Abstract
Introduction:
Rheumatoid arthritis (RA) is an autoimmune disease that is identified with chronic inflammation and progressive destruction of the joints. The defective activity of regulatory T cells (Tregs) plays a crucial role in RA development. Oleuropein (OLEU) is the most common polyphenolic compound in olive leaf extracts with numerous pharmacological activities. In this study, the potential effects of OLEU in shifting CD4+ T cells toward Tregs are evaluated in patients with RA.
Methods
32 healthy controls (HC) and 45 RA patients were included in two groups. The immunoturbidometric technique was used to measure serum levels of c-reactive protein (CRP) and rheumatoid factor (RF). Isolated CD4+ T cells from peripheral blood mononuclear cells (PBMCs) of HC and RA patients were cultured with appropriate concentrations of OLEU. The cytotoxicity effects of OLEU were determined using the MTT assay at 24, 48, and 72 h. The percentage of CD4+CD25 + FoxP3 regulatory T lymphocytes (Tregs) and the expressions of IL-10 and TGF-β were evaluated by flow cytometry and immunoassay techniques after treatment of cells with different concentrations of OLEU for 24 h. The serum levels of RF and CRP in patients with RA were 11.8 ± 5.32 IU/ml and 6.36 ± 5.82 mg/l, respectively.
Results
OLEU had a dose-dependent effect on the CD4+ T cells via increasing the frequency of CD4+CD25 + FoxP3 Tregs (p = 0.0001). Moreover, it induced the production of IL-10 (p = 0.0001) and TGF-β (p < 0.01) in both HC and RA patients.
Conclusion
The findings of this study suggest that OLEU may have immunomodulatory effects by inducing Tregs, and it might help in developing a novel nutrition strategy for management of autoimmune diseases such as RA.
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disease that typically results in disability, articular cartilage destruction, and bone resorption. 1 The hallmark of this systematic autoimmune disease is the breaking of immune tolerance to self-antigens leading to production of various autoantibodies including anti-cyclic citrullinated peptides antibodies (anti-CCPa), anti-carbamylated protein (anti-CarP) antibodies, and rheumatoid factor (RF).2,3 Both B-lymphocytes and T cells play crucial roles in the progression of many autoimmune/inflammatory diseases such as RA. 4 Regulatory T cells (Tregs) are a subpopulation of CD4+ T cells that specifically express higher levels of IL-2Rα (CD25) and transcriptional regulator forkhead box P3 (FoxP3). These cells play a key role in maintaining homeostasis and immune tolerance using various mechanisms such as production of regulatory cytokines interlekin-10 (IL-10), transforming growth factor-β (TGF-β) and IL-35, expressing co-inhibitory receptors cytotoxic T-lymphocyte-associated protein-4 (CTLA-4), and programmed cell death protein-1 (PD-1).5,6
Dysregulation of frequency and functions of Tregs might lead to development of inflammatory disorders and autoimmune diseases.7,8 Due to these potential functions, Tregs therapy has been studied as a promising option for prevention and/or treatment of autoimmunity. 9 Recently, many studies have indicated the protective role of Tregs in RA pathogenesis.2,4,10–12 In addition, a consistent number of studies have reported a decrease in the percentage of circulating Tregs in RA patients compared to healthy individuals.13,14 Clinical treatment of RA, essentially involves the use of medications to improve disease progression and minimize disease activity. The majority of these medications have shown adverse effects. 15 Therefore, novel therapeutic approaches should aim to re-establish self-tolerance before apparent tissue injury.
