Abstract
Growing evidences suggest that stroke is a systemic disease affecting many organ systems beyond the brain. Stroke-related systemic inflammatory response and immune dysregulations may play an important role in brain injury, recovery, and stroke outcome. The two main phenomena in stroke-related peripheral immune dysregulations are systemic inflammation and post-stroke immunosuppression. There is emerging evidence suggesting that the spleen contracts following ischemic stroke, activates peripheral immune response and this may further potentiate brain injury. Whether similar brain–immune crosstalk occurs in hemorrhagic strokes such as intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH) is not established. In this review, we systematically examined animal and human evidence to date on peripheral immune responses associated with hemorrhagic strokes. Specifically, we reviewed the impact of clinical systemic inflammatory response syndrome (SIRS), inflammation- and immune-associated biomarkers, the brain–spleen interaction, and cellular mediators of peripheral immune responses to ICH and SAH including regulatory T cells (Tregs). While there is growing data suggesting that peripheral immune dysregulation following hemorrhagic strokes may be important in brain injury pathogenesis and outcome, details of this brain-immune system cross-talk remain insufficiently understood. This is an important unmet scientific need that may lead to novel therapeutic strategies in this highly morbid condition.
Keywords
Introduction
Historically, stroke was thought to be a single-organ disease where the most important pathophysiologic processes occur within the central nervous system (CNS) and that these CNS processes determine stroke outcomes. Growing evidence now suggests that stroke is in fact a systemic disease with numerous extra-CNS manifestations including neurogenic cardiac dysfunction, 1 lung injury 2 and systemic immune dysregulations. 3 Stroke therapeutic strategies to date have focused on treating these two compartments separately – targeting the CNS for brain injury and treating systemic complications as their own entity. A paradigm shift is occurring with the emergence of data suggesting that the brain–body crosstalk in stroke may be an important part of the overall pathophysiology and may in fact impact stroke outcome.4,5
The brain–immune system interaction has been extensively studied in ischemic stroke animal models and human cohorts. Immuno-dysregulations following ischemic stroke include upregulation of systemic inflammatory response as well as immuno-suppression-associated post-stroke infections. 4 Developing systemic inflammatory response syndrome (SIRS) after ischemic stroke is associated with higher National Institutes of Health Stroke Scale (NIHSS) on hospital admission, worse Glasgow Coma Scale (GCS), and worse subsequent stroke outcome and higher mortality.6,7 Post-stroke immunosuppression, often characterized by lymphopenia upon stroke presentation, is strongly associated with increased susceptibility for post-stroke infections.4,8,9 Furthermore, ischemic stroke may be associated with an autoimmune process where systemic T-helper cell type 1 (Th1) reacts to brain-specific antigens such as myelin basic protein (MBP), which may further compound brain injury and worsen stroke outcomes.10,11 Taken together, these data suggest that the peripheral immune system dysregulation is not simply a sequelae of ischemic stroke but an active and important process that further promotes brain injury.
While much is known about ischemic stroke-related immuno-dysregulations, relatively little is known about the systemic immune and inflammatory reactions in hemorrhagic strokes. In this review, we examine the potential roles and impact of systemic inflammatory and immune responses in primary hemorrhagic strokes – intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH). Specifically, we will examine potential organ, cellular, and molecular mediators of immune function and human data on the effects of systemic inflammation and immune dysregulations in ICH and SAH complications and outcomes.
The spleen – An immune system checkpoint in ischemic stroke
The spleen is a major reservoir for peripheral immune cells. Splenic activation is associated with altered inflammatory responses, immune system dysregulation, neuroinflammation, increased infection risk and impaired neurological recovery.12,13 Pre-clinical studies showed that the spleen acutely decreases in size and changes its cellular composition upon ischemic stroke onset and the degree of splenic contraction positively correlated with stroke volume.14,15 Obliteration of splenic response pre- or post-stroke either by removing or radiating the spleen reduces infarct size and enhanced recovery in experimental ischemic stroke models.16,17 Human data corroborated the positive association between splenic contraction and initial stroke severity. 18
Pre-clinical ischemic stroke studies using middle cerebral artery occlusion (MCAO) models consistently show that spleen contraction in acute stroke is followed by an increase in number of spleen-derived immune cell first in the blood and then in the brain, suggesting possible CNS migration of peripheral immune cells.19,20 Different immune cells appear to migrate at different time points. Spleen-derived natural killer (NK) cells and markers of NK cell activation such as CD69 and interferon gamma (IFN-γ) peak in the brain one day after stroke onset,
21
while spleen-derived lymphocytes infiltrate the brain later, peaking at 96 h post MCAO.
19
Spleen-derived monocyte/macrophage enters the circulation and then the brain as early as 48 h following MCAO.15,19 As the spleen shrinks, spleen-derived monocytes decrease in number but the ratio of pro-inflammatory Ly-6Chigh to anti-inflammatory Ly-6Clow monocytes increases in the blood and subsequently the brain, reflecting transformation into a pro-inflammatory milieu.
20
Splenectomy decreased monocyte/macrophage infiltration into the ischemic brain and alleviated brain injury, suggesting these spleen-derived monocytes play an important role in ischemic stroke brain injury.20,22 Taken together, these data suggest that splenic contraction following ischemic stroke leads to mobilization and release of peripheral immune cells from this reservoir. These peripheral immune cells then infiltrate the CNS and may participate in brain injury processes or affect post-stroke recovery23,24 (Figure 1).
Peripheral Immune System Responses in Hemorrhagic Strokes: Brain-Body Interaction.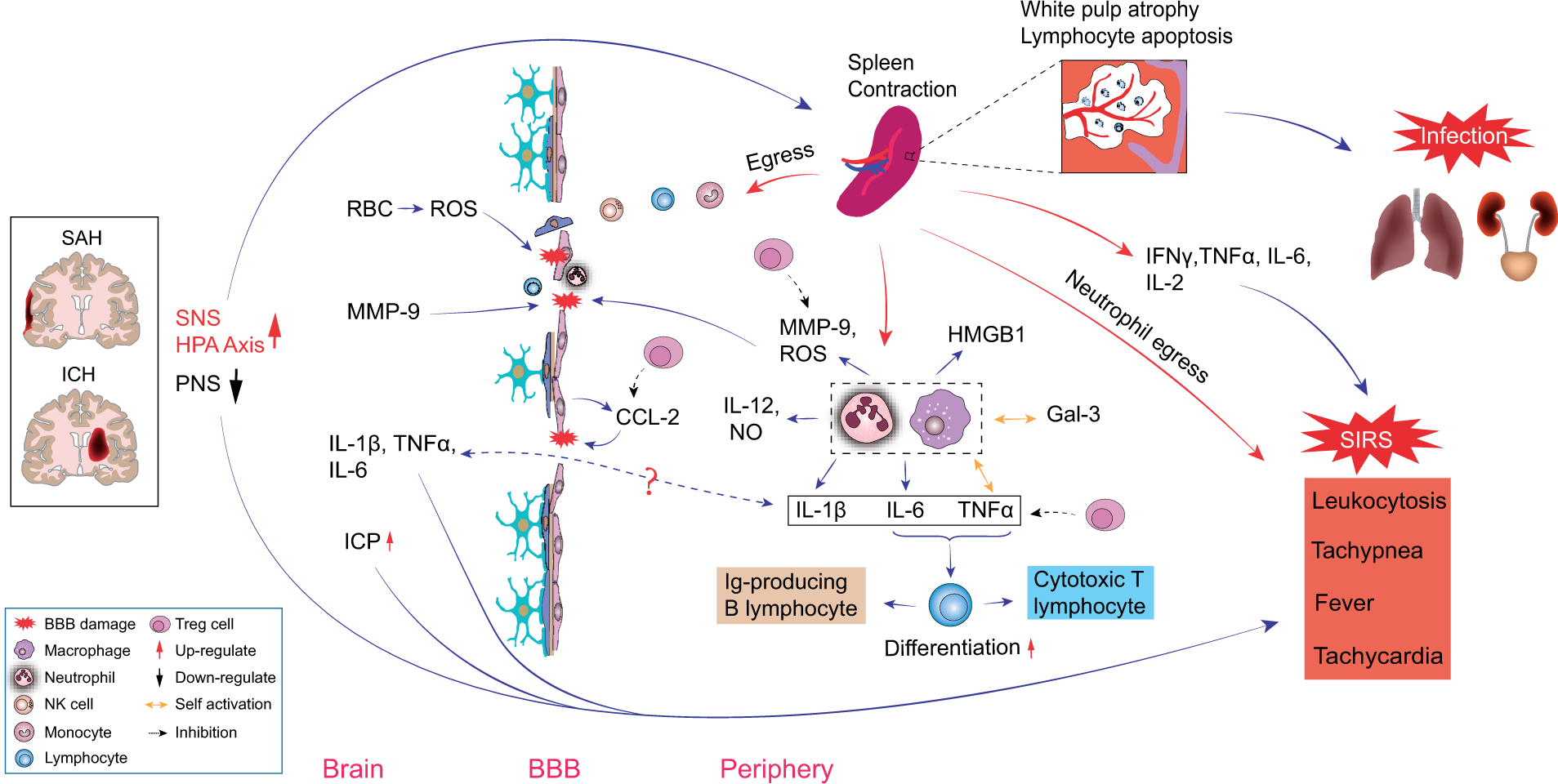
Spleen contraction following ischemic stroke also significantly alters the balance of pro- and anti-inflammatory mediators in the peripheral immune system. Within 6–22 h post MCAO, the spleen develops an inflammatory phase when activated splenocytes produce significantly higher levels of pro-inflammatory cytokines including tumor necrosis factor (TNF)-α, IFN-γ, interleukin (IL)-2, IL-6 and C-C motif chemokine ligand 2 (CCL2). 25 This is followed by a phase of immunosuppression at 22 h to four days post MCAO when both the spleen and the thymus develop dramatic reduction in cellularity, spleen-derived T lymphocyte becomes less responsive to mitogens, and splenocytes express more anti-inflammatory cytokine IL-1025 and less TNF-α, IFN-γ and IL-6. 15 At the same time, B lymphocytes numbers contract in both the spleen and the blood, while Regulatory T cell (T reg) numbers markedly expand. Tregs are a specialized subgroup of T lymphocyte that may regulate the immune system, inhibit inflammation and autoimmunity and possibly reduce immune-surveillance.26–28 T reg populations expand both within the spleen and in blood 96 h following MCAO. 15 While the mechanism of post-stroke immunosuppression and apparent susceptibility to infection is not known, it is postulated that splenic immune cell changes play an important role in post-stroke immunosuppression.
