Abstract
Endoplasmic reticulum (ER) stress is implicated in the pathogenesis of many diseases, including myocardial ischemia/reperfusion injury. We hypothesized that human umbilical cord mesenchymal stromal cells derived extracellular vesicles (HuMSC-EVs) could protect cardiac cells against hyperactive ER stress induced by hypoxia/reoxygenation (H/R) injury. The H/R model was generated using the H9c2 cultured cardiac cell line. HuMSC-EVs were extracted using a commercially available exosome isolation reagent. Levels of apoptosis-related signaling molecules and the degree of ER stress were assessed by western blot. The role of the PI3K/Akt pathway was investigated using signaling inhibitors. Lactate dehydrogenase leakage and 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) analysis were used for evaluating the therapeutic effects of HuMSC-EVs in vitro. The results showed that ER stress and the rate of apoptosis were increased in the context of H/R injury. Treatment with HuMSC-EVs inhibited ER stress and increased survival in H9c2 cells exposed to H/R. Mechanistically, the PI3K/Akt pathway was activated by treatment with HuMSC-EVs after H/R. Inhibition of the PI3K/Akt pathway by a specific inhibitor, LY294002, partially reduced the protective effect of HuMSC-EVs. Our findings suggest that HuMSC-EVs could alleviate ER stress–induced apoptosis during H/R via activation of the PI3K/Akt pathway.
Keywords
Introduction
Acute myocardial infarction is a significant cause of mortality worldwide. While timely reperfusion, such as primary percutaneous coronary intervention, has proven to be an invaluable therapy, subsequent reperfusion induces further cardiac damage during a process known as myocardial ischemia/reperfusion (MI/R) injury 1 . The mechanisms underlying I/R injury appear to be multifactorial, including elevated oxidative stress, intracellular Ca2+ overload, and endoplasmic reticulum (ER) stress 2 . The ER is the main organelle responsible for the regulation of intracellular homeostasis (including the translocation and post-transcriptional modification of proteins), lipid synthesis, calcium homeostasis, normal cell function, and survival 3,4 . Perturbations to any of these processes can result a large proportion of proteins that are misfolded and accumulate inside the ER lumen, resulting in ER stress 5 . The activation of intracellular signal transduction pathways by ER stress is defined as the unfolded protein response (UPR). Activation of the UPR triggers an adaptive program designed to restore homeostasis by increasing the folding capacity of the cell, reducing protein synthesis, and enhancing the clearance of abnormally folded proteins and damaged organelles 6 . But excessive ER stress induces apoptosis 7 . Previous studies revealed that ER stress–initiated apoptosis plays a very important role in MI/R injury 8,9 . Therefore, ER stress, as a potential target for the treatment of I/R-induced myocardial injury, has been an area of extensive research.
Mesenchymal stromal cells (MSCs) are present in a variety of tissues such as adipose tissue, peripheral blood, bone marrow, and umbilical cord 10 . This type of cell has been shown to protect against I/R injury through several mechanisms including immunomodulation, as well as antiapoptotic and proangiogenic effects 11 . Among these sources, human umbilical cord mesenchymal stromal cells (HuMSCs) are more suitable for replacement therapy because of fewer ethical issues, low immunogenicity, and high capacity for self-renewal 12 .Our group and others previously reported the potential therapeutic effect of HuMSCs in the treatment of cardiac injury 13,14 . Although transplanted MSCs exhibited low survival in the host, and only a small percentage of transplanted MSCs were able to reach the target site, MSCs transplantation has been shown to work well under certain conditions, primarily owing to the paracrine capability 15,16 .
Extracellular vesicles (EVs) are small, membrane-bound nanovesicles that range in size from 50 to 200 nm and contain nucleic acids, proteins, and bioactive lipids 17 . Several lines of evidence suggest that exosomes are active paracrine elements that play an important role in cell-to-cell communication 18 . Exosomes therefore have great potential to promote tissue repair 19,20 . Recently, Wang et al. 21 reported that exosomes derived from bone marrow mesenchymal stem cells protect against renal I/R injury by inhibiting ER stress. However, the relationships of HuMSC-derived extracellular vesicles (HuMSC-EVs) to MI/R injury and ER stress–induced apoptosis are unknown.
In this study, we used an in vitro hypoxia/reoxygenation (H/R) injury system to investigate whether HuMSC-EVs protects against MI/R injury through the regulation of ER stress.
