Abstract
Stroke is one of the most devastating diseases worldwide. In recent years, a great number of studies have focused on the effects of microRNAs (miRNAs) on stroke and the results demonstrated that the expressions of miRNAs are associated with the prognosis of stroke. In the present study, we review relevant articles regarding miRNAs and stroke and will explain the complex link between both. The miRNAs participate extensively in the pathophysiology following the stroke, including apoptosis, neuroinflammation, oxidative stress, blood–brain barrier (BBB) disruption and brain edema. The information about the stroke–miRNA system may be helpful for therapeutic and diagnostic methods in stroke treatment.
Introduction
Stroke is one of the most devastating diseases, ranking as the second leading cause of death worldwide 1,2 . The World Health Organization (WHO) defines stroke as ‘rapidly developing clinical signs of focal or global disturbance of cerebral function, with symptoms lasting for at least 24 hours, or leading to death with no apparent cause other than vascular origin’ 3 . According to the WHO, approximately 15 million people suffer from strokes around the world, of which 5.5 million people die from them annually 4 . The incidence of stroke, especially in developing countries, is increasing and poses a great societal burden 5 .
Stroke can be divided into two categories: ischemic stroke and hemorrhagic stroke. The core incident of stroke is the interruption of blood supply to the brain tissues, which further causes the shortage of oxygen and nutrients and leads to brain injuries. The pathological mechanisms in the process of stroke and neuroprotective effects of various drugs have been comprehensively studied throughout the years, which consists of cellular apoptosis, inflammation, oxidative stress, and brain edema 6,7 . However, no drugs with specific efficacy are currently available.
In recent years, hundreds of genes have been found to be associated with the pathogenesis of stroke, but only a few were fully shown to influence its susceptibility. In addition, many studies focused on the effects of microRNAs (miRNAs) on stroke, as previous studies demonstrated that the expression of miRNA was related to the prognosis of this condition 8 –10 .
miRNAs are characterized by single-stranded non-coding (nc)RNA, formed with 20–24 nucleotides 11 . The initial forms are primary miRNA transcripts (pri-miRNAs), which are transcribed from genomic DNA under the help of RNA polymerase II 12 . Then, the pri-miRNAs are transformed to functional secondary structures that contain stem-loops consisting of inverted repeats 12,13 . Precursor miRNAs (pre-miRNAs) leave from the pri-miRNAs which are cleaved by the endonuclease Drosha (also known as ribonuclease III) 14,15 . Next, pre-miRNAs are exported into the cytoplasm by exportin-5 with a Ran-GTP-dependent mechanism. Finally, pre-miRNAs are cleaved by the RNase III enzyme (Dicer) to become mature miRNAs 16 .
miRNA was first reported by Lee and colleagues in Caenorhabditis elegans (C. elegans) in 1993 17 . It exhibits its functions via mRNA degradation or translational repression 18 . The function of miRNA was initially identified in original tissue samples. Recently, the discovery of circulating fetal nucleic acids in maternal plasma 19 as well as detecting circulating extracellular miRNAs in the blood 20 suggests a broad opportunity for developing the use of circulating miRNAs as biomarkers for non-invasive molecular diagnostics. Circulating miRNAs remain rather stable even after suffering from rugged conditions, such as extreme temperatures, strong acids or alkalis, and long-term preservation 21,22 . In addition, the functions of miRNAs are also involved in the modulation of a variety of cellular processes and pathophysiology such as neuronal survival, differentiation, metabolism, death/apoptosis, cerebrovascular diseases and cancers 23 –28 .
miRNAs have been reported to play key roles in the regulation of molecular processes after stroke 29 –32 . The present study displays the complex relationship between miRNAs and stroke (Fig. 1). Therapeutic and diagnostic methods in stroke treatment may benefit from information about the stroke–miRNA system.
