Abstract
Introduction
Aging and metabolic disease enhance inhaled particulate toxicity. Nanoparticles (NPs) are rapidly coated with biomolecules forming a biocorona (BC), upon entering the body and may contribute to the susceptibility. Aging and metabolic syndrome (MetS) are progressive conditions resulting in biomolecule alterations over time potentially influencing susceptibility. We hypothesize NP-biomolecule interactions are altered during aging and throughout MetS progression.
Methods
C57BL/6J mice at 6 weeks of age were fed a healthy diet or a high-fat western diet. BALF was collected after 2, 4, 8, 12, 16, 20 or 24 weeks on diets. NP-biomolecules interactions were compared between healthy and MetS to determine age- and disease progression-related BC variations (proteins and lipids).
Results
Unique BCs were determined to form at each time point indicative of aging for the healthy and aging and disease progression for the MetS. Comparisons between healthy and MetS BCs at each time demonstrated distinct biomolecule interactions attributable to disease. Comparisons determined both unique protein and lipid content as well as quantitative differences. Proteins such as apolipoprotein A-IV, complement C3 and lipids such as PE (37:5), PE (O-38:5), PE (P-38:4), PC(40:7), PC(39:0), and PC(O-40:0) were identified on the MetS BC suggesting disease progression modifications. Proteins such as pulmonary surfactant protein A, fibrinogen alpha-chain and lipids such as CE (19:0)-NH4, DG (36:7), and DG (35:0)_C18:0 were increasingly present in the healthy BC over time, suggesting age-related interactions.
Discussion
Overall, unique BCs were identified demonstrating the impact of age and disease progression on BC formation which will aid in understanding initial pulmonary NP-biomolecular interactions potentially contributing to susceptibility.
Introduction
Nanoparticles (NPs) are engineered materials with at least one dimension in the range of 1-100 nm.1,2 They can be easily modified with unique physicochemical properties that can be applied to multiple fields including medicine, agriculture, biotechnology, housing, manufacturing, and electronics. With the growing development of nanotechnology, the potential of human NP exposure is a safety concern. Numerous safety assessments have demonstrated that inhalation of NPs and their accumulation/deposition in the lungs causes a variety of health effects including inflammation, oxidative stress, DNA damage, apoptosis, leading to impairment in lung function, acute lung injury, lung fibrosis and systemic effects.3–7 Iron oxide (Fe3O4) NPs are relatively biocompatible compared to other NPs and are widely utilized across various fields including biomedical and industrial processes due to their superparamagnetic properties. Their utilization in manufacturing processes causes potential occupational exposure.8–11 For example, a study assessing workplace exposure to Fe3O4 NPs reported personal breathing zone concentrations reaching 335 μg/m3 in facilities lacking localized engineering controls, highlighting a significant risk for workers involved in their production and handling. 12 An inhalation study in Wistar rats demonstrated increased pulmonary oxidative stress and lung inflammatory response at 24 h, 48 h and 14 days post exposure to Fe3O4 NPs at concentration of 640 mg/m3. 13 Pulmonary toxicity resulting from exposures is variable with distinct subpopulations demonstrating exacerbated responses. For instance, healthy old rats (20 months old) exhibited pulmonary and cardiovascular alterations including pulmonary inflammation, myocardial ischemic damage, atrio-ventricular blockage, and an increase in fibrinogen concentration along with an increase in blood viscosity compared to healthy young (3 weeks) and healthy adult (8 weeks) rats upon exposure to silicon dioxide NPs. 14 While the chronic disease conditions including cardiovascular disease such as obesity and metabolic syndrome (MetS) demonstrates an aggravated response to exposure. For example, high fat diet fed obese mice depicted increased pulmonary inflammation and induced oxidative stress after exposure to titanium dioxide NPs compared to animals fed a normal diet. 15 Currently, the mechanisms for these variations in exposure response remain unclear.
Following exposure to the biological environment, spontaneous adsorption of biomolecules such as proteins, lipids and/or other macromolecules occurs on the surface of NPs. This dynamic layer of biomolecules on the surface of NPs is termed the biocorona (BC). The formation of BC is highly dependent upon physicochemical properties of NPs as well as the nature of biological fluid, influencing the biological outcomes.16–18 Previously, we have demonstrated the unique formation of the NP-BC in serum and bronchoalveolar lavage fluid (BALF) due to underlying diseases such as obesity and MetS. These BCs resulted in modified immune cell interactions likely contributing to toxicity and susceptibility differences between individuals.19,20 Numerous other variations between individuals exist in our population that may contribute to differential responses following exposures.