Materials and Methods
Cell Culture
HuMSCs were isolated as previously described 13 . The study protocol was approved by the Institutional Review Board of Shantou University Medical College (Shantou, China). A total of six patients provided HuMSC. All patients provided signed informed consent. HuMSCs were cultured in Dulbecco’s modified Eagle’s medium (DMEM)/F12 media containing 10% exosome-depleted fetal bovine serum (Ca #C38010050, VivaCell Biosciences, Shanghai, China) in an incubator held at 37°C and filled with 5% CO2. The third passage of HuMSCs was used for exosome extraction. H9c2 cardiac cells were obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA), then cultured in DMEM supplemented with 10% fetal bovine serum, 100 U/ml streptomycin, and 100 U/ml penicillin, in an incubator held at 37°C and filled with 5% CO2.The PI3K/Akt inhibitor (LY294002) was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Extraction and Identification of HuMSC-EVs
HuMSC-EVs were isolated using ExoQuick-TC (System Biosciences,Palo Alto, CA, USA), according to the manufacturer’s protocol. Purified HuMSC-EVs were identified by transmission electron microscopy (JEOL JEM 1230; Tokyo, Japan). Exosomal markers such as CD63 and CD9 were analyzed by western blot and flow cytometry. Nanoparticle tracking analysis (NTA) was performed to analyze the exosome particles using a Nanoparticle Tracking Analyzer (ZetaView, PMX, Meerbusch, Germany). The protein concentration of the purified exosome fraction was measured with a bicinchoninic acid (BCA) protein assay kit (ThermoFisher Scientific, Waltham, MA, USA).
Internalization of HuMSC-EVs by H9c2 Cells
HuMSC-EVs were incubated with PKH26 (Ca#MIDI26, Sigma-Aldrich, Shanghai, China) for 15 min at 37°C in the dark and washed three times in 1× phosphate buffered saline with centrifugation at 100,000×g, followed by incubation at 4°C for 2 h. The purified EVs were then added to the culture medium and incubated with H9c2 cells for 12 h. Cell nuclei were labeled with 4’,6-diamidino-2-phenylindole (Ca#C1005, Beyotime Biotechnology, Shanghai, China). The uptake of HuMSC-EVs by H9c2 cells was analyzed using Olympus BX51 confocal microscopy (Olympus, Tokyo, Japan).
Generation of the H/R Model
To generate oxygen-glucose deprivation (OGD), the initial culture medium was replaced with buffer [pH 6.2, 137 mM NaCl, 12 mM KCl, 0.49 mM MgCl2·6H2O, 0.9 mM CaCl2, 4 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid, and 20 mM Na lactate] to generate OGD 22 . Then, H9c2 cells were exposed to pure N2 gas at 37°C for 3 h. H9c2 cells were then reoxygenated in fresh oxygenated culture medium for 1 h, in an incubator with 5% CO2. Four experimental groups were designed as follows: normal group, in which H9c2 cells were cultured under normal conditions; H/R group; H/R+ HuMSC-EVs group, in which H9c2 cells were first cultured with HuMSC-EVs (8 μg/ml) for 12 h, followed by exposure to H/R; and H/R + HuMSC-EVs + LY group, in which the PI3 K inhibitor LY294002 was added to cells at a concentration of 15 μmol/l for 30 min before H/R.
Cell Viability Assay
H9c2 cell viability was determined by 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide (MTT) assay. After exposure to H/R, MTT was added to the culture medium for incubation for 4 h at 37°C. MTT was then solubilized with 150 µl dimethyl sulfoxide. The optical density (OD) values were determined at 490 nm by a microplate reader. Relative cell viability was calculated as a percentage, as follows: viability (%) = (OD of assay − OD of blank)/(OD of control − OD of blank) × 100%. The experiment was repeated three times.
Assay of Lactate Dehydrogenase Activity
To determine the extent of cell injury induced by H/R injury, lactate dehydrogenase (LDH) activity in culture media was measured using a lactate dehydrogenase activity assay kit (Uscn Life Science, Wuhan, China), according to the manufacturer’s protocol. The experiment was repeated three times.
