Abstract
We were interested in evaluating the ability of the mesenchymal stromal cell (MSC) population, human umbilical cord perivascular cells (HUCPVCs), to undergo cardiomyocyte reprogramming in an established coculture system with rat embryonic cardiomyocytes. Results were compared with human bone marrow-derived (BM) MSCs. The transcription factors GATA4 and Mef 2c were expressed in HUCPVCs but not BM-MSCs at baseline and, at 7 days, increased 7.6- and 3.5-fold, respectively, compared with BM-MSCs. Although cardiac-specific gene expression increased in both cell types in coculture, upregulation was more significant in HUCPVCs, consistent with Mef 2c-GATA4 synergism. Using a lentivector with eGFP transcribed from the α-myosin heavy chain (α-MHC) promoter, we found that cardiac gene expression was greater in HUCPVCs than BM-MSCs after 14 days coculture (52 ± 17% vs. 29 ± 6%, respectively). A higher frequency of HUCPVCs expressed α-MHC protein compared with BM-MSCs (11.6 ± 0.9% vs. 5.3 ± 0.3%); however, both cell types retained MSC-associated determinants. We also assessed the ability of the MSC types to mediate cardiac regeneration in a NOD/SCID γ mouse model of acute myocardial infarction (AMI). Fourteen days after AMI, cardiac function was significantly better in cell-treated mice compared with control animals and HUCPVCs exhibited greater improvement. Although human cells persisted in the infarct area, the frequency of α-MHC expression was low. Our results indicate that HUCPVCs exhibit a greater degree of cardiomyocyte reprogramming but that differentiation for both cell types is partial. We conclude that HUCPVCs may be preferable to BM-MSCs in the cell therapy of AMI.
Keywords
Introduction
Cardiovascular disease is a leading cause of death globally (36). Cell-based therapies with bone marrow-derived mesenchymal stromal cells (BM-MSCs) may have therapeutic potential for cardiac regeneration. BM-MSCs tested in rodent and porcine models of acute myocardial infarction (AMI) show reduced infarct size and improved remodeling without causing a proinflammatory reaction (1,2,9,21). In clinical studies, intracoronary infusion of autologous BM-MSCs improves left ventricular (LV) function and myocardial perfusion after AMI (16,34). Mechanisms by which MSCs exert beneficial effects remain uncertain. Current opinion favors invoking paracrine effects to explain hemodynamic improvement (18,31). While transdifferentiation of donor cells into cardiomyocytes is no longer considered a likely possibility, it is unclear whether or not partial cardiomyocyte differentiation enhances the release of soluble growth factors and cytokines in mediating tissue regeneration.
Human umbilical cord perivascular cells (HUCPVCs) obtained from the region surrounding the blood vessels within the human umbilical cord are an attractive and readily available alternative source of MSCs for cell therapy (51). This cell type overcomes some of the limitations of BM-MSCs such as loss of multipotentiality and proliferative potential with increasing donor age (13,23,27). HUCPVCs have a higher clonogenic frequency than BM-MSCs and contain self-renewing stem cells with multilineage differentiation potential in vitro and in vivo (50). Differentiation capacity along the cardiac lineage, however, has not been studied. Recent reports have shown the cardiomyocyte reprogramming potential of stem cells derived from the human umbilical cord. It was found that umbilical cord blood-derived MSCs do not transdifferentiate into cardiomyocytes in vitro (38,49) and present a very limited regenerative capacity in a model of myocardial infarction (14). Some reports showed that stem cells derived from the whole umbilical cord tissue express α-cardiac actin (α-CA), cardiac troponin T (cTnT), and cardiac myosin after treatment with the demethylating agent 5-azacytidine (5-Aza), but quantitative data were lacking (45,55). Although 5-Aza activates the expression of different genes to induce phenotypic changes, its effects might not be specific. In the present work, we studied a human umbilical cord stromal cell population derived from the perivascular tissue (HUCPVCs) that contains a high frequency of progenitor cells which were either discarded or not specifically isolated in those previously described studies. Moreover, a study comparing BM-MSCs and HUCPVCs using a more physiological approach may establish whether the latter represent a feasible alternative for cardiac regeneration. We recently showed that bone marrow-derived murine MSCs can acquire cardiomyocyte markers but retain MSC properties when cocultured with rat embryonic cardiomyocytes (RECs) (48). Here, we used the same in vitro system to compare the ability of HUCPVCs and human BM-MSCs to undergo cardiomyocyte differentiation. We further compared the cells in vivo by intramyocardial injection after AMI in the nonobese diabetic/severe combined immunodeficient interleukin 2 receptor gamma chain (NOD/SCID gamma; NSG) mouse. We found that both cell types expressed α-myosin heavy chain (α-MHC), cTnT, α-CA, and atrial natriuretic protein (ANP) but retained MSCs characteristics after coculture with RECs. We observed that enhancement of cardiac gene expression as well as the extent of differentiation was significantly higher in HUCPVCs compared with BM-MSCs. We also showed that HUCPVCs resulted in superior improvement in ventricular function as assessed by echocardiography. Our data suggest that locally administered HUCPVCs may be particularly suited to cardiac regeneration after AMI.
Materials and Methods
Supporting information is available at the following link: http://dl.dropbox.com/u/58685630/Yannarelli_CT2012_Supporting_Information.pdf
Culture of HUCPVCs and BM-MSCs
Umbilical cords were donated by patients undergoing full-term cesarean section after written informed consent according to a protocol approved by the University of Toronto and Sunnybrook and Women's College Health Sciences Centre, Toronto. Both umbilical cord arteries were dissected including their associated perivascular tissue and were then looped and tied using silk sutures to prevent hematopoietic or endothelial contamination. These vessel loops were digested overnight at 37°C in a phosphate-buffered saline (PBS; Gibco, Gaithersburg, MD, USA) solution containing 1 mg/ml type 1 collagenase (Worthington Biochemical Corporation, Lakewood, NJ, USA). Cells were then removed from the supernatant, collected, washed, counted, and plated at a density of 4,000 cells/cm2. HUCPVCs were grown in minimum essential medium a modification (α-MEM) supplemented with 15% fetal bovine serum (FBS; both Gibco) at 37°C in a humidified incubator containing 5% CO2 (51). This extraction procedure results in a cell population (HUCPVCs) with a high frequency of colony-forming unit-fibroblast (CFU-F)-deriving cells, which were either discarded or not specifically isolated in other studies using whole umbilical cord tissue (51).
