Abstract
KMT2A rearrangement (KMT2A-r) in patients with acute myeloid leukemia (AML) is associated with poor outcomes; the prognostic factors after allogeneic hematopoietic stem cell transplantation (allo-HSCT) remain unclear. We investigated 364 adults with AML who underwent allo-HSCT between April 2016 and May 2022, and 45 had KMT2A-r among them. Propensity score analysis with 1:1 matching and the nearest neighbor matching method identified 42 patients in KMT2A-r and non–KMT2A-r cohorts, respectively. The 2-year overall survival (OS), relapse-free survival (RFS), cumulative incidence of relapse (CIR), and non-relapsed mortality rates of patients with KMT2A-r (n = 45) were 59.1%, 49.6%, 41.5%, and 8.9%, respectively. Using propensity score matching, the 2-year OS rate of patients with KMT2A-r (n = 42) was lower than that of those without KMT2A-r (n = 42; 56.1% vs 88.1%, P = 0.003). Among patients with KMT2A-r (n = 45), the prognostic advantage was exhibited from transplantation in first complete remission (CR1) and measurable residual disease (MRD) negative, which was reflected in OS, RFS, and CIR (P < 0.001, P < 0.001, and P = 0.002, respectively). Furthermore, patients with AF6 had poorer outcomes than those with AF9, ELL, and other KMT2A-r subtypes (P = 0.032, P = 0.001, and P = 0.001 for OS, RFS, and CIR, respectively). However, no differences were found in the OS, RFS, and CIR between patients with KMT2A-r with and without mutations (all P > 0.05). Univariate and multivariate analyses revealed that achieving CR1 MRD negative before HSCT was a protective factor for OS [hazard ratio (HR) = 0.242, P = 0.007], RFS (HR = 0.350, P = 0.036), and CIR (HR = 0.271, P = 0.021), while AF6 was a risk factor for RFS (HR = 2.985, P = 0.028) and CIR (HR = 4.675, P = 0.004). The prognosis of patients with KMT2A-r AML was poor, particularly those harboring AF6-related translocation; however, it is not associated with the presence of mutations. These patients can benefit from achieving CR1 MRD negative before HSCT.
Introduction
The KMT2A gene (formerly MLL) that encodes “histone-lysine N-methyltransferase 2A” is located at chromosome 11q23 1 . More than 100 different recombinations in KMT2A have been identified to date 2 . The fusion proteins encoded by KMT2A rearrangements (KMT2A-r) recruit chromatin-associated complexes, such as the super elongation complex and histone H3 lysine 79 methyltransferase DOT1L, positively regulate the expression of multiple homeotic (and other) genes, and consequently transform hematopoietic cells into leukemic stem cells3,4. In addition, 5%–10% of patients with acute leukemia have KMT2A-r 5 , characterized by occurring suddenly, progressing rapidly, and having a poor prognosis6,7. In patients with KMT2A-r acute myeloid leukemia (AML), the correlation between translocation partner genes (TPGs) and prognosis remains to be explored8–10, and whether mutations can be considered predictors of outcomes requires additional research11–13.
The role of hematopoietic stem cell transplantation (HSCT) in patients with KMT2A-r AML remains controversial. Some previous studies have reported the benefit of HSCT for patients with KMT2A-r AML14–16. However, other studies suggested that HSCT did not improve the outcomes17,18. Moreover, several studies found that HSCT during first complete remission (CR1) is strongly protective10,19,20, which made patients achieve a 2-year overall survival (OS) rate of approximately twice than that of those transplanted in CR2 21 . However, a few studies have underscored the importance of the expression of KMT2A-r and measurable residual disease (MRD) before HSCT for prognosis.
Therefore, this study aimed to explore the outcomes of allogeneic HSCT (allo-HSCT) in patients with AML who harbor KMT2A-r and the potential factors affecting prognosis. Since many previous studies were conducted in infants or children, this study focused on adults.
Materials and Methods
Study Population
This retrospective study included 364 patients aged ≥18 years with AML (diagnosed per the World Health Organization 2016 classification 22 ) who underwent allo-HSCT between April 2016 and May 2022; 45 of these individuals had KMT2A-r. Data were obtained from the patients’ medical records, including demographic information, laboratory test results, cytogenetic and molecular genetic data, and transplant-related parameters. The presence of chromosome 11q23 abnormalities was evaluated using chromosome banding or fluorescence in situ hybridization. KMT2A and gene mutations were detected using polymerase chain reaction and next-generation sequencing. Detectable KMT2A-r expression at any level (≥0.0001%) was defined as KMT2A-r positive in this study. Flow cytometry–based MRD (FCM-MRD) was assessed using six- to eight-color flow cytometry with a sensitivity of 0.01%. The MRD-positive status was defined as FCM-MRD ≥1% and/or KMT2A-r positive, and the MRD-negative status was defined as FCM-MRD <1% and KMT2A-r negative. Follow-up was completed on March 31, 2023. Informed consent was obtained from all patients according to the Declaration of Helsinki, and the institutional and national ethics committee approved this study.
Allo-HSCT
All patients received allo-HSCT from a human leukocyte antigen (HLA)–matched sibling, haploidentical relative, or unrelated donor. The myeloablative or reduced-intensity regimens were chosen based on the age at HSCT, performance status, comorbidities, and prior treatment strategies. For HLA-matched donors, the myeloablative conditioning (MAC) regimen mainly comprised busulfan (Bu) and cyclophosphamide (Cy; Bu 3.2 mg/kg/day i.v. on days −7 to −4 and Cy 60 mg/kg/day i.v. on days −3 to −2). In addition, rabbit anti-thymocyte globulin (ATG, Thymoglobulin; Genzyme, Cambridge, MA, USA) was administered to patients with matched unrelated donors (4.5−6 mg/kg total dose). For patients with HLA-haploidentical related donors, the MAC regimen included cytarabine (4 g/m2/day i.v. on days −10 to −9), Bu (3.2 mg/kg/day i.v. on days −8 to −6), Cy (60 mg/kg/day i.v. on days −5 to −4), and methyl-N-(2-chloroethyl)-N-cyclohexyl-N-nitrosourea (250 mg/m2 orally on day −3), and rabbit ATG (1.5 mg/kg/day i.v. on days −5 to −2). The main reduced-intensity conditioning regimen was Flu-Bu-ATG (fludarabine 30 mg/m2/day i.v. on days −10 to −5, Bu 3.2 mg/kg/day i.v. on days −6 to −5, and ATG 5 mg/kg/day i.v. on days −4 to −1). Absolute neutrophil count >0.5 × 109/L and platelet count >20 × 109/L without platelet transfusions were defined as neutrophil and platelet recovery, respectively. As previously described, cyclosporin A, short-term methotrexate, and mycophenolate mofetil were applied as prophylactics against graft-versus-host disease 23 .
Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 26 (IBM Corp, Armonk, NY, USA) and R statistical software version 4.2.0 (R Foundation for Statistical Computing, Vienna, Austria). Propensity score matching (PSM) was used to reduce the confounding and bias when comparing the outcomes between patients with and without KMT2A-r in this observational study. Propensity scores were calculated using logistic regression from baseline characteristics of recruited patients, including age at transplantation, white blood cell (WBC) count, disease status at HSCT, conditioning regimen, donor type, donor-patient sex, donor-patient blood group, and the time from diagnosis to HSCT. Propensity score analysis with 1:1 matching and a caliper control of 0.02 was performed using the nearest neighbor matching method. Continuous variables are expressed as medians and ranges and were analyzed using Student’s t-test and the Mann–Whitney U-test as appropriate. Categorical covariates are presented as counts with percentages and were analyzed using Pearson’s chi-square and Fisher’s exact tests. A waterfall plot was constructed to display gene mutations in each patient. The primary endpoints were OS, relapse-free survival (RFS), cumulative incidence of relapse, and non-relapsed mortality (NRM). OS and RFS were estimated using the Kaplan–Meier method, and the log-rank test was used to compare Kaplan–Meier curves. The cumulative incidence of relapse (CIR) and NRM were calculated by accounting for competing risks. Cox proportional hazards regression and competitive risk models were used to identify risk factors associated with OS, RFS, and CIR, and variables with P-values <0.15 on univariate analysis were subjected to the multivariate model. Statistical significance was set at P < 0.05.
Results
Baseline Characteristics of the Study Participants
The demographic characteristics of the 364 patients with AML who underwent allo-HSCT (including 45 with KMT2A-r) are presented in Table 1. The time from diagnosis to HSCT was 1 month shorter in the KMT2A-r group than in the non–KMT2A-r group (P = 0.001), and a less female-to-male donor-recipient combination was observed (P = 0.030). Subsequently, PSM identified 42 patients in each cohort (Table 2). No differences were observed in the basic characteristics between the two groups (all P > 0.05).
Baseline Characteristics of Patients With Acute Myeloid Leukemia.
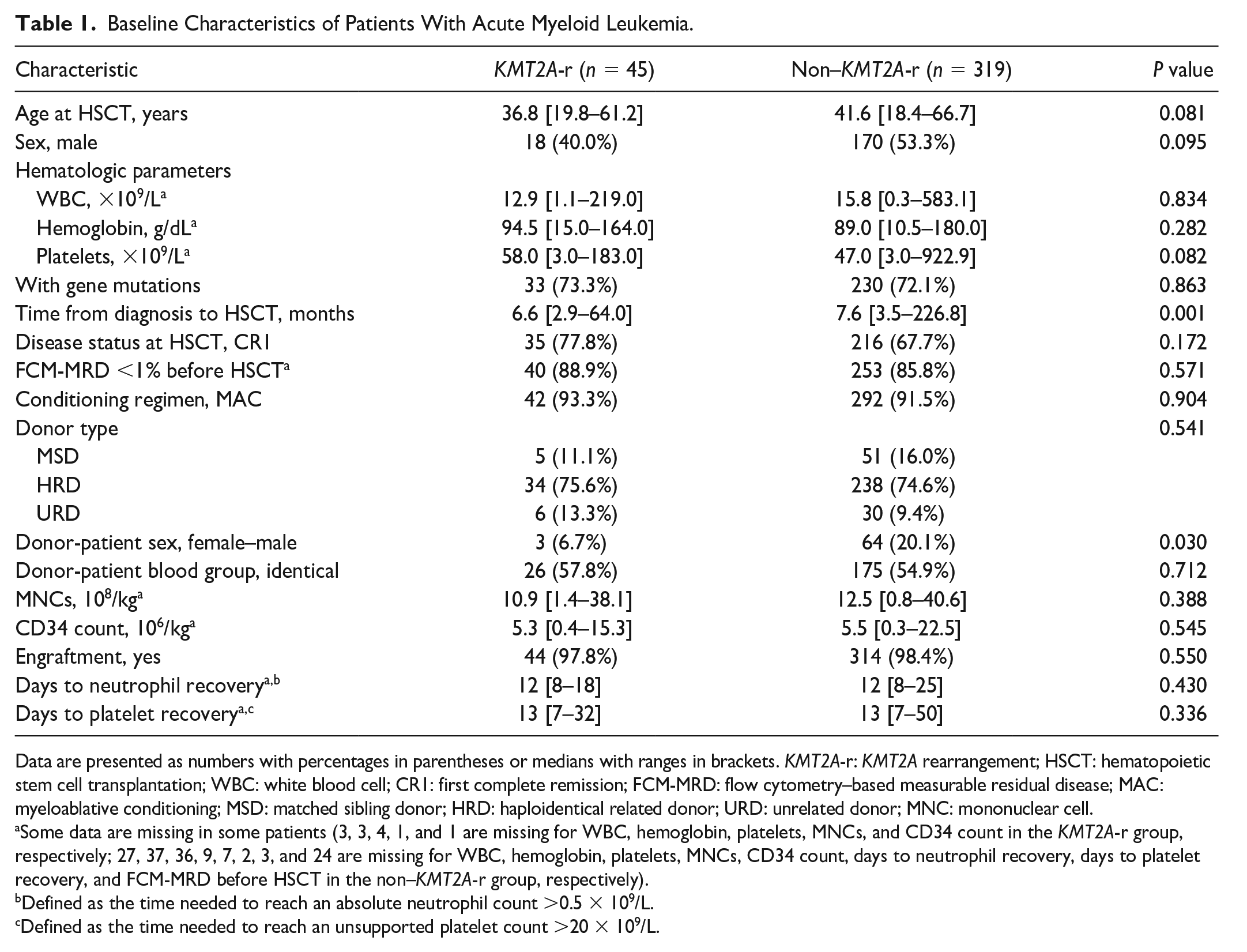
Data are presented as numbers with percentages in parentheses or medians with ranges in brackets. KMT2A-r: KMT2A rearrangement; HSCT: hematopoietic stem cell transplantation; WBC: white blood cell; CR1: first complete remission; FCM-MRD: flow cytometry–based measurable residual disease; MAC: myeloablative conditioning; MSD: matched sibling donor; HRD: haploidentical related donor; URD: unrelated donor; MNC: mononuclear cell.
Some data are missing in some patients (3, 3, 4, 1, and 1 are missing for WBC, hemoglobin, platelets, MNCs, and CD34 count in the KMT2A-r group, respectively; 27, 37, 36, 9, 7, 2, 3, and 24 are missing for WBC, hemoglobin, platelets, MNCs, CD34 count, days to neutrophil recovery, days to platelet recovery, and FCM-MRD before HSCT in the non–KMT2A-r group, respectively).
