Abstract
Myocardial ischemia-reperfusion injury (MIRI), which occurs when the blood supply is restored in the ischemic myocardium, is a major medical concern for patients with acute myocardial infarction (AMI). Despite the use of extracellular vesicles (EVs) from mesenchymal stromal cells (MSCs), which can be used to treat MIRI, the application of EVs still has limited use in clinical practice. Melatonin (MT), however, not only exerts a significant protective effect in the treatment of cardiovascular diseases but also enhances biological functions of MSCs through pretreatment. Therefore, in the current study, we sought to determine whether MT improves the paracrine effect of MSCs through pretreatment. Our research provides evidence to support the therapeutic effect of MT-pretreated MSCs-derived extracellular vesicles (MT-EVs) in ameliorating hypoxia/reoxygenation (H/R) injury in human umbilical vein endothelial cells (HuVECs). We also performed a metabolomic analysis using ultra-high-performance liquid chromatography/Q Exactive HF-X Hybrid Quadrupole-Orbitrap Mass (UHPLC-QE-MS/MS) to explore metabolism profiling of H/R cell model with MT-EVs or EVs from MSCs (NC-EVs) treatment. We found 932 differential metabolites (DEMs) in the MT-EVs group compared with the NC-EVs group. Metabolic profiling analysis showed these metabolites were engaged in the ABC transporters, nucleotide metabolism, purine metabolic pathway, and glycerophospholipid metabolism. Furthermore, we observed increased levels of palmitoylcarnitine (fatty acid-derived mitochondrial substrate) and gabapentin in the MT-EVs group, which may play a therapeutic role in HuVECs during H/R. In conclusion, the results demonstrated that MT-EVs can protect endothelial cells from H/R injury by affecting the metabolic pathways.
This study addresses how MT-EVs attenuated H/R injury by affecting metabolic pathways in HuVECs. EVs from MT-preconditioned HuMSCs (MT-EVs) were applied to treat HuVECs during H/R, ultimately increasing cell viability, attenuating cell injury, and reducing apoptosis. UHPLC-QE-MS/MS was used to explore the metabolism profiling of HuVECs treated. MT-EVs affected ABC transporters, purine metabolism, and glycerophospholipid metabolism in HuVECs. In addition, palmitoylcarnitine and gabapentin were upregulated. These may have therapeutic effects on HuVECs during H/R.
Keywords
Introduction
Acute myocardial infarction (AMI) is a major cause of cardiovascular death worldwide 1 . Timely and successful use of thrombolytic therapy or primary percutaneous coronary intervention (PCI) may save the affected myocardium in many patients with AMI and can often improve prognosis 2 . However, the myocardial reperfusion process can trigger myocardial necrosis and cause myocardial damage. This damage can result in myocardial ischemia-reperfusion injury (MIRI)3,4. Interestingly, the main pathological mechanisms of MIRI involve oxidative stress and the inflammatory response, mitochondrial damage, energy metabolism disorder, calcium overload, apoptosis, and autophagy 5 . Furthermore, because MIRI currently lacks effective treatments, developing therapies to alleviate MIRI remains an urgent medical need.
Furthermore, mesenchymal stromal cells (MSCs) have self-renewal properties, multi-differentiation potential, and immunomodulatory capacities, all of which may be important in the future treatment of cardiovascular diseases. Mesenchymal stromal cells, for example, can attenuate MIRI and reduce cardiomyocyte apoptosis in adult mice subjected to MIRI 6 . It has also been reported that paracrine action is a key pathway for the treatment of MSCs in tissue injury repair and regeneration, and extracellular vesicles (EVs) are important mediators of this process. EVs are membrane vesicles secreted by MSCs that carry proteins, lipids, RNAs, and other biologically active factors. These aid in the transfer of materials to target cells and can regulate biological functions7,8.
Recent research has shown that the biological function of MSCs and their paracrine effect can be enhanced by specific culture condition or interventions, such as hypoxic conditions, pro-inflammation, engineering modifications, cytokines, and drugs9,10.
Furthermore, the endogenous hormone mainly produced and released via the pineal gland, melatonin (N-acetyl-5-methoxytryptamine, MT), plays a crucial role in apoptosis, autophagy, proliferation, anti-inflammation, and anti-oxidation11–14. Recent studies have also demonstrated that MT significantly regulates and enhances the therapeutic effect of MSCs on clinical diseases such as liver fibrosis, diabetes, and osteoporosis15–17. Melatonin pretreated MSCs-derived EVs can reduce the fibrotic and promote regenerative effects of human limbal mesenchymal stem cells (hLMSCs), promote functional recovery after spinal cord injury in mice, and alleviate renal ischemia-reperfusion injury18–20. MT stimulated human mesenchymal stem cells (HuMSCs) to produce exosomes enriched with regeneration-related miRNAs and upregulated various functions of exosomes such as anti-inflammation, scavenging of ROS and anti-apoptosis 21 . Therefore, MT may be a novel strategy to enhance MSC-EVs in disease treatment.
