Abstract
Endothelial progenitor cells (EPCs) have been applied for cell therapy because of their roles in angiogenesis and neovascularization in ischemic tissue. However, adverse responses caused by EPC therapy have not been fully investigated. In this study, a human peripheral blood sample was collected from a healthy donor and peripheral blood mononuclear cells were separated using Ficoll-Hypaque. There were four experimental groups: 10 ml saline infusion group (injection rate; 3 ml/min), 10 ml saline bolus group (injection rate; 60 ml/min), 10 ml EPCs infusion group (2 x 105 cells/ml, injection rate; 3 ml/min), 10 ml EPCs bolus group (2 × 105 cells/ml, injection rate; 60 ml/min). Clinical assessment included physical examination and laboratory examination for intravenous human EPC transplantation in dogs. The results revealed no remarkable findings in vital signs among the dogs used. In blood analysis, platelet counts in saline infusion groups were significantly higher than in the EPC groups within normal ranges, and no significant differences were observed except K+, Cl– and blood urea nitrogen/urea. In ELISA assay, no significant difference was observed in serum tumor necrosis factor alpha. The serum concentration of vascular endothelial growth factor was significantly higher in EPC groups than in saline groups, and interleukin 10 was significantly up-regulated in the EPC infusion group compared with other groups. In conclusion, we demonstrated that no clinical abnormalities were detected after intravenous transplantation of human EPCs in dogs. The transplanted xenogenic EPCs might be involved in anti-inflammatory and angiogenic functions in dogs.
Introduction
In the current era, regenerative therapies are largely executed using various kinds of stem cells. One of the most commonly applied cell types are endothelial progenitor cells (EPCs). EPCs are defined as non-endothelial cells capable of acquiring the characteristics of mature endothelial cells 1 . They show clonal expansion, i.e., the ability of a single cell to multiply indefinitely, and stem characteristics, such as proliferative capability and resistance to stress 1 . They possess hematopoietic stem cell markers such as CD34+ and vascular endothelial growth factor receptor 2, and contribute to neo-angiogenesis 2 . Also, Rehman et al. suggested that EPCs isolated from monocytes and macrophages secreted angiogenetic growth factors to improve the process of angiogenesis 3 . Therefore, a large body of evidence demonstrates that EPCs are good candidates for regenerative medicine.
In this regard, transplantation of EPCs has been applied to various animal models for human diseases. In recent years, many studies have provided attractive therapeutic options for human EPCs to initiate myocardial neovascularization, improve myocardial blood flow in rats 4 , protect against cerebral ischemic injury in mice 5 , improve symptoms of preeclampsia, and treat stroke in rats 6,7 . In addition, infusion of EPCs derived from peripheral blood mononuclear cells in nude rats improved neovascularization in hind limb ischemia models 8 , while transplantation of ex vivo expanded EPCs significantly improved coronary flow reserve and left ventricular function in patients with acute myocardial infarction 9 .
However, before culture-expanded stem cells can be applied into the human clinic, the in vivo safety of these stem cells should be confirmed, because adverse effects and responses caused by stem cells or EPC therapy have been reported. It was reported that transplanted allogenic mesenchymal stem cells (MSCs) in dogs could induce adverse reactions such as pulmonary parenchymal edema and hemorrhage 10 . In addition, myocardial ischemia and microinfarction were observed after administration of autologous MSCs into dogs 11 , and interruption of blood circulation, embolism, and pulmonary sequestration, which caused death in mice after post-transplantation of human MSCs, were detected 12 . Also, allogenic EPC transplantation has been applied to humans for cell therapy; however, severe adverse reactions were observed, such as collapse, sepsis, and even death 13 , and there are no reports evaluating EPC transplantation in dogs. Thus, clinical assessment for EPC transplantation into dogs, which show similar physiological characteristics to humans, should be conducted prior to applying EPC transplantation for clinical therapeutic uses in human. Therefore, we performed intravenous transplantation of human EPCs to evaluate overall adverse responses instead of injecting to local sites to stimulate angiogenesis.
The aim of this study was to investigate 1) clinical evaluation, including physical examination and blood analysis; and 2) the levels of tumor necrosis factor-α (TNF-α), vascular endothelial growth factor (VEGF), and interleukin 10 (IL-10) in dog serum to identify the effects of EPCs on cytokine regulation. For this study, human EPCs were injected intravenously into dogs, and we conducted various laboratory examinations to evaluate the clinical characteristics caused by EPC transplantation.