Western Blot
H9c2 cells or HuMSC-EVs were lysed in RIPA lysis buffer, followed by whole-protein purification. Protein concentrations were quantified using the BCA method (Beyotime Institute of Biotechnology, Haimen, China). Total protein (30 μg) was subjected to 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred electrophoretically to polyvinylidene difluoride membranes (EMD Millipore, Billerica, MA, USA). Membranes were incubated with the primary antibody of interest for 16 h at 4°C, followed by incubation with horseradish peroxidase–conjugated goat anti-rabbit immunoglobulin G secondary antibody (1:2,000, Ca# BE0101, EASYBIO, Beijing, China) for 1 h at room temperature. Specific protein bands were observed with an ECL Plus chemiluminescence kit (EMD Millipore) and quantitatively analyzed with Image Pro Plus 6.0 software (Media Cybernetics, Inc., Rockville, MD, USA).
The primary antibodies used in this study were: CD9 (1:2,000, Ca#ab92726, Abcam, Cambridge, MA, USA); CD63 (1:1,000, Ca#ab134045, Abcam); Bax (1:5,000, Ca #ab32503, Abcam); bcl2 (1:2,000, Ca#ab196495, Abcam); cleaved-caspase 3 (1:1,000, Ca#9661, CST, Danvers, MA, USA); glucose-regulated protein 78 (GRP78; 1:1,000, Ca#3183, CST); C/EBP homologous protein (CHOP; 1:1,000, Ca#2895, CST); inositol-requiring protein 1α (IRE1α; 1:1,000, Ca#3294, CST); activating transcription factor 6 (ATF6; 1:1,000, Ca #ab203119, Abcam); Akt (1:1,000, Ca#4685, CST); p-Akt (1:1,000, Ca #4060, CST); PI3 K (1:1,000, Ca#4257, CST); and β-actin (1:5,000, Ca#0021, EASYBIO). The experiment was repeated three times.
Statistical Analysis
All statistical analyses were performed using SPSS 16.0 software (SPSS Inc., Chicago, USA). Data are expressed as means ± standard deviation. Statistical differences between groups were assessed with one-way analysis of variance, followed by Tukey’s multiple comparison test. A P-value <0.05 was considered statistically significant.
Results
Characterization of HuMSC-EVs
We characterized the HuMSC-EVs by transmission electron microscopy, which revealed the presence of spherical vesicles in the HuMSC-EVs isolated (Fig. 1A). The concentration of exosomes was 5.4 × 109 particles/ml. Particle diameter ranged from 27 to 139 nm (as measured by the NTA system; Fig. 1B). We next examined EV markers CD9 and CD63 by flow cytometry and western blot. As shown in Fig. 1C, D, the isolated HuMSC-EVs expressed high levels of CD9 and CD63. These findings suggested that EVs were successfully isolated from HuMSCs.

Characterization of HuMSC-EVs. (A) Images obtained by transmission electron microscopy showing the morphology of HuMSC-EVs (red arrows point to the EVs). (B) NTA of HuMSC-EVs. (C) Flow cytometric analysis of EV markers showing positive staining for CD9 and CD63. (D) Western blot analysis of EV surface markers (CD9 and CD63). (E) Internalization of HuMSC-EVs by H9c2 cells, as detected by fluorescence microscopy. HuMSC-EVs: human umbilical cord mesenchymal stromal cells derived extracellular vesicles; NTA: nanoparticle tracking analysis.
We also tested the internalization of HuMSC-EVs by H9c2 cells. We labeled HuMSC-EVs with PKH-26. After labeling, the HuMSC-EVs pellet showed a strong red color. PKH-26-labeled HuMSC-EVs were then incubated with H9c2 cells. As shown in Fig. 1E, we observed red fluorescence in the cytoplasm in nearly all H9c2 cells, indicating that large amounts of HuMSC-EVs had been taken up by cultured H9c2 cells.
HuMSC-EVs Alleviates H/R Injury
We assessed the viability of H9c2 cells via MTT assay. As shown in Fig. 2A, H/R injury significantly reduced cell viability, as expected, HuMSC-EVs improved cell viability in H9c2 cells subjected to H/R (## P < 0.01 vs control, **P < 0.01 vs H/R). LDH leakage is widespread use as a marker of cellular damage. According to Fig. 2B, LDH leakage increased in the H/R group compared with the control group, but was significantly decreased by HuMSC-EVs treatment (## P < 0.01 vs control, **P < 0.01 vs H/R). These findings revealed that HuMSC-EVs could promote cell survival and alleviate cell damage in H9c2 cells subjected to H/R injury.