The spleen and peripheral immune response in hemorrhagic strokes
The spleen in ICH – Animal models
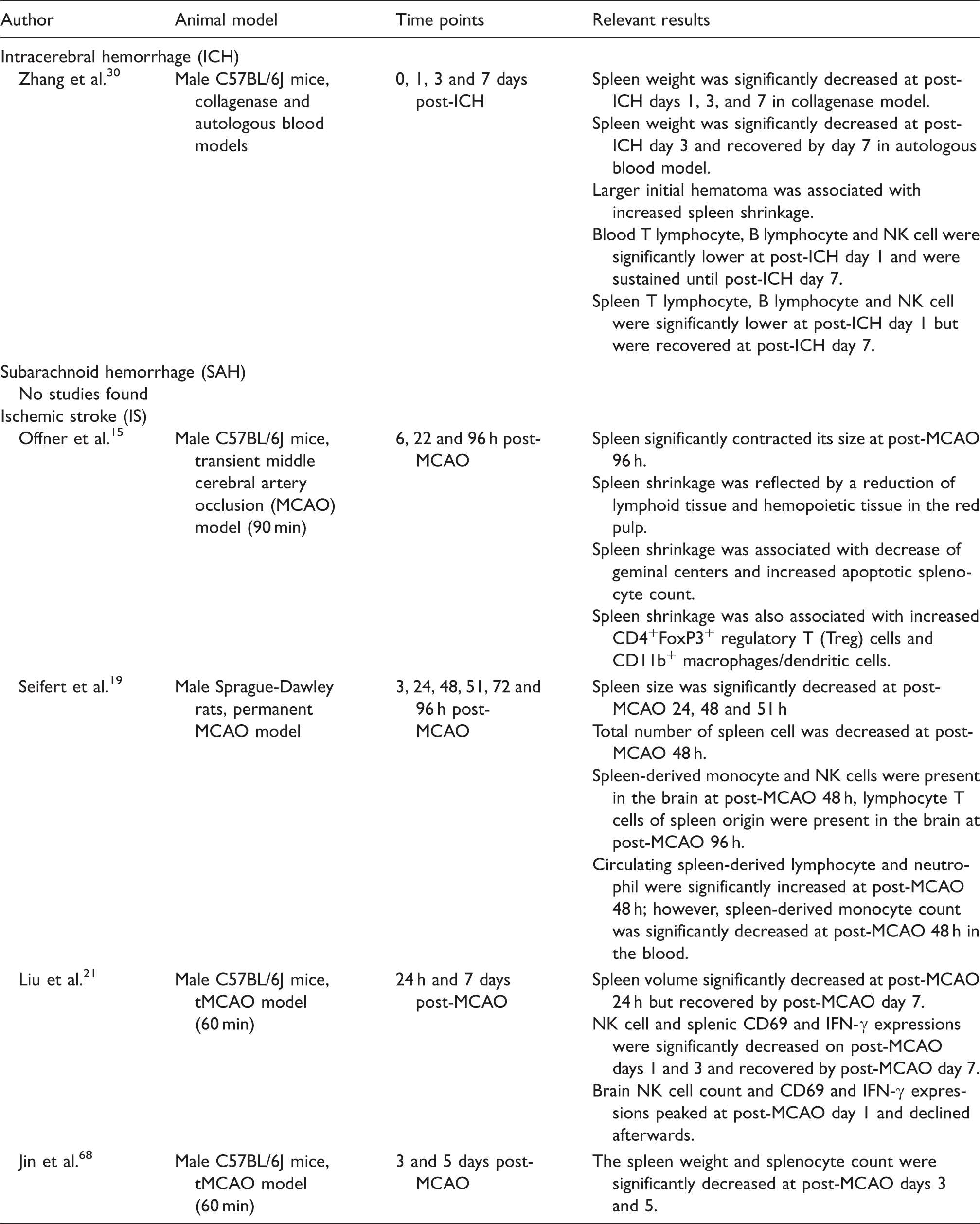
Compared to ischemic stroke, much less is known about splenic changes following hemorrhagic strokes. Few preclinical studies address the relationship between ICH, the spleen, and the peripheral immune system (Table 1). Illanes et al. 29 examined changes in immune cell composition, cytokine profiles and infectious complications after ICH in an autologous blood mouse model using three different ICH sizes (10 μl, 30 μl, 50 μl). Large ICH resulted in marked decrease in leukocytes and lymphocytes and a 10-fold increase in monocyte population in the blood and the spleen. Meanwhile, circulating spleen-derived T and B lymphocytes, helper and cytotoxic T lymphocytes all decreased significantly regardless of ICH size. This study did not report spleen size changes but noted that mice with large ICH had significantly lower splenocyte count. A second study demonstrated that ICH leads to splenic contraction in both collagenase injection and in autologous blood injection mice ICH models, where larger ICH volume was associated with more splenic shrinkage in the collagenase model. 30 There is evidence that the spleen may mediate neuroprotective therapies in ICH. Using a rat collagenase ICH model, Lee et al. 31 observed that human neural stem cell IV infusion 2 h after ICH induction significantly attenuated TNF-α, IL-6 and nuclear factor-kappa B (NF-κB) upregulation in both the brain and in the spleen, but splenectomy prior to ICH induction eliminated beneficial effects of neural stem cell transplantation.
The spleen in SAH – Animal models
We found no studies that examined the spleen in SAH models.
Human data
Splenic changes following ischemic and hemorrhagic strokes – Pre-clinical data.
Splenic changes following ischemic and hemorrhagic strokes – Human data.