Aging is associated with significant alterations in the composition of body biofluids and tissue proteomes, including those of the lung and BALF.21–23 Aging lungs exhibit increased abundance of extracellular matrix proteins, proteases, and inflammatory mediators, alongside reductions in proteins involved in repair and antioxidant defense.21,23,24 For instance, an integrated proteomic and transcriptomic analysis on whole lung tissue was performed to build a draft of an atlas of the aging mouse lung. 25 The proteomic analysis revealed an increase in abundance of immunoglobulins and major histocompatibility complex (MHC) class I, in the lung tissue of older mice (24 months old) compared to younger mice (3 months old), correlating with upregulation of proinflammatory pathways. 25 While aging drives significant shifts in the pulmonary (lung and BALF) proteomes, these changes could be further amplified in chronic disease states. Conditions like MetS, obesity, and cardiovascular diseases introduce additional disruptions in lipid metabolism, complement activation, and inflammatory signaling, can potentially compound age-related proteomic alterations.26–28 Specifically, a study reported changes in the proteome composition of BALF in human subjects with increasing body mass index (BMI) (mean BMI of 34.9 ± 7.5 kg/m2). 29 Proteins such as complement-associated proteins, coagulation factors and apolipoprotein B-100 were positively associated with the BMI of the cohort. 29
Understanding modifications in the formation of BC due to age and disease is necessary for protecting growing subgroups within our populations that may be at risk for enhanced toxicity. Our previous studies have demonstrated the formation of unique BC in on Fe3O4 NPs following incubation in high-cholesterol serum compared to the BC formed in normal serum. 30 In addition, a mouse study from our laboratory demonstrated unique NP-BC formation on Fe3O4 NPs following incubation in MetS BALF compared to healthy BALF. 20 The study further noted the alterations in cellular interactions and toxicity due to the formed BCs. It is noteworthy that this study examined the formation of BCs in MetS at a single time point. However, MetS is a progressive, multifactorial disorder characterized by a cluster of metabolic abnormalities, including impaired glucose tolerance, central obesity, insulin resistance, dyslipidemia, and hypertension. 31 Over time, these metabolic disturbances may trigger the alterations in biomolecules including proteins and lipids, amplifying the metabolic and inflammatory response and increase susceptibility to exposures. 32 Hence it is fundamental to investigate NP-BC interactions in terms of disease progression. Along with this most existing studies primarily examine biomolecular changes at the tissue level, often within a limited age range, a single time point and using comparisons between only two groups that may oversimplify the biological complexity of aging and its interpretation. Furthermore, there has been minimal investigation into age and disease progression alterations in the BALF proteome and lipidome, particularly under conditions of environmental exposure. Exploring these changes could provide critical insights into the biomolecular profile of particulate interactions in the lung and their role in modulating susceptibility.
We hypothesize that unique BCs are formed due to age and metabolic disease progression. To investigate NP–biomolecule interactions during the progression of age and disease conditions, mice were placed on either a healthy or high-fat diet at 6-weeks (wks) of age and BALF was collected at multiple time points (2, 4, 8, 12, 16, 20, and 24 weeks on the respective diet). BALF samples were used to generate NP–BCs
Materials and methods
Animal model
C57BL/6J male mice (Jackson Labs, Bar Harbor, ME, USA) at 6 weeks of age were placed on either a healthy diet with 10% of kcal coming from fat (D12450 B, Research Diets Inc., New Brunswick, NJ, USA), containing 51.6 mg/kg cholesterol or a high-fat western diet (HFWD) with 60% of kcal coming from fat (D12492, Research Diets Inc.), containing 279.6 mg/kg cholesterol. The selected HFWD is a well-established diet for producing a mouse model of MetS.33,34 Our previous work has generated consistent results with very little variability in inducing MetS by using these diets.20,35–38 Specifically, we have established that these diets result in approximately a 50% weight gain and over a 30% increase in total cholesterol levels and a 6% elevation in insulin without altering hemoglobin A1C.37,38 Mice were housed in a room with constant temperature, 12/12-h light/dark cycle. All animal procedures were conducted in accordance with the National Institutes of Health guidelines and approved by the Purdue University Animal Care and Use Committee.
Necropsy and BALF collection
16 mice (8-healthy and 8- high-fat diet (MetS)) were necropsied after 2, 4, 8, 12, 16, 20, or 24 weeks on their respective diet. Briefly, the lung was lavaged once with cold phosphate buffered saline (PBS) at a volume of 35 mL/kg of body weight. All individual BALF samples from the groups were centrifuged at 1000×g at 4°C for 10 min. The biomolecule-rich supernatants were isolated, pooled among all mice from each group, aliquoted into individual volumes of 500 µL and stored at −80°C for utilization in forming BCs. The use of pooled samples is standard for evaluations of the NP-BC and allows for fundamental understanding of the interactions formulating the NP-BC. Use of a pooled sample suggests variations in BC formation result from variability in interactions between biomolecules and the NP surface allowing for assessment of how factors such as aging and MetS progression broadly influence formation.
Model characterization
Blood was collected from healthy and MetS mice via cardiac puncture. Blood samples were centrifuged at 3500×g at 4°C for 10 min and serum was isolated. Collected serum samples were used to evaluate total cholesterol, high-density lipoprotein (HDL), and low-density (LDL)/very-low-density lipoprotein (VLDL) in a random set of samples to verify groups. All serum endpoints were quantified using commercially available kits (Bioassay Systems, Hayward, CA, USA) via the manufacturer’s protocols.
