Abstract
The use of allogeneic hematopoietic stem cell transplantation (HSCT) is recommended during the first complete remission of acute myeloid leukemia (AML) and high-risk myelodysplastic syndrome (MDS). However, only 30% of these cases have fully matched sibling donors (MSDs). Alternatively, matched unrelated donors (MUDs) and haploidentical (haplo) donors from first-degree relatives increase the access to transplantation, with some reported differences in outcomes. The current systematic review and meta-analysis was conducted with the aim of summarizing the results of those studies to compare the efficacy and toxicity of MSD-HSCT and MUD-HSCT versus haplo-HSCT for patients with AML or MDS. Articles published before September 15, 2018, were individually searched for in two databases (MEDLINE and EMBASE) by two investigators. The effect estimates and 95% confidence intervals (CIs) from each eligible study were combined using the Mantel–Haenszel method. A total of 14 studies met the eligibility criteria and were included in the meta-analysis. The overall survival rates were not significantly different between the groups, with pooled odds ratios of the chance of surviving at the end of the study when comparing haplo-HSCT to MSD-HSCT and comparing haplo-HSCT to MUD-HSCT of 0.85 (95% CI: 0.70 to 1.04; I 2 = 0%) and 1.12 (95% CI: 0.89 to 1.41; I 2 = 33%), respectively. The pooled analyses of other outcomes also showed comparable results, except for the higher grade 2 to 4 acute graft-versus-host disease (GvHD) for patients who received haplo-HSCT than those who received MSD-HSCT, and the better GvHD-free, relapse-free survival and the lower chronic GvHD than the patients in the MUD-HSCT group. These observations suggest that haplo-HSCT is a reasonable alternative with comparable efficacy if MSD-HSCT and MUD-HSCT cannot be performed. Nonetheless, the primary studies included in this meta-analysis were observational in nature, and randomized-controlled trials are still needed to confirm the efficacy of haplo-HSCT.
Introduction
Because of the relatively poor survival outcomes of intermediate- and high-risk acute myeloid leukemia (AML) and high-risk myelodysplastic syndrome (MDS) patients, hematopoietic stem cell transplantation (HSCT) during first complete remission (CR1) is the first option for those aiming for a cure 1 –7 . A matched sibling donor (MSD) is the best option for HSCT due to the lower rates of nonrelapse mortality (NRM) and graft-versus-host disease (GvHD), which contribute to a higher rate of long-term survival. However, only 30% of patients have full MSDs 8 . Although a matched unrelated donor (MUD) is a good alternative, the higher expense and the long waiting duration involved in identifying matched donors are common obstacles 9 .
Haploidentical (haplo) HSCT is a novel technique that employs stem cell transplantation from first-degree relatives that have at least 5/10 to 8/10 matched human leukocyte antigen (HLA). This approach may help to increase the availability of first-degree relatives, although studies have shown that a higher rate of GvHD and graft rejection as well as a slower immune reconstitution are its major disadvantages 10,11 . However, after the development of the two main methods, the Baltimore protocol (which applies post-transplantation cyclophosphamide to destroy alloreactive T cells) 12,13 and the Beijing method (which uses granulocyte colony-stimulating factor prime bone marrow plus peripheral blood stem cells [PBSC] combined with a myeloablative conditioning [MAC] regimen and anti-thymocyte globulin to enhance rapid engraftment without increasing GvHD) 14,15 , the aforementioned unfavorable events appear to occur less frequently. In fact, several comparative studies have suggested that haplo-HSCT has comparable outcomes to MSD-HSCT and MUD-HSCT 16,17 . The current systematic review and meta-analysis was conducted with the aim of summarizing the study results in order to compare the efficacy and toxicity of MSD-HSCT and MUD-HSCT versus haplo-HSCT for patients with AML and MDS.
Materials and Methods
Data Sources and Searches
Articles published before September 15, 2018, were identified in two databases (MEDLINE and EMBASE) by two investigators (CK and WO). The search terms consisted of the terms for haploidentical transplantation, AML, and MDS (provided as supplemental data S1). References of the eligible articles and some review articles were also manually examined to identify additional eligible studies. This process was conducted independently by the two investigators. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses Statement, which was used as a guideline for this meta-analysis, is provided as supplemental data S2 18 .