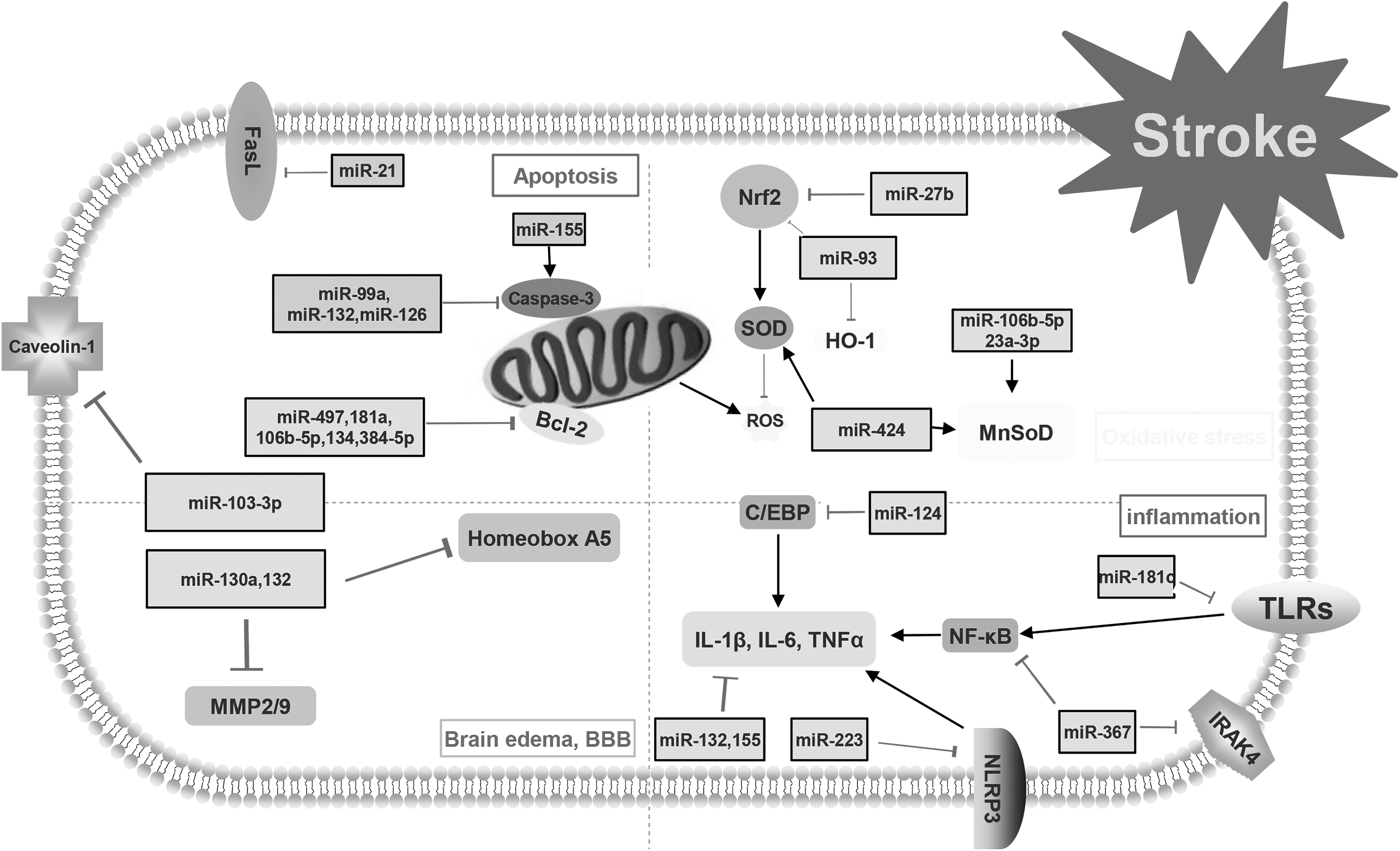
This figure demonstrates the mechanisms of post-stroke pathophysiology. Apoptosis, neuroinflammation, oxidative stress and blood–brain barrier (BBB) disruption with brain edema are the main avenues that research tries to focus on to decrease the consequences of stroke. Different types of microRNAs and their targets are involved in the pathophysiology of stroke.