Interestingly, however, there are no reports on the efficacy of EVs derived from melatonin-preconditioned HuMSCs on MIRI. Therefore, in the current study, we aimed to investigate whether MT-EVs could protect endothelial cells from H/R injury and sought to explore the molecular mechanisms of the bioactive metabolites we identified.
Materials and methods
HuMSC culture and EVs collection
We obtained ethical approval from the Institutional Review Board of Shantou University Medical College (Shantou, China). We performed culturing and gathering methods on the HuMSCs and EVs based on our previous research 22 . HuVECs were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). HuVECs were placed in an incubator at 37°C and charged with 5% CO2. The HuMSCs were cultivated in Dulbecco’s Modified Eagle Medium (DMEM)/F12 medium supplemented with exosome-depleted 10% fetal bovine serum (FBS), 100 U/ml penicillin, and 100 U/ml streptomycin.
EVs were isolated from third-generation HuMSCs using Exosome Isolation Reagent (GENSEED, Guangzhou, China) based on the manufacturer’s instructions. The cell supernatants were centrifuged at 2000 × g (10 min) to remove to remove cells and debris. After this step, we started the exosome extraction: (1) Transfer the supernatant to a new centrifuge tube and add 1/3 times the volume of Reagent A to the supernatant. (2) Mix by inverting or pipetting and leave for 15 min at 2°C to 8°C. (3) At room temperature, centrifuge at 13,000 × g for 10 min and aspirate the supernatant; centrifuge at 13,000 × g for 30 s and aspirate the residual supernatant. (4) Add Reagent B in the same volume as Reagent A (step 1), mix well by pipetting, centrifuge at 13,000 × g for 10 min, and discard the supernatant. (5) The precipitate is EVs and stored at -80°C.
EVs identification
EVs morphology was observed by a transmission electron microscopy (TEM, Tecnai G2 12, FEI). We used a Nanoparticle Tracking Analyzer (NTA, ZetaView PMX 120, Particle Metrix) for our Nanoparticle tracking analysis. We used Western blots to characterize EV-specific surface markers, including CD9, CD63, Alix, and Calnexin.
Internalization of EVs by HuVECs
To assess the uptake of EVs by HuVECs in vitro, EVs were labeled with PKH26 (Sigma, USA) following the manufacturer’s instructions. The steps were (1) Add Diluent C to 500 μL of EVs concentrate and mix by gently blowing with a pipette gun. (2) Add 2-μL PKH26 Dye to 500 μL Diluent C and mix well. (3) Quickly add 500 μL of EVs Solution (step 1) to 500 μL of Dye Solution (step 2) and immediately mix with a pipette gun. The final concentration of PKH26 was 2 × 10-6 M. (4) Incubate for 1 to 5 min and mix periodically. (5) Terminate the reaction by adding an equal volume of EVs-free serum (1 mL) and incubate for 1 min. (6) Re-extraction of EVs using Exosome Isolation Reagent, wash with PBS, and resuspend EVs in the buffer for use. The EVs were placed into culture medium, and cultivated with HuVECs for 12 h. The nuclei were stained with 4′,6-diamidino-2-phenylindole (Ca#C1005, Beyotime Biotechnology, Shanghai, China). EVs internalized by HuVECs were recorded and photographed using an Olympus BX51 confocal microscope (Olympus, Tokyo, Japan).
Establishment of H/R model in HuVECs and experimental design
The H/R model was developed in HuVECs 23 . Specifically, the culture medium was altered to a hypoxic medium, which was deficient in glucose and FBS, and the culture dish was inserted into a sterile anoxic box containing 95% N2 and 5% CO2 until the oxygen concentration in the box decreased to less than 1%. The box was sealed, and all HuVECs were cultivated in an incubator at 37°C for 8 h. After 8 h, the cells were incubated in normal medium with 4.5 g/ml glucose for 4 h at 37°C, followed by reoxygenation.
All groups were identified as follows: The normal group consisted of HuVECs incubated in normal circumstances; the H/R group; the NC-EVs group (H/R model treated with the HuMSC-derived EVs); the MT-EVs group (H/R model treated with MT-EVs, and especially HuMSCs were pretreated with 5 μM MT for 48 h before EVs were isolated). The EVs (8 mg/ml) were added to the culture before the ischemia/reperfusion. in the NC-EVs and MT-EVs groups.
Cell viability
HuVECs viability was detected by the enhanced cell counting kit-8 assay (C0041, Beyotime). Optical density (OD) values were measured at 450 nm with a microplate reader.