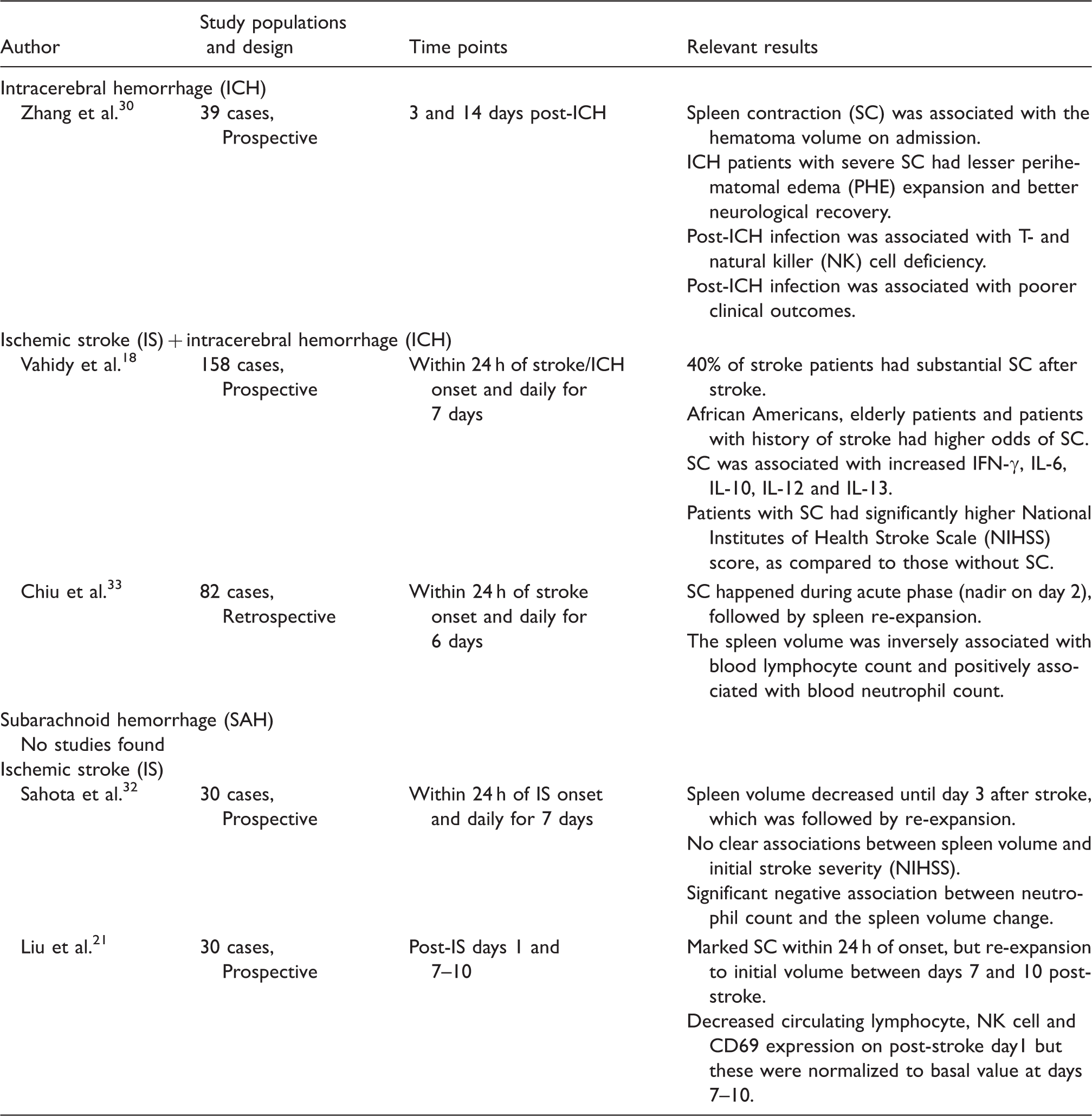
We found no human studies reporting on spleen size changes in SAH. Overall, there is a paucity of preclinical and clinical data on the peripheral immune and systemic inflammatory response in hemorrhagic strokes and their possible interplay with hemorrhagic stroke injury mechanisms and outcome.
Potential brain–spleen and brain–immune communications in hemorrhagic stroke
How do acute vascular brain injuries lead to changes in spleen size? Below are several postulated modes of brain–spleen communication:
The sympathetic nervous system/hypothalamus-pituitary-adrenal axis
Sympathetic nerve fibers make up 98% of total splenic innervation. 34 Rapid activation of the SNS/HPA axis following stroke onset is thought to be the first mode of communication between the CNS and the peripheral immune system.35,36 Human data from ischemic and hemorrhagic strokes showed that plasma catecholamines and cortisol levels acutely rise following stroke onset.30,37,38 In ICH animal models, pharmacologic inhibition of the SNS/HPA axis by β-adrenergic receptor blocker propranolol and glucocorticoid receptor (GCR) blocker RU486 reversed splenic contraction, confirming the key role of SNS/HPA in post-stroke splenic shrinkage. 30 While there are several ways the SNS/HPA can mediate splenic shrinkage, decreased splenic blood flow is not one of them. In a prospective human ICH study, serial abdominal magnetic resonance imaging (MRI) showed splenic capillary perfusion actually increased during splenic contraction. 30 Among all stroke syndromes, SAH is known to cause profound sympathetic activation and catecholamine surges.39–42 Theoretically, the SNS/HPA activation in SAH should result in splenic contraction. However, we found no studies that report splenic changes in SAH.
In addition to direct effects on spleen size, the SNS/HPA system activation may have spleen-independent effects on post-stroke immune deficiency and infection risks. Lymphocytes, NK cells and macrophages express β2 adrenergic receptor and GCR.43,44 Activation of these receptors promotes immune cell apoptosis, reduces cell proliferation and enhances cytokine production in animal ischemic stroke models.44–46 Activation of the SNS can also expand Treg cell population in the bone marrow and thereby directly affect immunomodulation. 47 Precisely how the SNS/HPA axis regulates spleen size and spleen-mediated peripheral immune response following ischemic and hemorrhagic strokes is not known. This remains an important knowledge and translational gap.
The parasympathetic nervous system
The parasympathetic nervous system (PNS) is another important mediator by which the brain may regulate the spleen and the peripheral immune system. 48 Though the PNS does not directly innervate the spleen, it may modulate splenic immune responses through its interfaces with the SNS. 49 PNS activation by the vagus nerve and by α7 nicotinic acetylcholine receptor (nAchR) stimulation inhibited inflammatory responses in an animal model of endotoxemia and sepsis. This effect was abolished in splenectomized animals, 50 suggesting that the spleen is essential in PNS modulation of systemic inflammation.
PNS activation appears to attenuate both systemic and neuroinflammation in ischemic 51 and hemorrhagic 52 stroke animal models. In a collagenase ICH rat model, PNS manipulation by intraventricular muscarine injection decreased high mobility group box-1(HMGB-1), TNF-α, IL-1β and Fas ligand (FasL) in both the brain and the spleen and improved neurological outcomes. 52 However, the PNS does not appear to play a role in post-ICH splenic contraction since PNS inhibition by α4β2 cholinergic blocker DhβE does not prevent splenic contraction following ICH. 30 In SAH models, PNS activation by intraperitoneal injection of α7 nAchR agonist decreased caspase-3 cleavage in neurons and improved neurological recovery in a rat filament-peroration model. 53 However, whether the spleen plays a role in PNS-mediated neuroprotection in SAH is not known. In human cohorts, PNS de-activation has been indirectly linked with unfavorable hemorrhagic stroke outcomes. Impaired baroreflex sensitivity (BRS) is a surrogate measure of PNS downregulation. Decreased BRS is independently associated with unfavorable 10-day outcome 54 and increased infection risks in ICH patients55,56 and with poor SAH outcome at three month. 57
Brain-derived antigens
The brain may directly activate the peripheral immune system by exposing it to CNS-specific antigens and generating an auto-immune response that potentiates secondary brain injury. CNS-specific antigen may come into contact with the peripheral immune system through the injured blood–brain barrier (BBB) or via CNS lymphatic drainage into lymph nodes.58,59 CNS-specific antigens such as microtubule-associated protein-2, N-methyl D-aspartate receptor subunit NR-2A and MBP have been found in the palatine tonsils and cervical lymph nodes from acute ischemic stroke patients, suggesting that the peripheral immune system can become exposed to CNS antigens possibly through lymphatic connections. 60 Ischemic stroke animal models found that brain-derived antigens can activate CD4+ and CD8+ T cells in the cervical lymph node and in the spleen 61 and shift the peripheral immune response into a Th1-like pattern which is associated with worse neurological outcome. 10 Compared with ischemic stroke, little is known about the effects of CNS-specific antigens exposure on peripheral immune activation in ICH and SAH where BBB injury is a prominent feature.
