Abstract
We investigated the safety of using umbilical cord-lining stem cells for liver regeneration and tested a novel method for stem cell delivery. Stem cells are known by their ability to repair damaged tissues and have the potential to be used as regenerative therapies. The umbilical cord’s outer lining membrane is known to be a promising source of multipotent stem cells and can be cultivated in an epithelial cell growth medium to produce cell populations which possess the properties of both epithelial cells and embryonic stem cells—termed cord-lining epithelial cells (CLEC). Hepatocytes are epithelial cells of the liver and their proliferation upon injury is the main mechanism in restoring the liver. Earlier studies conducted showed CLEC can be differentiated into functioning hepatocyte-like cells (HLC) and can survive in immunologically competent specimens. In this study, we chose a porcine model to investigate CLEC as a treatment modality for liver failure. We selected 16 immune competent Yorkshire-Dutch Landrace pigs, with a mean weight of 40.5 kg, for this study. We performed a 50% hepatectomy to simulate the liver insufficient disease model. After the surgery, four pigs were transplanted with a saline scaffold while seven pigs were transplanted with a HLC scaffold. Five pigs died on the surgical table and were omitted from the study analysis. This study addressed the safety of transplanting human CLEC in a large animal model. The transplant interfaces were evaluated and no signs of cellular rejection were observed in both groups.
Keywords
Introduction
Liver disease is a huge burden to public health globally. In 2010, mortality due to liver diseases accounted for over 2 million deaths, approximately 4% of all deaths in the world 1 .
Liver transplant remains the definitive cure for many liver diseases. Patients with chronic liver disease as well as severe acute liver decompensation can all benefit from liver transplant 2 . Approximately 25,000 liver transplants are performed worldwide every year 2,3 , with 1-year survival at 85–90% 3,4 and the 10-year survival rate at approximately 50% 4 . However, a scarcity of liver donors means selection is strict, and many patients die while waiting for a transplant 2 . Approximately 13,000–15,000 candidates are on the liver transplant waitlist in the United States; only 6,000 patients receive a liver transplant and 2,000 patients die waiting 2,5,6 . Demand for liver grafts far outweighs supply.
To address this problem, many attempts have been made to regenerate the damaged liver. Transplantation of stem cells gained importance at the turn of this decade 7,8 . However, the use of some stem cells, like embryonic stem cells, has provided a huge ethical dilemma. The use of other alternative human stem cell sources has therefore gained momentum 7,9 –11 . The stem cells used in this research were derived from the umbilical cord lining, which is regarded as biological waste post-delivery. This bypasses any ethical dilemma or debate 9 . Umbilical cord tissue is gaining attention as a novel source of multipotent stem cells because it is easily obtainable, ethically acceptable, and the cells are immunologically naïve 8,9,11 . Furthermore the immunosuppressive nature of cord-lining epithelial cells (CLEC) has been documented in recent studies 9,11,12 . Various studies have demonstrated that CLEC are strongly immunosuppressive in vitro and in vivo 9,11,12 .
We evaluated the toxicity of HLC in a xeno-transplanted model. The viability of hepatocyte-like cells (HLC) and signs of cellular rejection along the transplant interface in hosts after 3 weeks were investigated. The overall health of these animals was observed over 3 weeks.
We also evaluated the effectiveness of novel cell-delivery method, by placing a collagen scaffold directly on resected liver surface. Histological study of the collagen scaffold, before and after transplantation, was carried out.
Materials and Methods
Source of CLEC
Human umbilical cords were collected with informed consent of the mothers after normal deliveries. In total, 81 umbilical cords were collected from two academic hospitals in Singapore (KK Women’s and Children’s Hospital and National University Hospital, Singapore) with full Institutional Review Board approvals. The length of cord collected ranged from 20 cm to 30 cm. Stem cell isolation, cultivation and expansion of CLEC or mesenchymal stem cells were performed using our patented protocols and proprietary stem cell media, with a success rate of 100% for separation of desired cells. These cells went under full characterization and cryopreservation for research use.