Targeting Tregs can be an effective way to regulate the immune system via increasing their number and improving the inhibitory function of Treg cells in patients with RA. A great quantity of data has been published with a special focus on the therapeutic targeting of these cells and their products with the aim of overcoming the limitations of currently-used treatments. 10 The effects of a variety of nutritional supplements and herbal extracts have been investigated in RA models, and the majority of them are developed from traditional medicines.16,17
Oleuropein (OLEU) is the most prevalent and distinctive polyphenolic compound found in olive oil and the leaves of the olive tree, which has attracted scientific attention in recent years due to a variety of well-documented health effects. 18 Numerous researches have reported the remarkable physiological and pharmacological properties of OLEU. In vivo and in vitro studies have shown that OLEU has antioxidant, antimicrobial, anti-tumoral, and anti-inflammatory activities. 19 According to the current data, its efficacy in decreasing inflammation might be equivalent to that of ibuprofen and other nonsteroidal anti-inflammatory drugs (NSAIDs). 20 In addition, it has been shown that consumption of OLEU reduced the spontaneous development of osteoarthritis in Hartley guinea pigs and improved the clinical characteristics in a mouse model of collagen-induced arthritis. 21 Other studies have also demonstrated the anti-inflammatory, antioxidant, and immunomodulatory effects of OLEU in rats with induced experimental autoimmune encephalomyelitis (EAE) 22 and induced systemic lupus erythematosus (SLE) in mice, 23 and human synovial fibroblast cell line SW982. 24 However, only a few studies have explored the potential effects of OLEU in RA patients.25,26 The present study was conducted to determine whether OLEU could shift CD4+ T cells toward the Tregs and change the frequency and production of inhibitory cytokines TGF-β and IL-10 in Treg cells in RA patients and healthy controls (HCs).
Material and methods
Subjects
The procedure of this study was approved by the local Ethics Committee (IR.SHMU.REC.1397.213) of Shahroud University of Medical Sciences, Shahroud, Iran. All the experimental methods followed the guidelines of the Declaration of Helsinki (2008), as stated in the guidelines of Iran's Medical Ethics Committee, Ministry of Health. The informed consent was obtained from all participants before entering the study.
In this clinical experimental research, the minimum sample size was calculated to be 30 per each group based on previous studies and StataCorp. 2003. Stata Statistical Software: Release 8. College Station, TX: StataCorp LP. Forty-five patients with RA were selected from those referred to a rheumatology center, Shahroud, Iran from October 2020 to March 2021. The demographical data and clinical characteristics of each patient were obtained from medical records. The c-reactive protein (CRP) value was used to measure Disease Activity Score 28 (DAS 28) for RA patients. In addition, blood samples were taken from patients for performing CRP, RF, and anti-CCPa analyses by standard laboratory techniques.
Inclusion criteria
Exclusion criteria: Patients with common immunosuppressants such as infection diseases, neoplasia, immunodeficiency, uncontrolled blood hypertension, and hepatic or renal failure who were under the age of 18 and had a disease duration of less than 2 years were excluded from the study. In addition, 32 healthy subjects without any sign of health problems and familial history of autoimmune disorders served as the control group. Controls with elevated CRP or RF positive were excluded from the study. We also excluded from both groups any participants who had received vaccination 4 weeks prior to the study, had a specific diet, were pregnant, and breast-feeding.
Blood sampling and peripheral blood mononuclear cells isolation
Blood samples were obtained from patients with RA and healthy individuals, and stored in K2-EDTA tubes. Peripheral Blood Mononuclear Cells (PBMCs) were isolated from whole blood using Ficoll-Paque density gradient centrifugation. In brief, an EDTA-anticoagulated blood sample was diluted with an equivalent volume of phosphate-buffered saline (PBS) containing 0.05 M ethylenediaminetetraacetic acid (EDTA; Merck, NJ, USA). The diluted blood was applied on top of the Ficoll-Paque PLUS (GE Healthcare®, Buckinghamshire, UK). Gradients were centrifuged in a swinging-bucket rotor at 2800 rpm for 20 min at room temperature. The PBMCs were carefully removed by pipetting and were washed twice with PBS-EDTA by centrifugation at 1500 rpm for 10 min. To lyse contaminated red blood cells (RBC), PBMC pellets were suspended in ammonium-chloride-potassium (ACK) lysing buffer (Sigma Aldrich, Germany) and incubated for 10 min at room temperature with gentle mixing, which was followed by a PBS-EDTA washing. Trypan blue exclusion assay was used to evaluate viable cell numbers.
CD4+ T cells separation
Human CD4+ T cells were isolated from prepared PBMCs using the CD4+ T cells isolation Kit and AutoMACS Pro Separator (Miltenyi Biotech, USA). To determine their purity, isolated cells were labeled with a combination of phycoerythrin (PE) anti-human CD4 (Biolegend, USA; clone: SK3) and fluorescein isothiocyanate (FITC) anti-human CD3 (Biolegend, USA; clone: UCHT1) antibodies or matched isotype control antibodies for 30 min at 4°C, and they were analyzed using FACScalibur flow cytometry (BD Biosciences, USA), and FlowJo V. 10 software (Flowjo, LLC). Data showed a purity >90% for the isolated cells.