Human BM-MSCs were obtained from healthy male and female young adult volunteers (in their 20s and 30s) after informed consent according to a protocol approved by the University Health Network Research Ethics Board. The mononuclear cell fraction of the BM was isolated by Ficoll density gradient centrifugation (1.077; Sigma-Aldrich, St. Louis, MO, USA) and cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% FBS and 1% antibiotic-antimycotic solution (all from Gibco). Nonadherent cells were removed by replacing the media 24 h after seeding. The medium was then replaced every 2–3 days, and the BM-MSCs were subcultured at 70–90% confluence until passage 4 cells were obtained. Cell surface immunophenotype and multilineage differentiation potential were analyzed for both HUCPVCs and BM-MSCs at passage 4 to confirm that the cells conform with minimal criteria defining MSCs (11) (Supporting Information Fig. S1).
Lentiviral Vector Production and Transduction of HUCPVCs and BM-MSCs
A bidirectional HIV-1-based recombinant lentiviral vector (LV) was built from the plasmid pCCL.sin.cPPT. polyA.CTE.eGFP.minhCMV.hPGK.deltaNGFR.WPRE (3). Briefly, pCCL.EF1α.DsRed.α-MHC.eGFP (LV/α-MHC) was constructed in house using the ubiquitous promoter elongation factor 1α (EF1α) driving Discosoma sp. red fluorescent protein (DsRed) and the mouse α-myosin heavy chain (α-MHC) promoter driving enhanced green fluorescent protein (eGFP) expression. The human cytomegalovirus (CMV) immediate-early enhancer was used to enhance activity of the α-MHC promoter without affecting promoter specificity (20). Vesicular stomatitis virus-G protein (VSV-G)-pseudotyped lentivirus (LV), including an eGFP marking vector (LV/eGFP; pHR′. cPPT.EF1α.eGFP), was generated by transient transfection of 293T cells (ATCC, Manassas, VA, USA) with the three-plasmid system (LV plasmid construct, packaging plasmid pCMVDR8.91 and the VSV-g envelope-coding plasmid pMD.G) as described previously (57). The concentrated LV supernatants were serially diluted and titrated on 293T cells. The human cells were transduced at passages 3–4 using a multiplicity of infection (MOI) of 10 with LV/eGFP or LV/α-MHC in the presence of 8 mg/ml of protamine sulfate (Sigma-Aldrich) for 16 h. Infected cells were analyzed 72 h after transduction by flow cytometry (LV/eGFP) or fluorescent microscopy (LV/α-MHC) (Supporting Information Figs. S2 and S3). Cells were used 14 days after transduction for the assays.
Coculture of Human MSCs with Rat Embryonic Cardiomyocytes
Cardiomyocytes were isolated from rat embryos (day 20; Ontario Cancer Institute Animal Facility) and cultured as previously described with minor modifications (48). Briefly, ventricles were minced and digested using trypsin (0.08% w/v) and collagenase type 2 (0.035% w/v; both Worthington Biochemical Corporation). Preplating for 60 min was performed to eliminate contaminating fibroblasts. Nonadherent cells were collected and seeded on gelatin-coated plates (Sigma-Aldrich) in DMEM:F12 (1:1; Gibco) supplemented with 5% FBS and 10% horse serum (Gibco). d-Arabinofuranosylcytosine (Ara-C, 1 μg/ml; Sigma-Aldrich) was added to inhibit the proliferation of remaining fibroblasts. After 24–48 h in culture, cardiomyocytes were washed with PBS to remove Ara-C and fresh medium was added. Fourth passage BM-MSCs or HUCPVCs were plated onto the primary rat embryonic cardiomyocyte (REC) cultures and cocultured for up to 14 days. After 3 days, Ara-C was added (1 μg/ml) to inhibit nonmyocyte proliferation (4). Ara-C has no effect on DNA methylation and therefore does not improve cell differentiation unlike the other nucleoside analog, 5-azacytidine (25).
Conditioned Media and Transwell Experiments
To test whether direct contact with cultured cardiomyocytes is important in cardiomyocyte reprogramming, HUCPVCs were cultured alone in conditioned media obtained from HUCPVC and REC cocultures. Conditioned media were diluted 1:1 with fresh α-MEM supplemented with 15% FBS and replaced every 2–3 days for up to 14 days. In addition, cocultures were set up in a transwell system (0.4-μm transwell polyester pore membrane; Corning, Corning, NY, USA) in which the two cell types were physically separated and allowed to interact only by means of diffusible factors. RECs were cultured in the lower transwell chamber, and HUCPVCs were placed in the upper chamber at the same ratios used for coculture experiments.
Real-Time RT-PCR Analysis
Total RNA was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA) as described by the manufacturer. One microgram was reverse-transcribed into cDNA using random primers and MultiScribe RT (High-Capacity cDNA Reverse Transcription Kit; Applied Biosystems, Foster City, CA, USA). Quantitative RT-PCR was performed using human-specific primers for guanine-adenosine-thymine-adenine binding protein 4 (GATA4), myocyte enhancer factor 2C (Mef 2c), α-CA, ANP, cTnT, and α-MHC (Supporting Information Fig. S4 and Table S1). Samples were assayed in triplicate using Power SYBR Green master mix on an Applied Biosystems Sequence Detection System (SDS7900HT) with the following conditions: 1 cycle for 10 min at 95°C, 40 cycles with 95°C for 15 s and 60°C for 60 s, followed by a melting curve analysis. Results were analyzed using the Relative Quantification (2-DDCt) method by normalizing the threshold cycle (Ct) values against the reference gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (35).
Flow Cytometry
HUCPVCs and BM-MSCs were characterized by flow cytometry (Supporting Information Fig. S1). Cells were washed in PBS and incubated for 30 min in 2% FBS/PBS containing the following conjugated antihuman antibodies (at 1:50 dilution): cluster of differentiation 90-fluorescein isothiocyanate (CD90-FITC; Serotec, Raleigh, NC, USA), CD44-allophycocyanin (APC), CD105-phycoerythrin (PE) (both eBioscience, San Diego, CA, USA), CD11b-PE, CD34-PE, CD45-PE, CD49e-PE, CD73-PE (all BD Biosciences, San Jose, CA, USA). The cell suspensions were then washed twice and resuspended in 2% FBS/PBS for flow cytometric analysis (Cytomics FC500, Beckman Coulter, Brea, CA, USA).