The Wharton’s jelly and blood vessels were separated from the umbilical cord amniotic membrane by dissection. The isolated amniotic membrane of the umbilical cord, or cord lining, was cut into small pieces and explanted onto tissue culture dishes with proprietary medium, PTTe1, made up of Medium 171 (Cascade Biologics, Carlsbad CA, USA) supplemented with 2.5% v/v FBS (Lonza, Walkersville, MD, USA), 50 µg/mL insulin-like growth factor-1, 50 µg/mL platelet-derived growth factor-BB, 5 µg/mL transforming growth factor-b1, and 5 mg/mL insulin (R&D Systems, Minneapolis, MN, USA).
Cells were cultured initially in 100 mm dishes in the early stage of umbilical cord tissue culture, and 150 mm dishes were used subsequently for sub-culturing and stem cell expansion. Primary tissue culture was done in CO2 incubators.
Low passages (1 or 2) of CLEC were selected for single-cell cloning. CLEC colonies were expanded after single-cell cloning. For expansion of single-cell cloning colonies, cell scraping techniques were used, for all others, trypsinization was performed. For immunocytochemistry staining experiments, 3,000 cells were seeded into each well of 96-well plate. Colonies formed from single cells were monitored for 12 days and tested for hepatic stem cell markers. A small portion of cells was tested using immunostaining assays for AFP (R&D Systems, Minneapolis, MN, USA), CK19 (Santa Cruz Biotechnology, Dallas, TX, USA), CK18 (Santa Cruz Biotechnology, Dallas, TX, USA) and Albumin (R&D Systems, Minneapolis, MN, USA) (Fig. 1a–h). Cell colonies with high expression of hepatic stem cell markers of 60% and above were chosen for hepatogenesis.

Characteristic expression of hepatic-like stem cell markers in CLECs. Colonies formed from low passaged single cells were tested for hepatic-like stem cell markers by immunocytochemistry assays. CLEC-derived hepatic-like cells were incubated with primary antibody (AFP, CK19, CK18 and albumin) and pan-specific secondary antibody followed with DAB + substrate chromogen system. Oxidized DAB formed brown precipitate in positive cells. CLECs that were incubated without primary antibody served as negative controls. DAB-staining showed strong positive expression of AFP (1a), CK18 (1c), CK19 (1e) and albumin (1 g) and no expression in negative controls of AFP (1b), CK18 (1d), CK19 (1f) and albumin (1 h). These results unequivocally demonstrate that CLEC possess considerable expressions of hepatic stem cell markers. Cell colonies with above 60% expression were selected for hepato-genetic experiment; all images in 40× magnification.
Hepatogenesis
The chosen cell colonies were plated at a density of 5,000 cells/cm2 and cultured at 37°C in an atmosphere of 95% air and 5% carbon dioxide. Upon reaching confluency at passage 3 or 4, the PTTe1 medium was replaced with hepatocyte culture medium (HCM™ Bulletkit®, Lonza, Singapore). The CLEC colonies were cultured in HCM for 14 days, followed by hepatocyte maintenance medium (HMM™, Lonza, Singapore) for another 14 days to differentiate into HLC. The media were refreshed every 2 to 3 days.
Liver Insufficient Porcine Models
In this study, 50% left hepatectomy was performed on adult pigs to create a liver insufficient animal model. In total, 16 Yorkshire-Dutch Landrace pigs, with a mean weight of 40.5 kg on day of surgery, were initially selected for this study. All pigs were tranquilized by intramuscular injection of ketamine (1 mg/kg) (Ceva Animal Health Pte. Ltd, NSW, Australia). General anesthesia was subsequently induced using 5% isoflurane (Piramal Critical Care Inc, Bethlehem, PA, USA) and maintained at 3% isoflurane (Piramal Critical Care Inc, Bethlehem, PA, USA) until the entire surgery was completed.
The pigs were placed in supine position and surgical tools were inserted into the body via laparoscopic ports (see Fig. 2). The laparoscopic approach standardizes the amount of trauma experienced by the pigs and reduces blood loss. The open stage of the experiment was done through a mini-laparotomy upper midline incision. The liver was mobilized by division of peritoneal reflections to the diaphragm and division of the falciform ligament to completely mobilize the left liver lobes. The parenchyma was divided using electrical power device resecting the left two liver lobes completely, one lobe at a time.