Isolated CD4+ T cells were re-suspended in RMPI-1640 (Gibco, USA) medium supplemented with 10% fetal bovine serum (FBS) (Gibco) and 100 ug/ml penicillin-streptomycin. Then, they were transferred to 24-well tissue culture plates and incubated at 37°C for 72 h, after stimulation with 10 ug/ml phytohemagglutinin (PHA) (Gibco) to induce cell proliferation and increase the total number of purified CD4+ T cells. The results were compared to the cell culture of the control group, with and without PHA stimulation.
Oleuropein preparation
OLEU made from powdered olive leaf extract was purchased from Chymal Chemical Company (Sigma, USA, Cat: 12247-10MG) and prepared by dissolving it in dimethyl sulfoxide (DMSO) according to the manufacturer’s instructions and was stored until usage at −20 or −80°C.
Evaluation of the cytotoxic effects of Oleuropein on CD4+ T cells
The MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide) assay was performed to determine the cytotoxic effects of OLEU on CD4+ T cells. The MTT assay is a colorimetric method for determining the rate of tetrazolium conversion to formazan in live cells. The cultured human T cells were plated in 96-well plates and treated with 25, 50, 100, and 200 μg/ml concentrations of OLEU at time points of 24, 48, and 72 h at 37°C, 95% humidity, and 5% CO2. The wells containing OLEU solvent (DMSO) and PHA alone were used as controls. The MTT (Sigma Aldrich) was added to each well at a concentration of 5 mg/mL and the wells were incubated for 4 h at 37°C. The medium was removed at the end of the time period specified, and insoluble blue-violet dye formazan crystals were dissolved with 100 μl DMSO. The absorbance of the formazan solution was read at 570 nm using an enzyme-linked immunosorbent assay (ELISA) reader and the results were presented as percentage of optical density (OD) in both treated cells and controls. Half-maximal inhibitory concentration (IC50) was determined using linear regression. Three suitable concentrations of OLEU were selected to continue the study. 28
Flow cytometry
To determine whether OLEU has any effect toward shifting CD4+ T cells to CD4+CD25+FoxP3+ Tregs, the frequency of Tregs were determined in RA patients and healthy controls using flow cytometry. 29 Briefly, human CD4+ T cells, treated with various concentrations of OLEU (0, 50, 100, and 200 μg/ml), were collected after 24 h, washed, and then suspended in PBS containing 0.1% BSA (Sigma-Aldrich). The cells were stained with a combination of Allophycocyanin (APC) anti-human CD25 (Biolegend, USA; clone: BC96), phycoerythrin (PE) anti-human CD4 (Biolegend, USA; clone: SK3), and Alex Flour® 488 anti-human FoxP3 (Biolegend, USA; clone: 206D) antibodies or matched isotype control antibodies for 30–45 min at 4°C. After incubation, the cells were washed in PBS containing 0.1% bovine serum albumin (BSA) (Sigma-Aldrich, Germany), and were analyzed using FACScalibur flow cytometry (BD Biosciences, USA) and FlowJo V 10 software (Flowjo, LLC). Additionally, Fluorescence minus one (FMO) controls were used to ensure the correct cell population gating and fluorescence spread.
Cytokine detection by enzyme-linked immunosorbent assay
The cell culture supernatants from both untreated and treated CD4+ T cells with OLEU (50, 100, and 200 μg/ml) were collected after 24 h and spun down (1500 rpm, 5 min), and cell-free supernatants were stored at −80°C until they were used. The assays were done in duplicate using human IL-10 and TGF-β commercial ELISA kits (Bioassay Technology Laboratory, China) according to the manufacturer's instructions. Absorbance was determined at 450 nm with the ELISA reader, and concentrations of the cytokines were measured from standard curves and were adjusted by the dilution factor.
Statistical analysis
Data analysis was carried out using IBM SPSS 20 and Microsoft Excel 2016. Bonferroni adjustment and Dunn’s post hoc tests were performed on each pair of groups. A parametric two-way ANOVA test was used to determine the source of significant variations in the secretion of inhibitory cytokines and the frequency of Tregs among groups. Data was presented as mean ± standard deviation (SD) based on at least three independent experiments, and a difference of p < 0.05 was considered statistically significant.
Results
Demographic and laboratory characteristics
The demographic, clinical, and laboratory features of RA patients and healthy subjects.