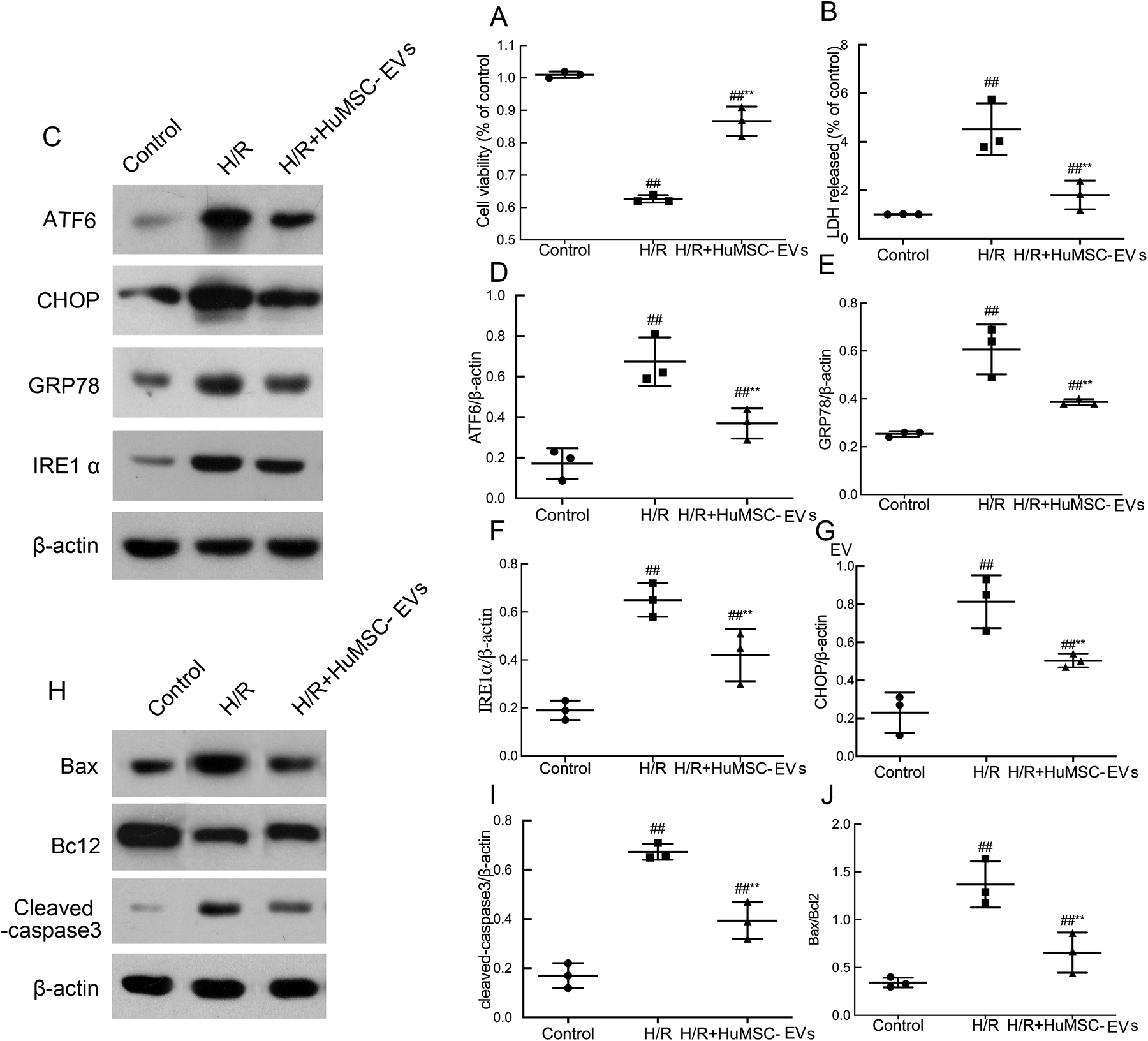
Effects of HuMSC-EVs on cell survival and ER stress in response to H/R insult. (A) HuMSC-EVs promote survival after H/R. (B) HuMSC-EVs decrease the release of LDH after H/R. (C-J) Analysis of protein expression and optical density for GRP78, CHOP, IRE1α, ATF6, cleaved-caspase 3, and Bax/bcl2 in H9c2 cells. Values are means ± SD. # P < 0.05 vs control; ## P < 0.01 vs control; *P < 0.05 vs H/R; **P < 0.01 vs H/R. β-Actin served as an internal control. Values are means ± SD. # P < 0.05, ## P < 0.01 vs control; *P < 0.05, **P < 0.01 vs H/R (n = 3/group). ATF6: activating transcription factor 6; CHOP: C/EBP homologous protein; ER: endoplasmic reticulum; GRP78: glucose-regulated protein 78; H/R: hypoxia/reoxygenation; HuMSC-EVs: human umbilical cord mesenchymal stromal cells derived extracellular vesicles; IRE1α: inositol-requiring protein 1α; LDH: lactate dehydrogenase; SD: standard deviation.
