Abstract
Mesenchymal stem cells (MSCs) have been shown to possess immunomodulatory properties. Systemic lupus erythematosus is an autoimmune disease that results in nephritis and subsequent destruction of renal microstructure. We investigated whether transplantation of human umbilical cord blood-derived MSCs (uMSCs) is useful in alleviating lupus nephritis in a murine model. It was found that uMSCs transplantation significantly delayed the development of proteinuria, decreased anti-dsDNA, alleviated renal injury, and prolonged the life span. There was a trend of decreasing T-helper (Th) 1 cytokines (IFN-γ, IL-2) and proinflammatory cytokines (TNF-α, IL-6, IL-12) and increasing Th2 cytokines (IL-4, IL-10). The in vitro coculture experiments showed that uMSCs only inhibited lymphocytes and splenocytes proliferation but not mesangial cells. Long-term engraftment of uMSCs in the kidney was not observed either. Together, these findings indicated that uMSCs were effective in decreasing renal inflammation and alleviating experimental lupus nephritis by inhibiting lymphocytes, inducing polarization of Th2 cytokines, and inhibition of proinflammatory cytokines production rather than direct engraftment and differentiating into renal tissue. Therapeutic effects demonstrated in this preclinical study support further exploration of the possibility to use uMSCs from mismatched donors in lupus nephritis treatment.
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune disease characterized by the production of antibodies to components of the cell nucleus with diverse clinical manifestations including vasculitis, immune complex type glomerulonephritis, and arthropathy. To date, lupus nephritis (LN) remains a leading cause of morbidity and mortality in patients with SLE (14). At present, glucocorticosteroids, cytotoxic drugs, and mycophenolate mofetil are the main therapeutic agents; long-term and high-dose usage inevitably results in toxic side effects. Therefore, it is imperative to find new therapeutic modalities for LN patients.
SLE is composed of a myriad of immune system aberrations that involve B cells, T cells, and cells of monocytic lineage, resulting in polyclonal B cell activation, autoantibody production, and immune complex formation (35,49). Numerous studies have investigated the cytokine profile in patients as well as in murine lupus model in vivo and in vitro. However, the exact pathogenesis mechanism of the immunological disturbance in cytokine remains elusive. T helper (Th) cells can be classified into different subsets based on their patterns of cytokine production. Th1 cells produce interferon-γ (IFN-γ) and interleukin-2 (IL-2). Th2 cells secrete IL-4, IL-5, IL-6, and IL-10. There have been studies showing that an imbalance towards specific Th subsets may play a significant role in the pathogenesis of SLE (9,12,21, 25,42,48,55). More recently, an imbalance towards Th1 predominance in the acceleration of lupus autoimmune disease has been reported in SLE patients and lupus-like autoimmune disease in mice (3,4,9,43).
Tumor necrosis factor-α (TNF-α) is a proinflammatory cytokine with immunoregulatory functions. In humans, several studies have indicated that levels of serum TNF-α are associated with SLE disease activity (3,43). TNF-α can also upregulate other proinflammatory cytokines such as IL-6. Treatment with anti-TNF-α and anti-IL-6 antibodies showed improvement in animal models of LN (4,15). IL-12 is produced by macrophages and dendritic cells and promotes the activated T cells to secrete INF-γ as well as to inhibit Th2-type cytokine production. Serum levels of IL-12 in SLE patients are elevated and related to the severity of LN (51).
Mesenchymal stem cells (MSCs) are multipotent cells capable of differentiating into a variety of mesenchymal lineages including cartilage, bone, muscle, tendon, ligament, and adipose tissue (26,28,29). Importantly, MSCs have been shown to possess immune-modulatory capabilities (6,7, 38,54,56). MSCs are hypoimmunogenic, often lacking MHC-II and costimulatory molecule expression. It has been demonstrated that MSCs can alleviate immune response by inhibiting proinflammatory cytokines such as TNF-α and IFN-γ. It has also been reported that MSCs inhibit the function of mature and immature T cells, inhibit the development of proinflammatory Th1, and promote anti-inflammatory Th2, resulting in a shift from a proinflammatory environment towards an anti-inflammatory environment (6,31,56).