Selection Criteria and Data Extraction
The inclusion criteria for this meta-analysis were as follows: (1) eligible studies must be randomized-controlled studies or cohort studies (either prospective or retrospective) that compared the efficacy of haplo-HSCT to any other stem cell sources for HSCT in AML or MDS; and (2) the studies must report the primary outcome of interest, which was the overall survival (OS) rate and/or leukemia-free survival (LFS) after HSCT. The secondary outcomes of interest (NRM, cumulative incidence of relapse [CIR], GvHD-free/relapse-free survival [GRFS], grade 2 to 4 acute GvHD, and chronic GvHD) were also extracted from the included studies for additional analyses, but they were not part of the inclusion criteria. This process was also independently performed by the two investigators. If different decisions regarding the eligibility were made, the studies in question were jointly discussed with a third investigator (PU), who served as a tiebreaker.
Data on the baseline characteristics of the included studies, along with the primary and secondary outcomes of interest, were extracted by the two investigators. This process was also conducted independently by the investigators, using a standard study record form. The extracted data were cross-checked to ensure their accuracy.
Outcome Definitions
The time between the stem cell infusion and the time of death or last follow-up was used for the calculation of the OS rate, while the time used for the calculation of the event-free survival rate was defined as the time interval from the stem cell infusion to the date of relapse, secondary malignancies, any infections after discharge from the bone marrow transplant unit, or death from any causes. LFS was defined as the time interval from the stem cell infusion to the date of relapse or death from any causes. Relapse was estimated in those patients who achieved CR using a cumulative incidence function with respect to competing risks. The NRM rate included all causes of death except death from relapsed disease. GRFS was defined as patients with grade 2 to 4 acute GvHD, chronic GvHD requiring treatment, relapsed disease, or death.
Quality Assessment
The included randomized-controlled studies were assessed for their quality using the Jadad Quality Assessment Scale 19 . The Newcastle–Ottawa Scale was used to evaluate the quality of the included nonrandomized studies 20 .
Statistical Analysis
The effect estimates and 95% confidence intervals (CIs) from each eligible study were combined using the Mantel–Haenszel method 21 . Cochran’s Q was calculated, and statistical heterogeneity among the included studies was estimated using the I 2 statistic. The four levels of heterogeneity were based on the value of I 2: (1) insignificant heterogeneity (I 2-value of 0% to 25%); (2) low heterogeneity (I 2-value of 26% to 50%); (3) moderate heterogeneity (I 2-value of 51% to 75%); and (4) high heterogeneity (I 2-value of 76% to 100%) 22 . Due to the high likelihood of between-study heterogeneity, a random-effects model was utilized rather than a fixed-effects model. Funnel plots were created to assess for publication bias. Statistical significance was determined to be P < 0.05. All statistical analyses were performed using Review Manager 5.3 software from the Cochrane Collaboration (London, UK).
Results
The aforementioned search strategy identified 1,646 potentially relevant articles (283 articles from MEDLINE and 1,363 from EMBASE). After exclusion of 261 duplicated articles, the titles and abstracts of the remaining 1,385 articles were reviewed. Of those, 1,348 were excluded for any one of the following reasons: (1) ineligible type of article (case report, review, meta-analysis, comments, and editorial); (2) the study was not on patients with AML or AML and MDS; (3) the study did not have a haplo-HSCT group versus an MSD-HSCT or MUD-HSCT group; and (4) the present study’s primary outcome of interest was not reported. The remaining 37 articles underwent a full-length article review, and 23 were excluded at this point due to the same reasons as the first round. Finally, 14 studies (1 prospective cohort study and 13 retrospective cohort studies) met the eligibility criteria and were included in the meta-analysis 15,23 –35 . Figure 1 demonstrates the literature review and selection process.

The literature review and selection process.
