 (Supplemental Fig. S4).
Microscopic Joint Inflammation Was Reduced After hUC-MSC Treatment
After end point, mice were sacrificed, and tissues were harvested so that histological analysis could be performed on the joints. Briefly, at the end point (Day 42), mice (Batch 1) were sacrificed and legs were harvested and processed, then assayed with H&E staining. The histological scores of TA group were significantly lower than those of CA group (P < 0.05) and had no significant difference compared to those of the Healthy group (P > 0.05), demonstrating a significant therapeutic effect of hUC-MSC on RA. A significant difference was also observed between CA group and Healthy group (P < 0.01; Table 2, Fig. 2). Collectively, arthritis symptoms were successfully established in mice, and hUC-MSC treatment showed improvements in several clinical parameters relating to arthritis.
IL-6 Level Was Declined by hUC-MSC Treatment
Blood was taken for cytokine analysis at multiple time points throughout the experiment. Briefly, murine blood (Batch 1) was harvested on the days before (Day 22) and after (Day 26) the hUC-MSC infusion, as well as on the baseline and the end point (Day 42). Concentrations of pro-inflammatory cytokines IFN-γ, IL-6, TNF-α, and IL-1β in plasma were measured with Luminex assay (Table 7). At the baseline, the cytokine levels were low and there was no significant difference between TA and CA groups. On Day 22, the concentrations of IFN-γ, IL-6, and TNF-α were highly increased with large variation among animals. The concentration of IL-1β also increased but not as high as the other cytokines. No significant difference was shown between TA and CA groups for all the cytokines (Supplemental Table S6).
Relative Concentrations of Cytokines (%, Mean ± Standard Error of the Mean).

#Significant reduction compared with Day 22; ###Significant declined by 80.0% (P < 0.0001) compared with Day 22; *Significantly lower compared with CA group.
CA: control assessment; IFN-γ: interferon gamma; IL-1β: interleukin 1 beta; TA: treatment assessment; TNF-α: tumor necrosis factor alpha.
Two days after hUC-MSC infusion (Day 26), the concentration of IL-6 in TA group had significantly declined by 80.0% (P < 0.0001) compared with Day 22, whereas no significant change was observed in CA group. The concentrations of TNF-α in TA group dropped significantly by 14.3% from Day 22 (P < 0.05) while a climbing trend was observed in CA group (increased by 13% compared to Day 22). The concentrations of IFN-γ and IL-1β also decreased compared with Day 22, but there was no significant difference between TA and CA groups.
At the end point (Day 42), the concentration of IL-6 was further reduced in both TA and CA groups. The decrease of IL-6 in TA group (by 93.4% compared to Day 22) was significantly steeper than that of CA group (by 64.7% compared to Day 22) (P < 0.05; Table 7 and Fig. 5A). The concentration of TNF-α was also declined in both TA and CA groups at the end point but without significant difference between TA and CA groups (Table 7 and Fig. 5B). For the concentration of IFN-γ and IL-1β, no significant difference was detected between TA and CA groups (P > 0.05; Table 7, Fig. 5C, D).

Concentrations of pro-inflammatory cytokines (IL-6, IFN-γ, TNF-α, and IL-1β) in each experimental group at baseline, Day 22, Day 26, and end point (Day 42). The concentrations of cytokines were determined by Luminex serology assay. Mean ± standard error of the mean. *P < 0.05 (TA group vs. CA group); # P < 0.05 and #### P < 0.0001 (Day 22 vs. Day 26).
No Side Effect Was Observed After hUC-MSC Administration
No abnormalities, including appearance, behavior, secretions, and excreta, were observed after infusion of hUC-MSC and no mice died from infusion of test agents. No erythema, edema, or exudation was observed at injection site, after infusion of test agent.