NP characterization
For this evaluation, 20 mg/mL iron oxide nanoparticles (Fe3O4 NPs) with a hydrodynamic diameter of 20 nm in aqueous 2 mM sodium citrate (NanoComposix, San Diego, CA, USA) were utilized. Similar to our previous publications, Fe3O4 NPs were diluted to a concentration of 25 µg/mL and characterized via assessment of hydrodynamic size, polydispersity index, and ζ-potential (ZetaSizer Nano, Malvern Panalytical Ltd, Malvern, UK) to confirm manufacturer’s specifications (n = 3/particle).36,38–42 The concentration of 25 µg/mL and methods have been previously utilized by our laboratory and others to characterize NPs.19,36–39,42
Formation of Fe3O4 NP BCs
To formulate the Fe3O4 NP-BCs, 125 µL of Fe3O4 NPs at a concentration of 1 mg/mL were diluted with 125 µL deionized water (diH2O), centrifuged at 15,000×g for 30 min, and the supernatant removed. The isolated Fe3O4 NPs were then resuspended thoroughly with either 500 µL BALF from healthy or MetS mice. NPs were then incubated at 4°C for 8 h while rotating. Following the incubation, NPs were washed 3 times via centrifugation at a speed of 15,000×g and resuspended in diH2O at their original concentration of 1 mg/mL. Pooled aliquots were utilized to produce samples for proteomic (n = 4/group) and lipidomic analysis (n = 4/group). These methods have been previously utilized and optimized by our laboratory for NP-BC formation.19,20,39 Similar to
Characterization of BC protein components
Protein components from healthy and Mets BCs were isolated and analyzed by liquid chromatography-tandem mass spectrometry (LC-MS/MS) using a similar method as described in our previous studies.19,43,44 Briefly, following the isolation of NPs with BCs by centrifugation at 21,130×
Proteomic statistical analysis
Protein were evaluated similar to previous evaluations of the NP-BC.20,40,43,44 LFQ intensities of proteins detected in three of four replicates from each group and with at least 2 MS/MS counts were considered as present for comparisons. Data were searched at 1% FDR (false discovery rate) for both peptide spectral match and proteins. Common contaminant proteins were removed before any subsequent analyses. A Venn diagram was produced using Venny 2.1.0 (https://bioinformatics.psb.ugent.be/webtools/Venn/, accessed on January 2025) to identify similarities and differences between the BCs. To quantify differences in abundance of shared proteins identified in both the healthy and MetS BCs, fold changes of intensities were calculated comparing each replicate to the mean intensity of the healthy group. Univariate analysis (pairwise t-test) was utilized to determine significantly different proteins using a p-value <0.05.
Characterization of BC lipid components
To assess the lipid components of the NP-BCs a separate set of samples were produced using the same incubation conditions used for evaluation of proteins. The multiple reaction monitoring (MRM) profiling method was used to profile ceramides (Cer), phosphatidylcholines and sphingomyelins (PC-SM), phosphatidylethanolamines (PE), phosphatidylglycerols (PG), phosphatidylinositols (PI), phosphatidylserines (PS), cholesteryl esters (CE), triacylglycerols (triglycerides) (TAG), and acyl-carnitines (AC) and present by their relative amounts. 45 Previously we have performed this procedure to examine components of the NP-BC following incubation in serum. 39 Following BC formation and NP isolation, NP-associated lipids were extracted via a modified Bligh-Dyer lipid extraction method. 46 The MRM profiling methods and instrumentation used have been recently described in previous reports.45,47–50 Specifically, isolated NP-BCs were resuspended in 200 µL water followed by adding 125 µL of chloroform and 225 µL of methanol. Then 125 µL of water and 125 µL of chloroform were added. The solution was separated into two phases by centrifugation. The bottom organic phase containing the lipids, was isolated. These extracts were dried and resuspended in 50 µL of methanol/chloroform 3:1 (v/v) and a 250 µL of 3:6.65:0.35 (v/v/v) mixture of acetonitrile, methanol, and ammonium acetate. This solution was diluted (200x) and 8 µL of each sample was injected using a micro-autosampler (G1277 A) directly to the ESI source of an Agilent 6410 triple quadrupole mass spectrometer (Agilent Technologies, Santa Clara, CA, USA). A capillary pump was connected to the autosampler and operated at a flow rate of 7 µL/min and a pressure of 100 bar. Capillary voltage on the instrument was 5 kV and the gas flow 5.1 L/min at 300°C. The MS data obtained from these methods was processed using an in-house script to obtain a list of MRM transitions with their respective sum of absolute ion intensities over the acquisition time. The parent m/z of the MRMs screened were based on the Lipid Maps Structure Database (https://www.lipidmaps.org/ accessed on September 2024), and the product ions were related to class-diagnostic fragments. 51 Free fatty acids were monitored only by the parent mass.
Lipidomic statistical analysis
Similar to previous studies, the respective ion intensity values were used for data analysis.20,40,43,44 There were 4 samples in each BC group. MRM signals were required to be 30% above the blank in all replicates to be considered for data analysis. For univariate analysis (pairwise t-test), p-value <0.05 was considered significant. Statistical analysis was performed utilizing Metaboanalyst 6.0 (https://www.metaboanalyst.ca/MetaboAnalyst/ModuleView.xhtml accessed on February 2025). Data on the relative amounts were auto scaled to obtain a normal distribution and evaluated by univariate analysis. Informative lipids were analyzed according to class, fatty acyl residue chain length and unsaturation level. Lipids thatere significantly different with >1 fold change between two BCs were graphed and shown through a histogram.
Results and discussion
Mouse model characterization
Mice provided the high-fat diet demonstrated increased body weight compared to mice on the healthy diet (Figure 1a). Serum levels of total cholesterol, HDL, and LDL/VLDL were higher in mice fed the high-fat diet compared to those receiving the healthy diet at each time point evaluated (Figure 1b, c & d). The diet-induced MetS mouse model is well established, characterized by phenotypic alterations such as increased body weight and elevated cholesterol, triglycerides, and fasting glucose.20,35–38,52 In our study, animals maintained on the high-fat diet exhibited a consistent increase in body weight, total cholesterol, and HDL levels at all time points, along with a rise in LDL levels starting from week 12, confirming induction of MetS, consistent with previous reports from our lab.36,38,53 Importantly, a study noted elevated insulin without changes in A1C, indicating MetS without progression to diabetes through 16 weeks on the high-fat diet.