Peripheral immune response as a potential therapeutic target
Targeting the spleen
Growing data suggest that stroke-spleen-peripheral immune system interaction has significant impact on ischemic stroke outcome and possibly on hemorrhagic stroke outcomes. Targeting spleen-mediated immune responses may be a novel therapeutic approach. Stem cells from umbilical cord blood and from the bone marrow are known to preferentially migrate into the spleen where they persist for days.62,63 In ischemic stroke animal models, stem cell transplantation has successfully reversed splenic contraction, increased splenic Treg cell population, 62 and protected against ischemic brain injury.14,63,64 These changes are associated with subsequent anti-inflammatory effects including down-regulation of brain TNF-α, IL-1β, IL-6 and IFN-γ and up-regulation of anti-inflammatory cytokines such as IL-10 within the spleen. 62
In autologous blood injection ICH models, intravenous infusion of bone marrow-derived mononuclear cells (MNCs) reduced peri-hematoma neuroinflammation and brain edema. Similar to ischemic stroke, MNCs appear to preferentially accumulate within the spleen. 65 Neural stem cells administered at 2 h after ICH induction also appear to accumulate and persist in the spleen for up to 35 days after transplantation and promote neurological recovery by decreasing TNF-α and IL-6 in the brain and the spleen. 31 To date, there are no reports on whether functional or physical splenectomy alters the results of stem cell transplantation in ICH models.
In addition to stem cells, other potential therapies that can target the spleen include SNS inhibitors and statins. In ischemic stroke models, sympatholytic agents agmatine, 66 prazosin and carvedilol 67 have effectively reversed splenic contraction and reduced brain injury. When administered immediately following stroke, simvastatin reduced splenic atrophy and splenocyte apoptosis, inhibited brain IFN-γ expression and attenuated pulmonary bacterial infection risks in a mouse MCAO model. 68 Interestingly, neuroprotective effects of these therapeutic interventions were abolished in splenectomized animals,31,62,68 suggesting that the spleen is essential in the mechanisms of action in these neuroprotective strategies.
Statins and their potential neuroprotective effects have been extensively studied in SAH. Multiple pre-clinical 69 and some small human clinical trials70–72 all found statins to be beneficial in reducing vasospasm and improving outcome in SAH. However, recent large phase 3 randomized clinical trial demonstrated no outcome benefit from statin use in SAH. 73 Sympatholytic agents have also been tested in SAH. β-blockers suppressed the elevation of CSF IL-6 in animal models 74 and small SAH human cohort studies suggest that they improve outcome.75–77 However, these results have not been validated in sufficiently powered clinical trials.
Targeting regulatory T cells
There is growing data supporting Tregs as master immune regulators in multiple organ systems including the brain. In ischemic stroke, Tregs regulate inflammation, balance brain pro-inflammatory (TNF-α and IFN-γ) and anti-inflammatory (IL-10) cytokine production, restore immune homeostasis, and are potentially neuroprotective.26,27,78–81 Tregs may protect BBB integrity by inhibiting matrix metallopeptidase (MMP)-9 production 27 and preserve endothelial function by inhibiting endothelial CCL-2 production. 82 Within the CNS, Tregs can shift the microglia/macrophage response toward a more favorable M2-like phenotype and secret CCN3 to promote oligodendrocyte progenitor cell differentiation. 83
Emerging data suggest that Tregs may also be neuroprotective in ICH84,85 and SAH. 86 In autologous blood injection ICH models, boosting Tregs via either adoptive transfer or with CD28 super-agonist antibody stimulation alleviated ICH-induced inflammatory injury by inhibiting microglia activation and modulating microglia/macrophage polarization toward a more favorable M2 phenotype.84,85 In a rat SAH model with autologous blood injection into the cisterna magna, adoptive Treg transfer decreased inflammatory reactions by hindering the activation of toll-like receptor 4/nuclear factor-kappa B (TLR4/NF-κB) signaling pathway. 86 Data on Treg changes following ICH have been consistent across human and animal studies, though the number of available studies on ICH is far fewer than that in ischemic stroke. In both animal and human studies, the proportion of circulating Tregs positively correlates with ICH hematoma size, extent of spleen atrophy and ICH disease severity.29,87 These characteristics make Treg a very attractive potential therapeutic target in acute brain injury. In vivo, Treg population can be successfully expanded by low-dose stimulants such as IL-2, IL-2 + anti IL-2 antibody JES6-1 complex 88 and monoclonal antibodies against DR3. 89 This ability to manipulate Treg population is an important step towards translation of Treg into human therapy in ICH and SAH.
While Tregs possess numerous attractive characteristics as a novel therapeutic target, important caveats exist in its translation into human therapy. The reactive expansion of Tregs following brain injury is thought to be an endogenous mechanism to protect the brain from overactivated immune response, but precise mechanisms remain incompletely understood. Animal models do not fully capture the complexity and heterogeneity in human host conditions and baseline immune status. Moreover, there are important differences between human data and animal model observations on Treg responses in ischemic stroke. While animal studies consistently reported Treg population expand following ischemic stroke 15 and that SNS activation stimulates Treg expansion, 47 human studies found that Treg decreases immediately following stroke, reaching nadir after two days, and then slowly re-expands. 90 An important next step towards potential translation of Treg therapy is to better characterize the peripheral immune and Treg responses to human hemorrhagic and ischemic strokes. Another concern in Treg cell therapy is the potential for immunosuppression and post-stroke infections. In ischemic stroke, animal studies found that adoptive Treg cell transfer preserved peripheral lymphocyte populations and did not lead to immunosuppression.27,78 No such study has been conducted in ICH or SAH where infection risks are relatively high and associated with worse outcome. 91 Furthermore, most animal studies focused on short-term outcomes. The long-term effects of Treg cell therapy remain unknown. One particularly important consideration in Treg cell therapy is the potential risk for malignancy over time. Longer term animal studies and translational studies are clearly needed to further understand the potential effects of therapeutic Treg expansion or Treg transfer in human ischemic and hemorrhagic strokes.
Peripheral immune system response in hemorrhagic stroke outcomes in clinical populations
The lack of biomarkers that reflect disease progression and predict clinical outcomes remains a significant unmet need in hemorrhagic strokes. Mediators of systemic inflammation and immune response are one of the largest class of biomarkers that have shown associations with clinical complications and outcomes in hemorrhagic strokes. These signals suggest that systemic inflammatory response is a prominent feature and that the peripheral immune response may impact secondary brain injury and recovery in hemorrhagic strokes. Here, we review available data on cellular and molecular mediators of peripheral immune response and inflammation in hemorrhagic strokes.
Peripheral immune cells and hemorrhagic stroke
Blood total leukocyte count
Circulating leukocytes and hemorrhagic strokes outcomes.
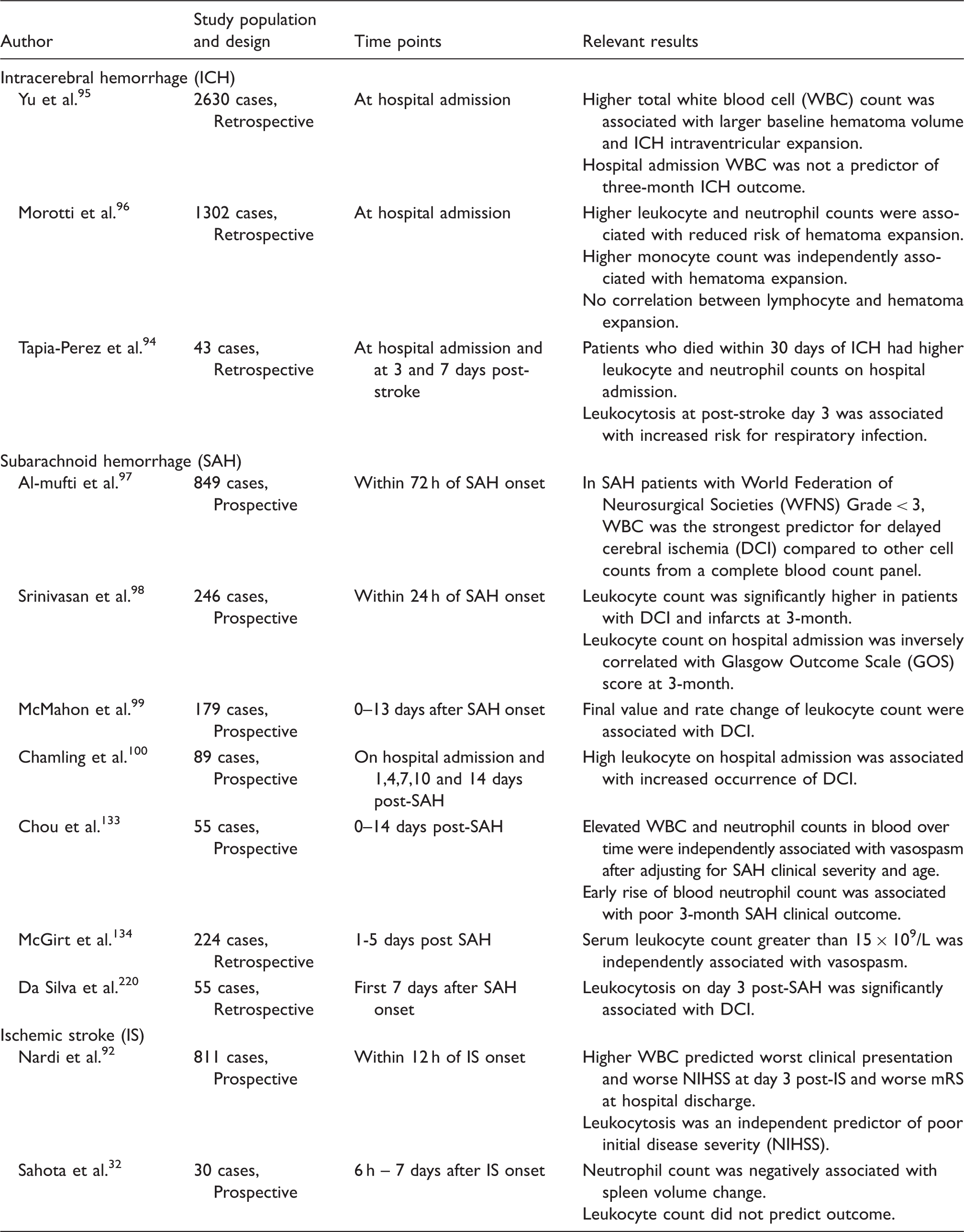
Unlike ICH, there are abundant prospective human data in SAH to suggest elevated circulating leukocyte is associated with poor outcome and SAH complications97–101 (Table 3). Across studies, early increase in blood leucocytes predicted delayed cerebral ischemia (DCI) and poor SAH outcome, while its association with cerebral vasospasm was inconsistent. In addition to single time-point elevations, leukocyte elevation throughout 0–14 days post SAH predicted SAH patients who develop subsequent vasospasm and poor long-term outcome. 101
Lymphocyte and monocyte count in blood
Circulating lymphocyte and monocyte and hemorrhagic stroke outcomes.
