Abstract
Extracorporeal shock waves (ESWTs) are “mechanical” waves, widely used in regenerative medicine, including soft tissue wound repair. Although already being used in the clinical practice, the mechanism of action underlying their biological activities is still not fully understood. In the present paper we tried to elucidate whether a proinflammatory effect may contribute to the regenerative potential of shock waves treatment. For this purpose, we exposed human foreskin fibroblasts (HFF1 cells) to an ESWT treatment (100 pulses using energy flux densities of 0.19 mJ/mm2 at 3 Hz), followed by cell analyses after 5 min, up to 48 h. We then evaluated cell proliferation, reactive oxygen species generation, ATP release, and cytokine production. Cells cultured in the presence of lipopolysaccharide (LPS), to induce inflammation, were used as a positive control, indicating that LPS-mediated induction of a proinflammatory pattern in HFF1 increased their proliferation. Here, we provide evidence that ESWTs affected fibroblast proliferation through the overexpression of selected cytokines involved in the establishment of a proinflammatory program, superimposable to what was observed in LPS-treated cells. The possibility that inflammatory circuits can be modulated by ESWT mechanotransduction may disclose novel hypothesis on their biological underpinning and expand the fields of their biomedical application.
Introduction
Extracorporeal shock waves (ESWTs) are “mechanical” waves, characterized by an initial positive very rapid phase, of high amplitude, followed by a negative pressure, producing a “micro-explosion” that can be directed on a target zone (body, tissue, or cells) in order to influence the cells in their behavior. ESWT technology is based on the production of low-pressure acoustic waves that interact directly with cells by mechanotransduction 1 . However, the mechanisms of action of ESWTs on cells are still largely unknown. Previous studies have shown that ESWTs have a pro-activator effect on cells 2 . For instance, ESWT treatment-mediated membrane hyperpolarization and Ras activation for osteogenesis in human bone marrow stromal cells 3 caused macrophage activation 4 and induced upregulation of angiogenesis by the activation of vascular endothelial growth factor 5 , endothelial nitric-oxide synthase, hypoxia-inducible factor 1a, and CD31 in diabetic mice 6 . ESWTs were also implicated in the expression of cytokines, chemokines, and matrix metalloproteinases with proangiogenic outcomes promoting wound healing 7 . Within this context, the main hypothesis is that the physical stimuli delivered through ESWTs can activate the cellular metabolism, by a mechanotransduction mechanism at cellular level 8 , and modulate cellular proliferation, migration, and senescence 9,10 . Other authors have shown that physical treatments modulated a proinflammatory milieu in cells, which is mediated by mechanotransduction 11,12 , similar to the proinflammatory activation mediated by lipopolysaccharide (LPS) 13 . LPS is a component of the outer membrane of Gram-negative bacteria, implicated in the induction of a strong immune response in normal animals. LPS exerts its biological effects on the host by binding to Toll-like receptor 4 (TLR4), a pattern recognition receptor that is widely distributed among lung parenchyma cells, including macrophages, epithelial cells, and fibroblasts 14 . In a recent study it was shown that LPS was able to directly induce secretion of collagen in primary cultured mouse lung fibroblasts via TLR4-mediated activation of the phosphoinositide3-kinase-Akt pathway, thus increasing the proliferation rate 15 . A class of molecules that is currently emerging as a major tuner of inflammation and immune responses are the heat shock proteins (HSPs). HPSs are also involved in cell-cycle control and signaling, and protection of cells against stress/apoptosis 16 . HSPs are often classified upon their molecular weight: hsp27, hsp60, hsp70, hsp90. In particular, HSP90 is associated with proteins of the mitogen-activated signal cascade, particularly with the Src kinase, with tyrosine receptor kinases, Raf and the MAP-kinase activating kinase (MEK) 8 . The Hsp90 system is a complicated machinery that is coactivated by Hsp70 and by a large number of cofactors 17 . Nevertheless, other studies showed that it can also be modulated alone. Fibroblasts are usually considered for their ability of producing and reshaping the tissue extracellular matrix 18 . Besides, fibroblasts play a major role in both secreting and “sensing” a wide-ranging spectrum of cytokines, and “trophic” mediators, involved in pro-, as well as anti-inflammatory responses 19 . Although the development of anti-inflammatory strategies has been extensively regarded as a major tool to afford tissue rescue, it is also essential to keep in mind that timely execution of proinflammatory patterning also plays a critical role in tissue repair. Based upon this central modulatory role of fibroblasts, and their ability to handle the homeostasis of neighboring cells in normal and diseased tissues, in the present study we used human skin fibroblasts as an in vitro model to elucidate the molecular mechanisms underlying a proinflammatory role of ESWTs. For this purpose, the action of ESWTs was dissected at the level of a number of crucial events involved in tissue regeneration, including cytokine production and release, ATP release, activation of reactive oxygen species (ROS), and HSP activation.