From top left. (a) Setup for hepatectomy on pig and insertion of various laparoscopic ports and endoscope on ADD2 pig. (b) Application of collagen scaffolds onto resected liver surface. Collagen scaffolds were inserted through the same opening the two left liver lobes were removed from. (c) Prior to application, the collagen scaffold was soaked for 5 min in either 0.9% NaCl (control) or 30 mL HLC suspension. (d) HLC suspension in a 20 mL syringe.
We tested a novel stem cell-delivery method. After surgical dissection, two pieces of 50 × 50 mm collagen scaffold (TissuFleece, Baxter, Compton, Newbury, UK) were applied directly onto the cut liver surface (Fig. 2). Prior to application, the collagen scaffold (TissuFleece, Baxter, Compton, Newbury, UK) was soaked for 5 min in either: 30 mL normal saline solution (0.9% NaCl) (Baxter Healthcare, Manilla, Philippines); pigs receiving this scaffold were treated as the control arm. 30 mL HLC suspension (see Fig. 2). On the day of the surgery, the HLC colonies were gently detached from the tissue culture surface with cell scrapers (Corning, NY, USA) and transported to the operating room. It was estimated that 10 million HLCs were transferred into solution. Pigs receiving this scaffold were treated as the study arm.
The remaining solution was extracted with a syringe (Becton, Dickson and Company, NJ, USA) and dispensed directly over the scaffolds, after they were positioned on the resected surface. Abdominal wall closure was performed with 1-0 PDS continuous suture observing Jenkins Rule. Skin was closed with 4-0 Monocryl (B Braun Surgical S.A., Barcelona, Spain) in a subcuticular suture. A portal catheter was inserted permanently into the external jugular vein to allow frequent blood collection. Anesthesia was discontinued and considered completion of surgery. All procedures followed aseptic precautions and were done under the approval of Institutional Animal Care and Use Committee (IACUC, Singapore).
Post-Operative Care
The pigs were housed in individual pens in NUS Comparative Medicine (CM) Vivarium, maintained at 25°C and ambient humidity, over a post-operative observation period of 21 days. For the first 7 days, the pig was given analgesic buprenorphine (Jurox Pty Ltd, Rutherford NSW, Australia) and antibiotics enrofloxacin (Bayer Co. Sdn Bhd, Selangor, Malaysia), amoxicillin (Sandoz, Kundl, Austria), clavulanic acid (Novem Healthcare Pte Ltd, Singapore) and carprofen (Laboratorios Pfizer Ltd, Sao Paulo, Brazil) to reduce localized pain and inflammation. The portal catheter was flushed with 1% heparin (Hanlim Pharm Co. Ltd, Gyeong-Gi, Korea) w/normal saline solution every other day for subsequent weeks to prevent deep vein thrombosis.
Biochemical Tests
A volume of 10 mL of venous blood was drawn from the pigs immediately after surgery (post-op), day 1 after surgery (POD1), day 2 after surgery (POD2), day 4 after surgery (POD4), day 6 after surgery (POD6), day 8 after surgery (POD8), day 15 after surgery (POD15) and day 21 after surgery (POD21) before euthanasia.
The blood was sent to National University Hospital and examined for prothrombin time (PT), international normalized ratio (INR), albumin (ALB), total bilirubin (TBIL), direct bilirubin (DB), indirect bilirubin (IB), aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP) and lactate dehydrogenase (LDH). The data were subsequently collected and tabulated respectively.
Computed Tomography and Liver Volumetric Study
Computed tomography (CT) scans of the liver were performed in NUS CM Imaging Facility, before the hepatectomy (pre-operatively), post-operative, POD8 and POD15. The pigs were under general anesthesia throughout the scans. Triple-phase liver CT protocol was performed.