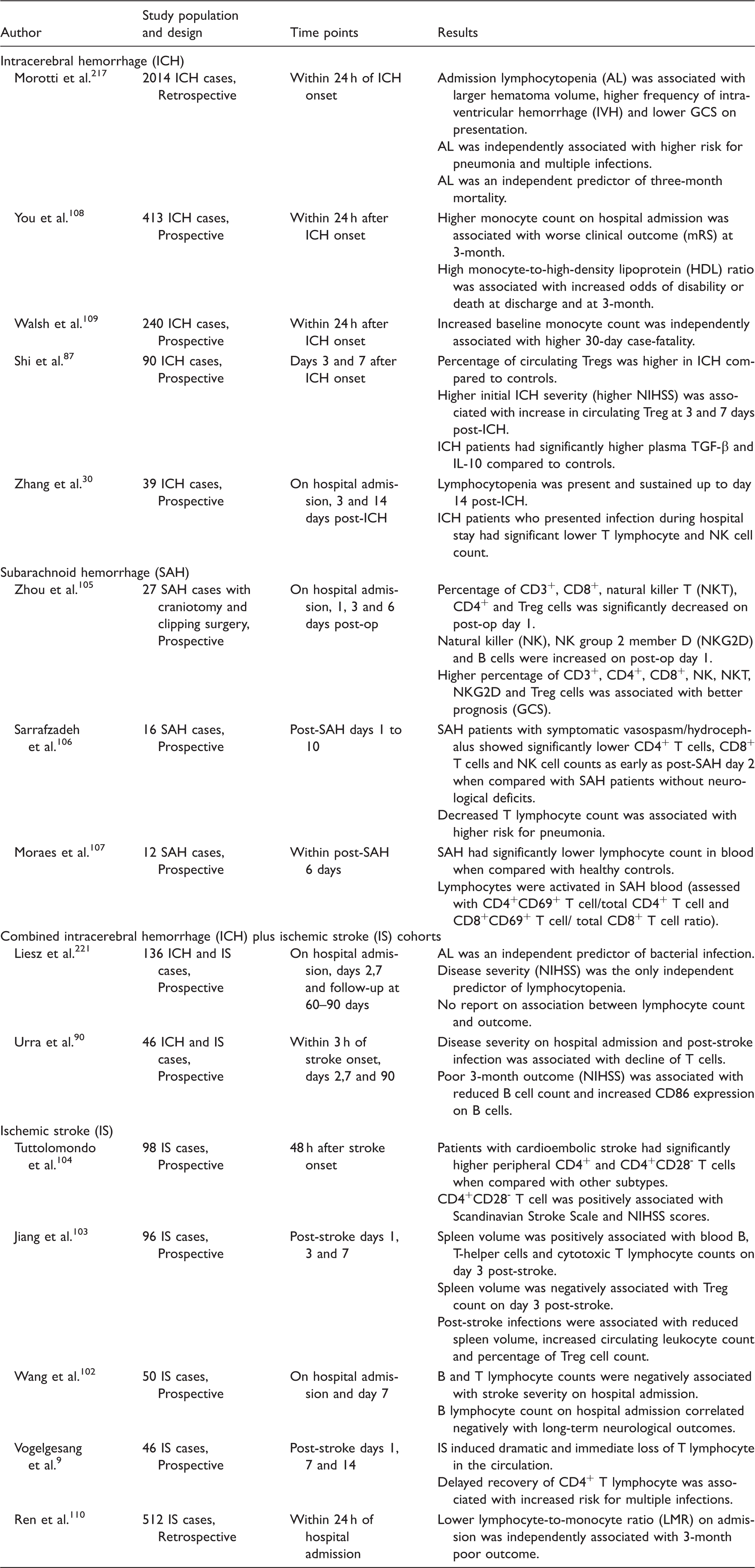
In SAH, few studies reported on changes in lymphocyte subpopulation. They consistently demonstrated significant decrease in circulating T lymphocytes during acute phase SAH.105–107 Specifically, acute SAH may be associated with significant decrease in natural killer T cells (NKT) and Tregs with simultaneous increase in NK, NK group 2 member D (NKG2D) and B lymphocytes 105 though data are inconsistent across studies. 106 Decreased T lymphocyte count may be associated with higher risk for pneumonia and increased proportions of CD3+, CD4+, CD8+, NK, NKT, NKG2D and Tregs may be associated with better prognosis in SAH. 107
While T and B lymphocytes decline, circulating CD4+CD25+FoxP3+ Treg cell population appears to expand following ICH along with elevated levels of serum IL-10 and TGF-β. Post-ICH Treg expansion appears proportional to initial ICH severity. 87 There are currently no data on whether Treg expansion in ICH has any effect on long-term ICH outcome. Unlike ICH and ischemic stroke, Treg counts appear to decrease in acute SAH, 105 though this must be interpreted with caution because ICH and SAH Treg studies are few and some have very small sample sizes.
In addition to lymphocyte subpopulations, monocyte counts also appear to be associated with ICH outcome. Two separate prospective human ICH studies reported increased monocyte count on ICH presentation to be associated with poor ICH outcome and higher mortality.108,109 Higher monocyte count, or decreased lymphocyte-to-monocyte ratio (LMR), has also been linked to poor outcome in ischemic stroke. 110
Taken together, the data to date suggest that the effect of leukocyte subpopulations on hemorrhagic stroke complications and outcomes is complex and different sub-populations need to be examined separately for their effects on hemorrhagic brain injury.
Neutrophil-to-lymphocyte ratio
Circulating neutrophil-to-lymphocyte ratio (NLR) and outcomes in hemorrhagic strokes.
Clinical features of systemic inflammation and immune dysfunction in hemorrhagic strokes
Systemic inflammation response syndrome in hemorrhagic strokes
Clinical SIRS criteria were originally selected to capture systemic inflammation in acute disease or injury such as sepsis.118–120 SIRS is defined as the presence of two or more of the following features: (1) temperature > 38 ℃ or < 36 ℃, (2) leukocytosis (>10,000/µL) or leukopenia (<4 000/µL), (3) tachycardia (>90 beats per min) and (4) tachypnea (>20 respirations per min). 121 Presence of SIRS is generally associated with longer intensive care unit (ICU) length of stay, increased number of ICU interventions and mortality. 118 In a general ICU population, incremental increase in number of SIRS criteria met at any given time predicted higher mortality in a linear fashion. 120 While SIRS is a validated and useful tool for measuring systemic inflammation severity, it has well recognized short-comings122,123 and alternative classification tools have been developed. 123 In critically ill patients with ischemic stroke, ICH, and SAH, SIRS remains by far the most common measure of clinical systemic inflammation in the literature.