Origin of miRNA Identified in Patients with Stroke
The functions of miRNA were initially identified in original tissue samples. Most of the studies focused on the relationships of miRNAs in brain tissues with underlying mechanisms of stroke, including cellular apoptosis, neuroinflammation, and oxidative stress 33 –36 . In recent years, research has focused on the profiles of circulatory miRNAs. Whole blood and plasma could both be adopted as sources of circulatory miRNAs (Table 1). Tan and colleagues firstly evaluated the expression of miRNAs between healthy individuals and individuals diagnosed with cerebral ischemia using microarray analysis of blood specimens and selective TaqMan Quantitative polymerase chain reaction (qPCR) of miRNAs 37 . The function of miR-25, miR-125b-2, miR-125b-627, miR-125b-27a, miR-125b-488, miR-145 has been explored in blood in patients with cerebral ischemia. These miRNAs might be important biomarkers for diagnosis and treatment of stroke. In addition, Wang and colleagues studied circulating miRNAs in plasma from patients with acute stroke and those with non-stroke diseases to test whether circulating miRNAs in plasma could serve as biomarkers in diagnosing and treating stroke 38 . Overall, some miRNAs have been identified to be promising peripheral biomarkers, including miR-106b-5p, miR-126, miR-30a, miR-4306, let-7b 38,39 . Wang and colleagues demonstrated that high level of miR-130a in serum contributed to serious brain edema and poor prognosis after acute intracerebral hemorrhage (ICH) 40 . Wang and colleagues compared the expression of circulating microRNAs in blood and hematoma samples. The results showed that the levels of 59 miRNAs obviously decreased after ICH and the Hsa-miR-21-5p were obviously downregulated in both peripheral blood and hematoma samples after ICH 41 .
The miRNAs in the blood and CSF of stroke patients.
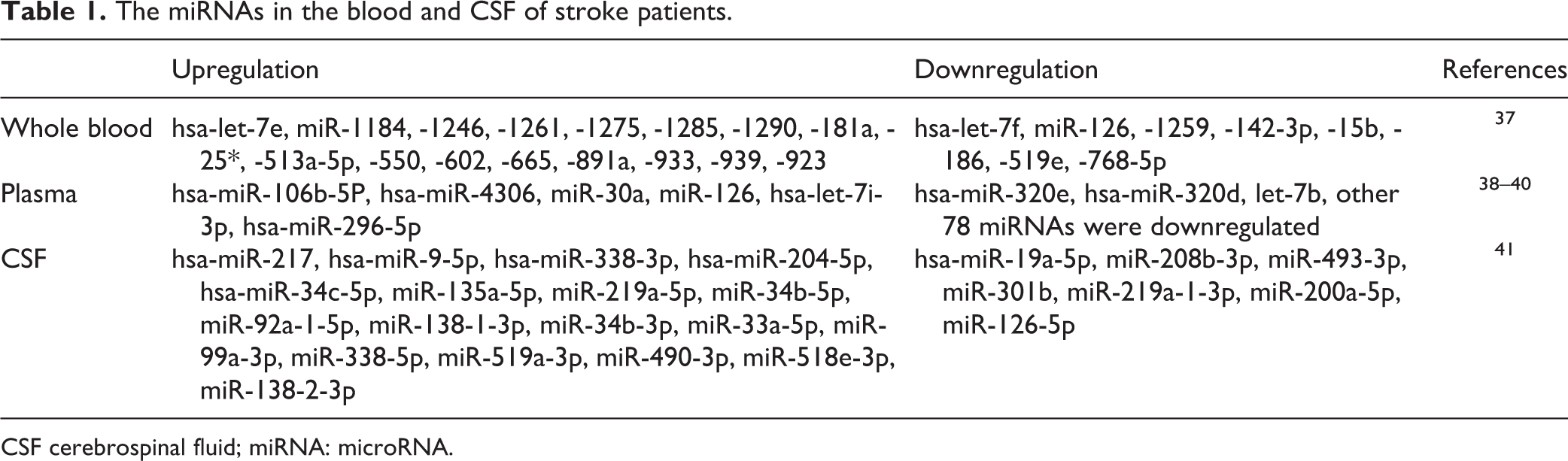
CSF cerebrospinal fluid; miRNA: microRNA.
In addition, other studies evaluated the expression profiles of miRNAs in cerebrospinal fluid (CSF) after stroke. Iwuchukwu and colleagues evaluated the miRNA profiles in cerebrospinal fluid of patients with ICH. Their results showed that 25 miRNAs display significant fold changes. The highly expressed miR-204-5p (2075-fold), miR-125b-5p (108-fold), miR-9-5p (299-fold), miR-338-3p (146-fold), miR-187-3p (21-fold), and miR-9-3p (42-fold) in CSF were associated with the regulation of matrix metalloproteinase-9 (MMP-9), interleukin (IL)-1b, IL-6, occludin, and selectin E, among others. miRNA profiles in CSF were physiologically contiguous with profiles in brain extracellular fluid 42 . However, only a small number of studies have been reviewed. This area of stroke research would benefit from more studies investigating the function of miRNAs in CSF of patients with stroke.