38
Characterization of body weight and serum lipid levels in healthy and MetS mouse models following 24 weeks on either a control or high-fat western diet. Healthy and MetS mice were characterized by examination of (a) body weight, (b) serum total cholesterol, (c) serum high-density lipoprotein (HDL) and (d) serum low-density lipoprotein (LDL) levels. Values are expressed as mean ± SEM, (n = 5/group). *denotes significant differences between healthy and MetS, (p < 0.05). Blue bars = Healthy BC, Red bars = MetS BC.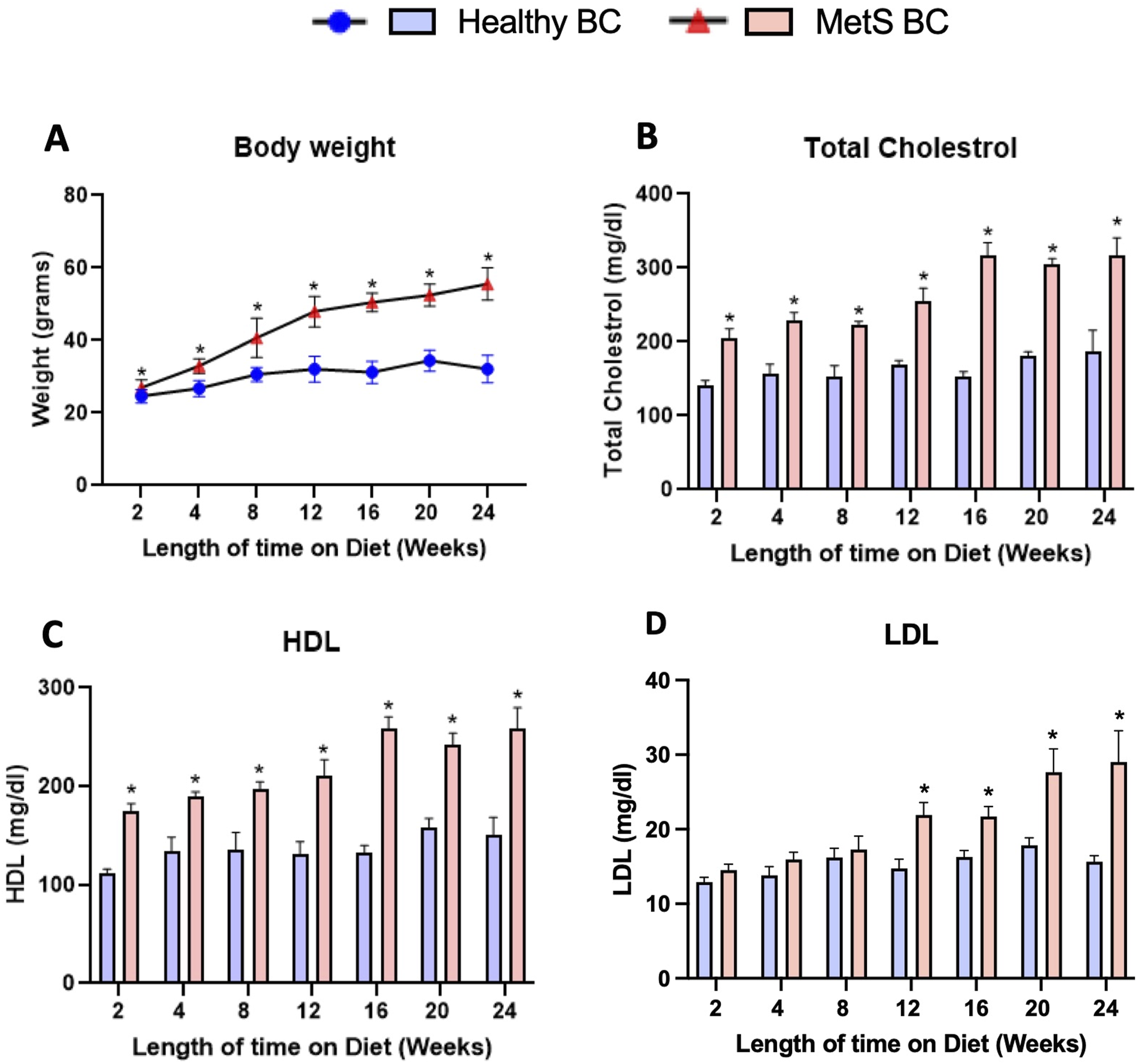
Characterization of Fe3O4 NPs
Fe3O4 NP characterization.

Biomolecular assessment in variations in BC formation with age and disease progression
BALF from mice fed with healthy or high-fat diets were collected at time points of 2, 4, 8, 12, 16, 20 or 24 weeks on their respective diets to evaluate the BCs that formed on Fe3O4 NPs. Protein (Supplemental Table 1) and lipid (Supplemental Table 2) components of the healthy and MetS BCs were identified and quantified. Differences between protein and lipid components of the BC, were evaluated based on age (healthy over time), age and disease progression (MetS over time) and disease progression (MetS compared to healthy at each time point). Comparison of BCs variations due to age and disease state progression will aid in understanding NP-biomolecular interactions that may contribute to toxicity and susceptibility following inhalation exposures in expanding subpopulations.