Baseline Patient Characteristics
A total of 9,800 patients were included (1,106 patients with haplo-HSCT, 3,109 with MSD-HSCT, and 5,585 with MUD-HSCT), with a slight male predominance (around 1.2 to 1.4 as many males as females across the studies) and ages between 2 and 76 y 15,23,25 –29,31,33 –35 . Majority of patients included in this cohort had AML (99.4%). Most studies categorized the AML disease into favorable, intermediate, and high risk by cytogenetic data, in accordance with the guidelines and recommendations of the National Comprehensive Cancer Network (NCCN) 15,23,26,28,31 , UK Medical Research Council 27,30 , the European LeukemiaNet, or the Southwest Oncology Group study 24,30,33,34 . It should be noted that Ciurea et al. used the cytogenetic risk as recommended by the NCCN, but ≥4 complex chromosomes were used to define the high-risk category 29 . Two studies defined the risk according to the NCCN guideline by using both cytogenetic and molecular abnormalities 26,28 . The other 62 patients were high-risk MDS.
The majority of patients in all three groups were in CR1 and had intermediate- to high-risk cytogenetics. In all, 78% of patients in the MSD-HSCT group and 88% in the MUD-HSCT group received PBSC, while 78% in the haplo-HSCT group received bone marrow plus PBSC. The characteristics of the studies that compared MSD-HSCT to haplo-HSCT are described in Table 1, whereas the characteristics of the studies that compared MUD-HSCT to haplo-HSCT are presented in Table 2. Supplemental Tables S1 and S2 detail the conditioning regimen and GvHD prophylaxis of the studies that compared MSD-HSCT to haplo-HSCT, and of the studies that compared MUD-HSCT to haplo-HSCT, respectively.
Characteristics and Participants of Studies that Compared Haplo-HSCT to MSD-HSCT.

AML: acute myeloid leukemia; BM: bone marrow; C: compatibility; CR1: first complete remission; CR2: second complete remission; EBMT: European Society for Blood and Marrow Transplantation; ELN: European Leukemia Net; fav: favorable; F/U: follow-up; Haplo: haploidentical; HCTCI: hematopoietic cell transplantation-specific comorbidity index; HSCT: hematopoietic stem cell transplantation: inter: intermediate; MAC: myeloablative conditioning; MSD: matched sibling donor; NA: not applicable; NCCN: The National Comprehensive Cancer Network; PBSC: peripheral blood stem cells; RIC: reduced intensity conditioning; S: selection; O: outcome.
Characteristics and Participants of Studies that Compared Haplo-HSCT to MUD-HSCT.

AML: acute myeloid leukemia; BM: bone marrow; C: compatibility; CR1: first complete remission; CR2: second complete remission; DRI: disease risk index; EBMT: European Society for Blood and Marrow Transplantation; fav: favorable; F/U: follow-up; Haplo:-haploidentical: HCTCI: hematopoietic cell transplantation-specific comorbidity index; HSCT: hematopoietic stem cell transplantation, inter: intermediate; MAC: myeloablative conditioning; MUD: matched unrelated donor; MDS: myelodysplastic syndrome; NA: not applicable; NCCN: The National Comprehensive Cancer Network; PBSC: peripheral blood stem cells; RIC: reduced intensity conditioning; S: selection; O: outcome.