Lactate dehydrogenase activity assay
We determined the level of cell impairment caused by H/R injury by identifying lactate dehydrogenase (LDH) activity. This was examined by assessing the culture medium through the LDH activity assay kit (Cloud-Clone Corp, Katty, TX, USA) based on the manufacturer’s instructions.
Western blot analysis
To perform the Western blot analysis, we isolated total protein from the HuVECs. The concentration of total protein was tested using the Qubit Protein Assay Kit (Q33212, Life Technologies). Then the total protein samples were electrophoresed on Express PAGE Gels (M42010c, Genscript) and transferred onto polyvinylidene difluoride membranes. The Membranes were hatched with primary antibody at 4°C overnight, then hatched with HRP-conjugated anti-mouse IgG (1:3000, 7076S, Cell Signaling) and kept at 4°C for 0.5–1 h. Specific protein bands were observed with an ECL Plus chemiluminescence kit (EMD Millipore) and quantitatively analyzed with ImageJ. We used glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as our control. We used the following primary antibodies: CD9 (1:1,000, 13174S, Cell Signaling); CD63 (1:1,000, ab134045, Abcam); Alix (1:1,000, sc-53540, SantaCruz); Calnexin (2433, Cell Signaling); Bax (1:3000, Ab32503, Abcam, Cambridge, MA, USA); and Bcl2 (1:3000, Ab196495, Abcam); cleaved-caspase 3 (1:5,000, Cst9664, CST).
Metabolomics analysis
The cell samples preparation process: take logarithmic growth phase cells, remove the culture medium, wash twice with the appropriate amount of pre-cooled PBS, add a small amount of PBS, scrape off the cells with a cell spatula, place them in a 1.5 ml centrifuge tube, centrifuge them for 1 min at 1000 × g, 4°C, and quench them with liquid nitrogen. After these step, we started the metabolites extraction: Add 100 μL of extraction solution (methanol: acetonitrile:water = 2:2:1 (V/V), including isotope-labeled internal standard mixture) to the cell samples; mix well and then put into a liquid nitrogen tank to freeze for 1 min, take out and thaw, mix well for 30 s. Repeat this step 3 times; ultrasonic for 10 min (ice-water bath) and then -40°C resting for 1 h; put the samples into a 4°C centrifuge (12,000 rpm, 13,800 × g, radius 8.6 cm) for 15 min and take the supernatant. Finally, the samples were centrifuged at 4°C, 12,000 rpm (centrifugal force 13,800 × g, radius 8.6 cm) for 15 min, and then the supernatant was taken.
We extracted six HuVECs metabolites samples (NC-EVs groups = 3; MT-EVs groups = 3) and transferred them into the ultra-high-performance liquid chromatography/Q Exactive HF-X Hybrid Quadrupole-Orbitrap Mass (UHPLC-QE-MS/MS) (Thermo Fisher Scientific, USA). The LC/MS raw data were processed by Soft Independent Modeling of Class Analogy (SIMCA) (V16.0.2, Sartorius Stedim Data Analytics AB, Umea, Sweden). We used principle component analysis (PCA) to examine the distribution of the samples and the reliability of the program for a complete analysis. We used the univariate (t-test) and orthogonal partial least squares-discriminant analysis (OPLS-DA), to explore the metabolome differences between groups. The variable importance projection (VIP) derived from the OPLS-DA model was applied to select different metabolites between groups according to the results of OPLS-DA. A metabolite was chosen as a DEM when it satisfied the conditions of P ≤ 0.05 and VIP ≥ 1. Differentially expressed metabolites were analyzed by MetaboAnalyst for powerful pathway enrichment. Then, the metabolites recognized in metabolomics were matched to Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways for biological elucidation of systemic functions at a higher level. We used commercial databases, including KEGG (http://www.genome.jp/kegg/) and MetaboAnalyst (http://www.metaboanalyst.ca/) to perform the pathway enrichment analysis.
Statistical analysis
We analyzed all experimental data using SPSS 27.0 software. GraphPad Prism 8.0 was used to prepare all diagrams. An unpaired t-test was applied to compare variation between groups, and multiple groups of samples were contrasted by performing one-way analysis of variance (ANOVA). To confirm the reliability of the experimental data, we replicated all experiments at least three times. We expressed all data as mean values ± standard deviation (SD); and P < 0.05 indicated significant differences.