New Zealand Black/White F1 (NZB/W F1) mice develop severe systemic autoimmune diseases that closely resemble SLE in humans (22). The mice manifest various immune abnormalities including antibodies to nuclear antigens and development of a fatal, immune complex-mediated glomerulonephritis. Ninety-eight percent of NZB/W F1 mice die before 1 year of age. NZB/W F1 mice with signs of LN are indicated by the presence of anti-dsDNA antibodies and proteinuria (32).
Umbilical cord blood (UCB) has been an excellent source for stem cells as banked UCB has been available for transplantation, and UCB contains not only hematopoietic stem cells but also MSCs (29,39,53). In view of the clinical application potentials, we sought to explore the therapeutic effects of MSCs in LN. The aim of this study was to determine the effectiveness of UCB-derived MSCs (uMSCs) transplantation on LN in NZB/W F1 mice as well as to explore the mechanisms of action. We hypothesized that uMSCs transplantation ameliorate LN and increase the survivorship of NZB/W F1 mice by reducing autoimmune activities through immunomodulatory effects.
Materials and Methods
Animal Model of LN
Twenty-three female NZB/W F1 mice were obtained from the Animal Center of the College of Medicine of National Taiwan University in a pathogen-free facility. All mice used in the analysis were housed in the same room and fed an identical diet. The animal experiments were approved by the Ethics Committee for Animal Research of the Taipei Veterans General Hospital.
Isolation and Culture of uMSCs
UCB was collected on delivery with informed consent and ethical approval for its use by Institutional Review Board, Taipei Veterans General Hospital. Isolation and characterization of MSCs from UCB was carried out as reported previously (29). Briefly, uMSCs were single cell derived and clonally expanded, and their surface immunophenotype and multilineage differentiation potentials were confirmed prior to the commencement of the transplantation experiments.
Transplantation Protocols of uMSCs
Mice were randomly divided into three groups. In group 1 (n = 8), 2-month-old NZB/W F1 mice with blood anti-dsDNA antibodies but no proteinuria were transplanted with uMSCs (1 × 106 cells) intravenously via the tail vein. Group 2 (n = 7) consisted of animals in which uMSC (1 × 106 cells) transplantation was performed at the age of 6 months. The 6-month-old NZB/W F1 mice presented with high levels of circulating anti-dsDNA antibodies and proteinuria >100 mg/dl. Group 3 (n = 8) was the controls. All mice were sacrificed at 8 months of age to evaluate the therapeutic effectiveness of uMSCs transplantation.
Enzyme-Linked Immunosorbent Assay (ELISA) for the Determination of IgG Anti-dsDNA Autoantibody Levels
Concentration of mouse IgG anti-dsDNA was measured by a commercially available ELISA kit according to the manufacturer's protocol (Alpha Diagnostic International, TX, USA) monthly.
Evaluation of Proteinuria and Renal Function
All mice were examined visually on a daily basis for signs of illness. Mice were evaluated monthly for proteinuria using dipstick tests with a scale of 0 to 4+, where 0/trace = negative, 1+ = 30 mg/dl, 2+ = 100 mg/dl, 3+ = 300 mg/dl, and 4+ = >2000 mg/dl. Once significant proteinuria (>100 mg/ml) was observed, urinalysis was performed weekly. Proteinuria scores above 3+ were considered the development of severe glomerulonephritis. Blood samples were harvested every month from retro-orbital sinus bleeding for serum creatinine level measurement.
Renal Histopathology
For histological analysis, formalin-fixed sections were stained with hematoxylin and eosin, periodic acid-silver methenamine, periodic acid Schiff, and Masson's trichrome. The glomerular pathology of 40 glomerular cross sections (gcs)/kidney was scored on a 4-point scale: 0 = normal (35–40 cells/gcs); 1 = few lesions with slight proliferative change and mild hypercellularity (41–50 cells/gcs); 2 = moderate hypercellularity (51–60 cells/gcs); and 3 = severe hypercellularity (>60 cells/gcs). The extent of glomerular sclerosis was graded on a 4-point scale as follows: 0 = normal; 1+ = <25% glomerulus; 2+ = 26–50% glomerulus; 3+ = >50% glomerulus.
Measurement of Serum Cytokine Levels
Blood was withdrawn weekly for 4 weeks to determine serum cytokines changes after treatment with uMSCs in group 1 animals. All specimens were stored at −70°C until testing. Measurement of cytokine levels of IFN-γ, IL-2, IL-4, IL-6, IL-10, TNF-α, and IL-12 in serum was carried out using sandwich ELISA reagent kits (R&D Systems, MN, USA).