Systemic inflammatory response syndrome (SIRS) and outcome in hemorrhagic strokes.

We identified three human studies that examined SIRS in ICH (Table 6). All three reported a positive association between SIRS and worse ICH functional outcome at hospital discharge and at three months.6,130,131 One reported that nearly 90% patients met SIRS criteria within 48 h of ICH, 130 suggesting that SIRS is highly prevalent in ICH and perhaps not a great tool to distinguish different degrees of systemic inflammation. However, other studies reported lower SIRS prevalence in ICH.6,131 SIRS being present on ICH hospital admission was associated with higher initial NIHSS, worse GCS, larger ICH volume, hypernatremia and the need for mechanical ventilation. One study reported significant associations between SIRS and mortality, where 97% of all patients who died from ICH had met SIRS criteria during their hospitalization. 6 Similar to SAH, it is not known whether SIRS is only a surrogate for initial ICH severity or whether it has independent impact on ICH outcome. A preliminary large rerospective cohort study of 2650 subjects showed that developing SIRS was independently associated with in-hospital mortality in ICH after adjustment for age and initial GCS. 132
Though clinical data to date are limited by retrospective study design, these data do suggest that SIRS is an important clinical feature in hemorrhagic strokes and may in fact independently contribute to post-stroke complications and outcome. What we don't know is whether it is the global SIRS syndrome or a particular component of SIRS that drives the effect on poor post-stroke outcome, and what the underlying mechanisms may be. There have been no prospective studies that examines whether reducing SIRS could improve hemorrhagic stroke outcome.
SIRS component: Leukocyte changes in SAH and ICH
A number of human studies have identified a positive association between circulating leukocyte elevation and complications and clinical outcomes following hemorrhagic strokes (Table 3).100,133–135
In SAH, the first descriptive studies examined leukocyte measurement at a single time point, typically upon hospital presentation. Leukocytosis exceeding 15 × 109/L within the first five days of SAH was associated with three-fold increase in vasospasm following SAH. 134 In a more recent study that examined leukocyte and neutrophil time profiles, elevated blood leukocyte count over the first 14 days of SAH was independently associated with subsequent angiographic vasospasm and worse SAH outcome 133 after adjusting for important confounders including age, Hunt and Hess grade, and mode of aneurysm treatment. In this SAH cohort, longitudinal regression analysis showed that patients who were destined to develop angiographic vasospasm and poor outcome had a different leukocyte count profile from the moment they presented with SAH and well before the development of vasospasm and poor outcome. This human data corroborates the hypothesis that patients at risk for complications such as vasospasm and for poor outcome have a different global immune and inflammatory response to the initial insult (SAH) over time, and this difference in immune response can distinguish between patients at risk for vasospasm and/or poor outcome.
The association between leukocyte count and ICH outcome is less clear. In a comprehensive literature search using PubMed, the following search criteria were used: “stroke” “intracerebral hemorrhage” “leukocyte” “systemic inflammation” “systemic inflammatory response syndrome”. We identified three studies that examined the relationship between leukocyte count and ICH outcome in human cohorts (Table 3). Some reported that leukocyte and neutrophil counts at hospital admission were significantly increased in ICH patients who died within 30 days. 135 Several studies failed to find a positive association between leukocyte elevation and ICH outcome. Yu et al. 95 found that elevated leukocyte count at hospital admission was not an independent prognostic factor in acute ICH. Another study reported that higher leukocyte count on hospital admission may even be favorable, as it was associated with lower risk of hematoma expansion in ICH patients. 136
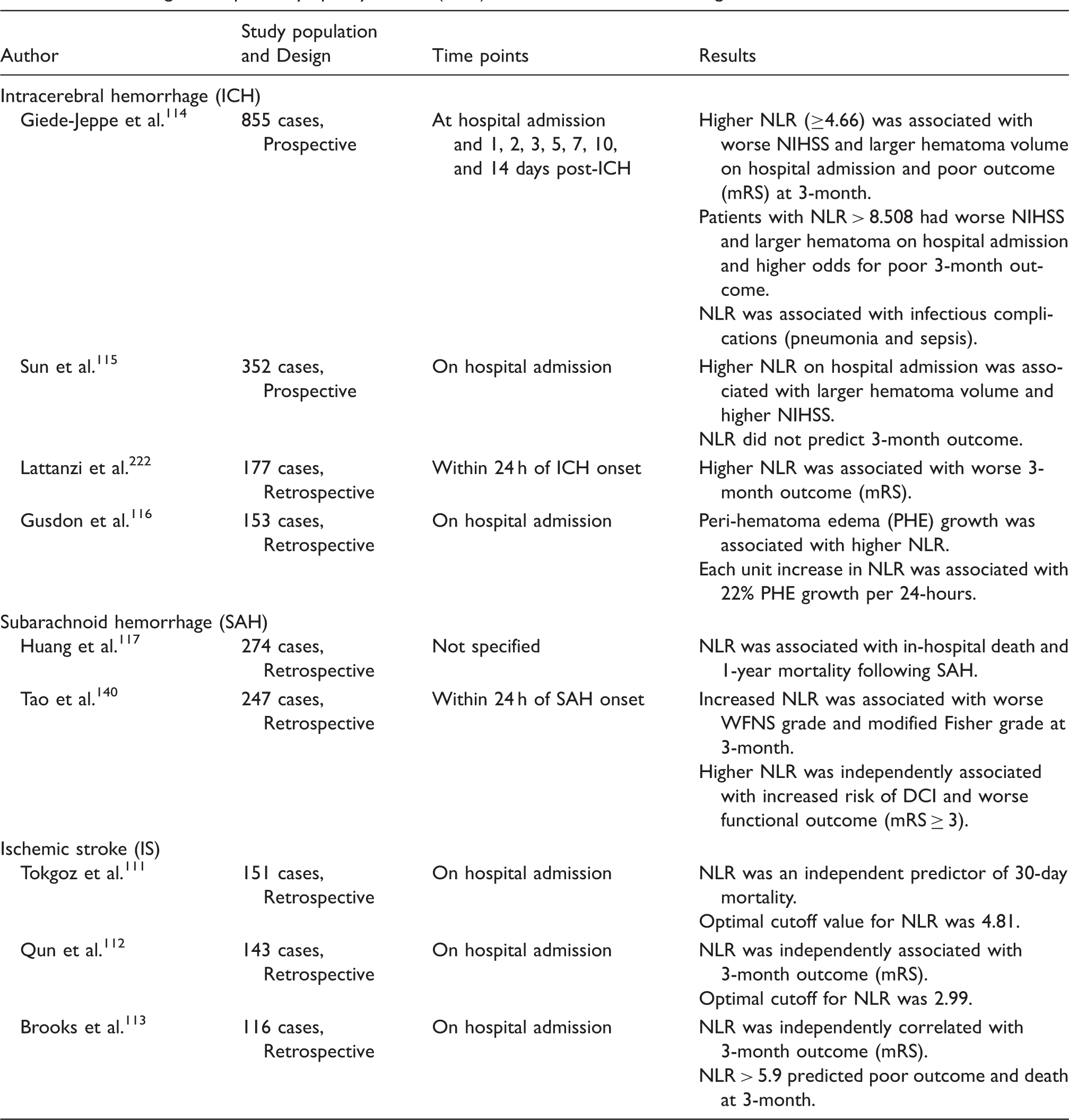
Neutrophil-to-lymphocyte ratio (NLR) has been proposed as a potential biomarker for systemic inflammation and mortality in sepsis. 137 In ischemic stroke, higher NLR is an independent predictor for mortality at three months. 138 In ICH, the two largest prospective studies both showed that higher NLR is associated with worse initial neurological status and larger hematoma volume on hospital admission. However, while one study reported association between higher NLR and worse neurological function at three month following ICH, others could not replicate this finding114,139 (Table 5). In addition to neurological function and hematoma size, higher NLR in ICH is also associated with infectious complications 114 and risk for developing peri-hematoma edema. 116 Interestingly, while NLR was predictive of ICH complications, total leukocyte counts in these patients did not show any such association, raising the possibility that the interaction of subsets of peripheral immune cells may play a role in ICH complications and outcomes, while leukocyte count as a global measure may be insufficient in reflecting the complex underlying pathophysiology. In SAH, only two retrospective studies examined NLR. These studies found higher NLR to be associated with SAH mortality, SAH clinical severity measured by World Federation of Neurological Surgeons (WFNS) grade and with DCI post SAH.140,141
Taken together, these findings suggest that peripheral inflammatory and immune cells responses may directly impact risks for complications following hemorrhagic strokes and they may represent novel therapeutic targets.