Results
Identification and internalization of EVs
Transmission electron microscopy demonstrated that both NC-EVs and MT-EVs presented a typical round-shaped morphology (Fig. 1a). Our Western blot analysis demonstrated that EVs expressed high levels of the known EVs molecular markers, including CD63, CD9, and Alix; EVs did not express negative marker (Fig. 1b). In addition, our NTA examined the distribution of particle size and concentration of EVs (Fig. 1c). The particle diameter of NC-EVs and MT-EVs were in the range of 87.4–209.8 nm and 86.7–204.5 nm, respectively, which is consistent with the range of EVs particle sizes; and the concentration was 5.0 × 107 particles/ml and 4.0 × 107 particles/ml, respectively. Collectively, these findings suggested the successful isolation of EVs.
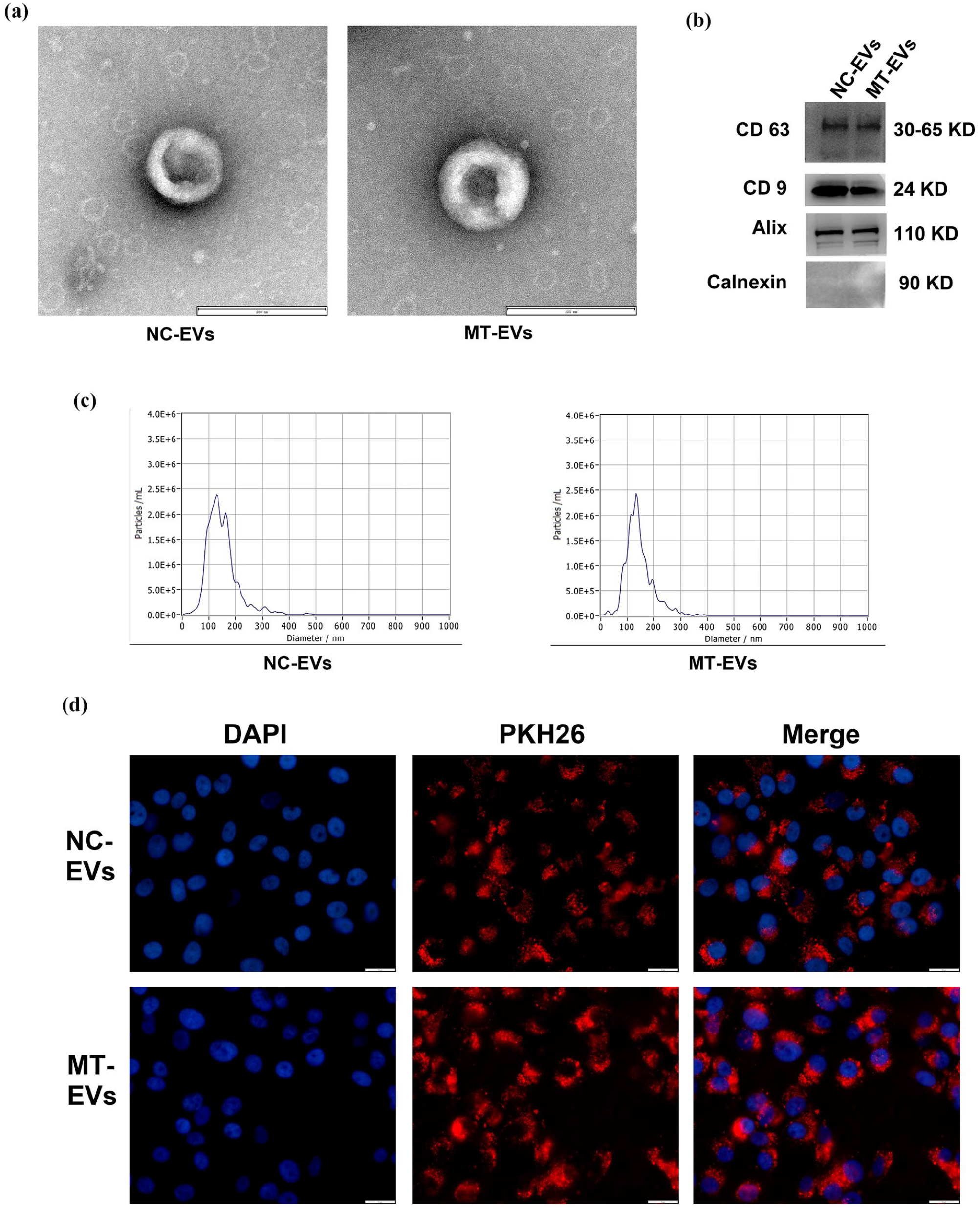
Identification and internalization of EVs. (a) Morphological characteristics of NC-EVs and MT-EVs (scale bar = 200 nm). (b) Western blot analysis of NC-EVs and MT-EVs surface markers (CD9, CD63, Alix, and Calnexin). (c) Nanoparticle tracking analysis of NC-EVs and MT-EVs. (d) Internalization of NC-EVs and MT-EVs by HuVECs as detected by fluorescence microscopy (scale bar = 10 um).
