Abstract
Introduction
Stroke is the third most common cause of death in the world and one of the leading causes of long-term disability, creating an urgent need for novel therapies 1 –5 . Currently, many neuroprotective drugs that showed promise in preclinical testing failed in clinical stroke treatment 6,7 . One of the main reasons is that these drugs mainly target pathogenic mediators in the protection of gray matter, but not white matter 8 . White matter plays an important role in signal transduction and communication across brain regions 9 –11 and is an important cause of cognitive decline and sensorimotor deficits in stroke 12 –14 . Therefore, strategies that battle both gray and white matter injury are urgently needed for stroke therapies.
Currently, the altered microstructures of white and gray matter caused by stroke are mainly detected by histological measurements which do not allow dynamic assessment at different time points
Materials and Methods
Experimental Animals
The male Sprague–Dawley rats, weighing 280 ± 20 g, were purchased from the Vital River Laboratory Animal Technology Co. Ltd. (Beijing, China) and housed three per cage under a controlled temperature (21 ± 2°C) and humidity (55 ± 5%) environment with a 12-h light/dark cycle. All experimental procedures were performed in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals and the local Ethical Committee at the Capital Medical University, China (Permit Number: No. 2013-X-16).
Preparation and Quantitative Analysis of XSECC
XSECC was supplied by Sanmenxia Sinoway Pharmaceutical Co. Ltd. (Henan, China). The formula consists of seven Chinese herbs, including Radix Astragali, Pheretima, Radix Paeoniae Rubra, Radix Angelica Sinensis, Rhizoma Ligustici Chuanxiong, Semen Persicae, and Flos Carthami. Herbal sources, preparation method and quantitative analysis of XSECC were stated in detail in our previous study 15 .
Animal Grouping, Stroke Model Induction and Drug Administration
A total of 30 rats were randomly divided into three groups (10 rats per group), including sham group, model group and XSECC treatment group. Focal cerebral ischemia was induced by permanent middle cerebral artery occlusion (MCAO) as previously described 23 . Briefly, the right common carotid, external carotid and internal carotid arteries were exposed. The external carotid and internal carotid arteries were temporarily clamped using microsurgical clips. A 4-0 monofilament nylon suture was inserted into the internal carotid artery and gently advanced from the lumen of the internal carotid artery until it blocked the origin of the middle cerebral artery. Sham-operated rats underwent the same surgery but no occlusion procedure. The rats in the XSECC group were treated with XSECC dissolved in sterile saline at a dose of 420 mg/kg/day for 30 days as previously described 15 . Rats in the sham and model groups were administered with normal saline (10 ml/kg/day).
Magnetic Resonance Imaging Protocols
The rats were subjected to magnetic resonance imaging examination using a 7.0 T magnet (Bruker, Pharma Scan, Germany) on the 3rd (
The cerebral blood flow (CBF) was measured by arterial spin labeling with echo-planar imaging fluid-attenuated inversion recovery sequences (TR=18,000 ms; TE = 25 ms; field of view = 3.0 × 3.0 cm; matrix size = 128 × 128; number of excitations = 1). The CBF was measured in the cortex, striatum and thalamus on both ipsilateral and contralateral sides. Then, the relative CBF (rCBF) was derived from the ratio of ipsilateral CBF to contralateral CBF 15 .
DTI was acquired using an axial single-shot spin echo-planar sequence (TR = 6300 ms; TE = 25 ms; 30 diffusion directions; two b values = 0 and 1000 s/mm2). A fractional anisotropy (FA) map, apparent diffusion coefficient (ADC) map and three eigenvector (λ1, λ2, λ3) maps were reconstructed with ParaVision software, version 5.1 (Bruker, Pharmascan, Germany). FA, ADC, λ1(axial diffusivity, λ//), λ2, and λ3 were analyzed in the regions of interest (ROIs) drawn on white matter (corpus callosum, external capsule and internal capsule) and gray matter (cortex, striatum and thalamus) at the level of bregma −1.4 mm. The radial diffusivity (λ⊥) was calculated using the equation: λ⊥ = (λ2+λ3)/2. The relative FA (rFA) or relative ADC (rADC) was calculated by comparing the parameters in the lesion ROIs with the mirrored ROIs of the contralateral side 24,25 . Diffusion tensor tractography (DTT) was reconstructed with Diffusion Toolkit and TrackVis software, Martinos Center for Biomedical Imaging, Massachusetts General Hospital, Boston, MA, USA. Relative mean tract length was derived from the ratio of ipsilateral mean tract length to contralateral mean tract length 26 .