The percentage of human cells expressing α-MHC after the coculture was also analyzed by flow cytometry. Cells were harvested using 0.2% trypsin, washed in PBS, and resuspended in 2% FBS/PBS. Cells (106) were stained with an APC-conjugated antihuman CD44 antibody (1:50; eBioscience), which does not cross react with rat CD44 and therefore was used to distinguish human from rat cells in the cocultures. Cells were then washed, fixed with paraformaldehyde (2%; Sigma-Aldrich), and permeabilized using 0.5% Triton X-100 for 10 min. After a blocking step with donkey serum (5% in PBS; Abcam, Cambridge, MA, USA), cells were stained 30 min with a mouse anti-a-MHC antibody (1:200; Abcam), which recognizes both human and rat epitopes. Finally, cells were incubated for 30 min with AF555-conjugated donkey anti-mouse IgG antibody (1:200; Molecular Probes, Eugene, OR, USA). Flow cytometry data were analyzed using FlowJo software (TreeStar, Ashland, OR, USA).
Immunocytochemistry
Immunocytochemical techniques were used to detect α-MHC, cTnT, and desmin in GFP-labeled human MSCs cocultured with rat embryonic cardiomyocytes for 14 days. Slides were washed in PBS, fixed with 4% paraformaldehyde, and permeabilized using 0.25% Triton X-100 for 10 min. For α-MHC, a monoclonal anti-α-MHC antibody (1:200; Abcam) was used with an AF555 anti-mouse IgG secondary antibody (1:200; Molecular Probes). For cTnT, a goat anti-cTnT polyclonal antibody (1:200; Santa Cruz Biotechnology, Dallas, TX, USA) was used along with an anti-goat IgG linked Cy3 secondary antibody (1:200; Abcam). For desmin, a rabbit anti-desmin polyclonal antibody was used (1:100; Abcam) in conjunction with an anti-rabbit IgG linked PE secondary antibody (1:200; Abcam). Nuclei were stained using ProLong® Gold Antifade Reagent with 4′,6-diamidino-2-phenylindole (DAPI; Molecular Probes). Detection of fluorescent signals for each of these markers was performed using Zeiss AxioObserver Z1 inverted microscope (Zeiss, Thornwood, NY, USA) and Metamorph software (Molecular Devices, Sunnyvale, CA, USA).
Experimental Acute Myocardial Infarction Model in Immune-Deficient Mice
Procedures conformed to the US National Institute of Health's guidelines for the care and use of laboratory animals with approval from the Animal Care and Use Committee of the Ontario Cancer Institute (OCI). The NOD SCID gamma (NSG) mice were obtained from Jackson Laboratory (stock number: 005557) and bred at the OCI Animal Facility. We chose this immune-deficient mouse model, which lacks B, T, and natural killer (NK) cells, to be able to study human MSCs in a xenogeneic model without rejection. Despite the immunodeficiency, this model is widely accepted for the study of myocardial infarction. Myocardial infarction was performed in 10-week-old female mice as previously described (52). Briefly, anesthesia was induced using isoflurane (Ontario Cancer Institute Animal Facility), and mice were mechanically ventilated using a TOPO ventilator (Kent Scientific Corporation, Torrington, CT, USA). An incision was made through the 5th intercostal space, and the chest was opened. A 7–0 silk suture (Ethicon, San Angelo, TX, USA) was passed underneath the left anterior descending coronary artery 1–3 mm from tip of the left auricle and ligation was performed. Ischemia was confirmed in all cases by myocardial blanching and ventricular arrhythmia. After MI, mice were randomly assigned to one of the following groups (n = 6 per group): (a) PBS IM; (b) BM-MSC 0.5×106 cells IM; (c) HUCPVCs 0.5×106 cells IM; (d) PBS IV; (e) BM-MSC 0.5×10 6 cells IV; (f) HUCPVCs 0.5×106 cells IV. Cell therapy was administered 15 min after AMI. Cells were suspended with 50 μl PBS using a 100-μm Hamilton syringe (Hamilton, Reno, NV, USA) and were injected into the infarct border zone. For IV administration, cells were suspended with 200 μl PBS and infused via tail vein using a 28-gauge needle (Terumo Medical Corporation, Somerset, NJ, USA). At 48 h after AMI, three mice per group were sacrificed to determine the presence of human cells by detecting human Alu sequences (see below). Expression of cardiomyocyte determinants was evaluated 7 days after AMI in an additional group of six mice injected IM with GFP-labeled human cells. See Supporting Information Figure S5 for a schematic diagram of the in vivo study design.
Echocardiography
Transthoracic echocardiographic imaging was performed by a blinded observer using a Vevo Ultrasound System (40 MHz transducer; Visualsonics Inc., Toronto, Canada). Hemodynamic parameters were acquired from optimized short-axis view of the left ventricle at the mid-papillary muscle level, using both B and M modes, in anesthetized mice (2% isoflurane) with heart rates between 500–700 bpm. The following measurements were obtained at baseline (before MI induction) and at day 14: left ventricular end-systolic diameter (LVESD), left ventricular end diastolic diameter (LVEDD), and septum thickness (Supporting Information Fig. S6). Fractional shortening was calculated with the formula: FS% = (LVEDD-LVESD)/LVEDD×100%.
Real-Time PCR Assays for Human Alu Sequences
DNA extracted from mouse heart, lung, spleen, and liver was subjected to real-time PCR for Alu sequences (Supporting Information Fig. S7 and Table S1). PCR was performed in 10-μl reactions containing 1× Taqman Universal PCR Master Mix (Applied Biosystems), 900 nM each of the Alu forward and reverse primers, 250 nM Alu TaqMan probe, and 100 ng target template. Reactions were incubated at 50°C for 2 min and at 95°C for 10 min followed by 40 cycles at 95°C for 15 s and 60°C for 1 min. Standard curves were generated by serial dilutions of human DNA prepared in a background of mice DNA. Total DNA in the samples was assessed by real-time PCR assays with primers that amplified both the human and the mouse gene for GAPDH. Samples were standardized by correction for total DNA and results presented as human DNA content per 100 ng of total DNA (31).