We also examined the internalization of NC-EVs and MT-EVs by HuVECs and marked NC-EVs and MT-EVs with PKH-26. After marking, the NC-EVs and MT-EVs pellet displayed an intense red color. PKH-26-tagged NC-EVs and MT-EVs were then incubated with HuVECs. We observed red fluorescence in majority of HuVECs cytoplasm, indicating that the cultured HuVECs had absorbed a large degree of NC-EVs and MT-EVs (Fig. 1d).
Effect of MT-EVs on the viability of H/R-induced HuVECs
We evaluated the viability of HuVECs via the CCK8 assay. H/R injury significantly inhibited cell viability, and NC-EVs and MT-EVs improved cell viability of HuVECs exposed to H/R (Fig. 2a; The average ± SD: Control 1.997 ±0.007, H/R 1.071 ± 0.010, NC-EVs 1.191 ± 0.027, MT-EVs 1.310 ± 0.010; ****P < 0.0001 H/R vs. Control, ##P < 0.01 NC-EVs vs. H/R, ####P < 0.0001 MT-EVs vs. H/R, &&P< 0.01 MT-EVs vs. NC-EVs). Moreover, the cell viability of HuVECs treated by MT-EVs was significantly higher than that treated by NC-EVs (P < 0.01). LDH leakage increased in the H/R group compared to the normal group but was significantly decreased by NC-EVs and MT-EVs treatment. Importantly, LDH leakage in the MT-EVs group was lower than that of the NC-EVs group (Fig. 2b; The average ± SD: Control 0.070 ±0.093, H/R 11.330 ±0.170, NC-EVs 4.590 ±0.108, MT-EVs 2.706 ±0.045; ****P < 0.0001 H/R vs. Control, ####P < 0.0001 NC-EVs vs. H/R, ####P < 0.0001 MT-EVs vs. H/R, &&&&P< 0.0001 MT-EVs vs. NC-EVs). These results demonstrate that NC-EVs and MT-EVs could promote cell survival and reduce the release of cell contents in HuVECs subjected to H/R injury, and MT-EVs had a stronger therapeutic effect.

Effects of NC-EVs and MT-EVs on cell survival in response to H/R insult. (a) NC-EVs and MT-EVs increase cell viability following H/R. (b) NC-EVs and MT-EVs decrease LDH release following H/R. (c) Analysis of protein expression and optical density for cleaved-caspase 3 and Bax/bcl2 in HuVECs. Error bars: Values are mean ± SD (n = 3 per group). **P < 0.01, ****P < 0.0001 versus Normal group; #P < 0.05, ##P < 0.01, ####P < 0.0001 versus H/R group. &&P< 0.01, &&&&P< 0.0001 versus NC-EVs group.
EVs reduced H/R-induced HuVECs apoptosis
To explore the impact of MT-EVs on apoptosis in HuVECs, we examined the Bax, Bcl-2, and cleaved-caspase 3 expression levels (Fig. 2c). In the H/R group, the expression level of Bax and cleaved-caspase 3 were upregulated, whereas Bcl-2 expression was downregulated. All EV treatment groups decreased the expression levels of these three proteins to baseline levels in HuVECs. In addition, Bax/Bcl-2, and cleaved-caspase 3 were lower in the MT-EVs group compared with the NC-EVs group (P < 0.01). Collectively, these findings show that NC-EVs and MT-EVs reduced apoptosis in HuVECs. Importantly, the effect of MT-EVs was superior to NC-EVs.
Multivariate statistical analysis
Multivariate analyses (PCA, OPLS-DA) that we used to evaluate the overall metabolite variation in HuVECs after NC-EVs or MT-EVs treatment. In PCA, the metabolites were clearly separated between the two groups (Fig. 3a, b). PC1 and PC2 accounted for 35.1% and 26.4% of the variance, respectively. Supervised OPLS-DA analysis demonstrated significant differences between the NC-EVs and MT-EVs groups, with all six samples falling within the 95% confidence interval, which indicated significant changes in the HuVECs metabolic profile between the two groups and clear clustering of the biological replicates within the same group (Fig. 3b). A permutation histogram test showed the performance parameters were high, and the OPLS-DA model exhibited both suitable model fit and prediction (R2Y = 0.999, Q2 = 0.759, P < 0.05; Fig. 3c). The OPLS-DA models were reliable and suitable for determining the metabolic differences.

Multivariate statistical analysis. (a) Score scatter plot for PCA model TOTAL with QC. (b) PCA of the metabolites identified in the NC-EVs and MT-EVs groups. (c) OPLS-DA score plot of NC-EVs group versus MT-EVs group. (d) Permutation histogram test of the OPLS-DA model.