Defined as the time needed to reach an absolute neutrophil count >0.5 × 109/L.
Defined as the time needed to reach an unsupported platelet count >20 × 109/L.
Characteristics of Patients With Acute Myeloid Leukemia After Propensity Score Matching.
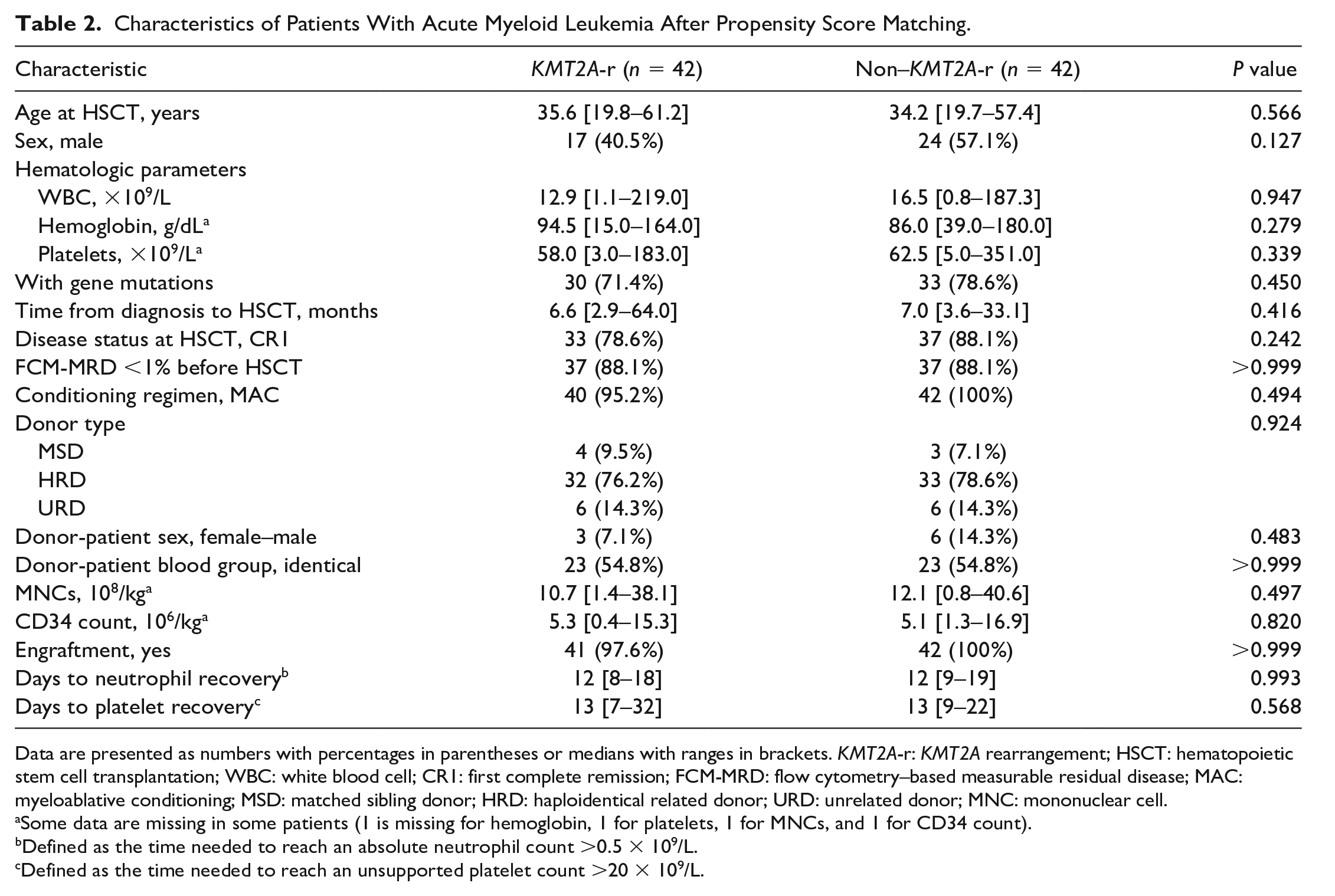
Data are presented as numbers with percentages in parentheses or medians with ranges in brackets. KMT2A-r: KMT2A rearrangement; HSCT: hematopoietic stem cell transplantation; WBC: white blood cell; CR1: first complete remission; FCM-MRD: flow cytometry–based measurable residual disease; MAC: myeloablative conditioning; MSD: matched sibling donor; HRD: haploidentical related donor; URD: unrelated donor; MNC: mononuclear cell.
Some data are missing in some patients (1 is missing for hemoglobin, 1 for platelets, 1 for MNCs, and 1 for CD34 count).
Defined as the time needed to reach an absolute neutrophil count >0.5 × 109/L.
Defined as the time needed to reach an unsupported platelet count >20 × 109/L.
Twenty-seven (60.0%) patients in the KMT2A-r group were alive at the last follow-up (March 31, 2023), among whom 24 were in CR1, and 22 were with MRD negative before HSCT. The distribution of KMT2A-r subtypes according to TPGs is described in Fig. 1A as follows: 13, 11, 7, 4, 3, and 3 patients with AF6, AF9, ELL, AF10, PTD, and AF1Q gene fusions, respectively. The remaining four patients were identified with other KMT2A-r subtypes, including two, one, and one with AF4, dup-MLL, and SEPT9, respectively. In addition, 33 (73.3%) patients had at least one somatic mutation, including the components of the RAS pathway [NRAS (n = 6), KRAS (n = 6), and BRAF (n = 1)], WT1 (n = 13), FLT3 (n = 6), TET2 (n = 6), and others (Fig. 1B). The median number of mutations carried per individual was 2 (range, 0–6).