Comparison of Outcomes of Haplo
-
HSCT Versus MSD
-
HSCT
Long-term outcomes, including OS, LFS, NRM, and CIR, were reported in six studies (a 2-y follow-up in four studies, a 3-y follow-up in three studies, and a 4-y follow-up in one study). The OS rate was not significantly different between the groups, with a pooled odds ratio (OR) of the chance of surviving at the end of the study when comparing haplo-HSCT to MSD-HSCT of 0.85 (95% CI: 0.70 to 1.04; P = 0.11; I 2 = 0%; Fig. 2A) 15,23 –27,35 . Similarly, the chance of achieving LFS was not significantly different between the groups (pooled OR: 0.91; 95% CI: 0.72 to 1.14; P = 0.40; I 2 = 23%; Fig. 2B) 15,23,25 –27,34,35 . The chance of developing NRM was also similar between the two groups (pooled OR: 1.37; 95% CI: 0.88 to 2.12; P = 0.16; I 2 = 62%; Fig. 3A) 15,23 –27,34 . Likewise, CIR was insignificantly lower in the haplo-HSCT group, with a pooled OR for developing at least one relapse of 0.80 (95% CI: 0.64 to 1.00; P = 0.05; I 2 = 0%; Fig. 3B) compared with those who received MSD-HSCT 15,23,25 –27,34,35 . The GRFS was also not different between the two groups (pooled OR: 0.88; 95% CI: 0.66 to 1.17; P = 0.38; I 2 = 0%; Fig. 3C) 25,27 . For the analyses of the adverse effects, the patients who received haplo-HSCT had a significantly higher chance of developing grade 2 to 4 acute GvHD than patients who received MSD-HSCT (pooled OR: 2.32; 95% CI: 1.52 to 3.56; P < 0.0001; I 2 = 67%; Fig. 4A) 15,23 –27,34,35 . The chance of developing chronic GvHD was similar for the two groups (pooled OR: 0.98; 95% CI: 0.53 to 1.82; P = 0.96; I 2 = 86%; Fig. 4B) 15,23 –25,27,34,35 .
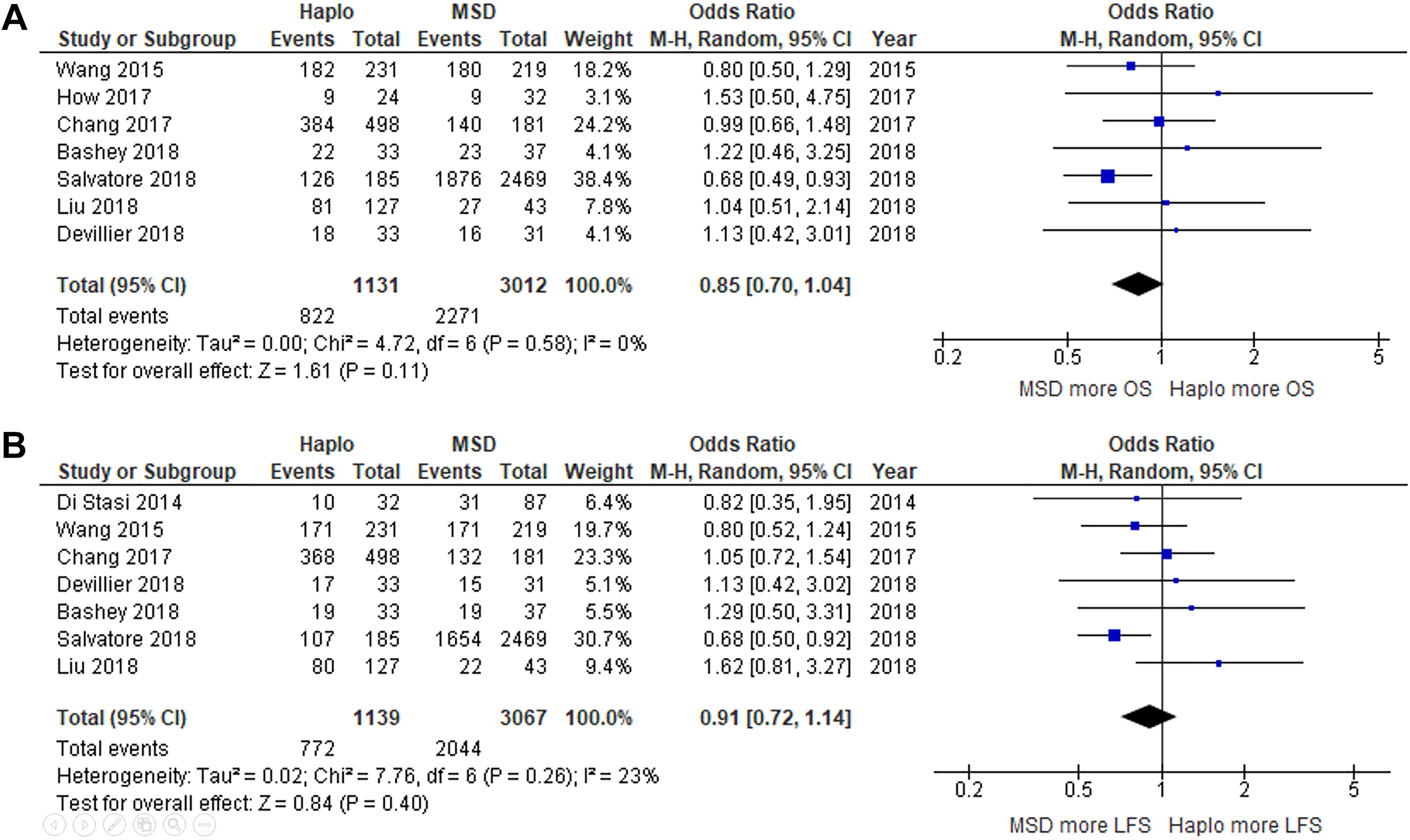
Forest plots of studies that compared (A) overall survival and (B) leukemia-free survival among patients who underwent haplo-HSCT versus MSD-HSCT.

Forest plots of studies that compared (A) nonrelapse mortality; (B) cumulative incidence of relapse and (C) GRFS among patients who underwent haplo-HSCT versus MSD-HSCT.