Metabolome profiling analysis
After databases searching and configuration by accurate molecular mass, the different expressed metabolites were selected to compare the MT- and NC-group. A total number of 7,729 metabolites were identified. These distinctive metabolites could be categorized into lipids and lipid-like molecules (31.93%), organic acids and derivatives (14.43%), organoheterocyclic compounds (13.31%), others (10.08%), and benzenoids (7.42%) (Fig. 4a). A total of 932 DEMs showed significant variations between two groups (P ≤ 0.05 and VIP ≥ 1). Among these, 166 metabolites were upregulated and 766 metabolites were downregulated in the MT-EVs group compared to the NC-EVs group (Fig. 4b). We also determined the top 10 upregulated and downregulated metabolites in the HuVECs (MT-EVs group vs. NC-EVs group) (Fig. 4c). Then, we determined clustering of various metabolites for the groups, and 151 metabolites expressed significant differences between the two groups (Fig. 4d).

Metabolome profiling analysis in HuVECs after MT-EVs treatment. (a) Pie chart with a summary of chemical components in the MT-EVs group. (b) Volcano plot. (c) Matchstick analysis for the top 10 upregulated and downregulated metabolites in the HuVECs (MT-EVs group vs. NC-EVs group). (d) Heatmap of hierarchical clustering analysis for NC-EVs group versus MT-EVs group.
Moreover, DEMs were analyzed for possible metabolic processes using KEGG analysis (Fig. 5a, b). 15 pathways were annotated, among which seven pathways, namely, ABC transporters, nucleotide metabolism, purine metabolism, glycerophospholipid metabolism, D-Amino acid metabolism, Aminoacyl-tRNA biosynthesis, and sphingolipid metabolism were associated with myocardial ischemia.

KEGG analysis in HuVECs after MT-EVs treatment. (a) KEGG pathway enrichment analysis. (b) Pathway analysis diagram.
Metabolites analysis
Kyoto Encyclopedia of Genes and Genomes pathway analysis suggested that MT-EVs treatment significantly influenced the glycerophospholipid metabolism and purine metabolism pathways; therefore, we analyzed changes in the abundance of metabolites mapping to these two metabolic pathways. We then compared the intensity values of the five metabolites from purine metabolism and the five metabolites from glycerophospholipid metabolism among groups (Fig. 6). The results indicated that inosine, adenine, adenosine, and guanosine were significantly downregulated and deoxyguanosine was upregulated in the MT-EVs group compared to the NC-EVs group. This suggested that the process of purine metabolism was inhibited (Fig. 6a). Phosphorylcholine, PE (four species), PC (nine species), LysoPE/LysoPE (five species), and LysoPC/LPC (13 species) were differentially downregulated by MT-EVs administration, suggesting the process of glycerophospholipid metabolism was inhibited. For compounds in which multiple metabolites were identified (e.g. PE, PC, LysoPE, and LysoPC), we concentrated our analysis on the molecules with the highest VIP values. We then compared the intensity values of fiver metabolites from glycerophospholipid metabolism among groups (Fig. 6b). Interestingly, palmitoylcarnitine and gabapentin showed high VIP values and substantial increments in the MT-EVs group, suggesting those metabolites had significant influences on the difference between the MT-EVs and NC-EVs groups (Fig. 6c).

Representative differential metabolites in HuVECs after MT-EVs treatment. (a) Purine metabolism pathway. (b) DEMs in the glycerophospholipid metabolism pathway. (c) Palmitoylcarnitine and gabapentin. Data are expressed as mean ± SD (n = 3 per group). *P < 0.05, **P < 0.01, ***P < 0.001. PE: Phosphatidylethanolamine; PC: Phosphatidylcholine; LPE: Lysophosphatidylethanolamine; LPC: Lysophosphatidylcholine.
Discussion
With the recent advancements in stem cell regenerative medicine, there is new hope in treating cardiovascular diseases. For example, MSC-derived EVs can exert cardioprotective effects in AMI 24 . However, concern remains in the precise way that cell-free therapy can enhance the efficacy of treatment with MSC-derived EVs 25 . In this study, we found that NC-EVs and MT-EVs protect HuVECs from H/R injury. NC-EVs or MT-EVs treatment reduced cleaved-caspase 3 and the Bax/Bcl-2 ratio, moderating cell apoptosis. LDH leakage is employed extensively as a biomarker of cytosolic injury. EVs’ treatment not only improved cell survival but it also alleviated LDH leakage. Specifically, MT-EVs were more effective than NC-EVs. To our knowledge, this is the first study to determine the cardioprotective effects of EVs derived from MSCs pretreated with MT.