Hematoxylin-eosin and Luxol Fast Blue Staining
Hematoxylin-eosin (HE) staining was used to evaluate the histopathological changes. On the 30th day after MCAO, rats were transcardially perfused with saline solution, followed by 1% glutaraldehyde and 4% paraformaldehyde (PFA) in 0.1 mol/l phosphate buffer. Subsequently, the brains were removed and fixed in 4% phosphate-buffered PFA. Then brain tissues were embedded in paraffin and cut into coronal sections. The paraffin sections were baked at 60°C for 30 min, dewaxed by xylene, dehydrated with gradient alcohol, washed with running water for 1 min, and stained with HE. Images were visualized under an optical microscope at 400× magnification. A total of six nonoverlapping regions were sampled for the cortex, striatum and thalamus. Cell counts were expressed as the mean number of viable neurons/mm2 as previously described 27 .
Luxol fast blue (LFB) staining was performed to visualize white matter structures through the detection of myelin on the 30th day after MCAO. The paraffin sections were first immersed in xylene followed by anhydrous ethanol gradient dehydration. Thereafter, the sections were stained overnight in 0.1% LFB solution (Sigma-Aldrich, St. Louis, MO, USA) at 60°C. After washing with 95% ethyl alcohol and distilled water, the sections were differentiated in 70% ethyl alcohol for 30 s. Differentiation was terminated by washing in distilled water until the unmyelinated tissue appeared white. Images were visualized under an optical microscope at 400×magnification. The severity of the myelin lesions was graded by LFB staining images as normal (0), disarranged nerve fibers (1), the formation of marked vacuoles (2) and disappearance of myelinated fibers (3) in accordance with a previous report 28 .
Statistics Analysis
All statistical analyses were carried out by using SPSS 17.0 software (SPSS, Chicago, IL, USA). Data are expressed as means ± standard deviation (SD). The one-way analysis of variance followed by a Fisher’s Least Significant Difference test was used for within-group comparisons. Pearson correlations were calculated to examine the linear association between DTI parameter FA and the number of surviving neurons or myelin damage grade. A significant difference was considered if the
Results
XSECC Improved CBF in the Striatum, Cortex and Thalamus after Stroke
The rCBF in the cortex, striatum and thalamus in the model group was significantly lower than in the sham group on the 3rd, 7th, 14th and 30th days post stroke (

The effects of XSECC on rCBF in the cortex, striatum and thalamus on the 3rd, 7th, 14th and 30th days after stroke.
XSECC Reduced the Injury of the Corpus Callosum, External Capsule and Internal Capsule in White Matter After Stroke
Serious damage occurred in the ischemic hemisphere in the model group evidenced by the abnormal signals on FA and ADC maps. The damage signals were weakened by XSECC treatment on the 3rd, 7th, 14th and 30th days after MCAO (Fig. 2).

The representative images of FA and ADC of the rat brains at the level of bregma −1.4 mm on the 3rd, 7th, 14th and 30th days. Serious damage occurred in the ischemic hemisphere in the model group evidenced by the obvious abnormal signals pointed with dotted lines on FA and ADC maps. The damage signals were weakened by XSECC treatment on the 3rd, 7th, 14th and 30th days.