RF: rheumatoid factor; CRP: c-reactive protein; Anti-CCPa: anti-cyclic citrullinated peptides antibody; DAS28: disease activity score in 28 joints; tDMARDs: traditional disease-modifying antirheumatic drugs; MTX: methotrexate; CsA: cyclosporine; LEF: leflunomide; SSZ: sulfasalazine; HCQ: hydroxychloroquine; NSAIDs: nonsteroidal anti-inflammatory drugs; bDMARDs: biological disease-modifying antirheumatic drugs; ETA: etanercept; ADA: adalimumab; RA: rheumatoid arthritis.
Cytotoxicity effects of Oleuropein
Treatment of CD4+ T cells with OLEU (25–200 μg/ml) for 24, 48, and 72 h resulted in a limited decrease in cell viability compared to the cells cultured with PHA or DMSO. However, OLEU treatment did not significantly effect on cell viability. Based on IC50 values, the results confirmed the lack of cytotoxicity of OLEU at the concentrations tested here at the three mentioned different time points (Figure 1(a)–(c)). Effects of OLEU on cell viability at time points (a) 24h, (b) 48h, (c) and 72h. The MTT results revealed that OLEU had no significant effects on cell viability in any of the examined concentrations at the three different time points. PHA: phytohemagglutinin; DMSO: dimethyl sulfoxide; OLEU: oleuropein; h: hours.
Effects of Oleuropein on CD4+ T cells’ shift toward CD4+CD25+FoxP3 tregs
There was a slight difference in the percentage of cell viability after treatment of cells with various concentrations of OLEU at the three mentioned time points. No considerable difference in cell viability percentage was identified between treatments with 25 and 50 μg/ml of OLEU. Therefore, 50, 100, and 200 μg/ml concentrations of OLEU were selected to continue the study. The frequency of CD4+CD25+FoxP3 Tregs were evaluated in HC and patients with RA using flow cytometry, before and after treatment of CD4+ T cells with OLEU. Gating strategy and flow cytometry analysis of Tregs frequency in one healthy individual and one patient with RA are demonstrated in Figures 2(a) and Figures 2(b). In RA patients the levels of CD4+CD25+FoxP3 Tregs was lower compared to HC, before treatment initiation of CD4+ T cells with OLEU (Figure 3a). A positive correlation was found between the frequency of CD4+CD25+FoxP3 Tregs and different concentrations of OLEU, in the sense that the percentage of CD4+CD25+FoxP3 Tregs was increased significantly, as OLEU dosage was increased (Figures 3(b) and (c)). Flow cytometry plots of CD4+CD25+ FoxP3 Tregs from one healthy individual and one patient with RA. (a) Isolated CD4+ cells from prepared PBMCs were stained with anti-CD4, anti-CD25, and anti-FoxP3 antibodies; (b) The frequency of CD4+CD25+FoxP3 Tregs was determined using flow cytometry. Quantitative analysis of CD4+CD25+FoxP3 Tregs frequency in HC and RA patients following treatment of cells with or without various concentrations of OLEU. (a) The prevalence levels of Tregs in HC and RA patients at baseline (before treatment with OLEU). Dose dependent effects of OLEU on Tregs frequency (b) in HC (c) and RA patients.* p < 0.0001. RA: rheumatoid arthritis; HC: healthy control; OLEU: oleuropein.