Images from contrast-enhanced portal venous phase were extracted and sent for data processing. The liver volume was determined and tabulated. On POD21, after the pig was euthanized via pentobarbital (150 mg/kg) (Jurox Pty Ltd, NSW, Australia), the liver (without the gallbladder) was harvested and its volume measured via the water displacement method. The measured volume was increased 13% to account for blood perfusion compared with CT-determined measurements. The results were tabulated.
Histological Procedures
On both POD8 and POD15, CT-guided core needle biopsy was performed, after the completion of the CT scan. The procedure was carried out by CM veterinarian, using a 16G 15 cm-long percutaneous biopsy instrument (Temno Evolution®, Carefusion). The pig was placed in left lateral recumbency and the biopsy needle was inserted between the right lower ribs; 40mm3 of core liver tissue was removed from the pig. CT images were referenced to prevent injury to major vessels. The core tissue was then sent for fixation in 10% neutral buffered formalin solution.
On POD21, after water displacement volume measurement, two samples were obtained from the site where the collagen scaffolds were placed and at a peripheral site away from the collagen scaffold. These samples were removed from the rest of the liver via a #10 scalpel blade (Heinz Herenz, Hamburg, Germany) and immersed in 10% neutral buffered formalin solution for fixation (Hanlim Pharm Co. Ltd, Gyeong-Gi, Korea).
Tissue fixation was carried out for a minimum of 48 h before sending to A*STAR Advanced Molecular Pathology Laboratory for automated tissue processing. This included processing fixed tissue into paraffin-embedded blocks, sectioning blocks into 5 µm thick samples and placing samples onto glass slides. For every block, one slide of H&E-stained and one slide of Masson’s Trichome-stained slide were requested.
Anti-Human Nuclei Staining
Anti-nuclei human antibody (Merck Millipore, Temecula, CA, USA) was used as the primary antibody. Immunohistochemical (IHC) staining was carried out on selected paraffin sections. Micrographs were taken and analyzed.
Results
Animal Profiles and Outcomes—no Significant Difference in Liver Regeneration and Recovery
Overall survival rate was 56.25% at 3 weeks (9 out of 16 pigs). Due to the complexity in performing the 50% hepatectomy procedure on porcine models, only 11 out of 16 pigs (68.8%) were able to be transplanted with the respective collagen scaffolds. The remaining pigs (5 out of 16) suffered on-table mortality due to uncontrolled intraoperative blood loss. These five pigs were omitted from the analysis of this study. Table 1 shows the profile and outcome of the 11 pigs that were transplanted with collagen scaffolds.
Overview of Animals’ Details and Post-Operative Survival.

Both sexes were equally represented in both treatment groups, and the weight difference between the two groups on the day of the surgery was 0.8 kg (2%). One pig from the control group and the study group suffered from ascites and were euthanized on post-operative day 16 and day 15, respectively. Postmortem examination performed by veterinarians attributed the cause to be intestinal adhesion and obstruction. All other pigs were euthanized at study end point of post-operative day 21.
A sample size of 60 porcine models is required with 30 in each group to demonstrate statistical differences between the two different groups. Due to limited resources we were unable to perform surgery on 60 porcine models. This initial study of limited animal number serves as a bridging study to future study with larger sample size.
Liver Function Test Results
Objective measures like the ratio between the concentrations of the enzymes AST and ALT, also known as the DeRitis ratio, PT, ALP and liver volumetry were used to measure liver regeneration and recovery.
The DeRitis ratio reflects hepatocellular injury and was noted to be higher in the study group compared with the control group in the first few days post-operation (POD1 to 4) (Fig. 3). ALP levels, which reflect biliary damage, were also noted to be higher in the study group compared with the control group up to the first week post-operation. (Fig. 3) These can be explained as expected post-op changes. There was no significant difference due to our small sample size. Further studies will need to be performed with larger sample sizes to investigate if there is significant biliary or hepatocellular damage.