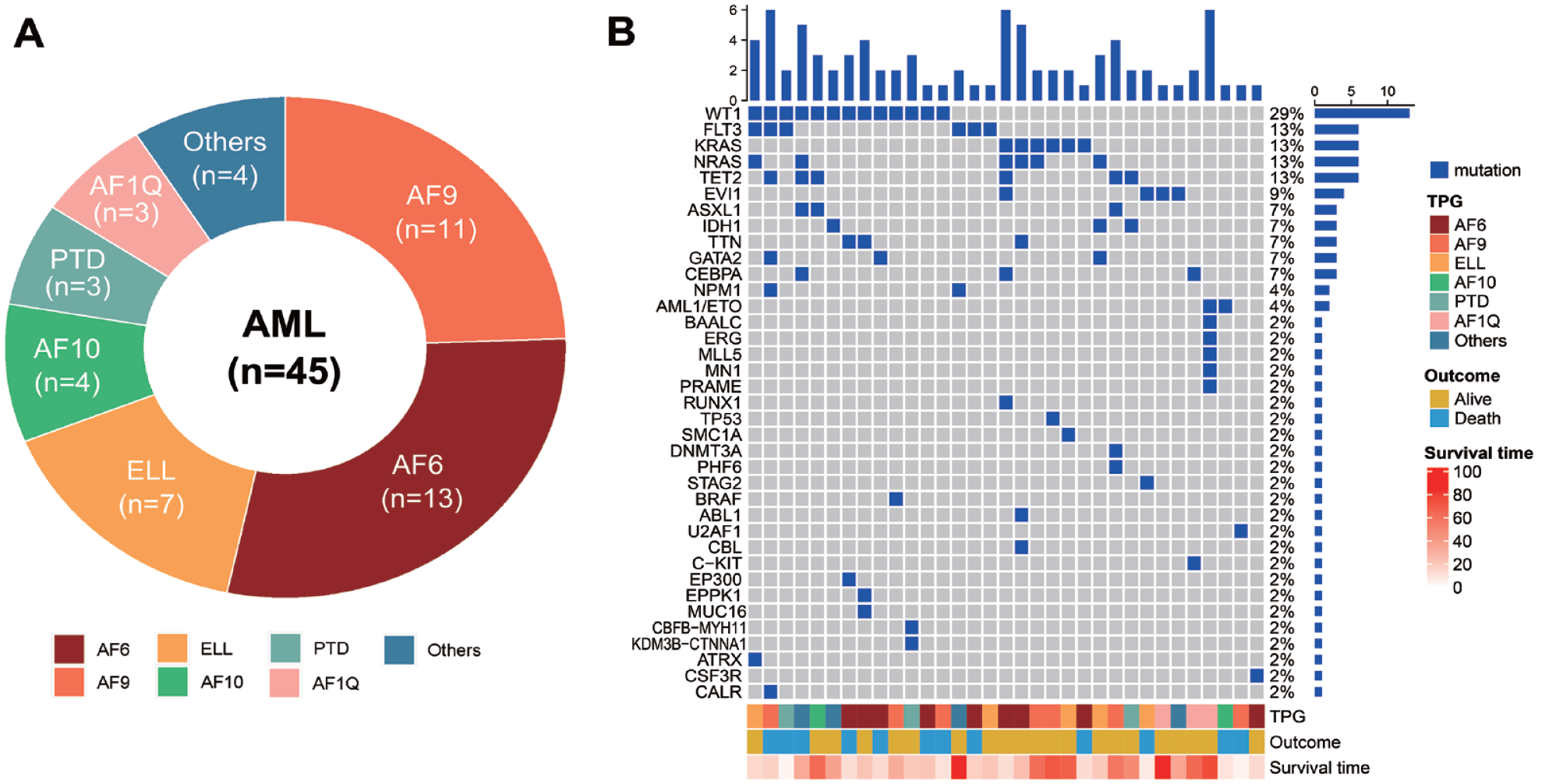
TPGs and the mutational landscape of the investigated patients with AML exhibiting KMT2A rearrangement. (A) TPG frequencies. (B) Landscape profile of somatic gene mutations; each column represents each sample. The name of the gene mutation is shown on the left, and its frequency is displayed on the right. The bar plot above the legend exhibits the number of mutational burdens, while the patients’ clinical features are summarized at the bottom. AML: acute myeloid leukemia; TPG: translocation partner gene.
Prognosis of Patients With KMT2A-r AML Compared to That of Those Without KMT2A-r AML
As shown in Table 1, 44 (97.8%) patients with KMT2A-r achieved engraftment. The median day to neutrophil and platelet recovery was 12 (range, 8–18) and 13 (range, 7–32) days, respectively. Their 2-year OS, RFS, CIR, and NRM rates were 59.1%, 49.6%, 41.5%, and 8.9%, respectively (Fig. 2).
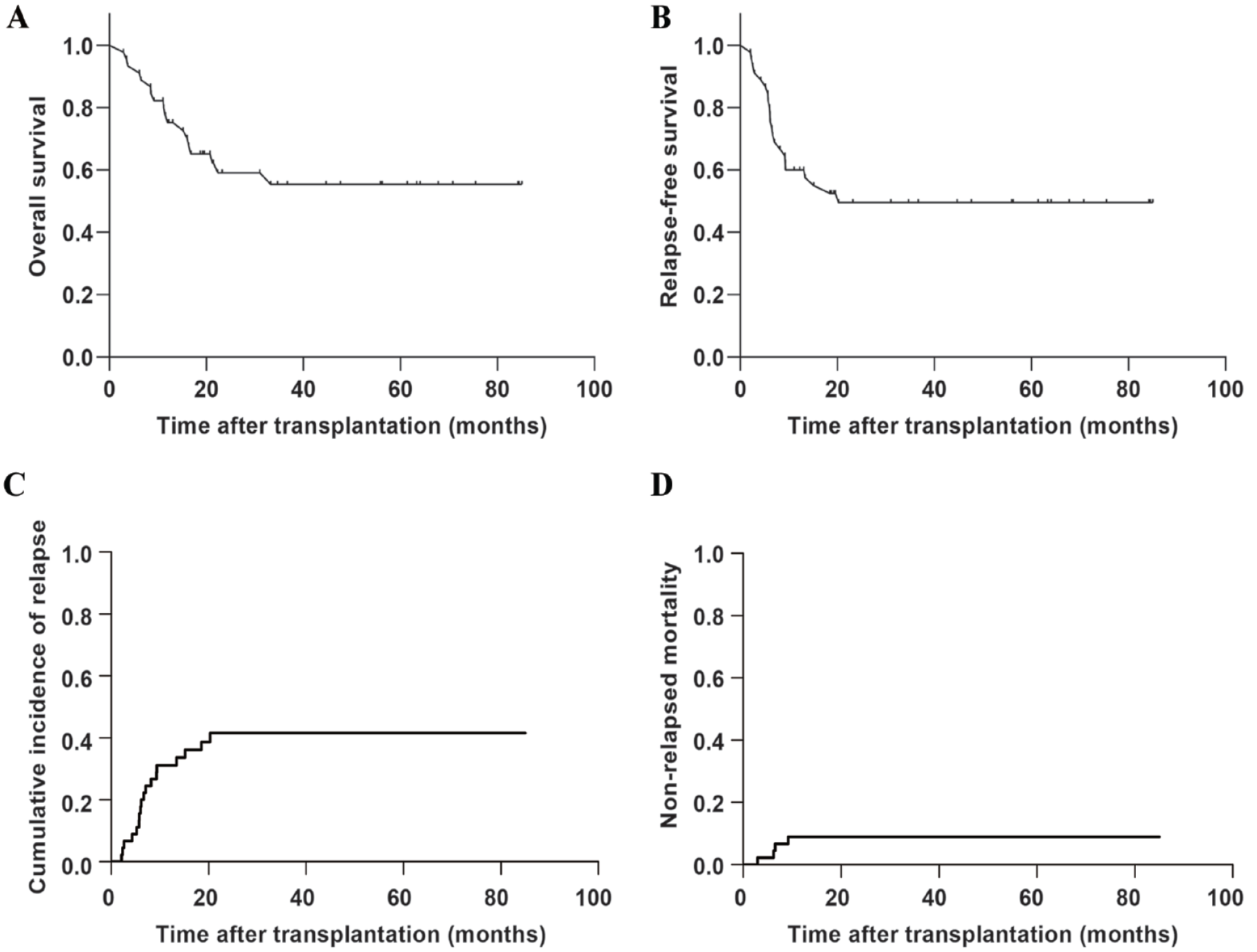
Outcomes of patients with acute myeloid leukemia exhibiting KMT2A rearrangement after hematopoietic stem cell transplantation. (A) Overall survival. (B) Relapse-free survival. (C) Cumulative incidence of relapse. (D) Non-relapsed mortality.