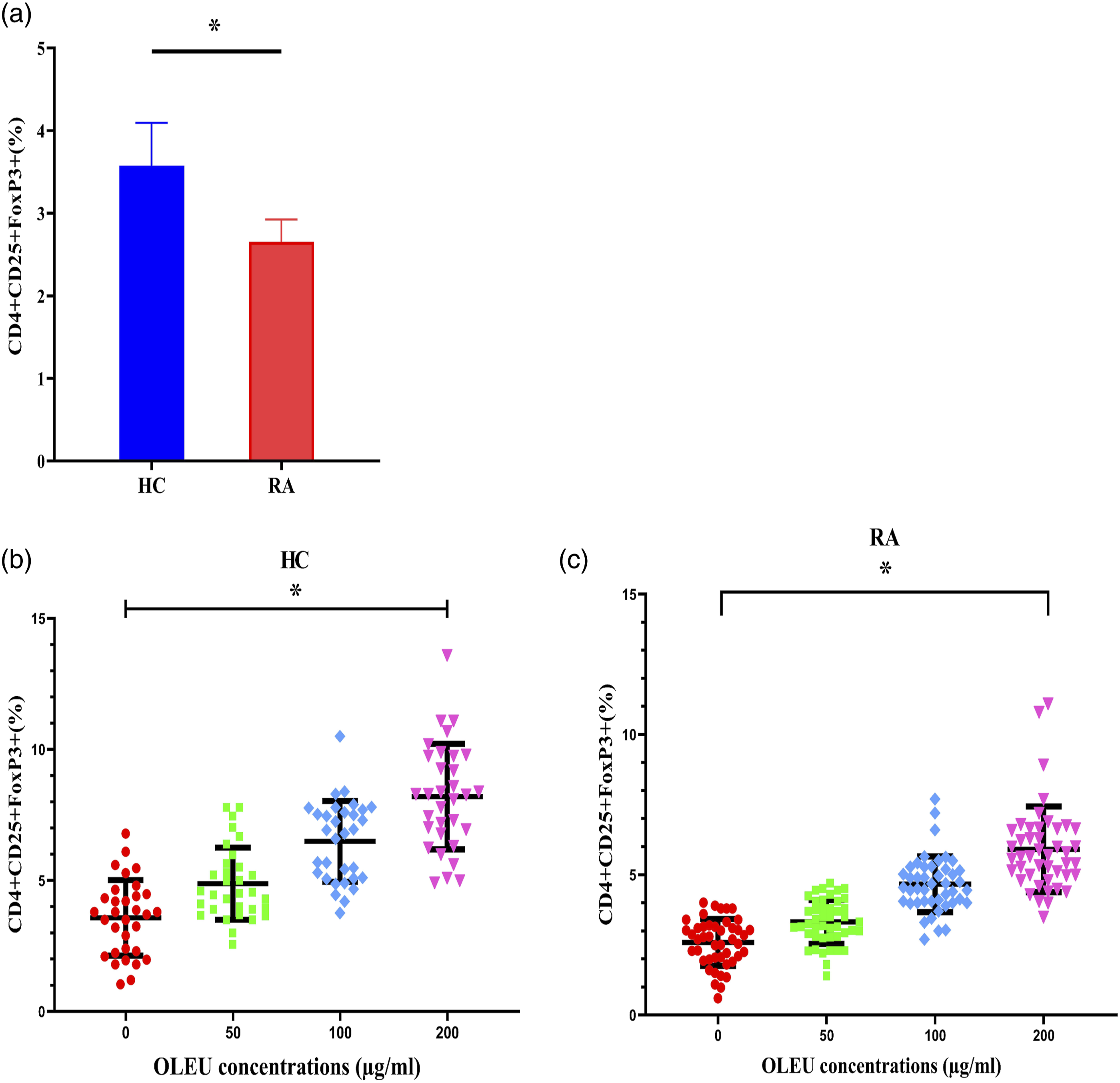
However, no significant statistical differences were identified in the frequency of CD4+CD25+FoxP3 Tregs between the HC and RA groups, after the treatment of cells with OLEU. Although majority of patients who had treated with most common DMARDs including methotrexate, etanercept, and adalimumab revealed lower levels of Tregs compared to HC at baseline (before the treatment with OLEU, Supplementary Figure 1).
Effects of Oleuropein on IL-10 and TGF-β productions
Levels of IL-10 and TGF-β were evaluated in the supernatants of cultured T cells with selected concentrations of OLEU using the ELISA method, to identify whether OLEU could change the levels of IL-10 and TGF-β, the two cytokines mainly involved in the immune response regulation and inhibition of development of autoimmune diseases. The levels of these cytokines were constantly lower in RA patients compared to HC at baseline (p < 0.01) (Figure 4(a) and (b)). At all the tested concentrations, OLEU caused a significant increase in IL-10 levels in both HC (p = 0.0001) and RA (p = 0.0001) groups (Figure 4(c) and (d)). The data provided similar results regarding the effects of OLEU on TGF-β production in HCs (p = 0.004) and patients with RA (p = 0.003) (Figure 4(e) and (f)). It was also observed that there is a positive linear correlation between the levels of IL-10 and TGF-β with different doses of OLEU (µg/ml). Data analysis revealed that there were no significant differences between RA and HC groups in the levels of IL-10 and TGF-β after treatment of the cells with OLEU, which was in consistent with the above findings. Supplementary Table 1 showed the frequency value of CD4+CD25+FoxP3 Tregs and the levels of measured cytokines in different concentrations of OLEU at both HC and RA patients as mean ± SD. OLEU increased the levels of IL-10 and TGF-β in HC and patients with RA. (a, b) The levels of IL-10 and TGF-β in HC and RA groups were determined using ELISA assay. These cytokines were significantly increased in both (c, e) HC and (d, f) RA patients in a dose-dependent manner, after treatment of cells with different concentrations of OLEU. * p < 0.01. RA: rheumatoid arthritis; HC: healthy control; OLEU: oleuropein.