Materials and Methods
Isolation and Culture of EPCs
A human peripheral blood sample was collected from a healthy donor
14
. Peripheral blood mononuclear cells (PBMCs) were separated by Ficoll-Hypaque (GE Healthcare Life Science, Piscataway, NJ, USA) density gradient centrifugation at 2500 rpm for 30 min, and were obtained from the interface between the plasma layer and ficoll-hypaque layer
3,15
. PBMCs were seeded into a fibronectin-coated T25 flask at 1–3 × 107 cells with Defined Keratinocyte-SFM (Gibco, Waltham, MA, USA)-based medium containing 0.2 mM ascorbic acid, 10 µg/ml
Flow Cytometric Analysis of EPCs
To evaluate the surface marker phenotype of the cultured EPC population, cells were detached and labeled for 1 h at 4°C with anti-CD144-PE, anti-CD31–FITC, or anti-CD105 as endothelial markers as previously described 16 ; anti-CD45–FITC or anti-CD14-APC as monocyte/macrophage markers; anti-CD133-APC as hematopoietic cell surface markers; fluorescent isotype-matched antibodies were used as negative controls. Cells were washed with Dulbecco’s phosphate-buffered saline (DPBS) three times and re-suspended with 200 µl DPBS, analyzed on a FACS-Calibur Instrument (Becton-Dickinson, Franklin Lakes, NJ, USA), and quantified using CellQuest software.
Uptake of Dil-Acetylated Low-Density Lipoprotein
Cultured EPCs were washed twice with DPBS and Dil-acetylated low-density lipoprotein (DIL-Ac-LDL) was added at a concentration of 10 µg/ml in medium. EPCs and DIL-Ac-LDL was incubated for 4 h at 37°C, blocking out light. The cells were washed twice with DPBS. The image was obtained using an inverted fluorescence microscope.
In Vitro Tube Formation Assay
The angiogenic activity of cultured EPCs was assessed using an in vitro angiogenesis assay kit (Chemicon, Temecula, CA, USA). Cells were harvested and washed with DPBS two times and then re-suspended at 2.5 × 104 cells/100 µl in endothelial cell basal medium-2 (Clonetics, Baltimore, MD, USA) supplemented with 2% fetal bovine serum (FBS). Suspended cells are seeded into pre-incubated solidify ECM matrix 2.5 × 104 cells/100 µl per well and then incubated at 37°C overnight for 16 h. Tube formation was confirmed by staining viable cells with calcein-AM (5 g/ml) (Molecular Probes, Eugene, OR, USA), and images were obtained using an inverted fluorescence microscope.
Transplantation of EPCs
The application of a minimal number of EPCs for transplantation is desirable because of safety, thus we used the lowest quantity of EPCs possible for this research. In many other studies, some inconvenience has occurred with the use of high doses of stem cells for transplantation. Previously, it was reported that a high dose of stem cells might be associated with cell clumping, forming aggregates, especially when injected through a narrow needle, and that such aggregates can cause pulmonary emboli or infarctions after application of stem cell transplantation 17,18 . Moreover, it has been suggested that multiple administration of high doses of stem cells affect alloreactive immune responses in recipient animals 19 . Also, according to previous research assessing the safety of adipose stem cell transplantation 20 , the high dose (2.5 × 108 cells/kg) of stem cell transplantation induced instant death in mice, although there were no specific pathological changes in these mice organs including lungs. Therefore, for these reasons, we chose a low cell dose for transplantation (1–2 × 106 cells/kg) 21,22 , and decided to apply a low dose of EPCs (2 × 106) for administration in this study. Also, to minimize the alteration of cell survival and viability effects, in this study we performed the cell transplantation as soon as we collected the EPCs from the dish. There were four experimental groups: 10 ml saline infusion (n = 3, injection rate; 3 ml/min), 10 ml saline bolus group (group B, n = 3, injection rate; 60 ml/min), 10 ml EPC infusion group (group C, n = 3, 2 × 105 cells/ml, injection rate; 3 ml/min), 10 ml EPC bolus group (group D, n = 3, 2 × 105 cells/ml, injection rate; 60 ml/min). The fresh saline or harvested EPCs resuspended in saline were infused into the animals through a peripheral catheter into the cephalic vein.
Clinical Assessment
Prior to human EPC injection, dogs were evaluated by physical examination. Body temperature, respiratory rate, heart sounds, and pulse rate were observed before injection, and every 5 min after injection up to 30 min. Neurological examination to monitor occurrence of adverse events was performed before injection and 30 min after injection. Heart rate, respiratory rate, body temperature, heart murmur, electrocardiography (ECG), neurological symptoms, and swelling of all four limbs were evaluated to assess adverse responses for 30 min. Lead II ECG tracings were recorded from each group of dogs, and for evaluation of neurological symptoms, cranial nerve reflexes (olfaction, menace, pupillary light reflex, cotton ball, strabismus, nystagmus, palpebral reflex, hearing, and gag reflex) and postural reflexes (proprioception, hemi-standing/walking, wheelbarrowing, placing reaction-tactile/visual, and withdrawal reflex) were examined.