The measurements of FA, ADC, λ// and λ⊥ were performed on the corpus callosum, external capsule and internal capsule in white matter on the 3rd, 7th, 14th and 30th days after MCAO (Fig. 3A). The FA value in the corpus callosum in the ischemic hemisphere of the model group was significantly lower than that of the sham group on the 3rd day (

The effects of XSECC on white matter on the 3rd, 7th, 14th and 30th days after stroke. (A) The regions of interest drawn on white matter (corpus callosum, external capsule and internal capsule). (B, C, D) The statistical analysis of FA, ADC, λ// and λ⊥ in the corpus callosum, external capsule and internal capsule on the 3rd, 7th, 14th and 30th days after stroke.
In the external capsule, the FA value in the model group was significantly decreased compared with the sham group on the 3rd, 7th, 14th and 30th days (
In the internal capsule, the FA value in the model group was significantly decreased compared with the sham group on the 3rd, 7th, 14th and 30th days (
XSECC Reduced the Injury of the Cortex, Striatum and Thalamus in Gray Matter After Stroke
The measurements of FA, ADC, λ// and λ⊥ were performed on the cortex, striatum and thalamus in gray matter on the 3rd, 7th, 14th and 30th days after stroke (Fig. 4A). The rFA in the cortex was significantly decreased in the model group on the 3rd, 7th, 14th and 30th days compared with the sham group (

The effects of XSECC on gray matter on the 3rd, 7th, 14th and 30th days after stroke. (A) The regions of interest drawn on gray matter (cortex, striatum and thalamus). (B, C, D) The statistical analysis of FA, rADC, λ// and λ⊥ in the cortex, striatum, and thalamus on the 3rd, 7th, 14th and 30th days after stroke.
In the striatum, the rFA value in the model group was significantly decreased compared with the sham group on the 3rd, 7th, 14th and 30th days (
In the thalamus, the rFA was significantly decreased in the model group on the 3rd, 7th, 14th and 30th days after stroke compared with the sham group (
XSECC Reduced the Injury of White Matter Fiber Bundles
Fiber tractography was reconstructed to trace the spatial distribution of white matter fiber bundles and observe the changes of the fiber bundles integrity on the 3rd, 7th, 14th and 30th days after stroke. The white matter fibers in the ischemic hemisphere were sparser than in the contralateral hemisphere with abnormal morphology, partial curl, and even missing in the rats of the model group on the 3rd, 7th, 14th and 30th days (Fig. 5A). XSECC could reduce nerve fiber injury and protect the integrity of white matter nerve fiber bundles over the course of 30 days after ischemic stroke (Fig. 5A). Statistical analysis results showed that the relative mean tract length in the model group was significantly decreased on the 3rd, 7th, 14th and 30th days after MCAO compared with the sham group (
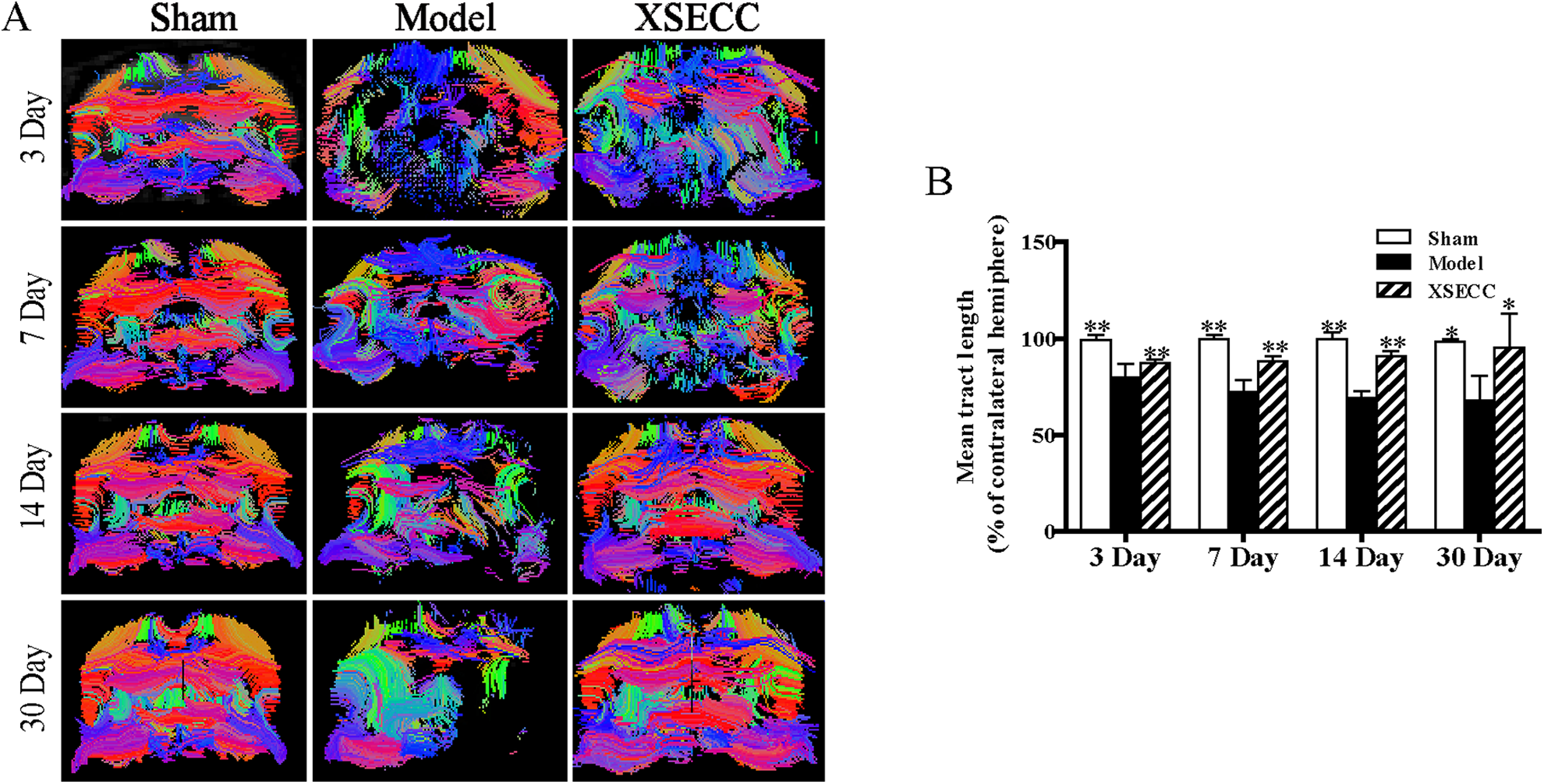