The Role of miRNAs in Stroke
The expression of miRNAs is activated in stroke and plays an important role in the regulation of prognosis of patients with stroke. The biological functions of miRNAs are quite complex and should be discussed within a specific condition. On one hand, activation of miRNAs could contribute to attenuating neurological deficits. miRNA 21, miR-99a and miR-497 were reported to reduce the ischemic volume and protect neuronal cells from apoptosis, thus improving the neurological functions in rat and in vitro model of ischemic stroke 43 –45 . Except for the functions mentioned above, overexpression of miR-424 and miR-let-7c-5p could also suppress the activation of microglia in cerebral ischemia 46 . As for hemorrhagic stroke, overexpression of miR-132, miR-126, miR-103 and miR-367 could attenuate neurobehavioral and neuropathological changes via improving BBB integrity, suppressing neuroinflammation and reducing neuronal apoptosis 47 –50 . Additionally, miR-210 overexpression could promote angiogenesis and neurogenesis in mouse brain and help to repair the injured brain tissues 51 . On the other hand, the role of miRNA could also be negative. Liu and colleagues found that silencing miR-155 expression could increase the proliferation, migration, and tube formation ability of human brain micro-vessel endothelial cells via decreasing cellular apoptosis and reactive oxygen species (ROS) production 52 . As for hemorrhagic stroke, Xu and colleagues found that miR-27b inhibition could alleviate neurological deficits via suppressing neuroinflammation and reducing cell death. In addition, miR-155 and miR-124 were reported to play roles in the polarization of macrophages 53 . miRNA was also involved in the regulation of synaptic plasticity. miR-134 could help to remodel neuronal structures via repressing the translation of Limk1-mRNA, a protein kinase that influences dendritic spine development 54 .
However, some types of miRNA were reported to exert multiple functions. For example, Xie and colleagues showed that miR-181a could significantly improve cellular survival in vitro by suppressing inflammation responses in monocytes and macrophages 55 . In the report of Moon, however, it was demonstrated that the inhibition of miR-181a could reduce forebrain ischemia-induced neuronal apoptosis 56 .
Underlying Mechanisms of the Regulation of miRNAs in stroke
miRNAs and Apoptosis or Cell Death
Apoptosis is one type of cell death characterized by energy dependence and programmed cell death 57 . The term ‘apoptosis’ was first described by Kerry and colleagues 58 . Apoptosis is of vital importance to normal physiological metabolism and growth and development. It maintains hemostasis by scavenging aging or damaged cells, and regulates the immune system by removing defective and excessive cells 59,60 . However, uncontrolled apoptosis may result in various pathological processes of different diseases including cancers, Alzheimer’s disease, and stroke 61,62 . The apoptosis process is initially triggered either by the extrinsic or intrinsic pathway. The extrinsic pathway is conducted via the activation of cell surface death receptors, including tumor necrosis factor (TNF)-α, Fas and TNF-related apoptosis-inducing ligand receptors, while the intrinsic pathway is related to the mitochondrial signaling pathway 63,64 . Stroke induces a mass influx of Ca2+ into the cell, which leads to the release of mitochondrial cytochrome c (Cytc) or apoptosis-inducing factor (AIF) 65 . The released Cytc binds to the apoptotic protease-activating factor-1 and procaspase-9 to form an apoptosome which activates caspase-9 and subsequently, caspase-3, leading to the damage of nDNA and finally to cell death. What’s more, AIF is translocated to the nucleus and induces large-scale (50 kb) DNA fragmentation and cell death in a caspase-independent manner 66 .