Splenocyte Isolation
Spleens were isolated aseptically in a laminar flow hood. Organs were cut into several pieces and clumps were further dispersed by drawing and expelling the suspension several times through a sterile syringe with a 19-gauge needle. Debris was filtered through a 100-μm cell strainer. Erythrocytes were lysed using 0.8% ammonium chloride solution for 10 min on ice and washed twice with PBS. Cell counts and viability were assessed by trypan blue stains.
Lymphocyte Isolation
Cervical lymph nodes were excised aseptically from mice. Single cell suspensions were made by pressing through a cell strainer. Cells were collected on RPMI-1640 containing 10% FCS and antibiotics. Cells were washed in medium and counted. Live cells were identified by trypan blue dye exclusion and were then counted.
Lymphocyte and Splenocyte Proliferation Assay
To analyze the effects on uMSCs on T lymphocytes and splenocytes proliferation, splenocytes and lymphocytes were cultured at 37°C with 5% CO2 in a 12-well flat-bottom plate at a concentration of 3 × 105 viable cells/well. The cells were directly cocultured with irradiated (2000 cGy) treated uMSCs at 1:3 ratios of uMSCs to splenocytes or without uMSCs in the presence of LPS (10 μg/ml), conconavalin A (Con A, 5 μg/ml), or CD3/CD28 (5 μg/ml) for 3 days and pulsed with 3(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium inner salt (MTS) 5 μg/ml per well for 4 h and the absorption value at 490 nm was measured. Lymphocytes and splenocytes without uMSCs were used as controls.
Mesangial Cells (MCs) Proliferation Assay
Primary MCs cultures were established from isolated glomeruli as described previously (45,50). Briefly, glomeruli were purified from minced renal cortex by three times sieving through meshes with 250, 150, and 75 μm pore sizes, then the glomeruli suspension was digested for 5–10 min at 37°C with type IV collagenase, and the dissociated glomerular cells were cultured in RPMI-1640 medium containing 20% FBS, 100 U/ml penicillin, 100 μg/ml streptomycin, L-glutamine (4 mM), β-mercap-toethanol (50 mM), and HEPES (25 mM). MCs were identified by their typically characteristic stellate morphology when subconfluent, then on becoming confluent they adopted the well-recognized elongated conformation, positive for both α-smooth muscle actin and vimentin stain, but E-cadherin negative. MCs (104/well) were cocultured with irradiated (2000 cGy) treated uMSCs at different ratios (1:1, 1:2, and 1:5 ratios of MCs to uMSCs) in 24-transwell plates at 37°C for 12, 24, and 48 h. MCs proliferation was also evaluated using a MTS cell proliferation assay.
Polymerase Chain Reaction (PCR) Experiment
To detect the existence of uMSCs in murine kidneys, partial nephrectomies were performed 2 weeks after administration of uMSCs to obtained kidney tissue for PCR analysis. Genomic DNA from mouse kidneys 2 weeks post-cell therapy with uMSCs and at the end of the experiments was analyzed by mouse and human-specific β2-microglobulin PCR to evaluate engraftment of human uMSCs to the kidneys. The primers for human β2-microglobulin were as follows: forward, GTGTCTG GGTTTCATCCATC and reverse, GGCAGGCATACT CATCTTTT. The mouse β2-microglobulin primers (forward: AGACTTTGGGGGAAGCAGAT, reverse: CAG TCTCAGTGGGGGTGAAT) were selected based on the published sequences (10,37). Amplification for β2-microglobulin was performed with initial denaturation at 95°C for 1 min, followed by 35 cycles of 30 s at 95°C, 1 min at 57.1°C, and 30 s at 72°C, with a final extension at 70°C for 5 min.
Statistical Analysis
Results of proteinuria were analyzed by chi-square analysis. Binomial regression analysis was used to calculate odds ratio for proteinuria during follow-up. Cumulative proteinuria and mortality were analyzed using the Kaplan-Meier method and log rank tests were performed for statistical comparisons of the curves obtained. Differences in serological parameters and quantitative histopathologic analyses between the control and uMSCs-treated groups were analyzed with Kruskal-Wallis test followed by Mann-Whitney U-tests with Bonferroni correction. A value of p < 0.05 was considered statistically significant except the Mann-Whitney U-test with the Bonferroni correction, for which a value of p < 0.017 was regarded as significant.