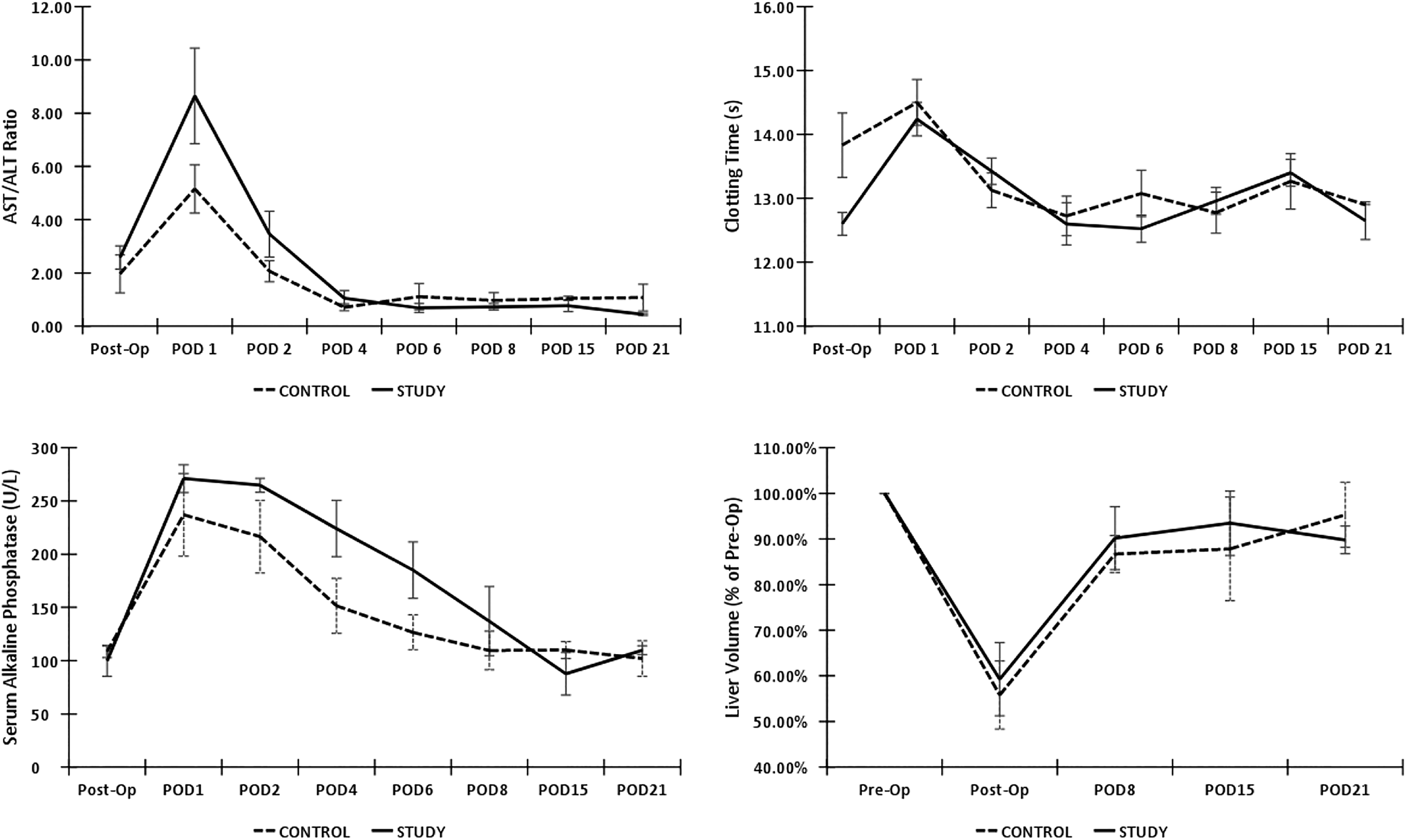
Comparison of average DeRitis ratio, prothrombin clotting time, ALP and liver volumetry for 21 days after hepatectomy between control and study group. Error bars represent one standard error. There were no statistically significant differences between the control and study groups in the aspects of cellular damage (De Ritis ratio), synthetic function (clotting time), biliary function (ALP) and mass regeneration of the liver (liver volumetry).