The effects of XSECC on nerve fiber injury on the 3rd, 7th, 14th and 30th days after stroke. (A) The representative images of fiber reconstruction of the rat brains at the level of bregma −1.4 mm on the 3rd, 7th, 14th and 30th days. White matter fibers in the ischemic hemisphere were sparser than that in the contralateral hemisphere with abnormal morphology, partial curl, and even were missing in the rats of the model group on the 3rd, 7th, 14th and 30th days. (B) The statistical analysis of mean tract length (% of contralateral hemisphere) on the 3rd, 7th, 14th and 30th days after stroke.
XSECC Reduced Histopathological Damage in White Matter After Stroke
The histopathological changes in white matter (corpus callosum, external capsule and internal capsule) on the 30th day after stroke were evaluated by HE and LFB staining (Fig. 6A). The well-defined white matter structures, including corpus callosum, external capsule and internal capsule, were seen in the sham group on HE and LFB staining images. However, the loss of white matter integrity in the external capsule and internal capsule was observed in the model group on both HE and LFB staining images. HE staining showed that rats in the model group had vacuolation in the external capsule and internal capsule. LFB staining displayed severe myelin disruptions in the external capsule and internal capsule in the model group. By contrast, vacuolation and myelin damage were alleviated in external capsule and internal capsule by XSECC treatment. Statistical analysis showed that the myelin damage grade was significantly increased in the external capsule and internal capsule of the model group when compared with the sham group on the 30th day after stroke (

The effects of XSECC on the histopathological changes in white matter (corpus callosum, external capsule and internal capsule) on the 30th day after stroke. (A) Representative HE and LFB staining images in the corpus callosum, external capsule and internal capsule from the sham, model and XSECC group rats. The well-defined white matter structures, including corpus callosum, external capsule and internal capsule, were seen in the sham group on HE and LFB staining images. However, the loss of white matter integrity in the external capsule and internal capsule were observed in the model group on both HE and LFB staining images. HE staining showed that rats in the model group had vacuolation indicated by arrows in the external capsule and internal capsule. LFB staining displayed severe myelin disruptions indicated by arrows in the external capsule and internal capsule in the model group. By contrast, vacuolation and myelin damage were alleviated in external capsule and internal capsule by XSECC treatment. Images were visualized under an optical microscope at 400× magnification. (B) The statistical analysis of the myelin damage grade on the 30th day after stroke.