Proteomic evaluation of the variations in BC formation with age
We assessed differential proteins as well as alterations in protein abundance within the NP-BC following NP incubation in BALF collected from healthy mice at different ages. These comparisons demonstrated that at each age there were proteins found to associate in common as well as age-specific accumulation of unique proteins (Figure 2). These findings suggest variability in the formation of the BC within the lung as mice age which may mediate age-specific susceptibility to inhaled exposure. Specifically, a slight decrease of shared proteins was observed between 8 and 12 weeks on healthy diet, which may indicate a transitional phase due to age-related changes in the physiological environment of the lung. Furthermore, there were more unique proteins present in the BC at 20 weeks on healthy diet compared to 16 weeks on healthy diet than for any other comparison suggesting another significant age transition phase. Many proteins were determined to associate with the NP surface at all ages examined. These commonly identified proteins included annexin A3, serotransferrin, serum albumin, pulmonary surfactant-associated protein A, tubulin alpha-1B chain, apolipoprotein A-IV, fibrinogen alpha chain, and complement C5 (Supplemental Table 3). The presence of these proteins within the NP-BC are unchanged irrespective of age within the healthy mouse group demonstrating them to be consistent components of the BC within the lung. Certain proteins such as signal transducing adapter molecule 2, nardilysin and low molecular weight phosphotyrosine protein phosphatase associated with NP surface at earlier ages and were not detected at later stages of aging (Supplement Figure 1a). In contrast proteins such as apolipoprotein D, immunoglobulin heavy variable 5-16, and immunoglobulin heavy variable V1-18 were detected at later ages, suggesting age-related shifts in BC formation (Supplement Figure 1b). Proteins identified as present in healthy BC at a given time point on diet (weeks) were compared to proteins identified in healthy BC in subsequent week. Venn diagrams were used to illustrate the number of unique and shared proteins.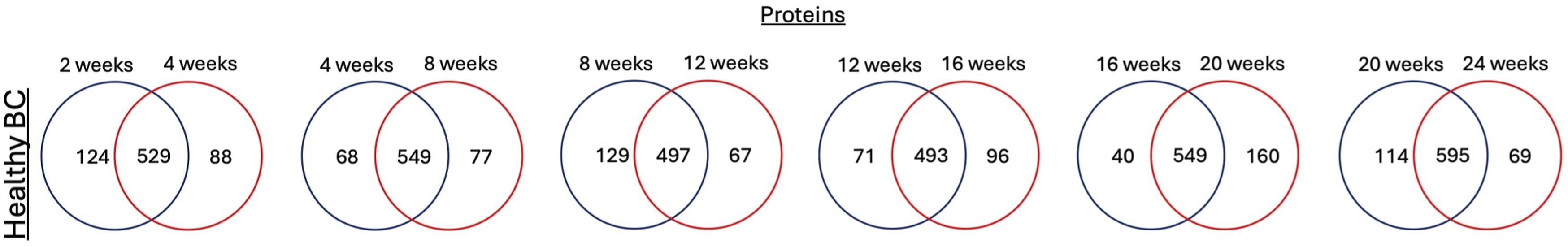
Although many proteins were determined to associate with the BC in common at different ages (Figure 2), there were differences in their abundance on the NP surface. Analysis of protein abundance changes over time in the healthy BC revealed a stepwise increase in the abundance of some of the proteins adsorbed on NPs from 2 to 24 weeks on healthy diet (Figure 3a). Examples of these included Annexin A4 (Anxa 4), Sulfotransferase 1 family member D1 (Sult1d1) and Plastin-3 (PlS3) in healthy BC. Similarly, certain proteins showed decreasing abundance for healthy BC associated with age. For example, these include Poly (rC)-binding protein 2 (Pcbp2), 3(2),5-bisphosphate nucleotidase 1 (Bpnt1), and in Glutathione synthetase (Gss) in healthy BC (Figure 3(B)). Further, proteins such as heat shock 70 kDa protein 13, glycine--tRNA ligase and ubiquitin-like modifier-activating enzyme 7 were consistent with age progression (Supplemental Table 4). Overall, these results demonstrate that BALF proteins adsorbed on NP-BC change over time with age. Aging is a complex and natural biological process that drives progressive physiological changes at both the tissue and cellular levels.58,59 Proteomic analyses comparing adult (23–48 years) and elderly (62–73 years) BALF have revealed significant age-related alterations in protein composition and associated biological pathways. For example, neutrophilic granule proteins such as proteinase 3; myeloperoxidase, cathelicidin, matrix metalloproteinase family proteins, lamin B1, a biomarker of cellular senescence were detected at higher levels in elderly BALF.
21
These alterations related to age can influence NP-BC formation which may influence subsequent cellular responses and toxicity. A recent study demonstrated age-dependent changes in proteins adsorbed onto NP surfaces across five major organs in mice, with the largest differences observed in younger animals (3 week old mice) compared to other age stages (16 weeks, 50 weeks and 80 weeks old), likely reflecting maturation during development.
60
Such tissue-level changes are expected to also impact biofluids such as BALF.
61
BALF from aged mice is likely to exhibit alterations in the abundance of key proteins, including plasma proteins, surfactant proteins, cytoskeletal components, and immune mediators. For example, gene expressions of protein (surfactant proteins A, B, D, regulatory protein of surfactant metabolism Abca3) in AE2 cells (progenitor cells producing and secreting surfactant proteins in lung) were altered in age-dependent manner demonstrating effect of age on biomolecular changes in biofluid composition.
62
Our data demonstrated proteins such as albumin, and serotransferrin, and surfactant proteins continue to dominate the BALF proteome across age groups, providing a stable baseline for protein adsorption onto NPs. Lung-specific proteins like pulmonary surfactant protein A (Sftpa1) persist with age but may undergo modifications affecting immune recognition.