Microscopic Examination of the Myocardium and Immunohistochemistry
Fourteen days after MI (n = 3 per group), mice were anesthetized with ketamine (Ontario Cancer Institute Animal Facility) and injected with KCl (0.08 mg/g; Sigma-Aldrich) in the inferior vena cava in order to obtain a diastolic arrest. The heart was harvested and the left ventricle (LV) along with its septum dissected out from the rest. A transverse cross section was done 0.5–1 mm apically from the ligation site, formalin-fixed, and embedded in paraffin. Three 4-μm-thick slices from different levels were cut, stained with Masson's trichrome (Sigma-Aldrich), and scanned with Aperio Scanscope XT (Aperio Technologies Inc., Vista, CA, USA). Total myocardial and fibrotic areas as well as septum thickness (average of 10 equidistant measurements) were manually traced and calculated using ImageScope software (Aperio Technologies) as previously described (28). The measurements were performed on three separate slices from different levels of each heart and the averages used for statistical analysis. Infarct size was expressed as percentage of total LV area (8). For immunohistochemistry, heart tissue sections were deparaffinized, rehydrated, digested with 1% pepsin (Worthington Biochemical Corporation), and treated with 3% aqueous hydrogen peroxide (Sigma-Aldrich) to block endogenous peroxidase. For GFP immunostaining, sections were incubated for 60 min with a rabbit polyclonal anti-GFP antibody (1:1,000; Molecular Probes). Consecutive slides were stained using a mouse monoclonal antibody to cardiac myosin heavy chain (α-MHC; 1:1,500, Abcam). To determine murine angiogenesis, mouse blood vessels were stained and quantified in three separate sections at different levels of each heart by using a rabbit polyclonal anti-CD31 antibody (1:100; Abcam). All the sections were then incubated with peroxidase-linked secondary antibodies, developed with 3′-diaminobenzidine (Sigma-Aldrich) and counterstained with Mayer's hematoxylin (Sigma-Aldrich). Slides were scanned with Aperio Scanscope XT, and the result was expressed as vessels per 0.2 mm2.
Statistics
Continuous variables were expressed as mean ± SD. Two-way analysis of variance with the post hoc Bonferroni test was used for comparisons to assess changes in cardiac gene expression and α-MHC promoter activity over time. The unpaired Student's t test was used to evaluate statistical significance between HUCPVCs and BM-MSCs for α-MHC protein expression and between the transwell system and conditioned media for the induction of reprogramming. One-way analysis of variance with the post hoc Tukey's test was used for intergroup comparisons in the AMI model. A value of p < 0.05 was considered statistically significant.
Results
HUCPVCs Express Higher Levels of Cardiomyocyte-Specific Genes Than BM-MSCs After Coculture with Rat Embryonic Cardiomyocytes
Reprogramming HUCPVCs and BM-MSCs towards the cardiomyocyte lineage was determined by analyzing the time course of cardiomyocyte-specific gene expression for up to 14 days after coculture with RECs (Fig. 1). Interestingly, HUCPVCs basally expressed the cardiac transcriptions factors GATA4 and Mef 2c, which peaked at day 7 of coculture with an enhancement of 14.8- and 7.4-fold, respectively, compared with baseline. BM-MSCs only expressed these two factors after coculture. In addition, transcript levels of GATA4 and Mef 2c after 7 days were significantly lower in BM-MSCs compared with HUCPVCs (7.6- and 3.5-fold, respectively) (Fig. 1A, B). Both cell types showed similar upregulation of α-cardiac actin (α-CA) and atrial natriuretic protein (ANP) after coculture; however, the expression of cardiac troponin-T (cTnT) and α-myosin heavy chain (α-MHC) were significantly higher in HUCPVCs versus BM-MSCs, especially at day 14 (2.8- and 5.3-fold, respectively) (Fig. 1E, F).
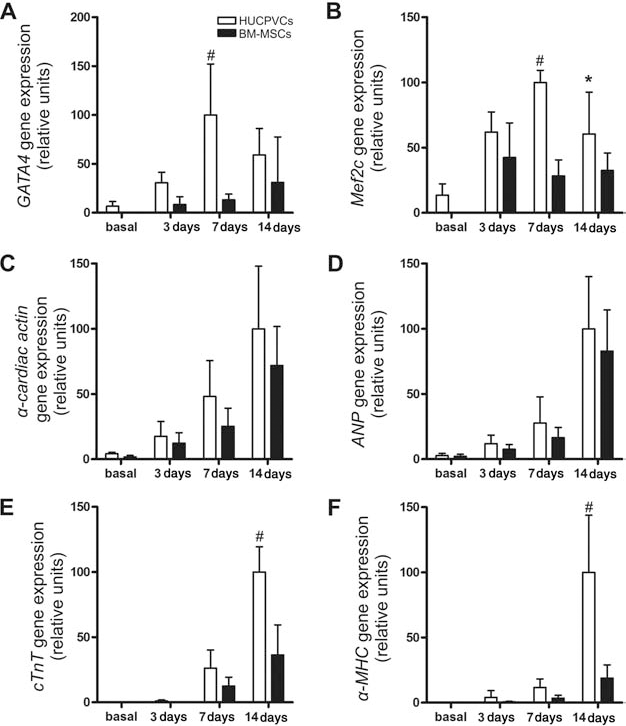
Expression of cardiac specific genes in human umbilical cord perivascular cells (HUCPVCs) and bone marrow-derived mesenchymal stromal cells (BM-MSCs) after coculture with rat embryonic cardiomyocytes for 3, 7, and 14 days. Quantitative realtime RT-PCR was performed to assess changes in gene expression of GATA4 (A), Mef 2c (B), α-cardiac actin (C), ANP (D), cTnT (E), and α-MHC (F). Results were normalized against the reference gene GAPDH and are presented as relative units taking maximum gene expression as 100 units. Data represent mean ± SD of six independent experiments. *p < 0.05 and #p < 0.001 between groups derived from two-way ANOVA after Bonferroni test. GATA4, guanine-adenosine-thymine-adenine binding protein; Mef 2c, myocyte enhancer factor 2C; ANP, atrial natriuretic peptide; cTnT, cardiac troponin T; MHC, myosin heavy chain; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
To determine the frequency of cells actively transcribing cardiomyocyte genes, HUCPVCs and BM-MSCs were transduced with a novel bidirectional lentiviral vector containing the constitutive promoter EF1α driving DsRed and the α-MHC promoter driving eGFP expression (Supporting Information Fig. S3). We observed that 8.6 ± 7.1%, 22.3 ± 8.2%, and 52.1 ± 16.8% of the HUCPVCs expressed GFP after 3, 7, and 14 days of coculture, respectively (Fig. 2). In contrast, only 29.3 ± 6.1% of the BM-MSCs were GFP+ at 14 days (p < 0.001 vs. HUCPVCs) (Fig. 2D).