SIRS component: Fever in SAH and ICH
Fever is a classic clinical sign of systemic inflammation and is generally associated with worse outcomes for many acute diseases142,143 and specifically in the neurocritical care patient population including patients with ischemic stroke, ICH, and SAH.142,143 Fever is prevalent in stroke patients, particularly those with ICH.130,144,145 Specifically in SAH, Pegoli et al. 146 showed that patients with fewer hours of fever during ICU stay had better clinical outcomes. In addition to outcome, fever also appears to be an independent risk factor for DCI following SAH. 147 SAH patients with higher clinical severity (Hunts-Hess grades > 2) and leukocytosis on presentation are at higher risk for developing central fever during the first seven days of their hospital stay. 148
Many studies discussed the role of fever in ICH, where higher fever burden is associated with neurological deterioration and lower health-related quality of life at follow-up.142,144,149 Extrapolating from these observations, many experts recommend fever control, or targeted temperature management (TTM) in critically ill patients with brain injuries. 150 Whether fever control can improve outcome and reduce hemorrhagic stroke complications has not yet been demonstrated. This is being evaluated in ongoing prospective clinical trials.
Cellular and molecular biomarkers of systemic inflammation and immune dysfunction in hemorrhagic strokes
There are abundant human data to suggest that cellular and molecular mediators of inflammation are upregulated in SAH and ICH and some are be independently associated with clinical outcomes. 151
Biomarkers of immune function and inflammation in SAH
Biomarkers of immune function and inflammation in subarachnoid hemorrhage (SAH).
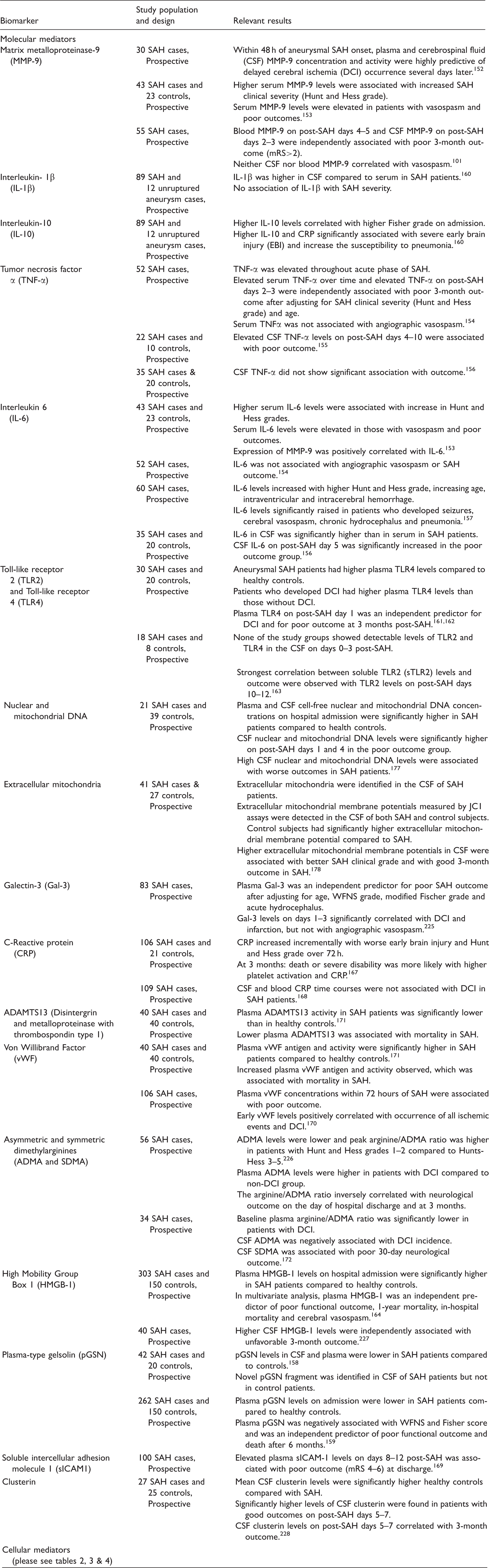
Plasma-type gelsolin (pGSN) is cleaved by MMP-9 and is postulated to have anti-inflammatory effects. SAH patients have lower blood and CSF pGSN levels compared to controls. 158 Lower plasma pGSN is an independent predictor of poor outcome and mortality after six months.158,159 Another mediator with anti-inflammatory effects, IL-10 appears to have elevated blood levels post SAH and is associated with early brain injury and increased risk of pneumonia in SAH. 160
Toll-like receptors (TLRs) are a class of innate immunity receptor known to recognize damage-associated molecular pattern (DAMP). TLR-4 is not detectable in the CSF following SAH161–163 but higher plasma TLR-4 levels have been linked to higher DCI risk in SAH. 162 HMGB-1 is an important nuclear protein that regulates DNA transcription. Immune cells such as macrophages and monocytes secrete HMGB1 which binds to TLR-2 and TLR-4, leading to NF-κB upregulation and subsequently stimulate macrophage cytokine release. In SAH, higher plasma and CSF levels of HMGB-1 are associated with worse outcome.103,164
Several studies explored C-reactive protein (CRP) as a potential biomarker in SAH. CRP is an acute phase reactant generally elevated in many acute conditions.165,166 In SAH, CRP appears to increase incrementally in parallel with SAH clinical severity and may be associated with death or severe disability at three months. 167 However, its association with DCI is inconsistent across studies (Table 7).167,168 Another non-specific inflammatory marker found to be elevated in SAH is soluble intercellular adhesion molecule-1 (sICAM1). Delayed increases in plasma sICAM-1 levels on post-SAH days 8–12 are associated with worse outcome at discharge. 169 Just like the case of CRP, the association between sICAM1 and DCI after SAH is inconsistent. 170 It is possible that these markers reflect generalized, nonspecific systemic inflammation but may not directly participate in the pathogenesis of brain injury in SAH.
Molecular mediators of endothelial function such as von Willbrand factor (vWF), ADAMTS13 (a disintegrin and metalloproteinase with thrombospondin type 1) and asymmetric dimethylarginine (ADMA) also appear to be important in SAH-associated inflammatory responses. vWF-antigen levels and activity in plasma are significantly higher in SAH compared to controls. 171 One prospective SAH cohort study with 106 subjects showed that early serum vWF increase is associated with DCI and poor outcome following SAH. 170 ADAMTS13, a metalloproteinase that cleaves vWF, is significantly lower in SAH than in controls. 171 Asymmetric dimethylarginine (ADMA), a competitive inhibitor of nitric oxide synthase (NOS), can induce vasoconstriction by reducing nitric oxide production and potentiate endothelial dysfunction and inflammation. Recent studies showed that higher plasma and CSF ADMA to be associated with SAH clinical severity (Hunts and Hess grade) and DCI, while lower plasma arginine/ADMA ratio was associated with DCI in SAH.171,172
Circulating cell-free deoxyribonucleic acid (cfDNA) is emerging as novel biomarkers in cancer as well as cardiovascular diseases. Primarily released due to cell death and necrosis, blood cfDNA was recently shown to be a potential biomarker for short-term 173 and long-term ischemic stroke outcome174,175 and may distinguish between ischemic and hemorrhagic strokes. 176 A study with 21 SAH patients and 39 health controls undergoing myelography found that SAH patients had higher blood and CSF nuclear and mitochondrial cfDNA levels and higher CSF nuclear and mitochondrial cfDNA levels were associated with worse SAH outcome. 177 In addition to extracellular DNA, a recent study demonstrated that extracellular organelles may also play an important role in hemorrhagic strokes and could be a candidate biomarker. This study reported presence of extra-cellular mitochondria in cell-free SAH CSF and detected active mitochondrial membrane potentials in CSF from control and SAH subjects, suggesting these mitochondria may be at least partially functional. CSF extracellular mitochondrial membrane potentials were significantly higher in control subjects compared to SAH. In SAH subjects, lower CSF extracellular mitochondrial membrane potentials were associated with worse clinical grade and independently associated with worse three-month SAH outcome. 178
Biomarkers of immune function and inflammation in ICH
Biomarkers of immune function and inflammation in intracerebral hemorrhage (ICH).