We compared the prognosis of patients without KMT2A-r using PSM. The rates and time of neutrophil or platelet engraftments were similar between the two groups (all P > 0.05; Table 2). The 2-year OS rate of patients with KMT2A-r was markedly lower than that of those with non–KMT2A-r (56.1% vs 88.1%, P = 0.003; Fig. 3A). Moreover, lower 2-year RFS and higher 2-year CIR rates of patients with KMT2A-r were observed (48.2% vs 80.9%, P = 0.002 and 42.3% vs 12.0%, P = 0.002; respectively), while no difference was found in NRM (9.5% vs 7.1%, P = 0.941; Supplementary materials: Table S1). Even in patients who underwent allo-HSCT during CR1 or those with FCM-MRD <1% before transplantation, individuals with KMT2A-r still displayed a lower 2-year OS rate (63.2% vs 89.2%, P = 0.035 and 61.7% vs 91.9%, P = 0.008; respectively; Fig. 3B, C). Great distinctions were also found for RFS and CIR (all P < 0.05; Table S1). Next, patients who achieved CR1 and FCM-MRD <1% before HSCT were designated for further exploration, and those in the KMT2A-r group were classified into two categories according to the KMT2A-r status before HSCT [KMT2A-r (+), n = 5; KMT2A-r (−), n = 26]. A difference was observed in the 2-year OS rate among patients with KMT2A-r (+), those with KMT2A-r (−), and the non–KMT2A-r group (60.0% vs 71.5% vs 93.9%, P = 0.026; Fig. 3D), although no significant differences were found in RFS or CIR among these three groups, which might be attributed to the small sample size of the KMT2A-r (+) group (all P > 0.05; Table S1).
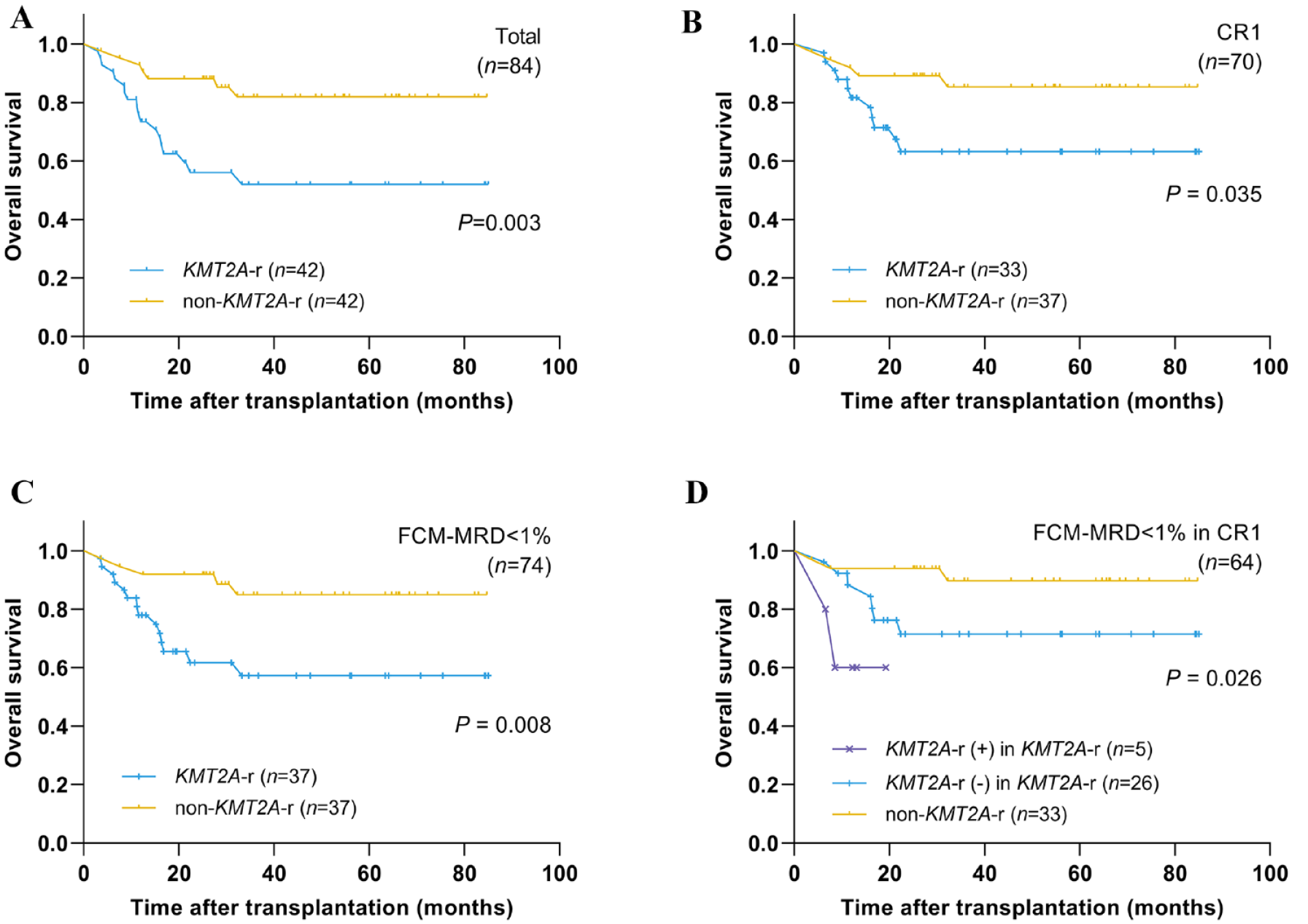
Comparison of overall survival after HSCT between patients with and without KMT2A rearrangement. (A) In all patients. (B) In patients who received HSCT while in CR1. (C) In patients who were detected with FCM-MRD <1% at HSCT. (D) In patients with FCM-MRD <1% and CR1 at HSCT. HSCT: hematopoietic stem cell transplantation; CR1: first complete remission; FCM-MRD: flow cytometry–based measurable residual disease.