Absorption of Collagen Scaffold into the Transplant Interface Observed in both Control and Study Groups
Before the application of the collagen scaffold onto the resected sites, a 10 mm × 10 mm sample of scaffold after soaking with HLC was removed for histological characterization. To understand how the collagen scaffold is being absorbed by the porcine liver after 21 days post-operation, we performed Masson’s Trichome staining on the scaffold–liver or transplant interface (Fig. 4). The stain highlighted collagen fibers and imparted a dark blue color to the collagen scaffold fibers. Fig. 4, 5 show the absorption of the collagen scaffold into the transplant interface of a pig from the control group. Fig. 4 also shows the absorption of the collagen scaffold into the transplant interface of a pig from the study group. This staining also allowed for localization of human antigens during IHC staining (Fig. 6).

H&E staining of collagen scaffold with HLC; clockwise from top left. (a) Hematoxylin and eosin (H&E) staining of collagen scaffold with HLC. Scaffold was soaked in HLC for 5 min. This figure shows the wavy thread-like structure of the collagen scaffold structure. Observed using 4× objective lens. (b) H&E staining of HLC displaying epithelial cell characteristics. Scaffold was soaked in HLC for 5 min. This figure demonstrates the epithelial cell characteristics of HLC. The squamous structure of HLC is clearly visible, located at the surface of the scaffold. Observed using 4× objective lens. (c) H&E staining of HLC before implantation. Scaffold was soaked in HLC for 5 min. The squamous structure of HLC is clearly visible, located at the surface of the scaffold. Observed using 10× objective lens. (d) H&E staining on inner layers of scaffold. Scaffold was soaked in HLC for 5 min. No HLC was observed in the inner layers of scaffold, suggesting that HLC only penetrated the collagen scaffold superficially. Observed using 4× objective lens. (e) Masson’s Trichome staining of collagen scaffold after POD21 in control pig. Section was removed from the scaffold–liver interface in ADD2. It was observed that the scaffold was being absorbed into the fibrous tissue of the liver as part of the regenerative process. Observed using 4× objective lens. (f) Masson’s Trichome staining of scaffold after POD21. Section was removed from the scaffold–liver interface in ADD4. It was also observed that the scaffold was being absorbed into the fibrous tissue of the liver as part of the regenerative process. Observed using 10× objective lens.

H&E staining of transplant interfaces; clockwise from top left. (a) H&E staining of transplant interface after POD21 in control. Section was removed from the scaffold–liver interface in ADD2. It was observed that the scaffold (extreme right) was being absorbed into the fibrous tissue of the liver as part of the regenerative process. No signs of specific or significant reactions were detected. Observed using 4× objective lens. (b) H&E-stained lobules adjacent transplant interface of control. Section was removed from the scaffold–liver interface in ADD2. Healthy lobular structures were seen next to the regenerating fibrous tissue. No signs of fatty changes or cellular rejections were detected. Observed using 4× objective lens. (c) H&E staining of transplant interface after POD21. Section was removed from the scaffold–liver interface in ADD7. It was observed that the scaffold (extreme right) was being absorbed into the fibrous tissue of the liver as part of the regenerative process. No signs of specific or significant reactions were detected. Observed using 4× objective lens. (d) H&E-stained lobules adjacent transplant interface. Section was removed from the scaffold–liver interface in ADD7. Healthy lobular structures were seen next to the regenerating fibrous tissue. No signs of fatty changes or cellular rejections were detected. Observed using 4× objective lens. (e) H&E-stained liver biopsy on POD1. Tissue was removed from ADD6’s right liver lobe via core tissue biopsy. Healthy lobular structures are visible and no signs of fatty changes or cellular rejections were detected. Observed using 4× objective lens. (f) H&E-stained liver sample on POD21. Tissue was removed from ADD6’s excised right liver lobe after euthanasia. Healthy lobular structures are visible and no signs of fatty changes or cellular rejections were detected. Observed using 4× objective lens. (g, h) H&E-stained pre-op liver sample. Tissue was removed from ADD6’s excised left liver lobe after hepatectomy procedure. Post-operative histological samples’ conditions were found comparable to that of pre-op sample, suggesting the insignificant toxicity of transplanted HLC. Observed using 4× objective lens.