HuMSC-EVs Reduced H/R-Induced ER Stress and Ameliorated H9c2 Cell Apoptosis
H/R is thought to damage cardiac cells by inducing severe ER stress and apoptosis 23 . To confirm the role of HuMSC-EVs in H/R-induced ER stress, the expression of ER stress markers (including GRP78, CHOP, IRE1α, and activating transcription factor 6) were measured by western blot analysis. As expected, levels of all markers investigated were significantly enhanced in the H/R group, indicating increased ER stress during H/R induction. Interestingly, the expression of these markers decreased significantly in the HuMSC-EVs group, compared with the H/R group (Fig. 2C; # P < 0.05, ## P < 0.01 vs control; *P < 0.05, **P < 0.01 vs H/R). To investigate the direct effect of HuMSC-EVs on apoptosis in cardiac cells, the expression levels of Bax, Bcl-2, and cleaved-caspase 3 were measured. In the H/R group, Bax and cleaved-caspase 3 were upregulated, while the expression of Bcl-2 was downregulated. HuMSC-EVs treatment restored the expression levels of all three proteins to baseline levels in H9c2 cells (Fig. 2H; # P < 0.05, ## P < 0.01 vs control; *P < 0.05, **P < 0.01 vs H/R). These data show that HuMSC-EVs reduce H/R-induced ER stress and ameliorate apoptosis in cardiac cells.
HuMSC-EVs Protects Against H/R-Induced ER Stress–Induced Apoptosis Through Activation of the PI3K/Akt Pathway in H9c2 Cells
Previous studies have shown that the PI3K/Akt pathway is activated by the delivery of exosomes 24,25 .To further investigate the mechanism by which HuMSC-EVs modulate ER stress, the expression of PI3 K, Akt, and p-Akt in H9c2 cells was measured by western blot analysis. As shown in Fig. 3A, pretreatment with HuMSC-EVs markedly upregulated the expression of PI3 K and p-Akt/Akt, compared with levels observed in the H/R group. Furthermore, a specific inhibitor of PI3K/Akt, LY294002, alleviated the inhibitory effects of HuMSC-EVs on the overexpression of GRP78, CHOP, IRE1α, and ATF6, suggesting that the inhibition of ER stress by HuMSC-EVs was largely abolished by treatment with LY (Fig. 3D). In addition, the expression of cleaved-caspase 3 was upregulated by cotreatment with LY (Fig. 3D, I). The MTT assay and LDH leakage results suggested that HuMSC-EVs decreased the apoptotic rate in H/R, while the blockade of PI3K/Akt signaling inhibited the antiapoptotic effects of HuMSC-EVs in vitro (Fig. 3J, K; # P < 0.05, ## P < 0.01 H/R vs control; *P < 0.05, **P < 0.01 H/R + HuMSC-EVs vs/H/R; △ P < 0.05, △△ P < 0.01 H/R + HuMSC-EVs + LY vs H/R + HuMSC-EVs). These results indicate that HuMSC-EVs protect against ER stress–related apoptosis at least in part through activation of the PI3K/Akt pathway in H9c2 cells.