63
We observed these modifications in differential abundance of Sftpa1, which increased initially from 2 to 4 weeks on healthy diet, decreased to its lowest at 12 weeks on healthy diet and increased again till 24 weeks on healthy diet (Supplemental Figure 2). Immune-related proteins, including complement C5 and fibrinogen, reflect age-associated shifts (upregulation in C5 and fibrinogen) in inflammatory tone,64,65 while metabolic and structural proteins such as alpha-enolase and tubulin indicate ongoing tissue remodeling and cellular turnover during aging.
66
Furthermore, studies have demonstrated that age may influence the pulmonary response to the particulate exposure. For example, study in C57BL6/J female mice revealed age-dependent inflammatory activation induced by multiwalled carbon nanotubes (MWCNT) exposure, where older mice (15 months old) have elevated BALF inflammatory proteins compared young ones (6–8 weeks old). It is unknown if unique BCs forming on NP surfaces contribute to these age-related modifications in exposure susceptibility. Together, these age-associated changes in BALF protein composition can modulate NP-protein interactions, influencing BC formation, clearance, and downstream biological responses in an age-dependent manner. Protein changes overtime of selected proteins found in healthy BCs. Representative proteins identified were found to be increased and decreased overtime in healthy BC (A & B). All selected proteins depicted in the figure are determined by 25% cutoff criteria (being present in 3 out of 4 replicate samples).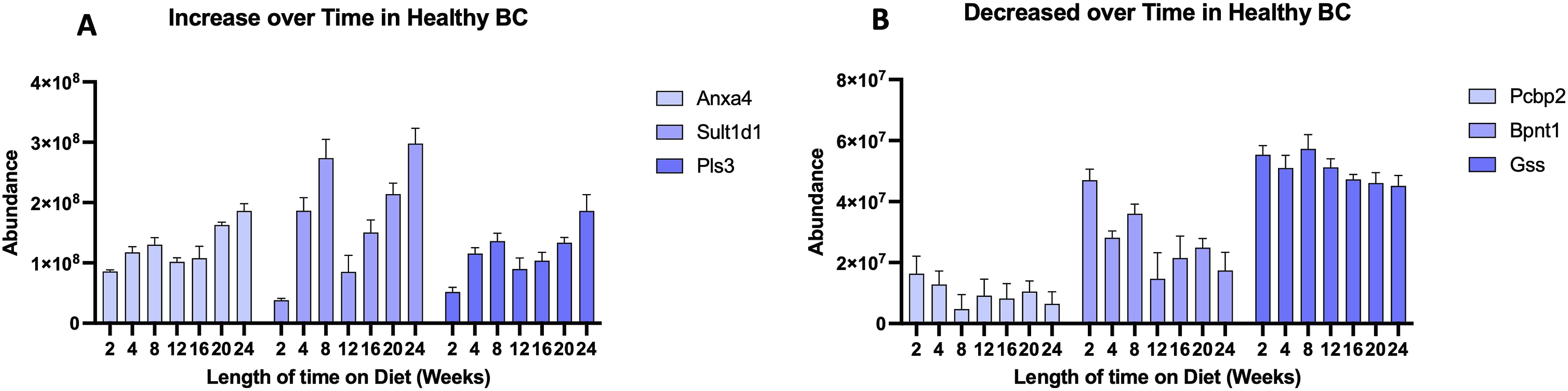
Proteomic evaluation of the variations in BC formation with age and disease progression
Unique BCs were identified on Fe3O4 NPs following incubation in MetS BALF collected at given time points (2 weeks to 24 weeks on the high-fat diet). This assessment allowed for the determination of protein-NP interactions modulating BC formation in a mouse model of MetS capturing both age-related and disease progression modifications. A subsequent comparison of MetS and healthy BCs will be discussed in later sections which will elucidate disease specific variations in BC formation at each time point therefore controlling for age-related alterations.
Comparisons determined unique BCs in each MetS group suggesting differential NP interactions due to age and MetS progression (Figure 4). These differences in BC likely relate to alterations in the biofluid dependent on age and time on the high-fat diet. An increase in unique proteins within the BC was observed at 12 weeks compared to 8 weeks on high-fat diet suggesting a transitional phase. Time points evaluated after 12 weeks on high-fat diet demonstrated more shared proteins than previous time points, although unique proteins were observed at each time point. Commonly shared proteins at each time point included alpha-enolase, apolipoprotein A-IV, apolipoprotein A-II, chitinase-3-like protein 1, serum albumin, heat shock protein HSP 90-beta, and complement C9 (Supplemental Table 4). The presence of these proteins within the BC are unchanged irrespective of age or disease stage demonstrating them to be consistent components of the BC within the lung. Certain proteins such as coatomer subunit beta, BRCA1-A complex subunit BRE, and Alanyl-tRNA editing protein were found on both healthy and MetS BC from 8 to 24 weeks on diet, highlighting the both age and disease-related NP-BC change overtime (Supplement Figure 3a). While proteins such as, protein arginine N-methyltransferase 1, GTP-binding protein Rheb, and surfeit locus protein 4 were absent in healthy BC but were found on MetS BC at later time points (8 to 24 weeks on high-fat diet) (Supplemental Figure 3b). The presence of these proteins within the MetS BC indicates a distinct pattern of BC formation, likely driven by alterations in the biological milieu associated with age and disease progression. Proteins identified as present in MetS BC at the given time point on high-fat diet (week) were compared to proteins identified in MetS BC in the subsequent week. Venn diagrams were used to illustrate the number of unique and shared proteins.