XSECC Alleviated Histopathological Damage in Gray Matter After Stroke
The histopathological changes in gray matter (cortex, striatum and thalamus) on the 30th day after stroke were evaluated by HE staining (Fig. 7A). It showed that tissues and cells with normal histomorphology in the cortex, striatum and thalamus in the sham group. By contrast, the brain tissues in the model group exhibited obvious pathological abnormalities with loosely arranged neurons, pyknotic nucleus and loss or dark color staining in the cortex, striatum and thalamus (Fig. 7A). However, these histopathological alterations were reversed following treatment with XSECC (Fig. 7A). Quantitative analysis revealed that the number of surviving neurons in the cortex, striatum and thalamus was markedly reduced in the model rats on the 30th day after stroke, as compared with the sham rats (

The effects of XSECC on the histopathological changes in gray matter (cortex, striatum and thalamus) on the 30th day after stroke. (A) Representative HE staining images in the cortex, striatum and thalamus from the sham, model and XSECC group rats. The tissues and cells with normal histomorphology were observed in the cortex, striatum and thalamus in the sham group. By contrast, the brain tissues in the model group exhibited obvious pathological abnormalities with loosed arranged neurons, pyknotic nucleus and loss or dark color staining as indicated by arrows in the cortex, striatum and thalamus. However, these histopathological alterations were reversed following treatment with XSECC. Images were visualized under an optical microscope at 400× magnification. (B) The statistical analysis of the number of surviving neurons in the cortex, striatum and thalamus.
Treatment with XSECC significantly increased the number of surviving neurons in the cortex, striatum and thalamus compared with the model group on the 30th day post MCAO (
Discussion
Ischemic stroke is accompanied by structural deformation and functional deficits
29
. DTI enable
In white matter, FA, the ratio of the anisotropy of water molecules to the entire dispersion tensor 33 , is closely related to the density and parallelism of fibers and the integrity of myelin, and can be used to reflect the integrity of white matter 22,34,35 . Therefore, it is widely used in the evaluation of white matter damage in human and animal models 24,36,37 . In this study, the decrease of FA caused by nerve fiber damage, axon degeneration and demyelination 26,38 in the corpus callosum only had obvious statistical significance on the 3rd day after stroke, while it had obvious statistical significance in the external capsule and internal capsule on the 3rd, 7th, 14th and 30th days after stroke. The results revealed that the corpus callosum was seriously damaged in the early stage while the external capsule and internal capsule were badly damaged at all stages of ischemic stroke. ADC is the diffusion rate of water molecules 39 , and a reduced ADC value means increased limits in tissue. λ// is the diffusion rate that parallels the axons and is mostly used to reflect the integrity of the axons 40 . In early ischemia, λ// will be significantly reduced due to severe cell edema and axonal damage 40 ; With ischemic time prolonging, λ// will increase due to the gradually aggravated vasogenic edema and the loss of nerve fiber 40,41 . λ⊥ is the diffusion rate perpendicular to the axons and the change of λ⊥ can reflect the pathological changes of demyelination 16,39,42 . Generally, λ⊥ will increase at the time of demyelination, myelination disorders and nerve fiber loss 39,43 . In our study, FA was found to be decreasing accompanied by the increases of λ//, λ⊥ and ADC in the external capsule on the 3rd day after stroke, which was caused by the severe vasogenic edema associated with demyelination, impaired axonal injury and loss of fibers 44 that resulted in an increased diffusion rate of water molecules 44 . FA decreased and ADC increased obviously in the external capsule on the 30th day after stroke, which was the result of the decrease of water molecule anisotropy and the increase of the diffusion rate, caused by the loss of fiber bundle and liquefaction and necrosis in a large area of the tissue 44 . A decrease in FA value was linked with decreased λ// in the internal capsule on the 3rd day after stroke, which may be due to the severe cell swelling that led to a narrow gap between the myelinated fiber bundles 26 . After the administration of XSECC, the FA value in the corpus callosum, internal capsule and external capsule increased notably after stroke. The increased FA might arise from an increased number of aligned fibers or greater myelination 40,45 . Additionally, the increased FA was linked with the decreased λ//, λ⊥ and ADC on the 3rd day and reduced ADC on the 30th day in the external capsule, indicating that drug treatment could reduce the damage of white matter axon and myelin, and maintain the integrity of the white matter structure and function.