Many studies have demonstrated that the expression of miRNA could modulate post-stroke neuronal survival by regulating the levels of target genes 67,68 . Liu and colleagues reported that miR-298 was upregulated in both brain and blood samples in both experimental models of cerebral ischemia and ICH 69 .
miR-21 is reported to be a potent antiapoptotic factor in biological systems. Buller and colleagues evaluated the expression level of miR-21 in vivo and in vitro. The results demonstrated that the level of miR-21 significantly increases after ischemic stroke through suppressing neuronal cell death through reducing Fas ligand (FasL)G, an important cell death-inducing ligand 43 . miR-155 was also reported to exert important roles in modulating cellular apoptosis by regulating caspase-3 expression. Knockdown of miR-155 significantly reduced apoptosis of brain micro-vessel endothelial cells 52 . miR-99a was reported to not only inhibit the activation of pro-caspase-3 and the expression of caspase-3, but also reduce neuronal apoptosis after ischemic stroke. Moreover, following cerebral Ischemia/Reperfusion (I/R), miR-99a reduced neuronal damage via regulation of the cell cycle and cellular apoptosis, which implied that miR-99a could be used as a new therapeutic agent targeting neuronal cell cycle re-entry following ischemic stroke 44 . In addition, the bcl-2 family plays a key role in the regulation of apoptosis. miR-497, miR-181a, miR-106b-5p, miR134, miR-384-5p were all reported to increase apoptosis by decreasing the level of bcl-2 proteins 45,55,56,70,71 .
Following hemorrhagic stroke, miR-132 was reported to exert a neuroprotective effect by decreasing neuronal death in ICH mice. The mice overexpressing miR-132 were less likely to suffer from neurological deficits 47 . Overexpression of miR-126 with lentivirus exhibits a protective role in ICH and antiapoptotic effects via downregulating the level of casepase-3 48 . In addition, Wang and colleagues showed that miR-103-3p was obviously upregulated in the experimental model of subarachnoid hemorrhage. miR-103-3p exerted its neuroprotective effect by reducing the neuronal death via decreasing caveolin-1 49 .
miRNAs and Neuroinflammation
Inflammation is a complex immune response following an injury. Under normal conditions, inflammation helps to scavenge necrotic cells or tissues, and initiates the tissue repair process 72 . However, excessive activation of immune responses is harmful to the organisms and can cause injury 73 . Recent studies have showed that neuroinflammation was a key factor in determining prognosis after stroke 50,74 –76 . Either ischemic or hemorrhagic stroke triggers the activation of microglial and the releasing of inflammatory factors 77,78 . The activation of microglial and subsequent inflammatory factors, such as TNF-α contribute to the progression of brain injury 53,79–80 . Additionally, there are also some peripherally-derived cytokines produced and secreted by natural killer cells, mononuclear phagocytes, T-lymphocytes and polymorpho-nuclear leukocytes which participate in neuroinflammation after stroke 81–82 .
Many miRNAs target several genes participating in the regulation of neuroinflammation 83 –85 . Zhao and colleagues demonstrated that lentiviral overexpression of miR-424 could significantly decrease brain injury after ischemic stroke through suppressing microglia activity 46 . In addition, miR-let-7c-5p was also reported to exert neuroprotection against neuroinflammation after ischemic stroke by inhibiting the activation of microglial and translational repression of caspase-3 86 . miR-124 is almost exclusively expressed in the central nervous system and referred to as ‘brain-specific miRNA’ 87–88 . In 2009, Laterza and colleagues showed that miR-124 was upregulated in plasma after brain injury (induced by middle cerebral artery occlusion) 88 . Ponomarev and colleagues found that miR-124 directly inhibited CCAAT/enhancer-binding protein alpha (C/EBP-α) and its downstream factor PU.1, promoting microglia quiescence. It also suppressed experimental autoimmune encephalomyelitis (EAE) by deactivating macrophages. In addition, Toll-like receptors (TLRs) are also reported to play important roles in neuroinflammation after stroke 89 –91 . Zhang and colleagues showed that miR-181c suppressed the expression of TLR4 by binding to its 3’UTR, therefore reducing the level of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and the production of downstream proinflammatory factors, which could be regarded as one potential therapeutic target for the treatment of ischemic stroke 92 .
As for hemorrhagic stroke, the mice with miR132 overexpression had better prognosis compared with the control group. miR132 overexpression suppressed the activation of microglia and the expression of proinflammatory cytokines 47 . In the experimental model of ICH, Yuan and colleagues demonstrated that miR-367 inhibited the level of IRAK4 through directly binding its 3’-untranslated region. In addition, miR-367 also suppressed the activation of NF-κB and the production of its downstream proinflammatory factors. miR-223 was reported to downregulate NLRP3 and inhibit inflammation through caspase-1 and IL-1beta, thus to improve neurological functions 93 .