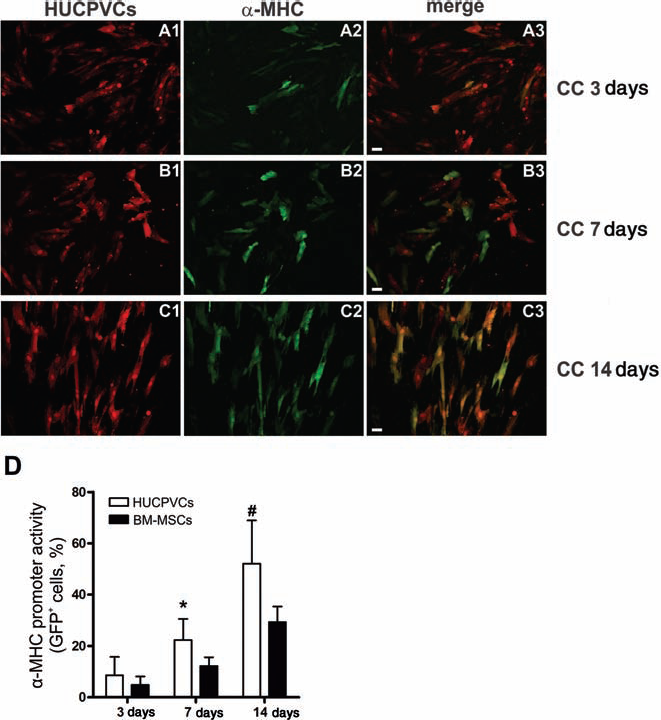
Characterization of α-myosin heavy chain (α-MHC) gene promoter activity in HUCPVCs and BM-MSCs after coculture with rat embryonic cardiomyocytes for 3, 7, and 14 days. Human cells were transduced with a bidirectional lentivector to constitutively express DsRed protein and α-MHC promoter-driven eGFP. The expression of eGFP was analyzed by fluorescence microscopy (original magnification: 200×, scale bar: 20 μm). Representative images of HUCPVCs at 3 days (A1–3), 7 days (B1–3), and 14 days (C1–3) of coculture. (1) DsRed-positive cells (red). (2) eGFP-positive cells (green). (3) Merge (double-positive cells are yellow). (D) α-MHC promoter activity is expressed as the percentage of the DsRed-positive cell population expressing eGFP. Data represent mean ± SD of 10 independent experiments. *p < 0.05 and #p < 0.001 between groups derived from two-way ANOVA after Bonferroni test. DsRed, Discosoma sp. red fluorescent protein; eGFP, enhanced green fluorescent protein; CC, coculture; MHC, myosin heavy chain.
HUCPVCs Express α-MHC and cTnT but Retain Expression of MSC Markers After Coculture with Rat Embryonic Cardiomyocytes
The expression of cardiomyocyte proteins was assessed by immunocytochemistry in coculture experiments carried out using GFP-labeled human cells (Supporting Information Fig. S2). As shown in Figure 3, HUCPVCs were able to express the cardiomyocyte proteins cTnT and α-MHC, but not the muscle cell marker, desmin. After 14 days of coculture, HUCPVCs did not show the characteristic cross-striated sarcomeric pattern (Fig. 3A, B) or the spontaneous contractions of cardiomyocytes when observed by microscopy, indicating that cardiomyocyte differentiation is partial under our experimental conditions. Similar results were obtained with BM-MSCs (data not shown). Of note, rat proteins were also recognized with these antibodies and provided an internal positive control (RECs are exclusively the red cells in Fig. 3A–C). When cocultures were assayed by flow cytometry (Fig. 3D, E), approximately one third of the total rat cells were cardiomyocytes (CD44 negative cells expressing α-MHC). Flow cytometry data also showed a higher frequency of HUCPVCs versus BM-MSCs expressing α-MHC protein after 14 days of coculture (11.6 ± 0.9% vs. 5.3 ± 0.3%, p < 0.001) (Fig. 3D, E). This result correlates with the enhanced α-MHC gene expression found in HUCPVCs compared with BM-MSCs (Fig. 1).

Expression of cardiac-specific proteins in eGFP-labeled HUCPVCs after coculture with rat embryonic cardiomyocytes for 14 days. Immunocytochemical analysis was performed using antibodies against α-MHC (A), cTnT (B), and desmin (C). (1) eGFP-positive cells (green). (A2) α-MHC-positive cells (red). (B2) cTnT-positive cells (red). (C2) Desmin-positive cells (red). (3) Total nuclei were stained with DAPI (blue). (4) Merge. Images are representative of three independent experiments (original magnification: 400×, scale bar: 10 μm). (D) Representative flow cytometry analysis of α-MHC protein expression in HUCPVCs and BM-MSCs after coculture for 14 days. APC-conjugated anti-human CD44 antibody was used to distinguish human from rat cells in the coculture system. (E) α-MHC-positive cells were calculated as percentage of the CD44+ population. Data represent mean ± SD of four independent experiments. Values of p derived from Student's t test. cTnT, cardiac troponin T; MHC, myosin heavy chain; APC, allophycocyanin; CD44, cluster of differentiation 44; PE, phycoerythrin.
Flow cytometry analysis demonstrated that both HUCPVCs and BM-MSCs retain the stromal determinants CD44, CD73, and CD105 (Fig. 4). It is noteworthy that both cell types were negative for the hematopoietic and endothelial markers CD45 and CD34, respectively (Supporting Information Fig. S1) and did not acquire the expression of CD34 after coculture (Fig. 4D, H).

HUCPVCs and BM-MSCs retain the stromal markers and do not acquire the endothelial marker CD34 after coculture with rat embryonic cardiomyocytes. The expression of stromal markers CD44 (A, E), CD73 (B, F), and CD105 (C, G) and the endothelial determinant CD34 (D, H) was assessed by flow cytometry in eGFP-labeled HUCPVCs (A–D) and BM-MSCs (E–H) after 14 days of coculture. The analysis was performed by gating the GFP-positive population (dotted rectangles). Solid gray histograms represent cells stained with fluorescent antibodies, while isotype-matched controls are overlaid in a dotted black line on each histogram. Data are representative of three independent experiments.