Outcomes of Different Subgroups of Patients With KMT2A-r
We investigated the prognosis of patients with KMT2A-r AML according to disease status, MRD status before HSCT, KMT2A-r subtypes, and the presence of mutations.
First, patients with KMT2A-r were categorized into two subgroups as follows: those who underwent allo-HSCT while in CR1 MRD negative and those transplanted while in CR1 MRD positive, or those with more advanced disease (transplanted while in CR2 or subsequent CR, during non-remission, or while relapsed, regardless of MRD status). The prognostic advantage from transplantation while in CR1 MRD negative was reflected in the 2-year OS, RFS, and CIR rates (73.7% vs 28.8%, P < 0.001; 66.9% vs 19.6%, P < 0.001; and 26.0% vs 68.6%, P = 0.002, respectively; Fig. 4A–C).
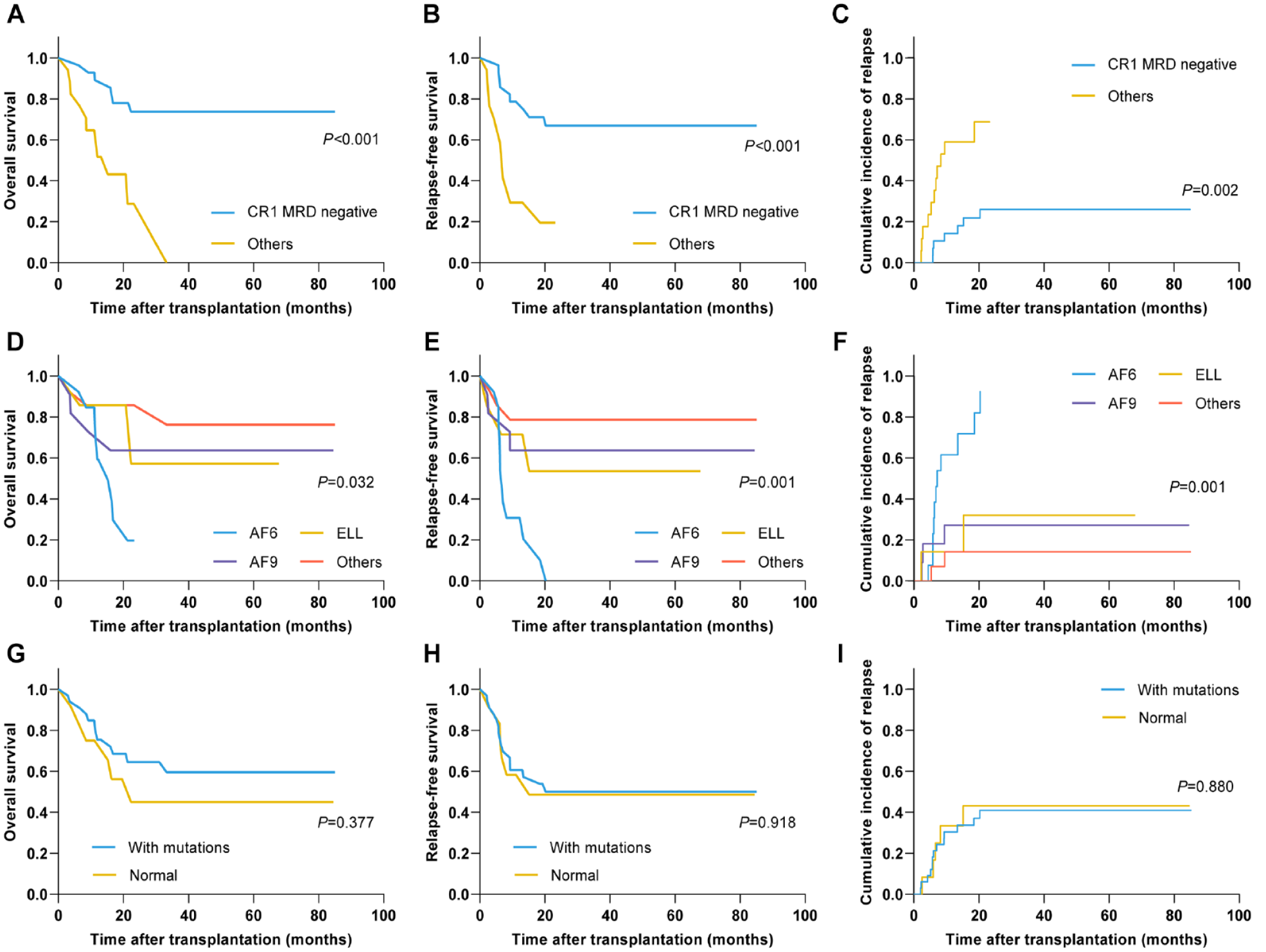
Prognosis of different subgroups in patients with acute myeloid leukemia exhibiting KMT2A rearrangement after transplantation. (A) OS, (B) RFS, and (C) CIR based on disease status and MRD status at transplantation. (D) OS, (E) RFS, and (F) CIR based on KMT2A rearrangement subtypes. (G) OS, (H) RFS, and (I) CIR based on the presence of mutations. The MRD-negative status was defined as FCM-MRD <1% and KMT2A rearrangement negative. OS: overall survival; RFS: relapse‑free survival; CIR: cumulative incidence of relapse; MRD: measurable residual disease; FCM-MRD: flow cytometry–based measurable residual disease; CR1: first complete remission.
When subgrouping was based on different KMT2A-r subtypes, only AF6 was significantly associated with unfavorable OS (P = 0.004, Supplementary materials: Fig. S1). Furthermore, we compared the outcomes of patients with AF6, AF9, ELL, and other KMT2A-r subtypes, and stark differences were observed (2-year OS rate: 19.7% vs 63.6% vs 57.1% vs 85.7%, P = 0.032; 2-year RFS rate: 0.0% vs 63.6% vs 53.6% vs 78.6%, P = 0.001; 2-year CIR rate: 82.1% vs 27.3% vs 32.1% vs 14.3%, P = 0.001; respectively; Fig. 4D–F). However, the OS, RFS, and CIR were not statistically different regardless of any harbored somatic gene mutations (all P > 0.05; Fig. 4G–I).