Results
Transplantation of uMSCs Reduced Severity of Proteinuria and Progression of Renal Function Deterioration
The incidences of proteinuria are shown in Figure 1A. In the control group, 62.5% of the mice showed significant proteinuria at 6 months of age, and 100% of the mice showed significant proteinuria at the age of 8 months, while none of the mice and 50% of mice in group 1 did not show significant proteinuia at 6 and 8 months of age, respectively. In group 2, 71.4% of the mice showed significant proteinuria at 6 months of age. Two mice had a reversal of proteinuria at 7 months of age. Compared to the control group, the mice treated with uMSCs transplantation had a significant decrease in proteinuria at 6 and 7 months of age (p = 0.007 and p = 0.039 by chi-square tests, respectively). Compared to the control group, group 1 showed a significant lower incidence of cumulative proteinuria (p = 0.002 by Kaplan-Meier survivorship analysis and log-rank tests). Odds ratio for proteinuria was reduced in group 1 at 7 months of age (p = 0.024, odds ratio = 0.048; 95% confidence interval = 0.003–0.665). Among the groups, there was a statistically significant difference in serum creatinine levels at 6 months of age (p = 0.037). Compared to the control group, mice in group 1 had a reduction in the progression of elevated serum creatinine levels at 6 months of age (p = 0.017) (Fig. 1B). There were no differences in reducing renal function between the control group and group 2 over the follow-up period.

uMSCs improve proteinuria, serum creatinine levels, and decrease anti-dsDNA titer. (A) Incidence of proteinuria in NZB/W F1 mice. Mice treated with uMSCs transplantation showed lower incidence of proteinuria compared to the control group at 6 and 7 months of age (p = 0.007 and p = 0.039 by chi-square tests, respectively). (B) Serum creatinine levels of the experimental animals. A significant difference was found in creatinine between group 1 and the control group at 6 months of age (∗p = 0.017). (C) Anti-dsDNA antibody titers. Group 1 showed lower levels with respect to the control group at 6 months of age (∗∗p = 0.046). There was a significant difference among groups (p = 0.019) and group 2 showed lower levels with respect to the control group (***p = 0.014) at 7 months of age.
Administration of uMSCs Reduced Anti-dsDNA Antibody Levels
IgG anti-dsDNA concentration increased gradually with age and peaked at 6, 7, or 8 months in the control group. Administration of uMSCs decreased the speed of increase of autoantibody concentration. Comparing the control and group 1, total IgG anti-dsDNA antibody levels were lower in group 1 at 6 months of age (p = 0.046) (Fig. 1C). There was no significant differences in serum levels of anti-dsDNA antibody among the three groups at 6 months of age (p = 0.118), while there was a statistical difference at 7 months of age (p = 0.019) (Fig. 1C). Comparing the mice in group 2 and the control group, uMSCs administration reduced IgG anti-dsDNA concentration significantly at 7 months of age (p = 0.014) (Fig. 1C).
TransplantaAtion of uMSCs Prolonged the Life Span of NZB/W F1 Mice
Mortality rates of the experimental animals are shown in Figure 2. The log rank statistic for an overall difference among the survival curves between the three groups was significant (p = 0.03). In the control group, 50% of the animals died before the age of 8 months whereas all of the mice in group 1 were alive at the age of 8 months. The survival rate in group 1 was statistically higher than that in the control group (p = 0.025). The survival rate also tended to be higher in group 2, with only one of the mice dying before the age of 8 months, although this result did not reach statistical significance.

Cumulative survival rate among the three groups. Kaplan-Meier analysis demonstrated significant difference in survival among the three groups (p = 0.03).
Administration of uMSCs Reduced Mesangial Proliferation and Sclerosis in Renal Pathology
Renal histological sections revealed endoproliferative change and glomerulonephritis with enlarged glomeruli and expansion of the mesangial matrix and marked mesangial proliferation in the control mice (Fig. 3A–D). In contrast, mice treated with uMSCs had milder changes (Fig. 3E, F). Figure 3G and H shows the comparison of histopathological scores of mesangial proliferation and sclerosis between uMSC-treated and control mice. The proliferation score in group 1 was lower (0.60 ± 0.20, p = 0.019) (Fig. 3G) than that of control group (1.66 ± 0.69). However, there was no significant difference among the three groups (p = 0.096). In addition, there was a significant differences between groups regarding sclerosis score (p = 0.047). The sclerosis score was lower in both group 1 and group 2 compared to the control group (p = 0.033 andp = 0.037, respectively) (Fig. 3H).