Soluble Factors Released by Rat Embryonic Cardiomyocytes Induce Cardiomyocyte-Specific Gene Expression in HUCPVCs
To test whether partial cardiomyocyte reprogramming is cell contact-dependent or can be mediated by soluble factors, we cultured HUCPVCs with conditioned medium obtained from the coculture experiments. As shown in Figure 5A, conditioned medium can induce the expression of cardiomyocyte-specific genes but to a lesser extent than does cell coculture. The enhancement of α-CA and ANP decreased by 2.5-fold, whereas α-MHC and cTnT upregulation was reduced fivefold compared with coculture experiments. The frequency of HUCPVCs showing an active α-MHC promoter was also significantly less in conditioned medium versus coculture (11.6 ± 5.6% vs. 52.1 ± 16.8% at 14 days, p < 0.001) (Fig. 5B, C). These data infer that cell to cell contact or labile short distance factors are required for cardiomyocyte reprogramming. When the coculture was set up using a transwell membrane that does not allow cells to interact but provides a viable REC layer continuously releasing paracrine factors, the expression of cardiac specific genes and α-MHC promoter activity were significantly enhanced compared with conditioned medium only (Fig. 5A–C). Moreover, the transwell system showed reprogramming efficiency comparable to cell coculture, suggesting the limited importance of cell to cell contact. The expression of cardiac specific genes and α-MHC promoter activity were significantly reduced after the removal of the conditioned medium (data not shown) or transwell inserts (Fig. 5D, E). These data suggest that HUCPVCs are not fully committed to the cardiac lineage and hence provide more evidence for partial cardiomyocyte reprogramming obtained under our experimental conditions.

Partial cardiomyocyte differentiation of HUCPVCs can be induced in a transwell system or with conditioned medium obtained from cocultures. (A) Expression of cardiac specific genes in HUCPVCs after culture with conditioned medium or using a transwell membrane for 3, 7, and 14 days. Quantitative real-time RT-PCR data are presented as the percentage of induction compared with results using the coculture system. Data represent mean ± SD of five independent experiments. *p < 0.001 compared to conditioned media derived from Student's t test. (B, C) α-MHC promoter activity in HUCPVCs after culture with conditioned media or using a transwell system for up to 14 days. HUCPVCs transduced with the bidirectional lentivector to constitutively express DsRed protein and α-MHC promoter-driven eGFP were analyzed by fluorescence microscopy. (B) Representative images after 14 days of culture (original magnification: 200×, scale bar: 20 μm). HUCPVCs with an active α-MHC promoter appear yellow (merge column). (C) Changes in α-MHC promoter activity were assessed over time. Data are expressed as the percentage of the DsRed-positive cells expressing eGFP and represent mean ± SD of five independent experiments. *p < 0.05, #p < 0.01, and ‡p < 0.001 between groups derived from Student's t test. (D, E) Partial cardiomyocyte reprogramming of HUCPVCs reverts to the parental MSC phenotype after removal of the rat embryonic cardiomyocyte (REC) layer. HUCPVCs were cultured with RECs using a transwell membrane system for 14 days. After that, the transwell inserts containing the HUCPVC monolayer were removed and cultured in minimum essential medium a modification (α-MEM) for 14 days. (D) Changes in cardiac specific gene expression after removal of RECs. Quantitative real-time RT-PCR data are presented as the percentage of induction compared with results using the coculture system. Data represent mean ± SD of five independent experiments. *p < 0.001 compared to transwell system derived from Student's t test. (E) Changes in α-MHC promoter activity after removal of RECs. Data are expressed as the percentage of the DsRed-positive cells expressing eGFP and represent mean ± SD of five independent experiments. *p < 0.001 compared to transwell system derived from Student's t test. CA, cardiac actin; ANP, atrial natriuretic peptide; cTnT, cardiac troponin T; MHC, myosin heavy chain.
Intramyocardial Injection of HUCPVCs and BM-MSCs Improves Cardiac Function After Acute Myocardial Infarction
The cardiac regeneration potential of HUCPVCs and BM-MSCs was assessed in vivo by intramyocardial injection in the AMI NOD/SCID gamma null mouse model. Using the same animal model, we previously showed that the best time point to evaluate the effect of cell therapy on cardiac function in the acute phase is 14 days after the infarct (10). Thus, echocardiographic and histological data were obtained 14 days after AMI in the present study. Cardiac function determined as fractional shortening (FS) was significantly higher in cell-treated mice compared with control animals, but more interestingly, HUCPVCs showed greater improvement than BM-MSCs (40.0% vs. 18.3% increase p < 0.05) (Fig. 6A). LVEDD, LVESD, and septum thickness were not different among groups (data not shown). It is noteworthy that no improvement in cardiac function was observed when the same dose (0.5×106 cells) was administered intravenously (Fig. 6B). Morphometric analysis of heart sections showed no significant differences in myocardial scar area and scar thickness among groups (Fig. 6C–E). The fate of MSCs was determined using real-time PCR for human-specific Alu sequences (Fig. 7A). More than 99% of the human cells were found in the heart 2 days after IM injection, and the number of lodged cells decreased by twofold at day 14. In contrast, when administered IV, HUCPVCs and BM-MSCs were trapped mainly in the lungs 2 days after infusion (99% and 90%, respectively) and only a very small proportion was detected in the heart (<2.5%) (Fig. 7A). To confirm the presence of MSCs in the infarct area, heart sections were analyzed by immunohistochemistry 7 days after the injection of GFP-labeled cells (Fig. 7B). GFP+ cells were detected by peroxidase-linked colorimetric staining to overcome potential false-positive immunofluorescence due to autofluorescence. GFP-positive cells were lodged exclusively in infarct and peri-infarct areas (Fig. 7B). Consecutive sections were stained for α-MHC using the same technique and, as shown in Figure 7B, more than 95% of GFP+ cells in the ischemic area did not express this cardiomyocyte marker at 7 days. We were interested in comparing the previous result with α-MHC promoter activity in vivo evaluated by the GFP signal from cells transduced with the LV/α-MHC construct. No reproducible data were obtained using confocal microscopy mainly due to the high autofluorescence of the heart sections in the green channel (data not shown). Heart sections were also evaluated for expression of the endothelial marker CD31 to quantify the extent of murine angiogenesis (Fig. 7C). Enumeration of blood vessels showed that HUCPVCs and BM-MSCs increased the number of capillaries in the peri-infarct area by 2.2- and 1.4-fold, respectively, compared with PBS controls (Fig. 7D). These results infer an induction of murine angiogenesis by the cells and suggest that the release of growth factors may play a role.