Blood Analysis
To assess adverse reactions to EPC transplantation, complete blood count (CBC) and serum chemistry panel tests were performed (IDEXX Laboratories, Kyunggi-do, Korea) on days 1, 7, 14, 21, and 28 post-injection of saline and EPCs. The CBC included red blood cell (RBC), white blood cell (WBC), packed cell volume (PCV), platelet (PLT), hemoglobin (HB), neutrophil (NEUT), basophil (BASO), and eosinophil (EOS). In addition, a serum chemistry panel test was performed, including sodium (Na), potassium (K), chlorine (Cl), alkaline phosphatase (ALP), alanine aminotransferase (ALT), aspartate aminotransferase (AST), blood urea nitrogen/urea (BUN/UREA), creatinine (CREA), phosphorus (P), and calcium (Ca).
ELISA Analysis
For enzyme-linked immunosorbent assay (ELISA) analysis, canine blood samples were collected, taking aseptic precautions, and allowed to clot to obtain serum, then centrifuged at 3000 rpm for 10 min to separate serum. Serum samples were stored at –80°C until use. The concentrations of TNF-α, VEGF, and IL-10 in serum were measured by ELISA Quantikine immunoassays (R&D Systems, Minneapolis, MN, USA) from each group. The assay was performed following the manufacturer’s instructions. Briefly, assay diluent was added to each well of the ELISA plates. Serum samples were added to each well, and incubated for 2 h at room temperature. To remove unbound antigen, wash buffer was applied to each well five times using a squirt bottle. After the last wash, conjugates were added to each well and incubated for 2 h at room temperature and washed five times with wash buffer. The substrate solutions were added to each well and incubated for 30 min at room temperature with protection from light. Lastly, stop solutions were added to each well and tapped gently to ensure thorough mixing. The spectroscopic absorbance of each well was measured in a microplate reader (Tecan Sunrise, Hayward, CA, USA) at 480 nm excitation/530 nm emission.
Statistical Analysis
Descriptive and inductive statistical analysis was carried out using GraphPad Prism 5.0 (Graphpad, San Diego, CA, USA). Differences among the groups were examined with two-way ANOVA and the Bonferroni post-test. Differences of P < 0.05 were considered significant and all data are presented as the mean ± standard error of the mean (SEM).
Results
Morphology and Phenotype of Culture Expanded EPCs
Mononuclear cells collected from adult peripheral blood of a healthy donor were seeded on a human fibronectin pre-coated plate with EPC culture medium containing 10% FBS. After 14–21 days, primary EPC colonies appeared as a small cluster, and then cells eventually proliferated to form a confluent cobblestone-shape monolayer (Fig. 1) 23 . Passaged EPCs maintained proliferation potential and cobblestone morphology, and other cells were removed at the end of the isolation period on day 28. To ensure that our modified procedure gave rise to true EPCs, cells were characterized phenotypically by flow cytometry as in a previous study 16 . Briefly, the immunophenotype of culture-expanded human EPCs was determined at each passage (3–5). The EPCs were positive for CD105, CD31, and CD144, but were negative for CD14, CD45, and CD133. Each passage of EPCs showed a homogeneous population of cells with high expression endothelial progenitor markers CD105, CD31, and CD144 and very low expression levels of monocyte/macrophage markers, hematopoietic cell marker CD14, CD45, and CD133. The expression of CD105 and CD31 continued to be ≥ 95%, ≥92% up to passage 5. CD144 was expressed ≥ 98% up to passage 5.

Morphology of EPCs from peripheral blood. (A) After 14 days of culture, EPC colonies appeared and showed cobblestone shape. (B) At passage 2, EPCs had a cobblestone appearance and unnecessary cells were removed by subculture.
Cultured EPC Characterization
Ac-LDL uptake has become routine to identify endothelial progenitor cells in EPC characterization. Dil-Ac-LDL enters the cells, becomes degraded by lysosomes, and subsequently accumulates in the lysosomal membranes 24 . Uptake of Ac-LDL was measured after incubation of cells with 10 μg/ml of Dil-Ac-LDL at 37°C in culture media for 4 h. All cells were positive for Dil-Ac-LDL uptake, indicating that they had maintained their endothelial cell function throughout the in vitro culture (Fig. 2A). A tube formation assay was performed on EPCs to assess the capacity for vasculogenesis, which is believed to be important in new vessel formation. Vasculogenesis, the ability to form tube-like structures, was assessed in vitro by seeding the cells on an ECM matrix. After 16 h, EPC cultures were able to organize themselves into similar tube-like structures by fluorescence microscopy (Fig. 2B).