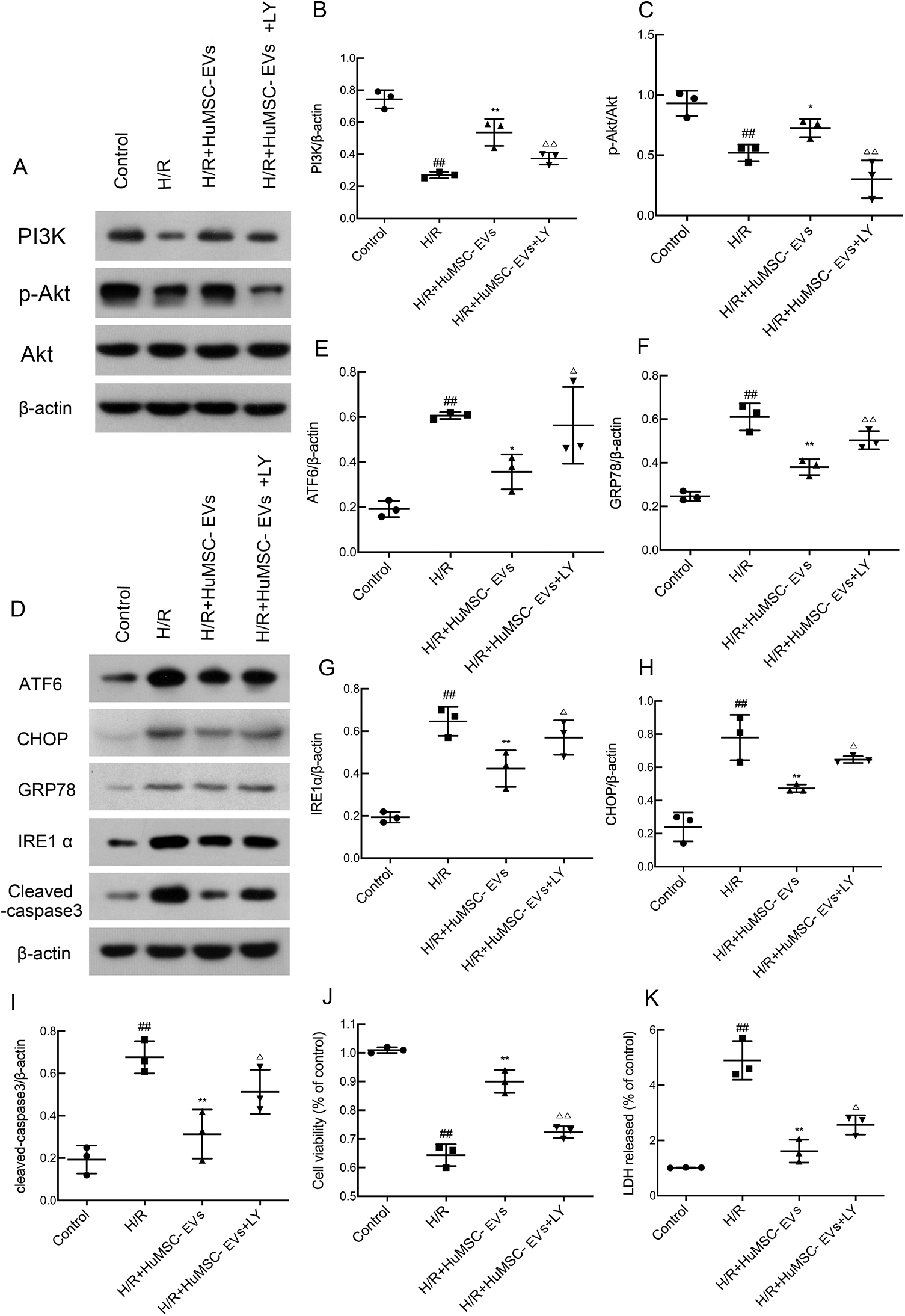
H/R induced ER stress through the activation of PI3K/Akt signaling in H9c2 cells. (A–C) Western blot and optical density analysis for PI3 K, Akt, and p-Akt. LY294002 (LY) is a broad-spectrum inhibitor of PI3K/AKT. (D–I) Analysis of protein expression and optical density for GRP78, CHOP, IRE1α, ATF6, and cleaved-caspase 3 in H9c2 cells. (J) H9c2 cell viability. (K) LDH release in H9c2 cells. Values are means ± SD. # P < 0.05, ## P < 0.01 H/R vs control; *P < 0.05, **P < 0.01 H/R + HuMSC-EVs vs H/R; △ P < 0.05, △△ P < 0.01 H/R + HuMSC-EVs + LY vs H/R + HuMSC-EVs (n = 3/group). ATF6: activating transcription factor 6; CHOP: C/EBP homologous protein; ER: endoplasmic reticulum; GRP78: glucose-regulated protein 78; H/R: hypoxia/reoxygenation; HuMSC-EVs: human umbilical cord mesenchymal stromal cells derived extracellular vesicles; IRE1α: inositol-requiring protein 1α; LDH: lactate dehydrogenase; SD: standard deviation.