Functional and histological evaluation 14 days after acute myocardial infarction (AMI). HUCPVCs and BM-MSCs were administered IM or IV after induction of MI in NOD/SCID gamma mice. (A, B) Fractional shortening (FS) was determined by echocardiography in IM (A) and IV (B) treated groups at baseline and 14 days after MI. Data represent mean ± SD (n = 3 per group). *p < 0.05 and #p < 0.01 compared to PBS group and ‡p < 0.05 compared to BM-MSCs group derived from one-way ANOVA after Tukey's post hoc test. (C) Heart sections stained with Masson trichrome (collagen scar light blue, myocytes red) were used for the morphometric analysis in IM injected mice. Representative photomicrographs (scale bar: 1 mm). (D) Scar area quantification in IM injected mice expressed as a percentage of total LV area. Data represent mean ± SD (n = 3 per group). (E) Scar thickness quantification in IM injected mice expressed in millimeter. Data represent mean ± SD (n = 3 per group). IM, intramuscular; I V, intravenous; LV, left ventricle.
Discussion
HUCPVCs have a higher clonogenic potential than BM-MSCs and can undergo adipogenic, chondrogenic, osteogenic, myogenic, and fibroblastic differentiation (50,51). HUCPVCs also share the same immune privileged status of BM-MSCs in both differentiated and undifferentiated state, allowing their use for allogeneic stem cell therapies (19). These characteristics, together with their ready availability, make HUCPVCs potentially useful for cell-based tissue regeneration. The ability of HUCPVCs to undergo cardiomyocyte differentiation and mediate cardiac regeneration, however, has not been previously investigated. We have shown that coculturing mouse MSCs with RECs induces cardiomyocyte-specific gene expression in up to 10% MSCs and is not due to cell fusion which occurs at a frequency of less than 1% (48). Using the same coculture system, we found that the reprogramming of HUCPVCs towards the cardiomyocyte lineage was greater than with BM-MSCs. HUCPVCs have a basal expression of the transcription factors GATA-4 and Mef 2c as well as the cardiomyocyte genes α-CA and ANP but do not express α-MHC and cTnT (Fig. 1). The spontaneous expression of cardiomyogenic markers such as α-CA, connexin (Cx)-43, sarcoplasmic/endoplasmic reticulum calcium ATPase 2 (SERCA-2), and stromal cell-derived factor (SDF)-1α has also been found in umbilical cord blood-derived stem cells (42). These data clearly indicate the absence of mature sarcomeric filaments but suggest that umbilical cord-derived cells are primed towards the cardiac lineage. The levels of the transcription factors GATA4 and Mef 2c increased in HUCPVCs after coculture. Similarly, BM-MSCs were induced to express these factors, but the levels were significantly lower than in HUCPVCs (Fig. 1). Although expression of α-CA, ANP, α-MHC, and cTnT increased in both cell types with coculture, gene upregulation was greater in HUCPVCs, consistent with the enhanced expression of Mef 2c and GATA4 (Fig. 1). Analysis of cardiomyocyte gene expression in mice with a truncated Mef 2c gene confirmed that α-CA, ANP, α-MHC requires Mef 2c for optimum transcription (33). Moreover, there is direct correlation between nuclear content of GATA4 and the expression of cardiomyocyte markers in human MSCs (4). In addition, Mef 2c can bind GATA4, potentiating its transcriptional activity over α-CA, ANP and α-MHC promoters (39). This effect was underscored by the significantly higher α-MHC promoter activity found in HUCPVCs compared with BM-MSCs (Fig. 2). Greater expression of Mef 2c and GATA4 may account for enhanced ability of HUCPVCs to undergo reprogramming towards cardiomyocytes. Of relevance, a recent report demonstrated that these two transcription factors directly reprogram fibroblasts into differentiated cardiomyocyte-like cells (24).
The enhanced activity of the α-MHC promoter was associated with a significantly higher frequency of HUCPVCs expressing α-MHC protein compared with BM-MSCs (Fig. 3). However, both cell types retained the expression of stromal determinants and did not present the cross-striated sarcomeric pattern of cardiomyocytes after 14 days in coculture, indicating that the reprogramming is partial under our experimental conditions (Figs. 4 and 5). These data are in agreement with our previous study showing that murine MSCs acquired cardiomyocyte markers (troponin I and α-actinin) but did not generate action potentials and retained the mesenchymal stromal phenotype in the same coculture system (48). Similarly, human BM-MSCs have also shown limited cardiomyogenic plasticity in vitro and in vivo (29,30,56). Martin-Rendon et al. (38) found that MSCs obtained from the umbilical cord and cord blood did not generate cardiomyocyte-like cells in vitro after treatment with 5-Aza. More recently, stem cells from umbilical cord tissue were induced to express cTnT and cardiac myosin with 5-Aza, although the frequency of reprogramming was not determined (45,55). It is possible that demethylating agents alone may not be sufficiently specific to reprogram human MSCs, inferring that other factors supplied by the cardiac environment, as in our studies, are required. In this regard, several reports have shown the effect of the cardiac milieu on BM-MSCs differentiation (4,15,29,30,48). The presence of soluble molecules capable of triggering the cardiomyocyte reprogramming of HUCPVCs was shown by the use of coculture conditioned medium and transwell membranes (Fig. 5). The latter system achieved a frequency and extent of differentiation similar to cell coculture, suggesting that cell–cell interactions were not necessary and labile short distance factors played a major role. The specific set of molecules released by RECs that are required to trigger cardiomyocyte differentiation has not been determined. Recent studies reported that not only cytokines and growth factors (46) but also microvesicle/exosome-mediated transfer of transcription factors and nucleic acids may be involved in directed reprogramming (54).
Whether BM-derived MSCs can acquire a cardiomyocyte phenotype to enhance cardiac regeneration is unlikely. The low rates of engraftment and cardiomyocyte differentiation reported suggest that reprogramming of MSCs into fully functional cardiomyocytes, if it takes place at all, is a highly uncommon event (32,41,47). In our study, less than 5% of the HUCPVCs expressed α-MHC in the ischemic area (Fig. 7B), indicating that cardiomyocyte differentiation of HUCPVCs in vivo as well is limited and may have coexpressed MSC determinants. It is increasingly believed that cell therapy with MSCs assists the heart by facilitating endogenous repair via paracrine effects, rather than through differentiation to regenerate cardiac tissue (44). Recent reports support the paracrine mechanisms of action of MSCs, which can modulate angiogenesis, cardiomyocyte apoptosis, fibrosis, and inflammation (18,31).