The role of miRNAs in the modulation of microglia and microphage polarization is also important for its anti-inflammatory effects in the brain. Some studies reported that miR-155 promotes skewing toward the M1 phenotype by targeting M2-associated genes. miR-155 clearly targeted multiple genes associated with the M2 phenotype and reduced the expression of M2-asociated proinflammatory factors such as Arg-1, IL-10, IL13Rα1 and CD206 94,95 .
MicroRNA and Oxidative Stress
The oxidative stress, which is mainly caused by the overbalance of pro-oxidants (ROS/RNS) and/or deficiency of antioxidant systems in cells, takes part in the pathogenesis of many diseases 96 –98 . The formation of free radicals and ROS during stroke involves several mechanisms, including high stimulation of N-methyl-D-aspartic acid (NMDA) glutamate receptors 99 , Ca2+ overload, mitochondrial dysfunction 100 –102 , and neuronal nitric oxide synthase (nNOS) activation 103 . Oxidative damage is a basic mechanism of brain injury after stroke. The brain is vulnerable when exposed to oxidative stress because of its highly oxygenated characteristics, with high levels of peroxi-disable lipids, low levels of antioxidants, and high iron content 104 . Several antioxidant and detoxifying enzymes such as glutathione peroxidase, superoxide dismutase (SOD), glutathione reductase, and glutathione-S-transferase, which maintain redox homeostasis in the brain, have been widely studied 105,106 . Nuclear factor erythroid-2-related factor-2 (Nrf2) was reported to have neuroprotective effects against brain injuries after stroke, such as hydrogen peroxide (H2O2) exposure, Ca2+ overload situations, and oxidative glutamate excitotoxicity via activation of antioxidant response elements 107 .
It was reported that 85 miRNAs could regulate the level of Nrf2 mRNA 108 . miR-93 was reported to inhibit the expression of Nrf2 and hemeoxygenase-1 (HO-1) after ischemic stroke 109 . In addition, for cerebral ischemia, miR-424 could decrease infarct volume via reducing the level of ROS and malondialdehyde in the cortex, and increasing manganese SOD (MnSOD) as well as extracellular SOD. Similarly, miR-424 could significantly reduce H2O2-induced injury in neuronal cultures, increase cell viability and MnSOD activity and decrease the level of lactate dehydrogenase leakage and malondialdehyde 110 . In addition, miR-106b-5p and miR-23a-3p could exert a neuroprotective effect against post-ischemic oxidative damages by increasing the expression of MnSOD 111–112 . In addition, miR-145 could inhibit the expression of SOD2 after ischemic stroke 113 .
Xu and colleagues showed that the inhibition of miR-27b could alleviate brain injury and upregulate the expression of Nrf2, Hmox1, SOD1 and Nqo1 after ICH via the Nrf2/ARE pathway 114 . The gene could be a potential therapeutic target in treating ICH. However, limited studies have been reported regarding the anti-oxidative effects of miRNAs in hemorrhagic stroke. Further research should be carried out to explore the roles of miRNAs in hemorrhagic stroke.
miRNA and Brain Edema
BBB is a continuous, non-fenestrated system which regulates the movement of many particles and cells, such as ion, toxins, and inflammatory cells. Any factors disrupting the BBB would deteriorate the condition of neurological diseases, including stoke, traumatic brain injury and neurodegenerative diseases 115–116 . The most common complication of BBB disturbance is vasogenic brain edema, which is reported to be related to the early expression of matrix metalloproteinase (MMP)-2,9 after brain injury 117–118 .
In the rat model of ICH, the expression of miR-130a was obviously upregulated in serum and perihematomal samples, which was consistent with the changes of brain edema after the induction of ICH. The application of miR-130a inhibitors could significantly attenuate brain edema, reduce BBB permeability, and improve neurological functions via increasing caveolin-1 and decreasing MMP-2/9. Moreover, Wang and colleagues also found the effects of miR-130a in rats with cerebral ischemia. Inhibition of miR-130a could alleviate brain edema, reduce infarct volume and BBB permeability, and enhance neurologic function by targeting Homeobox A5 119 . Similarly, Zhang and colleagues demonstrated that mice injected with lentivirus encoding miR-132 had significantly reduced brain edema and improved BBB integrity 47 . In the rat model of subarachnoid hemorrhage, miR-103-3p exerted its neuroprotective effects via increasing the level of caveolin-1 49 .
miRNA and Neurogenesis
Endogenous neurogenesis, the process of self-repairing, is increased after stroke. Neurogenesis is necessary for the neurological recovery for patients with ischemic or hemorrhagic stroke 120,121 .