Animals were weighed every other day for 18 days. Variations in animal weights prior to CIA disease induction were insignificant (Supplemental Table S7). After CIA model induction, all animals lost weight, and began to recover after 2 days. No significant difference in body weight was observed at any time point between TA and CA groups (P > 0.05, Supplemental Table S8), but by end point, both groups had lower average body weight than healthy mice (Supplemental Fig. S1).
Discussion
Stem cell therapies have the capacity to treat disease more directly than standard small-molecule drugs, which tend to only treat symptoms. MSC are a type of adult stem cell with immunomodulatory capabilities and were the active component of the hUC-MSC injection we evaluated in this study. MSC have been investigated for their beneficial therapeutic effects in multiple diseases, including myocardial infarction, connective tissue, autoimmune, inflammatory, lung, and kidney diseases 27,28 . Additionally, MSC have been evaluated in many animal studies and human clinical trials of rheumatic diseases 22,29,30 . There has been great interest in using MSC to treat patients with RA for many years 31,32 . We recently performed a meta-analysis of MSC in the treatment of RA and found that there was much variability between studies, and the origin of MSC may significantly impact treatment outcome 22 . For example, human umbilical cord and adipose-derived MSC may be more effective against RA than MSC from other sources. Overall, previous preclinical and clinical studies showed positive efficacy and tolerability and therefore warrant further investigation into the clinical benefits of MSC in RA 22,33 . This study aimed to evaluate the efficacy from a single infusion of hUC-MSC to treat RA in a CIA murine model via tail vein and thereby provide information for preclinical studies of BX-U001.
After a CIA model was established, the administration of hUC-MSC significantly reduced clinical scores of TA group (P < 0.01 starting from 6 days after hUC-MSC administration) compared to CA group. Clinical scores are a qualitative measure of joint appearance based on comparison to healthy animal joints. Generally, mice induced with a CIA model had swollen joints, and erythema around joints and extremities (especially in the hind legs). While paw thickness measurements alone showed no significant difference between TA and CA groups, these measurements are limited by the soft tissue surrounding the areas of inflammation and cannot tell us about the underlying joint structure. Clinical scoring includes color and overall appearance (straightness) of the joints, a general macroscopic evaluation that may indicate more about underlying joint health rather than the size of the joint alone.
In many studies of inflammation, histological evaluation can give insight to invasion of the immune system into specific tissue locations, and provide microscopic detail on structural damage, which macroscopic methods cannot achieve 34 . After treatment with hUC-MSC, the histological scores of the joints of TA group were significantly reduced compared to CA group (P < 0.001). The knee joint had significant inflammation and immune cell invasion in untreated CIA mice (CA group), and there tended to be reduced synovial fluid between the joints. Additionally, bone was clearly eroded in CA group joints, but had been at least partially protected in TA group joints. This type of damage is the typical manifestation of RA inflammation, and directly correlates with pain and overall joint function 35,36 .
It is essential to evaluate the systemic effects of hUC-MSC treatment in RA, because the high levels of inflammatory cytokines released into the blood during RA pathogenesis can produce damage in the heart, lungs, and other vital organs, despite their geographical distance from inflamed joints 9,10 . In particular, TNF-α and IL-6 are key inflammatory cytokines in both the initial and advanced stages of RA disease 29,30 . The serum levels of these key cytokines were significantly reduced in hUC-MSC-treated mice, compared to untreated controls. There was less of an effect of IFN-γ and IL-1β, which are also linked to RA pathophysiology. The link between MSC treatment and a reduction in TNF-α and IL-6 is likely attributable to their ability to influence systemic levels of M2 anti-inflammatory macrophages and upregulate the levels of FoxP3+ T-reg 29,37,38 . Immediately after intravenous injection, many MSC become trapped in the capillaries of the lungs, where they are engulfed by macrophages 39 . The effect MSC have on circulating macrophages, resulting from their engulfment, is heavily linked to the ratio of anti-inflammatory cytokines produced by these macrophages, and their downstream interactions with other immune cells 39 –41 . The detailed mechanisms of hUC-MSC action on RA are still being elucidated, but the reduction of TNF-α and IL-6 cytokines appears to be a key feature.