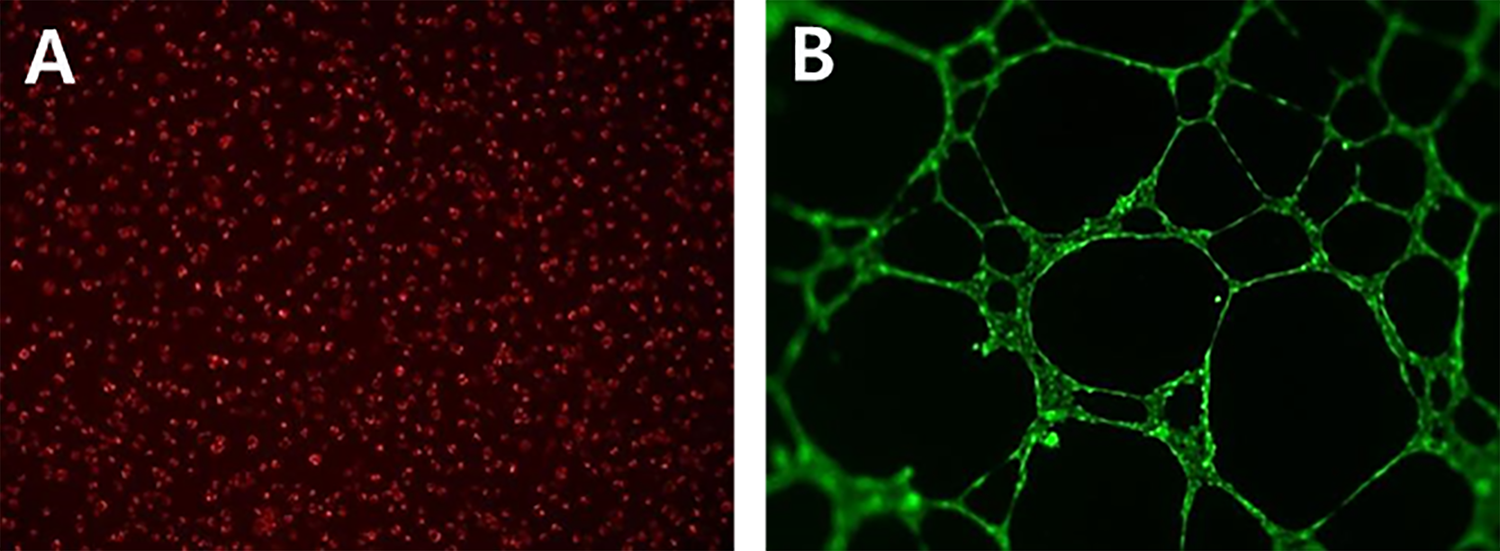
Characterization of EPCs from peripheral blood. (A) Endothelial progenitor cells determined by cellular uptake of DiI-labeled acetylated LDL. (B) Fluorescent image of endothelial progenitor cells forming vascular networks on ECM matrix after 16 h with calcein.
Clinical Assessment after EPC Transplantation
To evaluate any adverse responses of saline and EPC transplantation, auscultation of heart murmur, electrocardiography, neurological symptoms, and signs of swelling in the limbs were recorded (Fig. 3) for 30 min. There were no noticeable clinical findings in any of the dogs, and no significant differences in vital signs such as heart rate, respiratory rate, or body temperature were observed (p > 0.05) (Fig. 4). Also, no adverse responses, such as heart murmur or limb swelling, were detected, and all groups showed normal neurological responses for cranial nerve reflexes and postural reflexes. In addition, an ECG test was performed before and after transplantation to assess circulatory adverse responses such as arrhythmia and coronary artery anomalies. There were no significant differences before and after treatment in wave interval and height (P width, P height, PR interval, QRS width, R height and QT interval).

Physical examination outcome. (A) Lead II ECG tracings recorded from the each group of dogs (saline infusion, saline bolus, EPC infusion, and EPC bolus) after transplantation of saline/EPCs. (B) Observation of limbs swelling from each groups of dogs after transplantation of saline/EPCs. (C) Physical examination outcome for heart murmur and neurological symptom. For evaluation of neurological symptom, cranial nerve reflexes and postural reflexes were examined. NE: none exist.

Assessment of (A) heart rate (normal range: 100–160 beats/min), (B) body temperature (normal range: 38.0–39.2°C) and (C) respiratory rate (normal range: 10–35 breaths/min) after the saline/EPC transplantation. No significant differences of vital signs among the four groups (p < 0.05) and all groups were within normal ranges. 0, 5, 10, 15, 20, 25, and 30 min: time after the saline/EPC transplantation (infusion rate: 3 ml/min, bolus rate: 60 ml/min). Each bars represent the normal reference intervals.