Further, we performed untargeted metabolomics analysis to acquire a comprehensive metabolomic profile to reveal the potential pathways involving in the MT-EVs treatment. We analyzed metabolite statistics from NC-EVs and MT-EVs and found that 766 metabolites decreased and 166 metabolites increased in the MT-EVs group. They consisted mainly of lipid metabolites and organic compounds. Based on pathway analysis, compared to NC-EVs group, most DEMs were attributed to metabolic pathways including ABC transporters, nucleotide metabolism, purine metabolism, glycerophospholipid metabolism, D-Amino acid metabolism, aminoacyl-tRNA biosynthesis, and sphingolipid metabolism. D-amino acid oxidase was closely related to D-Amino acid metabolism and its disorder was found to oxidative stress 26 . Aminoacyl-tRNA synthetases exhibited important effects on aspects of endothelial cell migration, proliferation, and survival 27 . Sphingosine kinase catalyzed the phosphorylation of sphingosine to generate S1P and had cardioprotective properties28,29. Following, we focused on analyzing ABC transporters, glycerophospholipid metabolism, purine metabolism, palmitoylcarnitine, and gabapentin.
Our data demonstrated that ABC transporters enriched DEMs most (36.84%). ABC transporters were widely found in human cells and tissues, they belonged to five different families (A, B, C, D, and G) and were involved in regulating the transport of sugars, nucleosides, amino acids, fatty acids, lipids, and more 30 . It was reported that ABC transporters were associated with cardiovascular disease (CVD) and IRI. ABC transporters were found to be related to inflammatory regulation 31 . ABCA1 has been shown to act as an anti-inflammatory protein, mostly via the depletion of cellular cholesterol and lipids, which moderated the process of atherosclerosis 32 . Moreover, increased ABCA1 expression following ischemic preconditioning protected against cerebral IRI via suppression of a mitochondria-dependent apoptosis pathway 33 . ABCG2, a determinant of several tissue-specific stem/progenitor cell types that were involved in the vascular formation, maintenance, and regeneration, was strongly expressed by the primary murine EC, and it also expected promoting angiogenesis and antioxidant effect34,35. ABC-me (ATP binding cassette mitochondrial erythroid, ABCB10 or mABC2) was an important player in the protection from oxidative stress in cardiac recovery after ischemia-reperfusion through adjusting mitochondrial ROS generation 36 . Hence, ABC transporters may be involved in the protective effects of MT-EVs.
Previous studies have confirmed that hydrogen-rich water-modulated glycerophospholipid metabolism could alleviate MIRI 37 . In our study, phosphorylcholine, PC, LysoPC, PE, and LysoPE engaged in glycerophospholipid metabolism and reduced with MT-EVs treatment (Fig. 7); and the above metabolites were connected with phospholipase A2 (PLA2). PC and PE were the main components of cell membranes and were associated with membrane properties 38 . PLA2 was a lipolytic enzyme found in nearly all cell membranes and exerted a crucial function in sustaining normal membrane phospholipid metabolism39,40. Any element that altered the composition of membrane phospholipids compromised the stability of membrane structure, which in turn affected the function of membrane proteins 41 . For instance, PLA2 hydrolyzed PC to LysoPC. LysoPC induced Ca2+ overload in cardiomyocytes, which can lead to growth cone collapse and cell necrosis 42 . Moreover, several LysoPC were connected with superoxide dismutase dysfunction, phospholipase activation, and oxidative stress43–45. LysoPC could be considered toxic byproduct of the reactive oxygen produced during H/R43,46. In our study, MT-EVs treatment significantly decreased LysoPC, which may be associated with PLA2 inactivation. An investigation of lipidomic profiling in I/R-induced rat models found that several lipids including PC, PE, LPC, and LPE increased during I/R, while levels decreased after astragaloside IV treatment 47 . It is consistent with our findings. More studies are needed to clarify the effects of MT-EVs on glycerophospholipid metabolism in HuVECs during H/R.

Schematic of altered glycerophospholipid metabolism indicating the mechanism of MT-EVs treatment for ischemic-reperfusion injury. Blue indicates metabolites were significantly downregulated by MT-EVs treatment, and gray indicates no significant change before and after MT-EVs treatment.
Moreover, purine metabolites including inosine, adenine, adenosine, and guanosine also decreased in the MT-EVs group in our study. As previously described, energy metabolism disorder is one of the pathophysiological mechanisms leading to MIRI48,49. Purine metabolite accumulation provides a useful index of ATP depletion in ischemic diseases 50 . Previous studies indicated that ischemic myocardium accumulates purine metabolites 51 . However, the level of cellular production and release of purine metabolites declined during ischemia in the preconditioned myocardium. This may suggest that there could be a decrease in energy imbalance, as well as the cellular production and release of purine metabolites52,53. Research on the metabolic profiling (UPLC/Q-TOF-MS) in H/R cell models found that propofol treatment changed purine metabolism in HuVECs, suppressing 3′-GMP, guanosine, and hypoxanthine 54 . In our research, these data indicate that MT-EVs treatment significantly suppressed purine metabolism in HuVECs subjected to H/R, which could be associated with reduced energy demand and degradation of nucleotides.