While the hUC-MSC evaluated in this study did not treat every RA disease parameter tested, they did provide significant improvements in macroscopic (clinical scores) and microscopic (histology) pathophysiology, and reduced systemic levels of key inflammatory cytokines related to the RA. It is important to consider the application of cell therapies to disease as a supplement rather than a replacement to drug-based treatments. In many drug-resistant cases of autoimmune disease, MSC may provide a chance at treatment, where there is none. In RA patients who respond partially to drug treatments, MSC may provide an additional treatment option or alternative that might be less immunosuppressive or have as severe side effects as the current standard drug treatments methotrexate and TNF-α blockers.
Conclusion
A single intravenous infusion of BX-U001 hUC-MSC can significantly relieve RA disease symptoms in a CIA mouse model. hUC-MSC decreased clinical scores, histological scores, and serum levels of pro-inflammatory cytokines, indicating they have significant clinical potential. This new cell product showed no signs of adverse effects and will be further tested for safety and efficacy in clinical studies with human patients. The limitations of drug-based treatments may be overcome or at least supplemented by cell therapies, such as hUC-MSC.
Data Analysis and Statistics
Clinical scores were studied as the total scores of four legs whereas paw thickness was analyzed based on each leg separately due to the huge difference of the measurement between front and hind legs.
First, a linear mixed model (LMM) was used to examine the difference of outcome measures on infusion day (Day 24), among TA, CA, and Healthy groups, with adjustments for within-batch correlation. The LMM has the following form:

where
Second, a similar LMM followed by contrast testing was used to compare the 18-day change [from infusion day (Day 24) to the end point (Day 42)] of each outcome among the three groups. Unlike the previous model, the dependent variable

where
The slope of outcome change for each experimental group was calculated and the differences in slopes were compared using contrast testing. In order to investigate when a significant difference can be observed between TA and CA groups, the outcome measures on each measurement day were studied and compared. The difference between TA and CA groups was examined with a similar LMM model. Bonferroni correlation was applied to adjust for multiple-day comparisons.
For paw thickness, the abovementioned analysis first compared the overall mouse leg measurements among three groups without specifying the leg locations (front/hind). As paw thickness was quite different between front and hind legs, the measurements of front and hind legs were also studied separately. (1) Leg locations (front/hind) and (2) the interactions between leg locations and groups (TA/CA) were added into each of the above LMM, to allow for different treatment responses onto different leg locations.
The two batches were studied separately as a sensitivity analysis, to examine whether the results were robust and consistent in both batches. All the above analyses were repeated in each batch. A linear regression model was used instead of LMM for the comparisons on infusion day (Day 24), daily responses, and 18-day changes.
Supplemental Material
Supplemental_Materials - Preclinical Evaluation of a Single Intravenous Infusion of hUC-MSC (BX-U001) in Rheumatoid Arthritis
Supplemental_Materials for Preclinical Evaluation of a Single Intravenous Infusion of hUC-MSC (BX-U001) in Rheumatoid Arthritis by Linan Liu, Henry P. Farhoodi, Menglu Han, Guangyang Liu, Jingxia Yu, Lily Nguyen, Brenda Nguyen, Agnes Nguyen, Wenbin Liao and Weian Zhao in Cell Transplantation
Footnotes
Ethical Approval
Ethical Approval to perform and report this study was provided by the University of California Institutional Animal Care and Use Committee (IACUC) under protocol #AUP-90-082.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the University of California Irvine IACUC-approved protocols (#AUP-90-082).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH (R21CA219225 to WZ), the DOD (W81XWH-17-1-0522 to WZ), and a contract with Baylx Inc. (BI-206512). HPF was supported by the National Institute of Neurological Disorders and Stroke of the NIH (T32NS082174). The content of this paper is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