Materials and Methods
General Experimental Design
HFF1 Cell Culture
HFF1 are foreskin fibroblast cells type 1, purchased from ATCC (ATCC, Manassas, VA, USA). Cells were cultured and expanded in Dulbecco’s modified Eagle’s medium (Life Technologies, Camarillo, CA, USA), supplemented with 10% fetal bovine serum (Life Technologies, Camarillo, CA, USA), 400 mM glutamine (Euroclone, Milano, Italy), 100 U/ml penicillin, and 100 μg/ml streptomycin (Euroclone), at 37°C in a humidified atmosphere containing 5% CO2. In the present study, cells from passages 2–9 were used. The cell culture medium was changed every 3 d. An additional cell group of HFF1 was treated with 1 ng/ml LPS (Sigma-Aldrich, Hamburg, Germany), as a powerful positive inflammatory inducer, representing positive control of an inflammation-induced cell behavior (positive control of inflammation).
Shock Wave Setting and Cell Treatment
Shock wave device used was Ortho-Gold100 (Tissue Regeneration Technologies, LLC, manufactured by MTS Europe GmbH). Cells after synchronization were detached and a number of 1 × 106 for each group (control/ ESWT-treated) was resuspended in 1 ml medium in a 15-ml polypropylene tube, and shock wave treatment was performed using a water bath setup at 37°C in order to preserve aberrant changes in cellular behavior (Fig. 1). The distance between the shock wave applicator and the 15 ml tube containing the cells was 5 cm, as it was previously optimized 2 . The control group was allocated in a separate water bath without ESWT applicator, with the same temperature condition. ESWT treatment was applied to cells at 100 pulses using energy flux densities of 0.19 mJ/mm2 at 3 Hz, according to previous experiments and in vivo studies 2,20 . Depending on the analysis, cells were either plated in culture plates for further incubation or analyzed immediately.

Scheme of shock wave application in a water bath. One milliliter of cell suspension in a 15 ml polypropylene tube (C) was placed inside the water bath (B) at 37°C. The shock waves device (A) conducted the energy by the applicator connected to a water bath, and placed 5 cm far from tube 21 .
Viability Assay by BRdU Assay
For the BrdU proliferation assay, cells were seeded in 24-well plates after ESWT treatment and the proliferation assay (Roche, Merck KGaA, Darmstadt, Germany) was performed 24 h after treatment. According to the manufacturer’s protocol, BrdU labeling solution was added to the cell cultures exposed to ESWTs, as well as controls, at a final concentration of 10 μM and incubated for 2 h. This was followed by 90-min incubation with diluted peroxidase-conjugated anti-BrdU antibody. Absorbance was measured with a photometer (Spectra Thermo, TECAN Austria GmbH, Grödig, Austria) at a wavelength of 450 nm.