Blood Analysis after EPC Transplantation
To examine the possibility of systemic responses, we analyzed CBC and serum chemistry results on days 1, 7, 14, 21, and 28 post-transplantation of saline and EPCs (Figs. 5 and 6). The post-transplantation CBC results were within normal ranges for all animals. Also, no significant differences were observed among serial test results for the four groups except for one parameter: platelet numbers. The platelet counts showed significant differences between saline infusion groups (318.0 ± 47.3 × 103/μl) and the EPC groups (infusion: 184.7 ± 14.8 × 103/μl; bolus: 172.0 ± 12.7 × 103/μl) on day 1 post-transplantation (p < 0.05). However, there were no significant differences in platelet counts between the saline and EPC groups on days 7, 14, 21, and 28 post-transplantation.
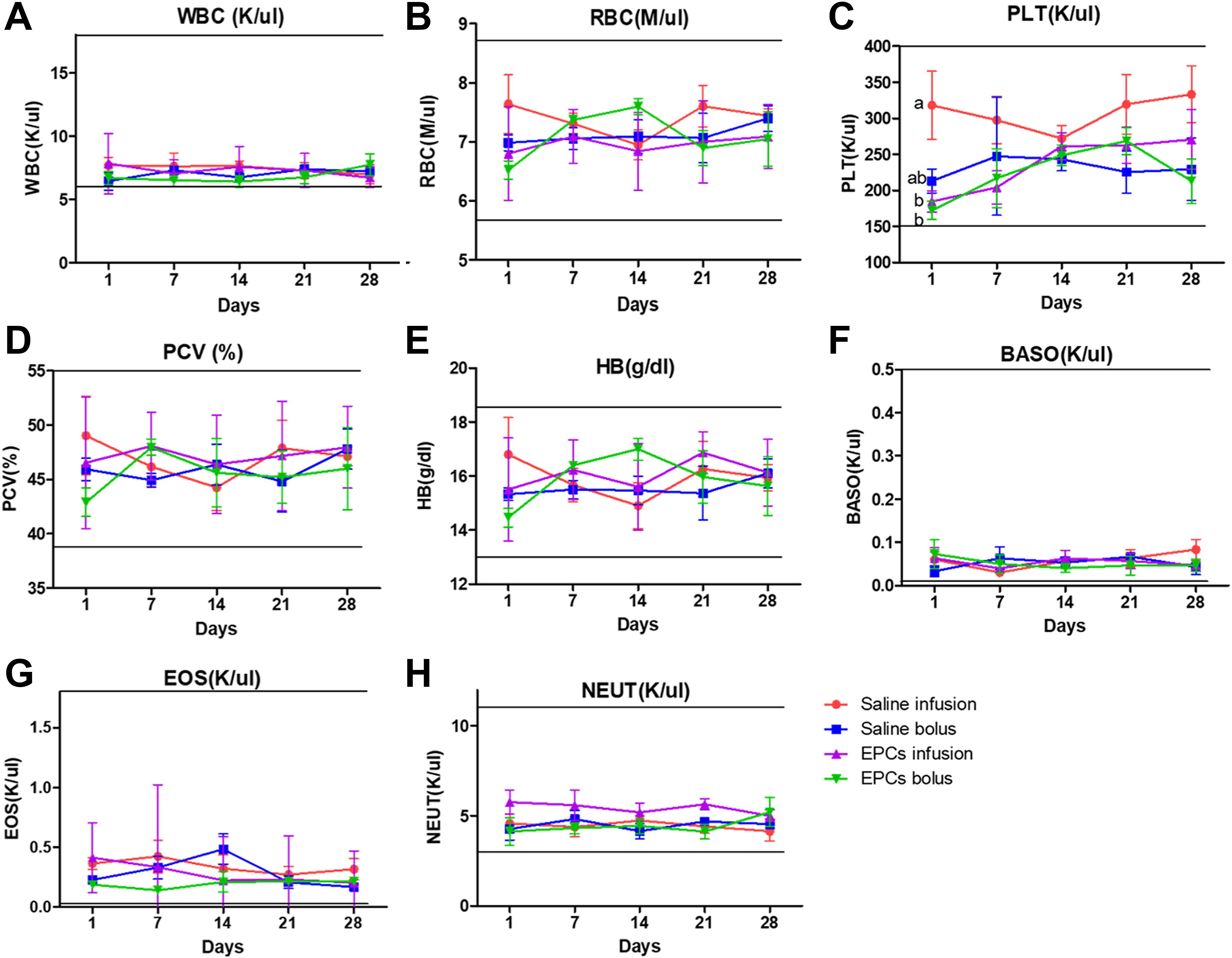
Evaluation of complete blood cell count after the saline/EPC transplantation.a, b Within groups, values with different superscript letters are significantly different (p < 0.05) and all groups were within normal ranges. 1, 7, 14, 21, and 28 days: time after the saline/EPC transplantation (infusion rate: 3 ml/min, bolus rate: 60 ml/min). Each group has three dogs and analyses were performed a total of five times on days 1, 7, 14, 21 and 28 post-transplantation in each dog. (A) WBC; white blood cell, (B) RBC; red blood cell, (C) PLT; platelet, (D) PCV; packed cell volume, (E) HB; hemoglobin, (F) BASO; basophil, (G) EOS; eosinophil, (H) NEUT; neutrophil. Bars represent normal reference intervals.