Similar to observations in SAH patients, elevated plasma levels of TLR-2, TLR-4, nuclear DNA, and HMGB-1 all showed associations with worse ICH ouctome.182–184 Specifically, higher blood TLR-2 and TLR-4 are associated with greater residual ICH lesion volume at three month and with worse clinical ICH outcome, suggesting these inflammatory mediators may have potential impact on post-ICH recovery and repair. 182 Higher plasma nuclear DNA levels in ICH patients correlated with initial clinical severity measures such as worse GCS and higher ICH volume at presentation and predicted poor ICH outcome. 183 Plasma HMGB-1 levels are significantly elevated in ICH and positively correlated with levels of downstream pro-inflammatory cytokines including IL-6 and TNF-α. Higher plasma HMGB-1 levels is associated with worse initial NIHSS and worse outcome in ICH. 184 Taken together, these data suggest that specific pro-inflammatory mediators are upregulated following ICH and their degrees of elevation appear to parallel initial ICH severity as well as subsequent clinical course. Whether these mediators independently contribute to ICH brain injury processes and whether interventions that change these serum biomarker levels translate into ICH outcome remain unknown.
Many acute phase reactants have been identified as potential biomarkers in ICH; however, their causal effect with ICH severity and outcome is not known. Higher plasma CRP at hospital admission is consistently associated with hematoma growth and poor ICH outcome.185,186 Hepcidin, an acute phase reactant and key iron regulator is elevated after ICH and associated with poor three-month outcome. 187 Elevations in serum ferritin, an acute phase response protein that regulates iron-storage, are also associated with poor outcome in ICH patients. 188 There are some data to suggest that ICH-associated changes in serum hepcidin and ferritin may mediate post-ICH anemia and thereby impact overall disease severity and outcome. 188 Whether elevations of these acute phase proteins in ICH represent non-specific ICH disease severity or whether they participate in brain injury processes remain an open question.
The consistent associations of inflammatory and immune mediators with disease severity and outcome in SAH and ICH support the notion that both innate and adaptive immune responses are important contributors to the overall outcome in hemorrhagic strokes.
Effect of age and sex on systemic inflammation in hemorrhagic strokes
Biological differences in sex and age have significant impact on the peripheral immune and inflammatory response to brain injury and subsequent disease course, responses to treatment, and outcome.189,190 Females have elevated immune response to antigen, experience higher incidence of inflammatory and autoimmune diseases, 191 express higher levels of TLR, have increased number and function of monocytes, macrophages, and dendritic cells,192–195 and have heightened adaptive immune response. 196 Ischemic stroke models demonstrated that female sex and estrogen are protective in young but detrimental in older, post-reproductive animals.197–205
Though systemic inflammation is highly prevalent in hemorrhagic strokes, very little is known about age and sex effects on immune response to ICH and SAH. One ICH model using collagenase injection compared 27 inflammation-related gene expressions between aged (10–11 months old) and young (3–4 months old) rats and found aged rats had less pronounced or delayed inflammatory response with delayed hematoma resorption. 206 No study specifically examined sex effects on systemic inflammation in ICH. One retrospective ICH human study found post-ICH sepsis is more common in males compared to females (4.8% vs. 3.2%). 207
SAH occurs more frequently in females, and yet most preclinical studies in SAH used male animals. One study examined sex differences in SAH neuroinflammation by applying an autologous blood injection SAH model and showed male rats had increased number of inflammatory cells and platelet aggregates in brain sections and more severe bleeding, worse early brain injury and increased mortality. 208 The second study examined inflammatory gene expressions in a filament perforation SAH rat model and found male rats had higher expressions of thrombomodulin and endothelial NOS 3 compared to females. 209 To date, no human studies have specifically reported on age and sex effects in SAH-associated systemic inflammation. Our group reported preliminary human data from large retrospective cohorts showing that younger age is associated with higher incidence of SIRS following ICH and SAH.129,132
Immunodeficiency after SAH and ICH
Both animal and human data suggest that ischemic strokes can lead to a secondary immunodeficiency syndrome (SIDS) resulting in increased susceptibility to infections and higher morbidity and mortality. 210 Ischemic stroke and traumatic brain injury (TBI) models have shown that the initial local and systemic pro-inflammatory response is followed by systemic immunosuppression characterized by lymphopenia, impaired T- and NK cell function,211,212 monocyte deactivation, a shift from Th1 to T-helper cells 2 (Th2) and development of spontaneous bacteremia and pneumonia. 45 One postulated mechanism for SIDS is that the initial acute systemic inflammatory response leads to overactivation of the SNS which causes rapid and sustained lymphopenia and lymphocyte and monocyte functional changes culminating in an immunodeficient state with higher susceptibility to infections.213,214
Whether and to what degree SIDS also occurs in ICH and SAH is not known. One human SAH cohort study showed that patients with high clinical severity demonstrated continuous suppression of the cellular immune response with lower CD4+, CD8+ and NK cell counts, 106 suggesting an immunosuppressed state following SAH. In ICH, the fact that increased sympathetic stimulation reflected by decreased BRS and heart rate variability is associated with unfavorable outcome and increased susceptibility to infections provides indirect evidence for SNS-associated immunosuppression.54–56 Earlier in the manuscript, we reviewed changes in circulating immune cell profiles after SAH and ICH, which are other surrogate measures of changes in immune function. There remain numerous unanswered questions in the role of peripheral immune response in hemorrhagic strokes. Specifically, very little is known about the long-term effects of immuno-dysregulation on repair and recovery in hemorrhagic strokes.
Infectious complications in hemorrhagic strokes
Infectious complications are known to worsen ischemic stroke outcomes and increase stroke recurrence risk.56,214,215 Up to 65% ischemic patients develop infections complications, with the predominant infections being urinary tract infections (UTIs) and pneumonia. 214 Similar to ischemic stroke, infectious complications are prevalent in ICH (30% patients) and independently predict in-hospital mortality and 30-day hospital readmission rate.207,216 Human cohort studies suggest the most common post-ICH infections are UTIs (14.8%) and pneumonia (7.8%). 207 Infections account for 22% of all hospital readmissions after ICH and the most common reasons for infection-related readmission are septicemia (11.8%), UTI (4%), pneumonia (3.5%) and gastrointestinal infections (1%). 215 Lymphopenia (lymphocyte count < 1000) within 24 h of ICH onset is an independently predictor of infectious complications and increased case-fatality at 90 days post ICH. 217 Similar to ischemic strokes and ICH, prevalence of infectious complications in SAH is 26–41% 218 with the most common infections being pneumonia (17%) and UTIs (9%). 219 Retrospective cohort studies reported that 44.8% of febrile SAH patients had infection which is associated with poor functional outcome. 143 Bacterial pneumonia is more prevalent in SAH patients with acute focal neurological deficits compared to SAH patients with minor symptoms such as headache, but the causal effect is not known. 106
The association between lymphopenia, post-stroke infection risks and hemorrhagic stroke outcomes suggests that post-hemorrhage peripheral immune suppression likely plays an important role in ICH and SAH pathophysiology. Changes in lymphocyte sub-populations may be the central connection between inflammation, immune deficiency, and brain injuries in hemorrhagic strokes.
Conclusion and future directions
We have systematically reviewed literature on the brain–immune system interactions in hemorrhagic strokes, highlighting similarities and differences from ischemic strokes. Many questions remain on how hemorrhagic strokes induce changes in the peripheral immune system and how the SNS, PNS, neuro-endocrine and autoimmune processes contribute to this brain–body cross talk. The mechanism and impact of post-hemorrhage immunosuppression on subsequent infectious complications and hospital readmission rates remain important knowledge gaps with little data. Exciting emerging data now suggest that peripheral immune organs such as the spleen and immune cells such as Tregs may be important mediators of the peripheral immune response and may represent potential novel therapeutic targets in hemorrhagic and ischemic strokes. The time is ripe to address these important knowledge gaps in understanding and targeting systemic immune responses to hemorrhagic strokes which continue to cause disproportionately high degrees of morbidity and mortality.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. S. Chou has been funded by the National Institute of Neurological Disease and Stroke (1K23-NS073806), the University of Pittsburgh Physician Foundation Award, and the University of Pittsburgh Dean's Faculty Advancement Award.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sherry Chou served as site principal investigator for Newton 2 clinical trial in SAH funded by Edge Therapeutics and as ad hoc consultant for Merck. S.C. is a co-inventor for the patent “MITOCHONDRIAL BIOMARKERS OF, AND THERAPEUTICS AQ15 FOR, CNS INJURY AND DISEASE” filed in the United States Patent and Trademark Office as Application 62/3817. The other authors declare that there are no conflicts of interests.