DTT, also known as fiber tractography, is a noninvasive imaging method to show the travel and spatial distribution of brain white matter fiber bundles three-dimensionally
In gray matter, the rFA, rADC, λ// and λ⊥ decreased in the striatum and rFA and λ// decreased in the cortex on the 3rd day after MCAO. The decrease in rFA was caused by the damaging of the cell membrane 46 and the decline in λ// and λ⊥ may be the result of severe cell swelling that limited the movement of water molecules 31 . On the 14th and 30th days post stroke, rFA in the striatum and cortex decreased significantly linked with increased rADC, λ// and λ⊥, which was caused by the necrotic liquefaction of infarcted tissue and increased permeability of the blood brain barrier with further development of vasogenic edema, allowing abundant proteins, electrolytes and water to enter the extracellular space and form cysts. This fluid-filled cystic area gave more space for the water free movement, resulting in a decrease in FA, and a significant increase in ADC, λ// and λ⊥ 47 . The ischemic injury was evident in the ipsilateral striatum and cortex following MCAO, while a secondary phase of injury also occurred in the ipsilateral thalamus remote from the primary injury in our study. The rFA in the ischemic far-thalamus was significantly lower on the 3rd, 7th, 14th and 30th days after stroke. The decreased rFA was accompanied by the increased rADC on the 14th and 30th days, which showed that obvious damage occurred in the distanced areas in the late stage of stroke, such as apoptosis, shrinkage and tissue liquefaction necrosis. Following treatment with XSECC, the rFA in the striatum, cortex and thalamus was significantly increased on 3rd, 7th, 14th and 30th days accompanied by the increased λ// on the 3rd day in the striatum and cortex, and reduced rADC and λ⊥ on the 30th day in the striatum, cortex and thalamus. The increased FA accompanied by other DTI parametric changes in gray matter potentially indicated that XSECC attenuated cell swelling and cell membrane damage in the early stage and mitigated tissue liquefaction necrosis in the late stage in gray matter after stroke.
Conclusions
The results of this study confirmed that DTI can accurately reflect the ultrastructure damage to white and gray matter after stroke. More importantly, we found that XSECC could alleviate the ultrastructural damage of white and gray matter after ischemic stroke, especially by reducing nerve cell damage and promoting the repair of axon and myelin injury, so as to promote the recovery of brain function.
Footnotes
Author Contributions
Jian Zhang, Shengpan Chen and Weilong Shi contributed equally to this work.
Ethical Approval
All procedures on animals in this study were in compliance with the ethical guidelines for researchers from the International Council for Laboratory Animal Science (ICLAS) and were approved by the Ethics Committee of Capital Medical University (No. 2013-X-16).
Statement of Human and Animal Rights
All experiments and animal rights were in compliance with relevant guidelines and regulations.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant nos. 81473745 and 81774381) and the Beijing Municipal Natural Science Foundation (Grant no. 7172034).