Transplantation of uMSCs ameliorates the destruction of renal microstructure. (A-D) Kidneys from control group mice at 2, 4, 6, and 8 months of age, respectively. A progressive increase of proliferation and enlargement of the glomeruli was noted. (E) Kidney histology from an 8-month-old mouse in group 1. (F) Kidney histology from an 8-month-old mouse in group 2. Hematoxylin-eosin stain; original magnification 400× for all the pictures. Scale bars: 150 μm. (G) Comparisons of histopathological scores of mesangial proliferation (∗group 1 vs. control group, p = 0.019) and sclerosis score (H) between uMSCs-treated and control groups (among the group, p = 0.047; ∗∗group 1 vs. control group, p = 0.033; ***group 2 vs. control group, p = 0.037).
Administration of uMSCs Changed Th Cytokine Polarization
After uMSCs transplantation, serum levels of Th1 cytokines, IFN-γ and IL-2, in group 1 were significantly lower compared to the control group (Fig. 4A, B). uMSCs administration also significantly decreased the concentrations of the proinflammatory cytokines TNF-a) (Fig. 4C), IL-6 (Fig. 4D), and IL-12 (Fig. 4E). On the other hand, there were marked increases in Th2 cytokines, including IL-4 and IL-10 (Fig. 4F and G, respectively) in group 1 following uMSCs transplantation.

Transplantation of uMSCs induced polarization of cytokine profile to Th2 in NZB/W F1 mice. Decrease of serum IFN-γ (A), IL-2 (B), TNF-α (C), IL-6 (D), IL-12 (E) and increase of serum IL-4 (F) and IL-10 (G) concentrations were noted in group 1 following uMSCs transplantation. Serum was obtained from the mice weekly after transplantation of uMSCs (∗p < 0.05).
uMSCs Inhibited Lymphocyte and Splenocyte Proliferation In Vitro
To analyze the effect of uMSCs on NZB/W F1 mice lymphocyte and splenocyte proliferation, we performed a 1:3 ratio of uMSCs to mitogen-stimulated lymphocytes or splenocytes by direct coculture in vitro. After 3 days of coculture, responder lymphocyte proliferation was measured by MTS assay. Responder lymphocytes treated with LPS, Con A, and CD3/CD28 cocultured with uMSCs exhibited a marked decrease in proliferation (Fig. 5A). uMSCs also significantly inhibited splenocytes proliferation induced by LPS, Con A, and CD3/CD28 in vitro (Fig. 5B).
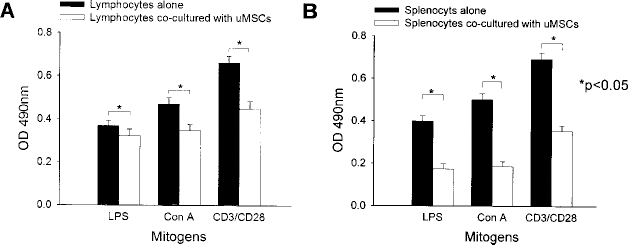
uMSCs inhibit mitogen-stimulated proliferation of lymphocytes and splenocytes. uMSCs inhibit proliferation of lymphocytes (A) and splenocytes (B) of NZB/W F1 mice induced by mitogens (LPS, Con A) and costimulatory factors (CD3/CD28) in vitro (∗p < 0.05).
uMSCs Did Not Inhibit MCs Proliferation and Did Not Engraft the Kidney Tissues
To evaluate the effect of uMSCs on murine MC proliferation, we performed mesangial proliferation assays. No significant inhibition of MC proliferation by uMSCs at different ratios (1:1, 1:2, and 1:5 ratios of MCs to MSCs) was noted when MCs and uMSCs were cocultured for 12, 24, and 48 h (Fig. 6). To assess engraftment of transplanted human uMSCs to mice kidneys, recipient mice received partial nephrectomies 2 weeks postadministration of uMSCs and bilateral total nephrectomies at the end of the study. PCR analysis for human and mice-specific β2-micorglobulin showed that human DNA was detectable in the recipient kidney 2 weeks postadministration of uMSCs (Fig. 7A). No human DNA was detected in the kidney tissues 8 months after cell transplantation (Fig. 7B).