Discussion
In this study, the effects of HuMSC-EVs on the survival of cardiac cell in response to H/R challenge and the underlying mechanisms were investigated. We found that HuMSC-EVs could protect H9c2 cells against ER stress–induced apoptosis under the stimulation of H/R. The antiapoptotic effect of HuMSC-EVs is partly mediated by the activation of PI3K/Akt signaling through the inhibition of hyperactive ER stress. Severe ER stress was induced by H/R treatment and resulted in increased expression of GRP78, CHOP, IRE1α, and ATF6, which promoted apoptosis in H9c2 cells. Supplementation with HuMSC-EVs attenuated the detrimental effects of H/R in H9c2 cells.
I/R injury is a major cause of morbidity, disability, and mortality worldwide 26 . In recent years, a number of studies have suggested that ER stress–mediated apoptosis plays an important role in the development of MI/R injury 27,28 . It has been shown that ischemic conditions interrupt ER homeostasis and activate the UPR 29 , which upregulates the expression of CHOP, a transcription factor 30 . The accumulation of CHOP protein induces the cleavage of caspase-3 31 and increases the Bax/Bcl-2 ratio 32 , resulting in cell apoptosis. In this study, the expression levels of GRP78, CHOP, IRE1α, and ATF6 were significantly upregulated in H/R-treated H9c2 cells, compared with normal control cells. CHOP overexpression was found to promote the activation of caspase-3 and to increase the Bax/Bcl-2 ratio, thus increasing the rate of apoptosis. These findings suggest that ER stress–mediated apoptosis plays an important role in the development of H/R injury.
EVs secreted by MSCs have been evaluated as a novel therapeutic modality for cardiovascular disease because they protect myocardium against I/R injury 33 . Accumulating evidence has shown that exosomal proteins or RNAs play an important role in modifying the behavior of recipient cells 34 . More importantly, EVs are easy to manipulate and may be transferred between individuals, or even across species 35 . In our study, treatment with HuMSC-EVs ameliorated ER stress, which protected H9c2 cells against excessive apoptosis, suggesting that HuMSC-EVs may effectively inhibit ER stress–mediated apoptosis.
The PI3K/Akt pathway plays a critical role in aspects of cellular physiology such as glucose homeostasis, lipid metabolism, protein synthesis, and cell proliferation and survival 36 . Impairing the PI3K/Akt pathway was found to aggravate ER stress–related apoptosis 37 . In the present study, we observed that H/R-induced ER stress inhibited activation of the PI3K/Akt pathway. The administration of HuMSC-EVs to H9c2 cells restored PI3K/Akt signaling, resulting in a decreased rate of apoptosis. However, the protective effects of HuMSC-EVs were partially eliminated when cells were cotreated with the PI3K/Akt inhibitor LY294002. The expression levels of GRP78, CHOP, IRE1α, ATF6, and cleaved-caspase-3 were all increased when PI3K/Akt signaling was blocked. It is reasonable to speculate that HuMSC-EVs ameliorated ER stress–induced apoptosis through activation of the PI3K/Akt signaling pathway. Although our results indicate that HuMSC-EVs treatment attenuated ER stress–reduced apoptosis in H9c2 cells through activation of the PI3K/Akt pathway, further investigation is required to fully reveal the mechanisms underlying the effects of HuMSC-EVs on ER stress.
In conclusion, we demonstrated that levels of ER stress and cell apoptosis are increased during H/R. HuMSC-EVs may ameliorate ER stress–induced H9c2 cell apoptosis after H/R stimulation in vitro.
Footnotes
Ethical Approval
The study protocol was approved by the Institutional Review Board of Shantou University Medical College (Shantou, China).
Statement of Human and Animal Rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board of Shantou University Medical College (Shantou, China).
Statement of Informed Consent
Informed consent was obtained from all individual participants included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by grants from the National Natural Science Foundation of China (Grant Nos 81671525 and 81070478), the Science and Technology Program of Shenzhen (Grant Nos JCYJ20150402092905162 and JCYJ20160429141742207), Shenzhen Public Service Platform of Molecular Medicine in Pediatric Hematology and Oncology, Medical Science and Technology Research Foundation of Guangdong Province (Grant No. A2017187), and The Medical and Health Technology Plan Project of Shantou (Grant No. 2018-121)..