Evaluation of serum chemistry analysis after the saline/EPC transplantation.a, b Within groups, values with different superscript letters are significantly different (p < 0.05) and all groups were within normal ranges. 1, 7, 14, 21, and 28 days: time after the saline/EPC transplantation (infusion rate: 3 ml/min, bolus rate: 60 ml/min). Each group has three dogs and analyses were performed a total of five times on days 1, 7, 14, 21 and 28 post-transplantation in each dog. (A) Na, (B) K, (C) Cl, (D) ALP, (E) ALT, (F) AST, (G) CREA, (H) Albumin, (I) BUN/UREA; blood urea nitrogen/urea, (J) Total protein, (K) Ca, (L) P. Bars represent normal reference intervals.
Meanwhile, the results of post-transplantation serum chemistry were within normal ranges for all animals. However, there were some significant differences among serial test results for the four groups with respect to several factors. On serial tests of serum chemistry, K revealed significant differences among groups on day 21, Cl showed significant differences on days 7 and 14, and BUN/UREA showed significant differences on day 1 following EPC transplantation. There was significantly increased K in the EPC infusion group (5.3 ± 0.2 mmol/L) compared with the EPC bolus group (4.7 ± 0.1 mmol/L) on day 21 post-transplantation (Fig. 6b) (p < 0.05). Also, on day 7 following EPC transplantation, the saline bolus group (116.7 ± 1.2 mmol/L) showed a significantly higher concentration of Cl– compared with that of the saline infusion group, and the EPC infusion and EPC bolus groups (113.0 ± 1.5, 111.7 ± 0.7, 111.0 ± 1.5 mmol/L respectively) (Fig. 6c). On day 14 following EPC transplantation, the EPC bolus group (117.0 ± 0.6 mmol/L) revealed significantly increased Cl compared with that of the saline infusion and bolus groups (113.0 ± 0.6, 114.0 ± 0.6 mmol/L, respectively) (Fig. 6c) (p < 0.05). Meanwhile, there was a significantly increased BUN/UREA in the EPC bolus group (12.3 ± 1.2 mg/dl) compared with the saline infusion group (6.7 ± 1.7 mg/dl) on day 1 following EPC transplantation (Fig. 6i) (p < 0.05).
ELISA Analysis
The concentrations of VEGF, IL-10, and TNF-α in serum of each group on days 1, 7, 14, 21, and 28 post-transplantation of EPCs were analyzed (Fig. 7). No significant difference was observed in serum TNF-α concentration among groups. However, the concentration of VEGF was significantly higher in the EPC groups on days 1, 7, 14, 21, and 28 following treatment (p < 0.05): EPC infusion 84.9 ± 4.9, 80.0 ± 0.0, 84.9 ± 6.4, 75.2 ± 2.4 and 80.0 ± 0.0 pg/ml, respectively; EPC bolus 77.6 ± 2.4, 80.0 ± 4.2, 75.2 ± 2.4, 82.4 ± 2.4 and 101.8 ± 8.4 pg/ml, respectively, compared with the saline groups: saline infusion 43.6 ± 7.3, 38.8 ± 10.6, 50.9 ± 0.0, 41.2 ± 2.4, and 46.1 ± 6.4 pg/ml, respectively. In addition, the serum concentration of IL-10 was significantly increased in the EPC infusion groups on days 1, 7, 14, 21, and 28 post-transplantation: 110.5 ± 9.1, 129.5 ± 37.4, 132.4 ± 34.3, 102.9 ± 19.0, and 99.1 ± 12.4 pg/ml, respectively, compared with the other three groups.

Concentration of (A) TNF-α, (B) VEGF, and (C) IL-10 from serum in each groups after the saline/EPC transplantation. A, B, C Within groups, values with different superscript letters are significantly different (p < 0.05). 1, 7, 14, 21 and 28 days: time after the saline/EPC transplantation (infusion rate: 3 ml/min, bolus rate: 60 ml/min). Each group has three dogs and analyses were performed a total of five times on days 1, 7, 14, 21, and 28 post-transplantation in each dog.