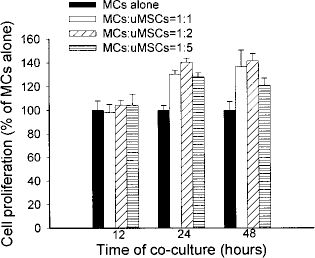
Effects of uMSCs on the growth of cultured NZB/W F1 mice MCs. No inhibitory effect of uMSCs on proliferation of MCs was found at different cell ratios after 12, 24, and 48 h of coculture. MCs: mesangial cells.

uMSCs did not engraft kidney tissues. (A) Human β2-microglobulin DNA was detectable in murine kidneys 2 weeks after administration of uMSCs by PCR analysis. (B) Human β2-microglobulin DNA was undetectable in the murine kidneys at 8 months after transplantation. DNA extracted from mouse B6 and human Hep3B cell lines was used as positive control for the mouse and human β2-microglobulin gene, respectively.
Discussion
In the present study, we clearly demonstrated that treatment of NZB/W F1 mice with uMSC transplantation markedly delayed renal function deterioration, reduced serum anti-dsDNA antibody level, and alleviated renal pathology change, as well as the development of proteinuria. The survival rate of mice treated with uMSCs at 2 months of age was 100% (8/8), while that of those treated at 6 months of age 86% (6/7), compared with 50% (4/8) in the control group. The difference in the survival rate between the control group and the mice treated at 2 months of age was statistically significant, but the difference between the control group and the mice treated at 6 months of age was not statistically significant. These findings support the view that early uMSCs transplantation may benefit SLE patients with LN, especially those at the early stages of the disease.
Preferential induction of Th1 or Th2 cell subsets has profound immunologic consequences, either pathogenic or protective, in autoimmune diseases. It has been established that T-cell abnormalities and aberrant T helper cytokine profiles play an essential role in the development of SLE, despite the debate regarding the dominance of Th1 or Th2 cytokines in SLE pathogenesis (9,12,21,25,42,48,55). Previous studies have shown that upregulation of Th1 cytokine IL-2 and IFN-γ together with a decreased production of Th2 cytokine IL-4 might upregulate autoantibody production by B cells and is associated with disease activity (9,12, 48,51,52). The administration of anti IFN-γ antibodies (19,41) or IL4pDNA (20) delays lupus development in NZB/W F1 mice, which also suggests the role of Th1 cytokines in SLE disease progression.
In addition to promising therapeutic potentials for tissue regeneration and repair, MSCs also possess immune-modulatory properties. We observed that uMSCs inhibit mitogen-stimulated lymphocytes and splenocytes in in vitro coculture studies. How uMSCs contribute to ameliorate progression of LN may be related to differential Th1/Th2 cytokine profiles. Compared to the control group, administration of uMSCs increased IL-4 and IL-10, and decreased IL-2 and IFN-γ, suggesting that uMSCs delay lupus autoimmunity by modulating T-cell differentiation and shifting Th1 to Th2 polarization. These findings are consistent with the concept that polarization of Th cytokines to Th2 may delay the onset of autoimmunity (24). Other proinflammatory cytokine networks, in particular TNF-α, IL-6, and IL-12, also play a critical role in the pathogenesis of SLE (3,51).
TNF-α is a central proinflammatory cytokine. It is able to upregulate IL-6, IL-1, and IL-18 production, which together may lead to renal inflammation and destruction (5). In addition, TNF-α is a growth factor for B lymphocyte through the autocrine loop (8). IL-12 is a 70-kDa heterodimer (IL-12 p70) produced by macrophages and dendritic cells. IL-12 p70 synergy with IL-18 stimulates the proliferation of activated T lymphocytes and their IFN-γ secretion. IL-12 triggers intrarenal inflammation and promotes a cytokine imbalance of peripheral cells toward a Th1 phenotype (13,33).