Discussion
The main purpose of arthritis therapy is development of high-quality, safe, and inexpensive drugs that specifically promote immune response tolerance and reduce the progression of RA. 30 Multiple medicines are currently used to treat RA patients. NSAIDs, immunosuppressive medications, and corticosteroids are the most widely used classes of medicines.31–34 The lack of efficacy, undesirable side effects, and poor clinical results observed in some of the patients have limited the use of these medications. 35 Recently, the potential effects of important natural plant-based compounds and their derivatives such as resveratrol, curcumin, matrine, quercetin, and sylimarin, have been extensively investigated on a variety of disorders including autoimmunity.16,36–43 The majority of natural/herbal medications can control different diseases by inhibiting pro-inflammatory mediators and increasing the frequency and activity of Tregs.36,44–46 Therefore, medicinal plants are important sources for development of new immunomodulatory and anti-inflammatory drugs. However, lack of sufficient knowledge about their mechanisms of action has restricted the clinical use of these compounds. Currently, various studies are being conducted to determine the efficacy of different herbal compounds, which is critical for scientific validation of using these products in alternative and/or complementary medicines. OLEU is a well-known natural compound present in olive leaf extracts that has shown effective pharmacological and biological effects.15,17,18 In addition, several studies have demonstrated the protective effects of OLEU against RA by modulating pro-inflammatory mediators.24,50,51
RA is a heterogenous disorder with various clinical symptoms. 52 Although pro-inflammatory cytokines play a crucial role in the RA pathogenesis, Treg cells are also involved in it by regulation of immune responses and maintaining self-tolerance.4,10 Therefore, manipulation of Tregs is a potential method for treating autoimmune diseases such as RA. The aim of this study was to examine the effects of OLEU on shifting CD4+ T cells toward Tregs in RA patients. For this purpose, the percentages of CD4+CD25+FoxP3 Tregs and the levels of IL-10 and TGF-β were analyzed in healthy individuals and RA patients, before and after treatment of T cells with OLEU. The outcome of this research revealed that the percentage of Tregs was lower in RA samples compared to HC, indicating that Tregs play a crucial role in RA development. The substantial defect in the frequency of Tregs has previously been identified in RA patients.13,14,53 However, the flow cytometry results showed no statistically significant difference in Treg frequencies between HC and RA patients, which might be related to the different effects of conventional and biological DMARDs on Tregs in RA patients.54–57 A number of studies have shown that treatment of RA patients with some but not all of DMARDs, such as methotrexate, tumor necrosis factor-α inhibitors (TNF-αi), adalimumab, and etanercept can restore the regulatory functions of Tregs and increase the frequency of these cells through various mechanisms.54,55,58,59 Therefore, the effects of OLEU on Tregs could be influenced by treatment of RA patients with various DMARDs. In addition, it was found that treatment of isolated CD4+ T cells with OLEU significantly increased the percentage of Treg cells in both HC and RA groups in a dose-dependent manner.