IHC staining of collagen scaffold with HLC; clockwise from top left. (a) IHC staining of collagen scaffold with HLC. Scaffold was soaked in HLC for 5 min. HLC is clearly visible with its stained cytoplasm and nucleus. Observed using 4× objective lens. (b) IHC staining of collagen scaffold with HLC (magnified). HLC cytoplasm is stained dark with black nuclear staining. Observed using 40× objective lens. (c) IHC staining of collagen scaffold after POD21. Dark cytoplasm is observed at collagen scaffold. Observed using 20× objective lens. (d) IHC staining of HLC after POD21. Dark cytoplasm is observed at collagen scaffold and black nuclear staining. Longitudinal cellular nuclear structure suggests evidence of HLC viability after 3 weeks post-transplantation. Observed using 40× objective lens.
No Significant Cellular Rejection Noted at Transplant Interface
The interface between the collagen scaffold and resection margin surface was also studied in both groups. Interfaces of both groups, 3 weeks post-operative, showed minimal and non-specific reactions. No signs of fatty changes or significant cellular rejection were observed adjacent the interfaces of both groups. Fig. 4 shows the interface from a control pig and a study pig. Healthy liver lobules were observed next to the regenerating fibrous tissue in control and study groups (Fig. 4).
Healthy Peripheral Liver Histology with no Cellular Rejection to Transplanted HLC
To investigate the overall condition of the liver, histological samples peripheral to the transplant interface were obtained and studied. Core tissue biopsy was performed on post-operative week 1 and week 2 to observe if any reactions were taking place due to the transplanted HLC. Week 3 samples were retrieved from the excised liver after euthanasia. Healthy lobular structures were seen and no signs of fatty changes or cellular rejections were detected (Fig. 5).
HLC Seen to Survive 3 weeks Post-Transplantation on Human Antigen Staining
It was observed that CLEC possess immunosuppressive properties and are able to survive in immunologically competent hosts. Such characteristics are still unclear for HLC. Staining for human antigens allowed the team to check the viability of HLC 3 weeks after transplantation in a xeno-transplanted host. Bright-field IHC was first performed on the scaffold with HLC for characterization (Fig. 6). Human antigen staining was then carried out on paraffin sections from the study group. Analysis was undertaken with Masson’s Trichome staining slides for scaffold localization. Human antigen staining also showed evidence of surviving HLC 3 weeks post-transplantation (Fig. 6). Longitudinal cellular nuclear structure suggests evidence of HLC viability after 3 weeks post-transplantation (Fig. 6).
Discussion
This study was done to prove the non-toxicity of transplanting HLC into an immunologically competent xeno-transplanted host, and to address the safety aspect of transplanting human CLEC in a large animal model. These data will be useful before moving on to human trials/therapy.
In creating our hepatic insufficiency model, we were limited by ethical and anatomical considerations 13 . The hepatectomy model cannot be too extensive, hence we opted for a 50% hepatectomy model.
The first objective of this study was to evaluate the toxicity of transplanting HLC into a xeno-transplanted host. From the liver function tests no statistically significant differences were found between the control and study groups in the aspects of cellular damage, synthetic function, biliary function and mass regeneration of the liver (Fig. 3). One pig died from intestinal adhesion. All other animals in the study group survived 3 weeks post-operation.
The transplant interfaces were evaluated and no signs of cellular rejection were observed in either group (Fig. 5). Healthy lobular structures were seen adjacent to the transplant interfaces, similar to other studies 14 . Histology performed on peripheral liver sites for post-operative week 1, week 2 and week 3 showed similar conditions as that of pre-transplantation of HLC.
Many factors can influence liver regeneration after hepatectomy 15 . However, these were not considered in this study and may have significant effects on the outcomes. Metabolic factors such as insulin (a potent hepatotrophic factor via stimulation of growth factors) have been quoted widely to affect liver regeneration 15 –17 . There are many other causes that affect liver regeneration, such as nutrition and infection, that were not studied here 15 . Furthermore, the existing liver conditions of the pigs were not determined. Other factors such as hyperbilirubinemia, renal insufficiency, cardiopulmonary compromise and thrombocytopenia have also been linked to affecting liver regeneration 18 .