ROS Production—Electron Paramagnetic Resonance
HFF1 cultures were treated with or without 0.19 mJ/mm2 100 pulses shock waves. HFF1 treated for 24 h with 1 ng/ml LPS served as positive controls. After treatment cells were seeded in 24-well plates, in a final volume of 250 µl. The cells were observed at 1, 4, 8, 12, 24, and 48 h. Ten minutes before every measurement, PPH (1-hydroxy-4-phosphono-oxy-2,2,6,6-tetramethyl-piperidine), a spin probe for oxygen radicals specific for superoxide radicals, and peroxynitrite were added at a final concentration of 500 μM. Fifty microliters of medium was put into oxygen-permeable capillary tubes (Noxygen Science transfer and Diagnostics, Elzach, Germany) and measured at room temperature with e-scan EPR spectrometer (Bruker, Germany) with the following settings: microwave frequency: 9.762741 GHz, modulation frequency: 86.00 GHz, modulation amplitude: 1.4 GHz, center field: 3487.920 G, sweep width: 200 G, microwave power: 54.10 mW, number of scans: 1, receiver gain: 2.24 × 10.
ATP Release
Immediately after shock wave pulses or after 0, 5, 10, 15, 30, 60, and 120 min, cells were lysed to measure ATP production. Lysates were pipetted into a 96-well microplate and 100 µl ATP Assay reagent (Promega Cell Titer-Glo Luminescent Cell Viability Assay, Klaus, Austria) was added and shaken for 2 min. Lysates were incubated for further 10 min, after which sample luminescence was measured by a luminometer (SPECTRA star Omega, Germany). Sample data were compared to an ATP standard calibration curve.
Enzyme-Linked Immunosorbent assay (ELISA) on HFF1
HFF1 were cultured for 24 h in the absence or presence of 1 ng/ml LPS, inducing an inflammatory response to be used as a comparative positive control for shock wave–treated cells 22 . After ESWT treatment, cells were seeded in 24-well plates in a final volume of 250 µl. The culture medium was collected at different time points (1, 4, 8, 12, 24, and 48 h) for the ELISA analysis. The levels of interleukin 6 (IL-6) and IL-8, monocyte chemoattractant protein 1 (MCP-1), and TGF-alpha released into the culture supernatant were measured using the human Ready-SET-Go kit (eBioscience, Life Technologies, Camarillo, CA, USA), according to the manufacturer’s protocol.
RNA Extraction, cDNA Synthesis, and Gene Expression Analysis by Real-Time Polymerase Chain Reaction (PCR)
Total RNA was isolated using Trizol reagent according to the manufacturer’s instructions (Sigma-Aldrich, Hamburg, Germany), as previously described 23,24 . Total RNA was dissolved in RNAase-free water and quantified using a Nanodrop spectrophotometer. A 20-µl reaction volume containing 1 µg total RNA was reverse transcribed using HiCapacity cDNA reverse transcription kit (Applied Biosystem, Foster City, CA, USA) with oligo (dT) primers. Quantitative PCR reaction was performed using i5 Biorad with kapa green (Biorad, Hercules, CA, USA).
After an initial denaturation step at 95°C for 15 min, temperature cycling was initiated. Each cycle consisted of 95°C for 15 s, 53°C –59°C for 30 s, and 72°C for 15 s, the fluorescence being read at the end of this step. All primers used in this work were from Invitrogen and are listed in Table 1. PCR products were confirmed by melting curve analysis and electrophoresis. All measurements were done as technical quadruplicate of biological replicates. Biological replicates were obtained from independent cell culture samples at different passages. Relative expression was determined using 2-ΔΔCt method with glyceraldehyde 3-phosphate dehydrogenase as housekeeping gene.
Primers List.

HSP: heat shock protein; IL: interleukin; MCP-1: monocyte chemoattractant protein 1; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Statistics
Statistical differences were evaluated by using two-way analysis-of-variance tests with Tukey’s correction. All data sets are presented as mean ± standard deviation. P-values less than 0.05 were considered as significant. All statistical analyses were performed with GraphPad Prism 6.0 software (GraphPad, San Diego, CA, USA). All experiments were performed seven times with HFF1 with three technical replicates. We considered *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001.