Prognostic Predictors for Patients With KMT2A-r AML After Allo-HSCT
The factors that may affect the OS, RFS, and CIR of patients with KMT2A-r after allo-HSCT were analyzed using univariate analyses, including achieving CR1 MRD negative before HSCT, age at transplantation, sex, WBC, platelets and hemoglobin at diagnosis, KMT2A-r subtypes (AF6 vs others), the presence of mutations, time from diagnosis to HSCT, conditioning regimen (MAC vs reduced-intensity conditioning), donor type (haploidentical related donor vs HLA-matched donor), donor-patient sex (female–male vs others), identical donor-patient blood group, mononuclear cells, and CD34 count. Variables with P < 0.15 in univariate analyses were subjected to multivariate analysis, which are summarized in Table 3.
Univariate and Multivariate Analyses of Prognostic Factors in Patients With KMT2A-r After Undergoing Allogeneic Hematopoietic Stem Cell Transplantation.
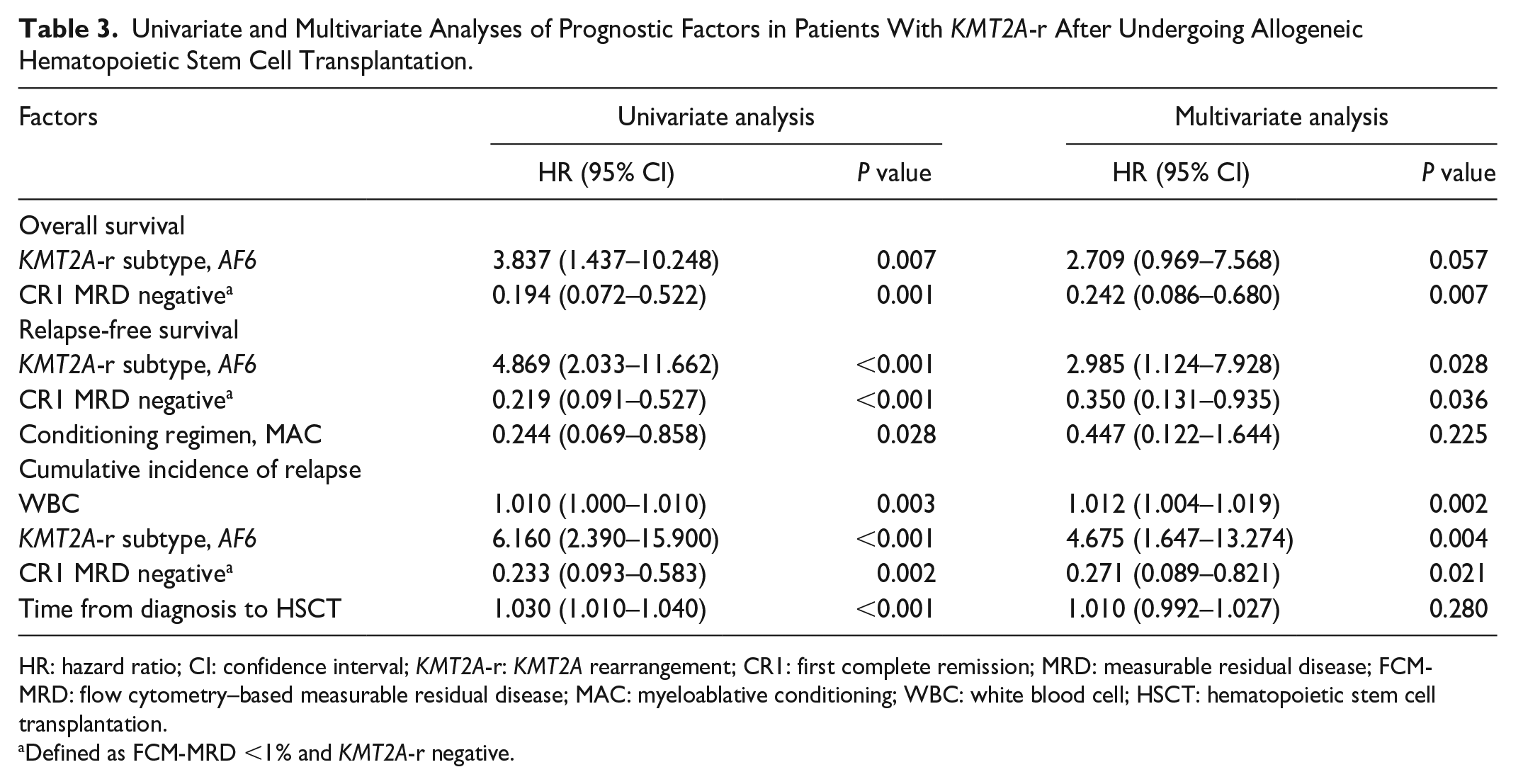
HR: hazard ratio; CI: confidence interval; KMT2A-r: KMT2A rearrangement; CR1: first complete remission; MRD: measurable residual disease; FCM-MRD: flow cytometry–based measurable residual disease; MAC: myeloablative conditioning; WBC: white blood cell; HSCT: hematopoietic stem cell transplantation.
Defined as FCM-MRD <1% and KMT2A-r negative.
Notably, achieving CR1 MRD negative before HSCT predicted higher OS [hazard ratio (HR) = 0.242, 95% confidence interval (CI) 0.086–0.680, P = 0.007], improved RFS (HR = 0.350, 95% CI 0.131–0.935, P = 0.036), and lower CIR (HR = 0.271, 95% CI 0.089–0.821, P = 0.021). In contrast, patients harboring AF6 experienced lower RFS (HR = 2.985, 95% CI 1.124–7.928, P = 0.028) and higher risk of relapse (HR = 4.675, 95% CI 1.647–13.274, P = 0.004), and their OS tended worse despite no statistical significance (HR = 2.709, 95% CI 0.969–7.568, P = 0.057). Moreover, at first diagnosis, WBC had a slight impact on CIR (HR = 1.012, 95% CI 1.004–1.019, P = 0.002).
Discussion
Patients with AML carrying KMT2A-r are deemed high risk17,24, making them more prone to relapse 25 . Similarly, among our study population, patients with KMT2A-r also had relatively inferior prognoses. Using PSM, their 2-year OS and RFS rates were approximately 30% lower, and the 2-year CIR rate was approximately 30% higher than that in those without KMT2A-r. In addition, as a curative treatment for AML, the importance of HSCT appears self-evident. A study that recruited 184 adults with KMT2A-r AML observed a clear advantage in patients undergoing transplantation with a 2-year OS and RFS rates of 58.12% and 52.97%, respectively, compared to those who did not 15 . We observed an analogous result in adult patients with KMT2A-r AML undergoing allo-HSCT, with the 2-year OS and RFS rates of 59.1% and 49.6%, respectively.
Given the high risk of relapse in patients with KMT2A-r and the late toxicities of HSCT, as 36.3% of post-transplantation events are reportedly HSCT related 26 , it is essential to explore strategies for improving the prognosis after HSCT.