Discussion
Stem cell therapies have been used to replace damaged cells and tissues in the field of regenerative medicine. In particular, EPCs have shown potential therapeutic effects in a variety of fatal diseases that cannot be treated with conventional therapies 25,26 . However, deleterious effects of human EPC therapy have not been fully reported. Recently, the occurrence of adverse responses such as death soon after discharge, collapse, sepsis, and development of breast cancer were reported after delivery of human EPCs over-expressing endothelial nitric oxide synthase into patients with pulmonary arterial hypertension 13 . Also, the intravascular transplantation of cultured stem cells or progenitor cells caused pulmonary thrombus formation in arterioles and venules due to stem cells obstructing circulation in mice and rats 12 , and transplanting rat stem cells to adult rat brains elicited acute inflammatory responses 27,28 . Likewise, transplanted EPCs need to be assessed clinically before applying them for various therapies because of potential adverse effects after transplantation. In addition, it was reported that accidental bolus injection of stem cells would cause death in mice, while slow injections of cells into mice did not induce any adverse events 20 , and bolus or infusion rates of stem cells after transplantation exhibited significantly different levels of serum proteins in dogs 29 . Therefore, to establish the possibility of adverse effects, in the present study EPCs were transplanted into dogs intravenously with different injection rates at a constant volume (2 × 106 cells/10 ml).
The EPCs collected from human peripheral blood were cultured and formed monolayers with a typical cobblestone-shape of endothelial appearance which is consistent with previous studies 30,31 . In the present study, the immunophenotype of EPCs showed the expression of endothelial cell markers (CD105, CD31, and CD144) and the absence of monocyte, macrophage, and hematopoietic markers (CD14, CD45, and CD133). In addition, our results showed that human EPCs were positive for Ac-LDL uptake, which has been commonly referred to as conventional endothelial phenotype 32,33 . Also, in our results, tube-like structures, which have been used to demonstrate the characteristics of endothelial cells in putative EPCs, were detected in EPCs 34 . Taken together, these findings indicate that the cells could fulfill the requirements for EPCs, and that cultured EPCs can be used to reliably obtain a large number of EPCs for transplantation.
The incidence of adverse reactions after intravenous xenogenic injection of human EPCs in dogs was evaluated through various laboratory studies: physical examination, blood analyses, and ELISA assays. Some studies concluded that transplanting xenogenic EPCs derived from humans into rats and mice showed no adverse reactions, with successfully transplantation into injured areas 35,36 . According to our results, transplantation of human EPCs at a concentration of 2 x 106/10 ml into dogs did not induce any adverse reactions. In addition, no clinical adverse events occurred during detailed observation for 2 months.
We analyzed the CBC, serum chemistry, and several results, including platelet count, K, Cl, and BUN/UREA, revealed significant differences among the experimental groups. It has been reported that the role of EPCs in vascular injury is to interact with platelets, and the function of EPCs could be mediated by platelet-derived growth factors 37 . One report using hypertensive-hypercholesterolemic hamsters suggested that treatment with EPCs reduces platelet activation by modulating thrombogenic properties 38 . Also, platelets recruit EPCs by involving platelet P-selectin, and activated platelets secrete stromal cell-derived factor-1, which supports the migration of EPCs by adhesion 39 , perhaps explaining why platelet numbers are reduced after EPC transplantation. In our study, the number of platelets decreased significantly in the EPC groups compared with the saline infusion groups on day 1 post-transplantation. This may be because EPCs down-regulated platelet activation by altering thrombogenic characteristics. Also, we may assume that canine platelets could support recruitment and migration of human EPCs by adhesion, thus the amount of platelets could be reduced temporarily after EPC transplantation. Taking these results together, the transient effect of human EPCs on reducing platelet counts might have possible application as an anti-thrombotic therapy for patients suffering from thrombosis and embolism, although further studies are needed to fully understand the relationship between EPCs and reduction in platelet numbers.
Analysis of serum chemistry showed that K, Cl and BUN/UREA, important factors for evaluating the condition of the kidneys, showed significant differences among groups on particular days after EPC transplantation, although the results were within normal ranges for all groups. A report suggested that the level of BUN, which represents a uremic toxin burden, varies widely depending on the method of measurement 40 . Thus, correlations between the level of BUN and EPC transplantation might be only suggestive, and are hardly conclusive. In line with published results, the findings in our study of significant differences in levels of K, Cl, and BUN/UREA might be caused by measurement variations.
We evaluated the concentrations of TNF-α, VEGF, and IL-10 after EPC transplantation; VEGF and IL-10 showed significant changes. IL-10 is an anti-inflammatory cytokine that plays an important role in regulating immune responses. Several investigators have demonstrated an interaction between EPCs and IL-10. Bone marrow-derived macrophages (BMDM) co-cultured with EPCs produced higher concentration of IL-10 than BMDM cultured alone in an in vitro system 41 . Additionally, administration of EPCs into sepsis animal models augmented plasma IL-10 concentrations 42 , which suggested that EPCs could participate in modulation of anti-inflammatory cytokines in injured animal models. In the present study, the EPCs infusion group showed significantly increased IL-10 compared with the other groups, which indicated that slow injection of EPCs might mediate some effects on healthy dogs by increasing the concentration of IL-10.