Forest plots of studies that compared (A) acute GvHD and (B) chronic GvHD among patients who underwent haplo-HSCT versus MSD-HSCT.
Comparative Outcomes Between Haplo-HSCT and MUD-HSCT
Long-term outcomes, including OS, LFS, NRM, and CIR, were reported in eight studies (a 1.5-y follow-up in one study; a 2-y follow-up in five studies; a 3-y follow-up in three studies; and a 5-y follow-up in one study) 24,25,28 –35 . The OS rate was not significantly different between the groups, with a pooled OR comparing the chance of surviving at the end of the study when comparing haplo-HSCT to MUD-HSCT of 1.12 (95% CI: 0.89 to 1.41; P = 0.35; I 2 = 33%; Fig. 5A) 24,25,28 –33,35 . Similarly, the chance of achieving LFS was not significantly different between the groups (pooled OR: 1.21; 95% CI: 0.87 to 1.68; P = 0.26; I 2 = 46%; Fig. 5B) 25,28,31 –35 . The chance of developing NRM was also similar between the two groups (pooled OR: 0.85; 95% CI: 0.55 to 1.32; P = 0.47; I 2 = 65%; Fig. 6A) 24,25,28 –31,33,34 , the same as the chance of developing CIR (pooled OR: 0.91; 95% CI: 0.65 to 1.27; P = 0.59; I 2 = 61%; Fig. 6B) 25,28 –34 . However, the GRFS was significantly better in haplo-HSCT group (pooled OR: 1.40; 95% CI: 1.10 to 1.78; P = 0.007; I 2 = 0%; Fig. 6C) 25,27,32 . The chance of developing grade 2 to 4 acute GvHD was not different between two groups (pooled OR: 0.82; 95% CI: 0.65 to 1.03; P = 0.09; I 2 = 29%; Fig. 7A) 24,25,28 –35 ; in contrast, the patients in the haplo-HSCT group had a significantly lower chance of developing chronic GvHD than those in the MUD-HSCT group (pooled OR: 0.59; 95% CI: 0.48 to 0.73; P < 0.00001; I 2 = 15%; Fig. 7B) 24,25,28 –35 .

Forest plots of studies that compared (A) OS and (B) LFS among patients who underwent haplo-HSCT versus MUD-HSCT.

Forest plots of studies that compared (A) nonrelapse mortality; (B) cumulative incidence of relapse and (C) GRFS among patients who underwent haplo-HSCT versus MUD-HSCT.