Disease burden plays a crucial role in prognosis. The protective effect of achieving CR1 during transplantation to survival in patients with AML and 11q23 abnormalities has been confirmed 19 . However, a recent study by the International Berlin–Frankfurt–Münster Study Group on children with KMT2A-r AML demonstrated that HSCT during CR1 did not improve OS; in contrast, FCM-MRD negative at the end of induction 2 was strongly protective 27 . Moreover, MRD status at transplantation has a clear influence on survival in patients with AML. For instance, Jentzsch et al. found a distinct outcome separation between patients with AML with MRD positive and negative before transplantation 28 . These increase hematologists’ incentive to achieve MRD negative before HSCT rather than CR1. Furthermore, recently, there has been an increased emphasis on monitoring KMT2A-r expression during treatment29,30. It was reported that the 3-year CIR rate of patients with KMT2A-r detectable at HSCT was approximately 40% higher, and the 3-year RFS and OS rates were approximately 50% lower than those of individuals without KMT2A-r expression at HSCT 31 . In addition, the detection of PTD, a specific KMT2A-r subtype, was found to be more sensitive than FCM-MRD since it became positive before FCM-MRD in relapsed patients 32 . Bai et al. combined KMT2A-r expression and FCM-MRD to evaluate MRD status and found the adverse impact of MRD positive on prognosis 14 . These findings imply that the validity of predicting prognosis can be improved by monitoring FCM and KMT2A-r simultaneously.
In our study, patients with KMT2A-r had poorer outcomes than those without KMT2A-r, regardless of whether they received allo-HSCT in CR1 or achieved FCM-MRD <1% during HSCT. Regarding patients who met CR1 and FCM-MRD <1% simultaneously, worse OS was observed in those detected with KMT2A-r (+) before HSCT than in those with KMT2A-r (−) and the non–KMT2A-r group, which implies the importance of combining FCM-MRD and KMT2A-r to define MRD. Subsequently, we considered disease and MRD statuses before transplantation when investigating the prognostic factors of adult patients with KMT2A-r AML. As expected, CR1 MRD negative during HSCT had a substantial protective impact on OS, RFS, and CIR. In addition, since we focused on KMT2A-r status during transplantation, more studies aimed at different monitoring timepoints after HSCT or the monitoring frequency and threshold should be conducted.
The potential relationship between KMT2A-r subtypes and prognosis has attracted some attention. TPG-based heterogeneity has been reported among patients with KMT2A-r AML 18 . Particularly, AF6-related translocation t(6;11) was associated with dismal outcomes33,34. An international retrospective study that recruited 756 children with KMT2A-r AML showed that those with AF6 had a substantially lower 5-year event-free survival rate than those with AF1Q (11 ± 5% vs 92 ± 5%) 18 . We also found the adverse outcomes of patients harboring AF6 with 2-year OS, RFS, and CIR rates of 19.7%, 0%, and 82.1%, respectively, and AF6 was an independent risk factor for relapse and shorter RFS based on Cox regression analyses. One explanation is that AF6-mediated myeloid transformation relies on dimerization, such as other dimer-type KMT2A fusion proteins 35 , and the enhanced RAS signaling caused by endogenous AF6 sequestration within the nucleus confers additional oncogenic effect on AF6 36 .
Moreover, several investigations have provided insights into the mutational landscape of KMT2A-r AML. The mutational burden is low in these patients, with any such mutations most commonly found in RAS signaling pathway components37–39. Similarly, RAS mutations were comparatively common in our cohort. These mutations are frequently subclonal, possibly promote proliferation, and are lost upon relapse38,40. Given the association between KRAS mutations and poor OS in patients with KMT2A-r AML 11 , RAS signaling pathway might be a novel potential therapeutic target, particularly in those with AF6 36 . Another research reported that mutations do not significantly influence prognosis, as evidence of poor OS is found only in patients with two or more mutations 10 . In our study, because of the similar survival curves of patients with and without mutations as well as the nonsignificant HR on Cox regression analysis, we did not confirm a clear impact of the mutation presence on the outcomes of patients with KMT2A-r AML. Nevertheless, our study comprised a relatively small number of patients and did not examine the influence of different gene mutations; hence, studies with larger sample sizes are needed to draw more accurate conclusions.
This study had some limitations. First, our data were analyzed retrospectively. Second, our sample size was relatively small. Therefore, additional large-cohort prospective studies are needed to validate our findings. Moreover, since all recruited patients in our study underwent allo-HSCT, the role of autologous transplantation in the prognosis of patients with KMT2A-r AML has not been analyzed; thus, future studies are needed to explore this aspect.
In conclusion, the prognosis of patients with KMT2A-r AML was poor, particularly for those harboring AF6-related translocation; however, the presence of gene mutations had no significant effect on the prognosis. CR1 MRD negative before HSCT was the most important prognostic factor. Therefore, CR1 and MRD negative should be achieved as far as possible before HSCT for patients with KMT2A-r AML.
Supplemental Material
sj-docx-1-cll-10.1177_09636897231225821 – Supplemental material for Outcomes of Allogeneic Hematopoietic Stem Cell Transplantation in Adult Patients With Acute Myeloid Leukemia Harboring KMT2A Rearrangement and Its Prognostic Factors
Supplemental material, sj-docx-1-cll-10.1177_09636897231225821 for Outcomes of Allogeneic Hematopoietic Stem Cell Transplantation in Adult Patients With Acute Myeloid Leukemia Harboring KMT2A Rearrangement and Its Prognostic Factors by Bingqian Jiang, Yanmin Zhao, Yi Luo, Jian Yu, Yi Chen, Baodong Ye, Huarui Fu, Xiaoyu Lai, Lizhen Liu, Yishan Ye, Weiyan Zheng, Jie Sun, Jingsong He, Yi Zhao, Guoqing Wei, Zhen Cai, He Huang and Jimin Shi in Cell Transplantation
Footnotes
Acknowledgements
The authors are grateful to all the clinicians and patients who contributed to this study.
Author Contributions
JBQ analyzed data, interpreted data, and drafted the manuscript; ZYM and LY analyzed and interpreted data; YJ, CY, YBD, FHR, LXY, LLZ, and YYS collected data; ZWY, SJ, HJS, ZY, WGQ, and CZ organized data; SJM and HH designed the research and supervised the manuscript. All authors read and approved the final manuscript.
Availability of Data and Material
All data used and analyzed are available from the corresponding author upon reasonable request.
Ethical Approval
This study was approved by the ethics committee of the First Affiliated Hospital of Zhejiang University School of Medicine and the First Affiliated Hospital of Wenzhou Medical University.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Informed consent was obtained from all patients included in the study according to the Declaration of Helsinki.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Key Program: No. 82130003; General Program: No. 82070179, No. 81970158).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