Lodgment of human cells, cardiac reprogramming, and angiogenesis. (A) Tissue distribution of human Alu sequences 2 days after AMI. HUCPVCs and BM-MSCs (0.5×106 cells) were administered IM or IV after the induction of MI. Presence of Alu sequences in mouse hearts 14 days after IM injection (inset). Values represent the amount of human DNA (ng) in 100 ng of total DNA per organ sample as determined by real-time PCR. Data expressed as mean ± SD (n = 3 per group). Li, Liver; Lu, Lung; S, Spleen; MI, Myocardial infarction; PI, Peri-myocardial infarction. (B) Presence of HUCPVCs in the infarct area and expression of α-MHC 7 days after AMI. Peroxidase-based immunohistochemical staining to detect eGFP and α-MHC in heart sections of mice receiving eGFP-labeled HUCPVCs IM after the induction of MI. Slides were counterstained with Mayer's hematoxylin. Representative photomicrographs (scale bar: 100 μm). Note decrease in α-MHC content in the infarct area. Arrows point to the HUCPVCs cells. (C) HUCPVCs IM administration induces murine angiogenesis. Representative immunohistochemical staining for CD31 in cardiac sections 7 days after AMI (scale bar: 100 μm). (D) Quantitation of the number of vessels per 0.2 mm2 of infarcted and peri-infarcted area. Data represent the mean ± SEM (n = 3 per group). *p < 0.001 compared to PBS group and #p < 0.05 compared to BM-MSCs group after one-way ANOVA with Tukey's post hoc test.
The release of soluble growth factors and cytokines is influenced by the developmental status and properties of the MSCs and by their interaction with the local environment (5,26). In fact, immunoselection for stromal precursor antigen-1 (STRO-1) generates a MSC population with improved multilineage differentiation potential and the capacity to produce soluble factors with favorable cardiovascular reparative properties (43). Cardiac lineage prespecification has been shown to enhance the therapeutic benefit of human BM-MSCs in a murine model of chronic myocardial infarction (5). Moreover, a recent study found that the selective removal of transplanted BM cells that commit to cardiovascular lineages produce a significant deterioration of the ejection fraction in a mouse model of AMI, indicating a functional contribution of partial reprogramming in vivo (56). These data suggest that the multipotency of MSCs is an important aspect for both mechanisms proposed for their role, partial reprogramming, and paracrine effects. Following those assumptions, the greater multipotency of HUCPVCs compared with BM-MSCs suggest superior in vivo outcome for the former. We have previously shown that the IV infusion of HUCPVCs and BM-MSCs is associated with a similar improvement in cardiac function after AMI (10). However, the role that partial cardiomyocyte differentiation plays in cardiac regeneration cannot be assessed in that experimental design as the MSCs located mainly to the lungs. Consistent with a link between partial cardiomyocyte reprogramming and paracrine effects, we found here that improvement in cardiac function was significantly higher in mice treated with HUCPVCs versus BM-MSCs after intramyocardial injection (Fig. 6A). Our results also suggest that the release of factors by MSCs is the major mechanism mediating short-term functional recovery. It is noteworthy that HUCPVCs showed more improvement over BM-MSCs in the enhancement of angiogenesis, which may explain, in part, the better functional outcome of HUCPVCs (Fig. 7C, D). It is also well established that MSCs exhibit immunomodulatory properties in vitro and in vivo (6,7,17). This is particularly important in the acute MI setting characterized by a proinflammatory infiltration of neutrophils and macrophages that diminish heart function (40). The anti-inflammatory proteins TNF-stimulated gene 6 protein (TSG-6) and interleukin (IL)-10 released by MSCs improve myocardial infarction in mice (10,31). The concept of a trophic effect of these cells also opens the possibility of using MSC conditioned medium for cardiac tissue regeneration. This approach has shown favorable results, although the degree of repair observed with cell injections was not achieved with single applications of factors secreted by MSCs (22,46,53).
Interestingly, we observed that the advantage of HUCPVCs over BM-MSCs reported here was lost when the same dose of cells (0.5×1 06) was administered intravenously (Figs. 6B and 7A). These data may represent a dilution of the paracrine secretion that reached the heart, as other studies usually administer 2×1 06 MSCs IV to show functional improvement (10,31). Another possible explanation is that the cardiac microenvironment may partially reprogram MSCs to efficiently release the necessary growth factors and cytokines required for cardiac regeneration (5,56). A few reports have demonstrated that HUCPVCs share a similar cytokine and growth factor gene expression profile as BM-MSCs under basal conditions (12,37). Whether partial cardiomyocyte reprogramming specifically affects the expression of these molecules, however, is still unknown. In addition, basal expression of cardiomyocyte transcription factors may also play an important role in HUCPVC-mediated cardiac repair. Further studies are required to determine whether there is a direct association between enhanced partial reprogramming of MSCs and functional improvement of the injured heart.
Conclusion
In summary, we showed that HUCPVCs undergo partial cardiomyocyte reprogramming to a greater extent than do BM-MSCs. Interestingly, HUCPVCs have basal expression of the cardiac transcription factors, GATA4 and Mef 2c, suggesting that they are primed towards differentiation along the cardiomyocyte lineage. Our results also infer a greater ability of HUCPVCs to enhance angiogenesis in vivo, a possible explanation for the short-term functional improvement we demonstrated. HUCPVCs appear to be an attractive alternative source of MSCs in the local treatment of injured myocardium. Further in vivo studies with this enriched MSC population are warranted to address the molecular mechanisms involved and the long-term impact of therapy.
Footnotes
Acknowledgments
This work was supported by the Orsino Translational Research Laboratory of Princess Margaret Hospital. A.K. holds the Gloria and Seymour Epstein Chair in Cell Therapy and Transplantation of University Health Network and the University of Toronto. Author contribution: G.Y.: conception and design, collection and assembly of data, data analysis and interpretation, manuscript writing, final approval of manuscript; V.D.: collection and assembly of data, data analysis and interpretation, final approval of manuscript; N.P. and CJ.L.: collection and assembly of data, final approval of manuscript; J.M.: financial support, administrative support, final approval of manuscript; A.K.: conception and design, financial support, administrative support, data analysis and interpretation, manuscript writing, final approval of manuscript. The HUCPVC technology, developed at the University of Toronto, was licensed by the University to Tissue Regeneration Therapeutics Inc (TRT). TRT supplied the HUCPVCs used in this study. A.K. is on the Scientific Advisory Board of TRT and holds stock.