Results
Cell Proliferation
The BrdU assay showed that cell proliferation was significantly increased in HFF1 during the first 24 h following shock wave therapy of 0.19 mJ/mm2 or LPS, as compared to the negative control group (Co) (Fig. 2).

Cell proliferation analysis with BrdU was performed in HFF1 at 24 h after exposure in the absence (Co, gray bars), or presence of ESWT 0.19 mJ/mm2 treatment 100 pulses (ESWT, orange bars). LPS was administered as a positive control (LPSCo, white bars) (***P < 0.01).
ATP Release After Shock Wave Treatment
Figure 3 shows ATP concentration, detected in supernatants from HFF1 cells, treated in the absence (control) or presence of ESWTs, or with LPS as a positive control. The same figure shows that HFF1 cultured in the presence of LPS released a significantly higher amount of ATP, as compared to control cells. Interestingly shock wave treatment caused a significantly higher release of ATP as compared to both controls and LPS-treated cells (Fig. 3).
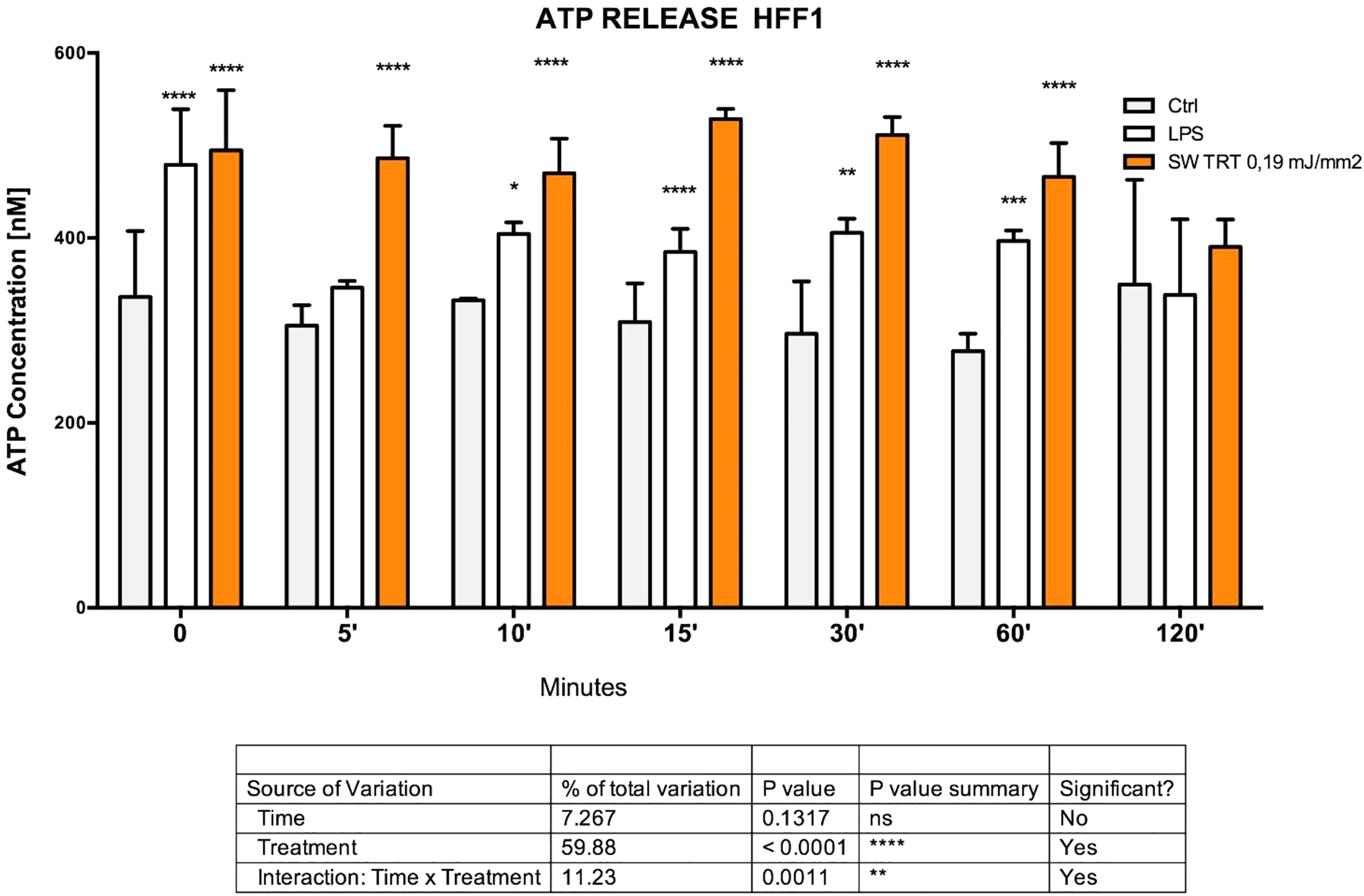
ATP concentrations in HFF1 supernatants were determined at 0, 5, 10, 15, 30, 60, and 120 min after ESWT 0.19 mJ/mm2 treatment 100 pulses (orange bars) or no treatment (gray bars), LPS (white bars) was administered as a positive control.
ROS Produced in HFF1
Figure 4 shows that ROS were gradually released after 1 h of ESWT treatment, peaking at 8 h, then gradually decreased in HFF1. The same figure shows that also LPS-treated cells stimulated ROS release; nevertheless, ESWT was able to induce the production of a higher amount of ROS as compared to both the negative and positive (LPS-treated cells) control.