Although many proteins were determined to associate in common between the assessed time points, differences in protein abundance were identified. On analyzing BC-protein abundance changes overtime for MetS mice, there was step wise increase in abundance of specific proteins adsorbed on NPs from 2 to 24 weeks on high-fat diet. For example, these included Plastin-3 (PlS3), Heparin cofactor 2 (Serpind1), Acid ceramidase (Asah1) and others in MetS BC (Figure 5a). Similarly, certain proteins showed decreasing abundance for MetS BCs from 2 to 24 weeks on high-fat diet. For example, these included Filamin-A (Flna), Fibrinogen beta chain (Fgb), Inositol monophosphatase 1 (Impa1) and others in the MetS BC (Figure 5b). Studies have demonstrated NP-BC formation can be influenced due to disease-induced physiological differences.39,55 These studies often evaluate only single time points where disease has been established and compare to a healthy model. Many diseases are progressive, developing over time with increasing severity of symptoms. The progression of the disease state results in biofluid variations in proteins which may contribute to BC alterations. Furthermore, during the progression of the disease individuals also age which can impact the BC. MetS is a progressive disease developing over time due to sedentary lifestyles and poor diets. It has been demonstrated that as symptoms of MetS increase so do exacerbations in inflammatory responses to inhaled exposures.
20
Our data demonstrated proteins such as alpha enolase, apolipoprotein IV and apolipoprotein II that were commonly found in MetS BC across all the time points. These proteins are elevated in MetS conditions compared to healthy.67–69 Since these proteins were elevated at all time points assessed they do not appear to be age-related and are present due to MetS. Association of apolipoproteins on the surface of NPs suggests interactions with lipid transport proteins in shaping the NP interface, potentially affecting cellular uptake, biodistribution, and interactions.
70
Literature has demonstrated that adsorption of apolipoproteins on the NP surface can facilitate transport across the blood-brain barrier.71–73 Together, this suggests that MetS BCs may result in differential biodistribution of exposures across lipid-rich cellular membranes. HSP90, a molecular chaperone protein central to cellular stress response, protein folding, and stabilization of signaling proteins, indicates that cellular stress and proteostasis imbalance may influence the MetS and NP BC composition.
74
Finally, complement C9, a terminal component of the membrane attack complex, underscores the role of immune surveillance and inflammatory activation that modulate the bio-nano interface.
75
Together, these proteins illustrate how age and disease progression influence the BC which may contribute to differential effects of exposures based on MetS severity. Protein changes overtime of selected proteins found in MetS BCs. Representative proteins identified were found to be increased and decreased overtime in MetS BC (A & B). All selected proteins depicted in the figure are determined by 25% cutoff criteria (being present in 3 out of 4 replicate samples).
Comparison of variations in protein content of the BC due to disease
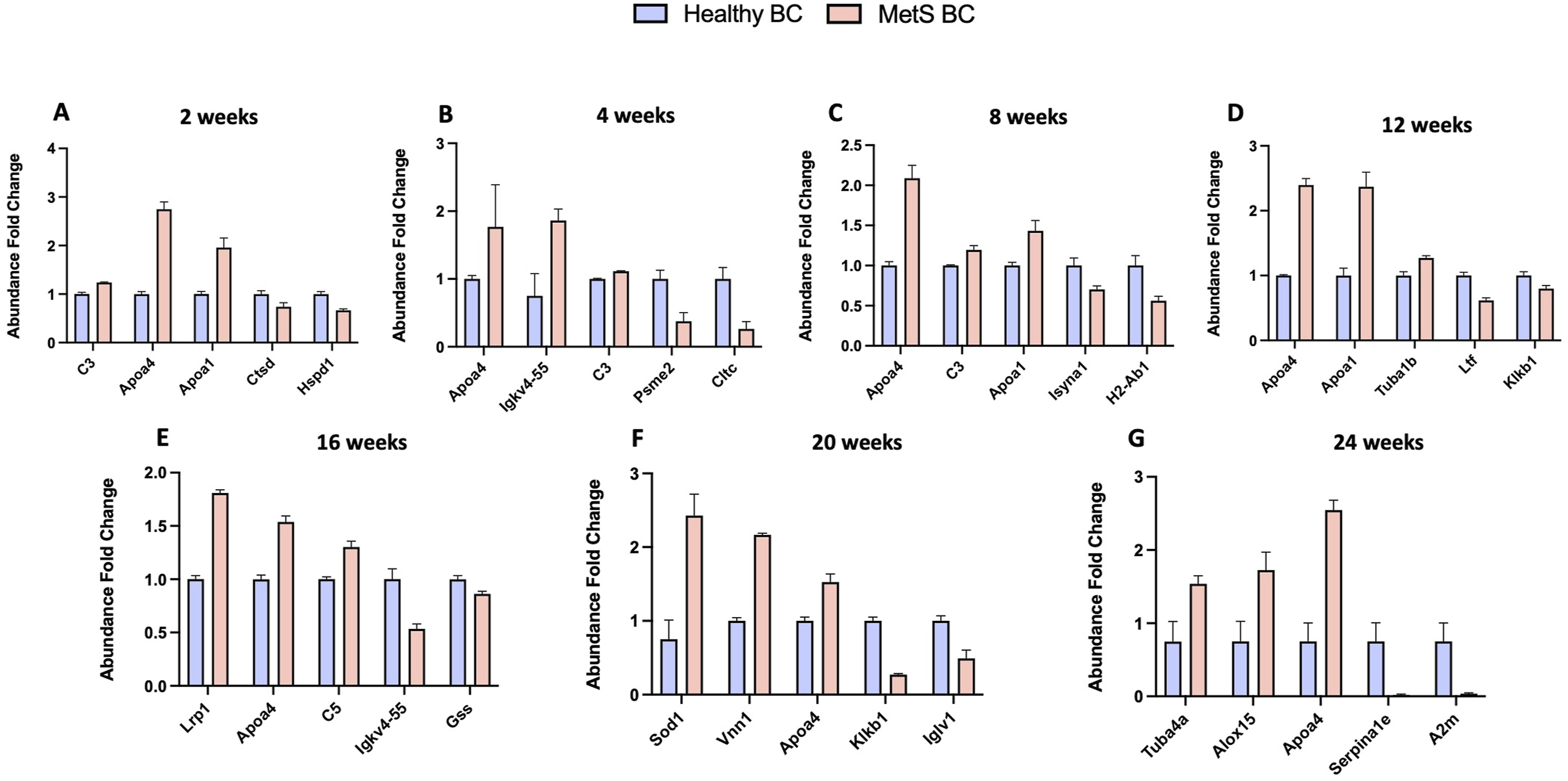
Importantly, both age and disease progression are contributing factors in comparing healthy and MetS BC and may independently or in combination drive the observed differences in BC composition. To isolate disease-specific variations in the protein composition of the BC from age-related variations, we compared healthy and MetS BCs at each individual time point. On comparing healthy BC with MetS BC, the number of shared proteins were increased up to 12 weeks on diets and remained stable till 20 weeks on diets (Figure 6). For example, these included tubulin alpha-4A chain, apolipoprotein A-IV, complement C3, protein S100-A6, and serotransferrin (Supplemental Table 5). Further, individually, more unique proteins were identified on healthy BC compared to MetS BC at each given time point. At 2 weeks on diets, 172 unique proteins were