To investigate the underlying mechanisms of the OLEU’s effects on Tregs, IL-10 and TGF-β concentrations in the supernatants of cultured T cells were assessed following the treatment of cells with OLEU. OLEU had a similar effect on the levels of these cytokines and significantly increased the production of IL-10 and TGF-β in HC and RA groups. IL-10 and TGF-β are the most important inhibitory cytokines that are primarily produced by Tregs and are eventually involved in the Tregs’ function. 60 This should be noted that there are contradictory reports on the levels and functions of these cytokines in RA. Some studies have found decreased levels of IL-10 in the serum of patients with RA compared to healthy individuals, while others have found higher IL-10 levels. 61 The meta-analysis demonstrated a link between the levels of this cytokine and susceptibility to RA with IL-10 polymorphism. In this study, RA patients with IL-10 -592AA and −592CA genotypes showed lower levels of IL-10 protein. 62 In addition, IL-10 has dual effects in the pathogenesis of RA. Although IL-10 down regulates the pro-inflammatory cytokines TNF-α and IL-6, it also enhances autoantibody production by B cells stimulation.39,40
TGF-β is a type of multipotent cytokine with regulatory and inflammatory functions, which is secreted by resident cells in the synovium of RA patients. 65 Higher levels of this cytokine have been detected in the serum and synovial fluid of patients with RA. 66 In addition, the importance of TGF-β in the RA pathogenesis has already been established by numerous studies. The effects of TGF-β on RA progression are affected by environmental conditions as well as local or systemic injection of TGF-β. Local administration of TGF-β in animal models of RA induces the incidence of RA and joint damage by increasing the production of metalloproteinase and proinflammatory cytokines. In contrast, systemic administration of this cytokine reduces the severity of RA by activating regulatory T cells.65,67 Therefore, treatment of T cells with OLEU at concentrations of 50, 100, and 200 µg/ml enhances the frequency of CD4+CD25+FoxP3 Tregs and induces the production of IL-10 and TGF-β cytokines in HC and patients with RA in a dose-dependent manner. These results indicated that OLEU can modulate the immune response by targeting Tregs, and thus it could be utilized as an effective natural medication for prevention or treatment of autoimmune diseases. However, various functions of IL-10 and TGF-β in RA have limited the pharmacological application of OLEU in RA patients. Further research is required before using this major bioactive component (obtained from olive leaf extract) for management of RA.
There are some limitations to the current study. First, an in vitro experiment was carried out to assess the potential effects of OLEU on Tregs in RA patients and determine the optimal concentrations of this natural product. Although the experimental results provided important information about the effects of OLEU on the frequency and functions of Tregs, they failed to predict the adverse effects of OLEU on immune responses and its physiological concentration range. Second, this study included RA patients treated with various DMARDs, which may have influenced the observed effects of OLEU on Tregs. In the future, a combination of in vitro and in vivo pre-clinical studies using onset-new RA patients will be required to clarify the safety, efficacy, and appropriate concentrations of OLEU.
Conclusions
The current study revealed the potential effects of OLEU on shifting CD4+ T cells toward CD4+CD25+FoxP3 Tregs and inducing the production of IL-10 and TGF-β in both healthy individuals and patients with RA in a dose dependent manner. Considering that the major aim of the current treatment methods for autoimmunity is to reduce inflammation and maintain self-tolerance, OLEU could be used in developing a new nutrition strategy for management of autoimmune disorders. However, use of OLEU in RA therapy needs more research.
Supplemental Material
sj-pdf-1-iji-10.1177_03946320221086084 – Supplemental Material for Dose-dependent effects of oleuropein administration on regulatory T-cells in patients with rheumatoid arthritis: An in vitro approach in International Journal of Immunopathology and Pharmacology
Supplemental Material, sj-pdf-1-iji-10.1177_03946320221086084 for Dose-dependent effects of oleuropein administration on regulatory T-cells in patients with rheumatoid arthritis: An in vitro approach by Zahra Yousefi, Zahra Mirsanei, Fatemeh S Bitaraf, Sepideh Mahdavi, Mehdi Mirzaei and Reza Jafari in International Journal of Immunopathology and Pharmacology
Supplemental Material
sj-pdf-2-iji-10.1177_03946320221086084 – Supplemental Material for Dose-dependent effects of oleuropein administration on regulatory T-cells in patients with rheumatoid arthritis: An in vitro approach in International Journal of Immunopathology and Pharmacology
Supplemental Material, sj-pdf-2-iji-10.1177_03946320221086084 for Dose-dependent effects of oleuropein administration on regulatory T-cells in patients with rheumatoid arthritis: An in vitro approach by Zahra Yousefi, Zahra Mirsanei, Fatemeh S Bitaraf, Sepideh Mahdavi, Mehdi Mirzaei and Reza Jafari in International Journal of Immunopathology and Pharmacology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Shahroud University of Medical Sciences, Shahroud, Iran (Research Grant: 97137).
Ethics approval
Ethical approval for this study was obtained from * local Ethics Committee at Shahroud University of Medical Sciences, Shahroud, Iran. (IR.SHMU.REC.1397.213)*.
Informed consent
Written informed consent was obtained from all subjects before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