IL-6 is a cytokine with pleiotropic effects. It promotes the differentiation of B cells into immunoglobulin production cells, as well as the differentiation of T cells into effective cells. Elevation of serum IL-6 levels is observed in SLE patients with serositis, infection, and during disease exacerbation (15,43,46). Treatment of NZB/W F1 mice with anti-IL-6 mAb improves lupus (34). Data from our study revealed that the serum levels of TNF-α, IL-6, and IL-12 significantly decreased after uMSCs transplantation. Previous studies have shown that MSCs may work through direct interaction with immune cells or by secreted transforming growth factor-β, in-doleamine 2,3-dioxygenase, and prostaglandin E2, thereby promoting Th1 to Th2 polarization and anti-inflammatory dendritic cell type 2 signaling (1). From the above evidence, therapeutic advantages of uMSCs in this study were clearly conferred from their immunomodulatory capabilities.
Mesangial proliferation is one of the predominant pathological features of LN (44). Activation and proliferation of mesangium with release of proinflammatory materials such as prostaglandin, oxidants, and proteases is associated with the initiation of an inflammatory reaction and further glomerular injury in kidney diseases (16,18). MSCs are multipotential precursor cells that are capable of differentiation into cardiac myoblasts, hepatocytes, and neuroectodermal cells. Bone marrow (BM)-derived MSCs can also engraft the damaged kidneys and differentiate into tubular epithelial cells in the acute renal failure model. Repopulation of MCs in a rat anti-Thy-1 model has also been reported (27).
The mechanism of how uMSCs alleviate lupus renal disease in NZB/W F1 mice appears to be more complex. To evaluate whether uMSCs delay renal function deterioration only by immunomodulation, or whether it could also involve the inhibition of MCs proliferation or regeneration, we examined the in vitro inhibitory effect of uMSCs on the proliferation of mice MCs. We observed that uMSCs failed to inhibit the proliferation of MCs at different ratios. The finding of only temporary detection of human-specific β2-microglobulin 2 weeks after transplantation indicated that there was no permanent engraftment of uMSCs to the kidneys. Consequently, according to our results, we postulate that, by immune-modulatory effect rather than differentiation into renal resident cells, uMSCs contribute to the therapeutic effect and this explains poor engraftment of uMSCs to the kidney. It is consistent with the previous findings that the cytoprotective effect of systemically infused MSCs was mediated via soluble factors that acted in the paracrine manner on target tissues (23).
MSCs are hypoimmunogenic and can evade the host immune elimination (30). In animal experiments, it has been shown that human BM-derived MSCs administered intravenously are able to engraft in animals with or without a pretransplant total body irradiation (2,17). The therapeutic potential of human uMSCs was recently investigated by Morigi et al. in immunodeficient NOD/SCID mice with cisplatin-induced acute renal failure (36). Based on these facts, we used uMSCs to treat immune-competent NZB/W F1 mice.
It has recently been shown that MSCs from SLE patients demonstrated early signs of senescence, and autologous MSCs treatment had no effect on disease activity in SLE patients (11,40). Nevertheless, Sun et al. reported that BM-derived MSCs from related donors seem to be effective in ameliorating disease progression in four steroid-refractory patients (47). The above findings favored the use of allogenic rather than autologous MSCs for SLE treatment, and the current xenogenic study also support the use of HLA-mismatched MSCs. Importantly, it would be valuable if the therapeutic efficacy of UCB- and BM-derived MSCs for LN can be compared using the same experimental model.
In conclusion, we describe a xenogenic experimental cell therapy model of using uMSCs for LN treatment. Therapeutic effects of uMSCs relate to their immunomodulatory properties. Improvement of LN in NZB/W F1 mice caused by uMSCs is also associated with their inhibitory effects on T lymphocytes and a predominant inhibition of Th1 cytokines and proinflammatory cytokines TNF-α, IL-6, and IL-12 instead of directly affecting proliferation of MCs. Intravenous uMSCs transplantation may serve an alternative treatment for LN in SLE patients.
Footnotes
Acknowledgments
The authors acknowledge financial support from the Taipei Veterans General Hospital (VGH97F-013, VGH98E1-001, and VGH98C1-023) and the National Science Council (NSC95-2745-B-010-003-MY3, NSC97-2627-B-010-010, and NSC97-3111-B-010-003). This study was also supported by a grant from the Ministry of Education, Aim for the Top University Plan. The authors thank Professor Bor-Luen Chiang, M.D., Ph.D., from College of Medicine, National Taiwan University for his suggestions about the experiments. We also thank the staff from the Animal Facilities of the Department of Medical Research and Education, Taipei Veterans General Hospital for their help.