We expected all xeno-transplanted human cells to be eventually rejected by the competent host in our experiments. We did not investigate the percentage of surviving HLC after post-operative week 3. Although evidence of viable HLC can be seen from bright-field IHC, they could not be quantified.
In earlier studies, our team investigated different cell-delivery methods. HLC transplanted into the liver via the portal vein caused portal vein thrombosis and portal hypertension, due to the “sticky” epithelial nature of HLC. This led to high morbidity and poor outcomes. Intraperitoneal infusion was also attempted. However, the transplanted HLC could not be localized to the liver, making further analysis difficult.
Although there was no significant statistical difference in enhancement effects on liver regeneration for this small sample size study, we did note a faster recovery trend for the study arm. The study arm subjects may have suffered from a higher level of trauma during the hepatectomy, suggested by the higher DeRitis ratio in the first few days after the surgery. The ratio for the study arm dropped faster to reach the same level as that of the control arm at post-operative day 4. Certain ALP isoenzymes are clinically observed to be elevated, which indicates favorable liver regeneration.
Nevertheless, several factors can be improved to achieve the desired enhancement effect. First, a significant amount of HLC may have “escaped” from the collagen scaffold during the initial period of transplantation. This is due to the superficial attachment of the HLC to the collagen scaffold observed (Fig. 4). Furthermore, fibrous tissue formed on the transplant interface, a part of the regenerative process of the host liver (Fig. 4e, 4f, 5). This could have impeded any mitogenic effects HLC might have had on the host hepatocytes or in assisting in hepatocyte functions, in later periods of transplantation. A multiple-layered sandwich collagen scaffold may be a solution to these problems. HLC would be confined in the scaffold and, similar to a delayed-release tablet, as the initial layers of collagen are being absorbed, HLC will continue to be exposed to host hepatocytes as the fibrous tissue degenerates during the later periods of the regeneration process.
Second, the injury caused to the host liver may have not been severe enough. Proliferation of hepatocytes is the main mechanism of liver repair and regeneration. Liver stem cells only come into play when there is insufficient hepatocyte proliferation activity. The porcine subjects used in this study were healthy, and strong organic hepatocyte regeneration may have overpowered any regenerative effects HLC had on the host liver. A 50% hepatectomy procedure was the most severe surgical intervention the team could carry out, because of survival risks for the subject. It is suggested that hepatotoxins, such as CCl4, be administered in future studies to create a more severe liver insufficient model.
This study proved that the collagen scaffold can be used as a viable, unique cell-delivery vehicle. HLC transplanted in this way can survive for up to 3 weeks post-transplantation in a xeno-transplanted host. However, the effectiveness of cell delivery was noted to be low. Histology showed HLC attaching to the collagen scaffold only superficially; inner layers of the collagen scaffold were not penetrated with HLC (Fig. 4d). Therefore, the collagen sandwich extra-cellular matrix, a system which has been well studied for the maintenance of mature hepatocyte function, is suggested for future HLC works. This too prevents the HLC from “escaping” to non-intended sites and mechanical injuries during surgical application.
Conclusion
We achieved our primary objective of showing the non-toxicity of transplanting HLC into an immunologically competent xeno-transplanted host. There was also no rejection seen even in a xenograft model. This bodes well for future applications for allografts, where a lesser degree of immune reaction is expected.
A unique cell-delivery method was tested in this study, by applying a collagen scaffold with HLC directly on the liver parenchyma surface. This was also proven to be effective, with transported cells surviving for up to 3 weeks post-transplantation. This approach can be used in future experiments. Further experiments will be required to prove the efficacy of HLC on liver regeneration.
Footnotes
Ethical Approval
Ethical approval to report this case was obtained from Institutional Animal Care and Use Committee (IACUC), Singapore (IACUC Application Number: 2014-00657).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Institutional Animal Care and Use Committee, Singapore (IACUC Application Number: 2014-00657) approved protocols.
Statement of Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Phan TT is the founder and major shareholder of CellResearch Corp Group of Companies Pte.Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research project was funded by National Medical Research Council of Singapore and CellResearch Corporation Group of Companies Pte. Ltd.