It was reported that when EPCs were transplanted into a chronic kidney disease animal model, secretion of TNF-α could be potentially decreased 43 . Also, intravenous injection of bone marrow stem cells could modulate the host immune response by stimulating release of IL-10 44 . Although, in our study, no significant differences were observed in concentrations of TNF-α among the saline and EPC groups, there were significance differences in IL-10. Intense elaboration of pro-inflammatory cytokines such as TNF-α by macrophages and monocytes results in the onset of sepsis 45,46 . This pro-inflammatory cytokine surge occurs with a compensatory release of regulatory cytokines, including IL-10 47,48 . Furthermore, it seems that stem cells may alter the inflammatory response via promoting increase in IL-10 production 44,49 . We may assume that EPCs would not be involved in increasing TNF-α concentration when EPCs transplantation is performed in non-diseased animals of normal status. Also, we could speculate that that IL-10 expression might play a role in mediating the beneficial effects of intravenously injected EPCs in healthy dogs free of disease.
VEGF and signal transducer and activator of transcription 3 (STAT3) are essential factors involved in neovascularization during ischemic injury. Previous studies have demonstrated that the level of VEGF could be increased by activated STAT3 when IL-10 was treated in myocardial infarction 50 . Also, STAT3/VEGF in the myocardium could be induced by IL-10, and with synergy of STAT3 and VEGF in EPCs, so IL-10 led to angiogenesis activation 51 . In line with these results, our study showed that the level of IL-10 was significantly higher in the EPC infusion group, which could augment the VEGF level in EPCs. Thus, we could speculate that VEGF signaling is involved in neovascularization of EPCs along with IL-10. Likewise, in this study, the EPCs groups showed significantly increased VEGF levels compared with the saline groups, and we confirmed that EPCs could increase VEGF in intact animals. Although our results suggest the safety of transplantation of human EPCs transplantation in dogs, which would be a potential pilot study for clinical use in humans, the efficacy of EPCs from the present results in dogs cannot be determined due to the following limitations of the study design. Firstly, the number of dogs in each group was relatively too small to have adequate statistical strength for observing the frequency of adverse effects. Secondly, the observation time after transplantation was somewhat short, hindering us from verifying the possibility of adverse effect occurrence.
In conclusion, dogs with EPC transplantation have reduced platelets, increased VEGF, and increased IL-10, without any clinical adverse events. Therefore, we suggest that the transplantation of human EPCs would appear to be safe to use for direct intravenous transplantation in dogs, and that this would provide a potential pilot study for clinical use in humans. In addition, EPCs may represent a novel therapeutic tool for stimulating anti-inflammatory and angiogenic factors.
Footnotes
Acknowledgments
This research was supported by Nature Cell (#550-20150030), global PhD Fellowship Program through the National Research Foundation (NRF) funded by the Ministry of Education (NRF-20142A1021187), Cooperative Research Program of RDA (CCAR, #PJ013954022018), Korea Institute of Planning and Evaluation for Technology (IPET) (#316002-05-4-SB010), and Research Institute for Veterinary Science, the BK21 plus program.
Author Contributions
Seok Hee Lee and Jeong Chan Ra contributed equally to this work.
Ethical Approval
This study was approved by Institutional Animal Care and Use Committee of Seoul National University, Republic of Korea (approval number; SNU-150331-4) and the Life Ethics Committee of the Biostar Stem Cell Technology (RBIO 2015-12-001), and all patients gave informed consent for inclusion in the study and obtaining for human endothelial progenitor cells. All experimental research on humans is in compliance with the Helsinki Declaration.
Statement of Human and Animal Rights
In this experiment, 12 healthy, 4-year-old, intact male beagle dogs (weight range: 9.00 to 11.55 kg) without clinical signs of any disease were used. All dogs were monitored continuously throughout the research period and fed with a constant amount of commercial adult dry food (Natural Balance, Natural Balance Pet Food, Burbank, CA, USA) and water daily. Also, all dogs were housed in an identical manner since birth in individual cages. All of the experimental procedures involving animals were conducted in accordance with “The Guide for the Care and Use of Laboratory Animals” published and approved by the Institutional Animal Care and Use Committee of Seoul National University, Republic of Korea (approval number; SNU-150331-4). In human rights, the procedures and the experiments were performed with appropriate consent and ethical approval from Life Ethics Committee of the Biostar Stem Cell Technology (RBIO 2015-12-001).
Statement of Informed Consent
This research was approved by the Life Ethics Committee of the Biostar Stem Cell Technology (RBIO 2015-12-001), and all patients gave informed consent for inclusion in the study and obtaining for human endothelial progenitor cells. All experimental research on humans is in compliance with the Helsinki Declaration.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