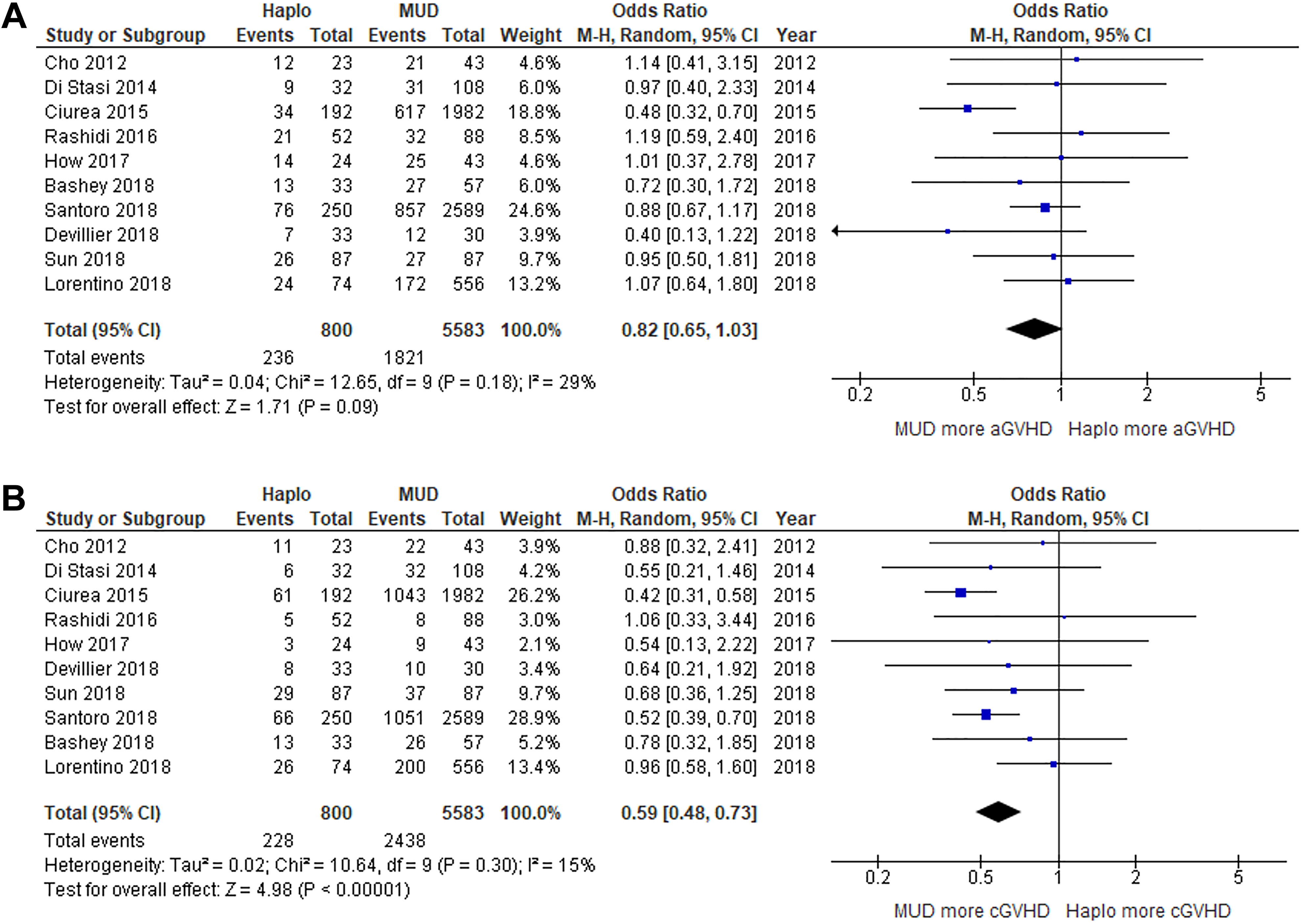
Forest plots of studies that compared (A) acute GvHD and (B) chronic GvHD among patients who underwent haplo-HSCT versus MUD-HSCT.
Sensitivity Analysis
A sensitivity analysis was conducted by excluding the studies by Wang et al.