Neurotrophic factors are small polypeptide molecules, which take part in cell proliferation, differentiation, migration and development of the nervous system. Previous studies have shown that neurotrophic factors such as nerve growth factor, brain derived neurotrophic factor (BDNF), ciliary neurotrophic factor, and glial-derived neurotrophic factor and insulin-like growth factor-1 (IGF-1) could reduce neuronal death and brain injuries 122–123 .
In the experimental model of cerebral ischemia, miR-Let7f was found to provide IGF-1-like neuroprotection 122 . Moreover, inhibition of miR-134 could significantly attenuate ischemic injuries via improving the expression of BDNF and Bcl-2. miR-30-5p and miR-107 could also regulate the expression of BDNF and be potential therapeutic targets in neuroprotection 124 . What’s more, endogenous neural stem cells (NSCs) and neural precursor cells (NPCs) could be activated and migrate to the injured area 125 . miR-21 could regulate the function of NPCs via Wnt and transforming growth factor (TGF)-β signaling pathways. In addition, miR-34a negatively regulated NPC proliferation after cerebral ischemia 126 . Furthermore, miR-126 could improve the neurorestorative effects induced with umbilical cord blood cells after stroke, according to studies completed in type-2 diabetic mice by Chen and colleagues 127 .
miRNA and Angiogenesis
The focus during the treatment of stroke should not only be placed on the regeneration of neural cells, but should also on the supporting tissues. This includes blood vessels to increase angiogenesis after stroke. Zhang and colleagues studied the structural changes after stroke, and they found that vascular volume was increased from 3% prior to stroke to 6% at 90 days after stroke 128 . miRNAs that regulate the process of angiogenesis could be regarded as potential therapeutic targets in the treatment of ischemic stroke 129 . Zheng and colleagues found that miR-210 played a key role in promoting angiogenesis after cerebral ischemia, partly via increasing the level of vascular endothelial growth factor (VEGF). In addition, Zeng and colleagues also showed that miR-210 could induce vascular endothelial cell migration and tube formation under hypoxia in vitro 51 . In the rat model of ICH, Ma and colleagues demonstrated that miR-129-5p could inhibit the HMGB1-RAGE signaling pathway and thus restrain the revascularization 130 . Overexpression of miR126 protected against ICH, which may be involved in the process of angiogenesis via upregulating the protein levels of VEGF-A 65 . Thus, promotion of angiogenesis could be regarded as a promising therapeutic approach for stroke via pharmacological modulation of these miRNAs.
Conclusions
In this review, we have introduced the basic knowledge of stroke and miRNAs. In addition, we further explored available concrete mechanisms of miRNAs in the post-stroke pathophysiological process to guide us in better understanding the processes involved in stroke pathology, including apoptosis, neuroinflammation, oxidative stress, BBB integrity, brain edema, neurogenesis, and angiogenesis (Table 2). With improved understanding comes the potential for a new therapeutic agent. However, several problems should not be ignored. First, the expression profiles of miRNAs in varied samples may change depending on the disease state, making it difficult to find appropriate endogenous controls. Second, the studies regarding the miRNAs in patients with subarachnoid hemorrhage (SAH) were limited. Lastly, more studies should also be carried out to explore the functions of miRNAs in CSF. Finally, till now, most studies were limited to experimental models while clinical research was scarce. More clinical studies should be carried out based on clinical medical ethics.
The roles of different miRNA in stroke.

BBB: blood–brain barrier; miRNA: microRNA
Footnotes
Acknowledgments
Sheng Chen, Jingwei Zheng, Weilin Xu and Liansheng Gao have equally contributed to this work as co-first authors.
Ethical Approval
This study was approved by the review board of the second affiliated hospital of Zhejiang univeristy.
Statement of Human and Animal Rights
Statement of Human and Animal Rights is not applicable.
Statement of Informed Consent
Statement of Informed Consent is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81500992), Natural Science Foundation of Zhejiang(LQ16H090002), Medical and health key project of Zhejiang Province (2016RCA015), China Postdoctoral Science Foundation (2017M612010) and National Natural Science Foundation of China (81701144).