HFF1 ROS measurement analysis with electron spin resonance using PPH was performed at 0, 1, 4, 8, 12, 24, and 48 h after ESWT 0.19 mJ/mm2 treatment 100 pulses (orange bars) or no treatment (gray bars). LPS (white bars) represents the positive control. Orange asterisks are related to ESWT treatment, black asterisks are related to LPS treatment.
Cytokine Release
IL-6 was detected already after 4 h in cells treated with LPS, but no significant differences could be detected between ESWTs treated and the negative control at this time point (Fig. 5). Nevertheless, IL-6 was upregulated in cells treated with ESWTs after 8 h, similar to what was observed in LPS positive control. At 12 and 24 h, IL-6 was still increased above the control levels in both LPS-treated and ESWT-exposed cells (Fig. 5).

Effect of shock wave treatment on cytokine production in HFF1 at different time points (1, 4, 8, 12, and 24 h) in response to shock wave treatment. The levels of IL-6 (A) and IL-8 (B), and MCP-1 (C) were measured in cell supernatants using ELISA. Orange asterisks are related to ESWT treatment, black asterisks are related to LPS treatment, both compared to untreated control at the given time point.
The level of IL-8 was significantly upregulated already after 4 h following LPS treatment, as well as in cells that received ESWT treatment; however, in ESWT-exposed cells IL-8 declined at subsequent time points with similar expression levels as those detected in unexposed cells (Fig. 5).
The timely pattern of MCP-1 expression reproduced the expression of IL-8 and IL-6 at 8 h, with a significant upregulation in both ESWT- and LPS-exposed cells, when compared to unexposed controls. Consistent with the time course expression of IL-8, MCP-1 levels in ESWT-treated HFF1 declined at 12 and 24 h with no significant difference compared to values observed in untreated controls (Fig. 5).