15
, Chang et al.
23
, and Liu et al.
26
from the full analysis (two studies were excluded at a time). This sensitivity analysis was conducted because of a concern that some patients may have been recruited to more than one of the three studies (the three studies were conducted at the same institute during the overlapping period of time), resulting in double-counting of the same patients. We found that the results of most analyses were not significantly different from the full analysis, either with the exclusion of the study by Wang et al.
15
, Chang et al.
23
, or Liu et al.
26
except: the LFS outcome for the haplo-HSCT versus MSD-HSCT analysis became significant when studies by Chang et al.
23
and Liu et al.
26
were excluded. The new analysis found that the chance of achieving LFS for patients who received haplo-HSCT was significantly lower than for patients who received MSD-HSCT (new pooled OR: 0.76; 95% CI: 0.61 to 0.96; P = 0.02; I
2 = 0%). the CIR outcome for the haplo-HSCT versus MSD-HSCT analysis became insignificant when studies by Chang et al.
23
and Liu et al.
26
were excluded, with the new pooled OR comparing haplo-HSCT to MSD-HSCT of 0.84 (95% CI: 0.64 to 1.11; P = 0.22; I
2 = 0%).
Subgroup Analysis
We selected data, which included only patients with CR prior to HSCT and reanalyzed the primary outcome. The pooled OS and LFS for haplo-HSCT versus MSD-HSCT analysis were not different from the full analysis. Similarly, the pooled OS and LFS for haplo-HSCT versus MUD-HSCT analysis were not different from the full analysis (supplemental data S3A).
Because post-transplant cyclophosphamide is now widely used for GvHD prophylaxis especially in patients with haploidentical transplantation 36 , we selected data that included only patients who received this GvHD prophylaxis protocol and reanalyzed the acute and chronic GvHD outcomes. The pooled acute and chronic GvHD for haplo-HSCT versus MSD-HSCT analysis and haplo-HSCT versus MUD-HSCT were not different from the full analysis (supplemental data S3B).
Evaluation of Publication Bias
Funnel plots of the primary outcome analysis (OS) were constructed for both haplo-HSCT versus MSD-HSCT and haplo-HSCT versus MUD-HSCT comparisons (supplemental data S4). The plots were relatively symmetric, which was not suggestive of the presence of publication bias.
Discussion
Currently, there are three methods of GvHD prophylaxis in haplo-HSCT including post-transplant cyclophosphamide, antithymocyte globulin 36 , and alpha-beta T cell deplete 37 . However, this meta-analysis included only the first two methods. This is the first systematic review and meta-analysis to summarize all available studies that compared the efficacy and complications of haplo-HSCT with the standard HSCT (MSD-HSCT and MUD-HSCT) for patients with AML and MDS. The patient characteristics of MSD versus haplo-HSCT were comparable in terms of their age and sex distribution. The haplo group included more cases of active AML disease and AML in the second CR than the MSD arm. However, the MSD arm had about 10% more intermediate- to high-risk cytogenetic patients than the haplo group. As around 80% of the patients in the haplo arm were from China, which uses the Beijing protocol, a higher proportion of patients in this arm received the MAC regimen and bone marrow plus PBSCs; conversely, a higher proportion in the MSD group received PBSCs.
In the comparison of MUD and haplo-HSCT, patients had comparable ages, sex, distributions of cytogenetic risk, and proportions receiving the MAC and RIC regimens. Similar to the former comparison, the haplo-HSCT group had a higher proportion of AML in the second CR and active diseases. Moreover, the MUD group used PBSCs at a higher ratio than the haplo-HSCT patients did.