Notably, palmitoylcarnitine is also known as an important intermediate in mitochondrial fatty acid oxidation 55 . Early observations revealed micromolar concentrations of palmitoylcarnitine enhance postischemic cardiac function through plasma membrane stabilization. This may act to protect the myocardium from deterioration by reperfusion after ischemia 56 . Furthermore, Tominaga et al. 57 found that application of 1 µM palmitoylcarnitine given to permeabilized ventricular rat myocytes resulted in hyperpolarization of the mitochondrial membrane and the induction of ROS production. Current research suggests that ROS in the cardiovascular system can be protective and detrimental58,59. Large amounts of ROS during reperfusion can be related to damage, while a controlled amount of ROS during pretreatment stimulation or at the start of reperfusion in a posttreatment regimen activated protective signaling and attenuated ischemia-reperfusion injury58,60–62. As an active cardioprotective metabolite of Intralipid, palmitoylcarnitine inhibited complex IV, stimulated ROS production, activated the RISK pathway, and exerts a highly potent cardioprotective effect during myocardial ischemia/reperfusion 63 . In the current study, we found that MT-EVs treatment increased palmitoylcarnitine in HuVECs subjected to H/R. These findings suggest that MT-EVs may exert cardioprotective effects through increasing levels of palmitoylcarnitine in HuVECs (Supplementary Material).
Gabapentin, 1-(aminomethyl)cyclohexane acetic acid, is an analog of the neurotransmitter gamma-aminobutyric acid (GABA), and extensively used clinically as an anticonvulsant drug64–66. Recent studies have found that gabapentin has therapeutic effects on ischemic diseases67–70. Li et al. 68 found that gabapentin inhibited M1 macrophage polarization and attenuated cardiac remodeling after myocardial infarction through the peroxisome proliferator-activated receptor-γ pathway. In addition, Yan et al. found that during cerebral artery occlusion in a rat model, GBP treatment inhibited IR-induced neuronal death, increased antioxidant levels, and reduced neuronal autophagy in the infarct penumbra by up-regulation of the PI3K/Akt/mTOR signaling pathway. At the same time, its effects could be reversed by LY294002 (a PIK3 inhibitor) 69 . Wu et al. 70 found that in vivo experiments, gabapentin had a protective effect against myocardial ischemia-reperfusion by the activation of the PI3K/Akt signaling pathway and the up-regulation of GABAARδ in the spinal cord. Here, our study provides evidence that gabapentin was increased in ME-EVs. While the role of gabapentin in HuVECs H/R model remains unclear, further research is necessary to verify gabapentin’s effects in HuVECs subjected to H/R.
Conclusion
In summary, we provide evidence that EVs derived from MT-preconditioned MSCs could protect HuVECs from H/R injury by affecting intracellular metabolism, including ABC transporters, glycerophospholipid, and purine metabolism. Furthermore, our findings suggest that palmitoylcarnitine and gabapentin may be important molecules mediating this therapeutic effect. This may suggest that further research is needed to better understand the role that palmitoylcarnitine and gabapentin can have in the treatment of MT-EVs, as well as in associated pathways.
Supplemental Material
sj-tif-1-cll-10.1177_09636897251347389 – Supplemental material for Extracellular vesicles from melatonin-preconditioned mesenchymal stromal cells protect human umbilical vein endothelial cells against hypoxia/reoxygenation detected by UHPLC-QE-MS/MS untargeted metabolic profiling
Supplemental material, sj-tif-1-cll-10.1177_09636897251347389 for Extracellular vesicles from melatonin-preconditioned mesenchymal stromal cells protect human umbilical vein endothelial cells against hypoxia/reoxygenation detected by UHPLC-QE-MS/MS untargeted metabolic profiling by Liwei Cai, Qian Ma, Danli Zhuo, Qingcheng Chen, Yiming Zhuang, Shengkun Yuan, Xin Lu, Changyi Zhang and Yu Zhou in Cell Transplantation
Footnotes
Ethical considerations
The study was approved by the Institutional Review Board of Shantou University Medical College (Shantou, China).
Statement of human and animal rights
This article does not contain any studies with human or animal subjects.
Statement of informed consent
Informed consent was obtained from all subjects involved in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by The Medical and Health Technology Plan Project of Shantou, grant number 240425206497638.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The original contributions presented in the study are included in the article/supplementary materials, further inquiries can be directed to the corresponding authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