Gene Expression Analysis
Figure 6 shows that IL-8 transcription was strongly enhanced in HFF1 that had been cultured for 4, 8, 12, and 24 h with LPS. ESWT-treated cells exhibited a significant increase in IL-8 mRNA expression only at 4 h after exposure, with a progressive decline at 12 and 24 h. MCP-1 gene expression was significantly increased after 4 h in HFF1 that had been treated with LPS, compared to untreated cells. ESWT slightly enhanced MCP-1 gene expression at 4 and 8 h. After 12 or 24 h, the transcriptional enhancement was only observed in LPS-treated cells (Fig. 6).
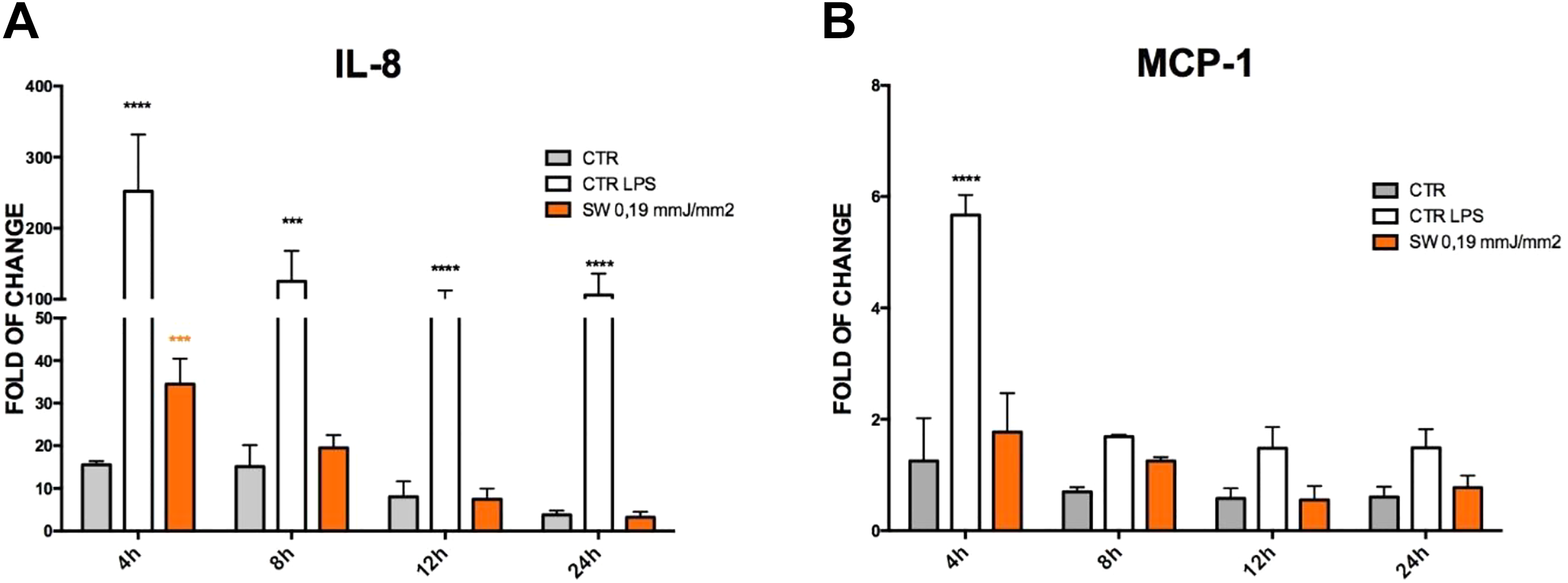
Effect of shock wave pulses on IL-8 and MCP-1 gene expression on HFF1. Cells were exposed (orange bar) or not (gray bars) to 100 pulses 0.19 mJ/mm2, cell treated with 1 ng/ml LPS are positive control (white bar). The amount of IL-8 (A), MCP-1 (B) mRNA was assayed during 4, 8, 12, and 24 h and was normalized to glyceraldehyde 3-phosphate dehydrogenase. The data were plotted as fold change relative to the mRNA expression in control cell at 0 h (mean ± SD; n = f3), orange asterisks are related to ESWT treatment, black asterisks are related to LPS treatment.