The pooled analysis found that the chance of survival at the end of the study, which ranged from 1.5 to 5 y, of patients who underwent haplo-HSCT was not significantly different from patients who underwent either MSD-HSCT or MUD-HSCT. In addition, analyses of the secondary outcomes showed comparable results for haplo-HSCT and conventional HSCTs, except for the higher grade 2 to 4 acute GvHD among patients who received haplo-HSCT than those who received MSD-HSCT, and the lower chronic GvHD and better GRFS than patients in the MUD-HSCT group. These observations suggest that haplo-HSCT is a reasonable alternative with comparable efficacy if MSD-HSCT and MUD-HSCT cannot be performed. The significantly better GRFS in the haplo-HSCT group is probably an unintended benefit of a higher degree of HLA disparity of this approach, which introduces a higher immunogenicity 38 . However, after excluding the three studies that might contain duplicated cases, a sensitivity analysis revealed that patients who received MSD-HSCT had significantly better LFS outcome than those with haplo-HSCT. We therefore believe that MSD-HSCT remains the first donor’s choice for AML and MDS patients.
Nonetheless, the current systematic review and meta-analysis study has several limitations that may jeopardize the validity of the results. The most importation limitation was the observational nature of the included studies, as none were randomized-controlled studies. Therefore, it is very likely that the baseline characteristics of the patients in each group were not perfectly similar, and the observed results could therefore be skewed by the unequal distribution of confounders and/or effect modifiers. The between-study heterogeneity was also high in several pooled analyses, which was likely to be due to the difference in background populations, follow-up protocols, conditioning regimens, GvHD prophylaxis regimens, and stem cell sources across the included studies. In addition, when time to transplant is taken into account, patients who receive haplo-HSCT might have access to treatment more rapidly than those using other donors' sources. That might affect the outcome of transplantation. We found that some of the included studies reported no significant difference in the duration from diagnosis to the date of transplantation between the two groups of patients. However, the rest of the studies did not provide these data; we therefore cannot completely conclude that waiting time prior to HSCT was not significantly different between both groups.
Conclusion
The current study found that the OS and several secondary outcomes of patients with AML and MDS who received haplo-HSCT were not significantly different from MSD-HSCT and MUD-HSCT. This suggests that haplo-HSCT is a reasonable alternative. However, the primary studies included in this meta-analysis were observational in nature. Therefore, randomized-controlled trials are still needed to confirm the efficacy of haplo-HSCT.
Supplemental Material
Supplementary_data_1 - Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis
Supplementary_data_1 for Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis by Chutima Kunacheewa, Patompong Ungprasert, Ployploen Phikulsod, Surapol Issaragrisil and Weerapat Owattanapanich in Cell Transplantation
Supplemental Material
Supplementary_data_2 - Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis
Supplementary_data_2 for Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis by Chutima Kunacheewa, Patompong Ungprasert, Ployploen Phikulsod, Surapol Issaragrisil and Weerapat Owattanapanich in Cell Transplantation
Supplemental Material
Supplementary_data_3 - Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis
Supplementary_data_3 for Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis by Chutima Kunacheewa, Patompong Ungprasert, Ployploen Phikulsod, Surapol Issaragrisil and Weerapat Owattanapanich in Cell Transplantation
Supplemental Material
Supplementary_data_4 - Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis
Supplementary_data_4 for Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis by Chutima Kunacheewa, Patompong Ungprasert, Ployploen Phikulsod, Surapol Issaragrisil and Weerapat Owattanapanich in Cell Transplantation
Supplemental Material
Supplementary_Table_S1 - Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis
Supplementary_Table_S1 for Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis by Chutima Kunacheewa, Patompong Ungprasert, Ployploen Phikulsod, Surapol Issaragrisil and Weerapat Owattanapanich in Cell Transplantation
Supplemental Material
Supplementary_Table_S2 - Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis
Supplementary_Table_S2 for Comparative Efficacy and Clinical Outcomes of Haploidentical Stem Cell Transplantation to Other Stem Sources for Treatment in Acute Myeloid Leukemia and Myelodysplastic Syndrome Patients: A Systematic Review and Meta-Analysis by Chutima Kunacheewa, Patompong Ungprasert, Ployploen Phikulsod, Surapol Issaragrisil and Weerapat Owattanapanich in Cell Transplantation
Footnotes
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval
The need to obtain ethical approval by an institutional board review was waived as this study did not directly involve human subjects.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Not applicable because this study did not directly involve human subjects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