Figure 7 shows that the mRNA levels of the analyzed HSPs were differentially modulated by LPS and ESWTs. In particular, HSP90 gene expression exhibited an increase after 4 and 8 h in both ESWT- and LPS-treated cells, being then lowered to control values in both experimental groups at 12 and 24 h (Fig. 7). Transcription of HSP60 was increased in ESWT-treated HFF1 over both control and LPS-treated cells at 4 h, with no significant changes among the investigated groups at 8, 12, and 24 h. Conversely, HSP27 gene expression not only increased at 4 and 8 h in SW- and LPS-treated cells, but its overexpression persisted at similar levels in both experimental groups at 24 h, as compared to unexposed cells (Fig. 7). HSP70 transcription was only slightly increased at 4 and 8 h in ESWT-exposed cells, with no significant change at subsequent time points, where only LPS proved effective, as compared to untreated controls (Fig. 7).

Effect of shock wave pulses on HSP gene expression on HFF1. Cells were exposed (Bordeaux bar) or not (gray bars) to 100 pulses 0.19 mJ/mm2. Cells treated with 1 ng/ml LPS are positive control (white bar). The amount of HSP90 (A), HSP27 (B), HSP60 (C), and HSP70 (D) mRNA was assayed at 0, 4, 8, 12, and 24 h and was normalized to glyceraldehyde 3-phosphate dehydrogenase. The data were plotted as fold change relative to the mRNA expression in control cell t0 (mean ± SD; n = 3).
Discussion
Although the effectiveness of ESWTs has been clearly demonstrated in clinical applications, the underlying molecular mechanisms are not fully clarified yet. ESWTs were initially used to disintegrate renal stones 25 , afterwards the use of ESWTs was completely updated into a pro-regenerative treatment in musculoskeletal disorders 26,27 , soft tissue wounds repair, 28,29 and neurological pathologies 30 . ESWT action was found to trigger the secretion of IL-6, IL-8, MCP-1, and tumor necrosis factor alpha (TNF-α) in a way that is strictly related to the applied “dose” 31,32 . In a study published by Weihs et al., different dosages of ESWTs enhanced cell proliferation in C3H10T1/2 murine mesenchymal progenitor cells, primary human adipose tissue–derived stem cells, and human T cell line, and improved wound healing via ATP release-coupled Erk1/2 and p38 mitogen-activated protein kinase (MAPK) pathways 2 . This is in line with the report of Chen et al. who showed that the ESWT mechanism is mediated by ATP release and P2 receptor activation that promote cell proliferation and tissue remodeling via Erk1/2 activation 20 . In addition, the implication of PI-3K/AKT and nuclear factor kappa B (NF-kB), as well as TLR3/TLR4 signaling pathways has been reported following ESWT cell treatment 33 . Recently, it was observed that ESWTs can modulate the expression of IL-6, IL-8, MCP-1, and TNF-α in human periodontal ligament 31 . In the present study, we show that ESWT action can trigger the activation of proinflammatory pathways. It is known that during stress, cells respond with various reactions, including inflammation 34 . Our observation that the LPS-induced inflammatory response was coupled with HFF1 proliferation is in line with the report from He et al., who observed significantly enhanced proliferation in lung fibroblast exposed to LPS 35 . Recently, other authors described the effect of ESWTs on the modulation of IL-6, IL-8, MCP-1, and TNF-α in periodontal ligament fibroblasts 31 . In the present in vitro experiments, we hypothesized that ESWTs at 0.19 mJ/mm2 may modulate early proinflammatory response (initiation phase). Hence, the hypothesis that ESWT-primed mechanotransduction may have enhanced fibroblast proliferation through a proinflammatory induction is inferred by the observation that as early as 4–8 h after delivery, ESWT was able to enhance both the protein and gene expression of various cytokines, including IL-6, IL-8, and MCP-1. ILs are produced by numerous cell types, including macrophages, monocytes, fibroblasts, endothelial cells, and smooth muscle cells 36 . Interestingly, unlike the LPS-mediated cytokine overexpression, the effect produced by ESWT did not occur as a long-lasting phenomenon. Moreover, ESWT therapy has been found to promote TLR3 stimulation, alongside TLR-4, and downstream signaling, leading to the production of cytokines and chemokines that in turn modulate a macrophage-mediated inflammatory response 37 . Additional evidence shows that LPS promotes ATP release as well 33 by the activation of macrophage cells. In other studies, the ability of ESWTs to induce ATP release, which in turn activates Erk1/2, an upstream effector for Mek1/2, and p38 MAPK, was consistently demonstrated 2 . Here we confirmed these results, by showing that ESWTs mainly enhanced cell proliferation, acting on ATP release (Fig. 3). ROS physiologically play a role as secondary messengers during the repairing process, and appear to be important in coordinating the recruitment of white blood cells during healing 38 . Endogenous cellular ROS can arise from mitochondrial oxidative phosphorylation during ATP production, from the endoplasmic reticulum or from a class of enzymes known as oxidoreductases 39 . ROS have an established role in inflammation and have been implicated in the activation of signaling pathways such as MAPKs, NF-kB, and guanylate cyclase 40 . Recently, it was observed that ROS also played a role in the activation of the NLRP3 inflammasomes, a pathway for generation of active caspase-1 and secretion of mature IL-1 41,42 .
The transient cytokine response to ESWTs and the associated cell proliferation may be placed within the context of a stress-induced resilience mechanism. Therefore, the stress associated with a low-level and/or short-lived inflammatory response may be funneled into an organized protective response, like the observed cell proliferation. ESWTs have been applied as a successful therapy in heart failure and cardiovascular diseases in humans 43 –45 . Moreover, in vivo experiments carried out in rats in the presence of LPS or ESWTs resulted in post-infarct cardiac tissue regeneration 46,47 .
To demonstrate that the mechanism, by which cells convert mechanical signals into biochemical responses, may involve HSP recruitment, we analyzed the gene expression of specific HSPs. HSPs, also known as chaperones or stress-induced proteins, have a crucial role during proteins folding/unfolding, assembly of multiprotein complexes, and transport/sorting of proteins into correct subcellular compartments 17,48,49 .
In our experiments, HSP60 and HSP90 were upregulated after 4 h of ESWT treatment. It was recently shown that HSP60 plays a regulatory role during proinflammatory activation via TLR4–p38 MAPK axis 50 , as well as HSP90 17 . These findings again could be strictly associated with previous observations concerning the involvement of ESWTs in the Erk1/2 and p38 MAPK activation pathways 2,20 and with the modulatory action of ESWTs on the plasticity of TLRs 5 . On the other hand, HSP70 did not show any significant change during ESWT treatment, being probably not a target of the ESWT effect. Overall, the ability of ESWTs to trigger similar increase in HSP gene expression as LPS may be viewed as the induction of a protective arm from oxidative stress, apoptosis, and cell death during an inflammatory response. In the case of ESWTs, the increase in HSP transcription coupled with the short-term increase in cytokine expression may prove successful in activating a resilient protective mechanism. Such acquired resilience may on the contrary largely fail when HSP induction occurs within a persistent inflammatory response, as in the case of LPS.
Further understanding of the coupling of transient inflammatory and cytoprotective responses by ESWTs may help taking a glimpse into the complex patterning through which physical energies support tissue regeneration.
Footnotes
Author Contributions
VB, SC, SC, and SS designed and performed the experiments, VB, SC, and MM writing—original draft preparation, MM, PD, SC, FB, and CV writing—review and editing, CV and HR supervision.
Acknowledgments
We thank Adelheid Weidinger for her valuable support in ROS analysis.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