identified on healthy BC while 22 unique proteins were adsorbed on MetS BC. This trend was followed across all the given time points but not at 12 weeks. Particularly at 12 weeks on high-fat diet, the number of unique proteins were higher in MetS BC compared to healthy BC, which may represent the transitional phase of metabolic state altering the dynamics of protein environment. (Supplemental Table 5). This suggests that distinct BCs are formed in disease condition due to differences in the composition of the BALF. Beyond identifying the shared proteins, we also measured their fold-change in abundance across time. This analysis provides a more nuanced view of how biomolecule–NP interactions evolve with physiological state. Notably, these alterations align with disease progression, suggesting that specific biomolecular shifts within the BC may serve as indicators of altered biomolecular environment as disease progresses. Differential abundance was determined for statistically significant proteins (p value <0.05) between healthy and MetS BC (Figure 7). The proteins that were significantly abundant in MetS BC are complement 3, apolipoprotein A-IV, apolipoprotein A-I, complement 5, Tubulin alpha-4A chain etc. Compared to healthy BC while some proteins such as cathepsin D, clathrin heavy chain 1, plasma kallikrein etc. Were significantly lower in abundance in MetS BC. Specifically, protein apolipoprotein A-IV was consistently present with higher abundance in MetS BC in approximately all the given time points. This suggests that protein apolipoprotein A-IV may dominate over other proteins influencing the biological identity of NPs. Apolipoprotein A-I and apolipoprotein A-IV are glycoproteins and major protein components of chylomicrons in the post-prandial plasma of humans and rodents.76–78 They are involved in the modulation of triglyceride-rich lipoprotein assembly and metabolism, reversing cholesterol transport promoting cholesterol efflux by activating-lecithin-cholesterol acyltransferase (LCAT) and cholesterol ester transfer protein (CETP), and induction of satiety in mice.
79
A proteomic analysis of plasma obtained from individuals with obesity and mice that were kept of eucaloric high-fat diet (64E% fat) for 6 weeks reveals apolipoprotein A-IV to be a significantly elevated protein.
80
Apolipoprotein A-I and apolipoprotein A-IV abundance increases in underlying disease conditions such as MetS, aligning with our current data demonstrating their increased presence in the BC. Moreover, NP-protein complexes enriched with apolipoprotein A-I were able to interact with certain scavenger receptors (SRs) such as SR-B1 which are expressed on the surface of many cells including macrophages.20,81 An investigation demonstrated increased accumulation of SR-B1 in BCs and enhanced uptake of NPs, suggesting a possible NP biomolecule interaction between the enriched protein on BC and SRs of cells.
81
In line with our assessment, the altered biomolecular components due to the underlying condition can govern the identity of formed BC, which can vary depending upon the advancement of MetS. Comparison of BC composition between healthy and MetS conditions. Proteins identified as present in healthy BC were compared to proteins within the BC formed from MetS BALF for the given time points. Venn diagrams were used to illustrate the number of unique and common proteins. Relative abundance comparison of selected proteins found in healthy and MetS BCs. A value larger than 1 denotes a higher abundance in MetS BC compared to the healthy BC whereas a value less than 1 denotes lower abundance in the MetS BC compared to the healthy BC. All proteins depicted in the figure are significantly different between BCs based on p < 0.05 (n = 4/group). Blue bars = Healthy BC, Red bars = MetS BC.
Lipidomic evaluation of the variations in BC formation with age
To gain a comprehensive understanding of NP-BC formation we performed similar assessments for lipids as for proteins, enabling a holistic view of NP-biomolecular alterations associated with age and disease progression. We determined BCs consisted of differential lipids as well as alterations in lipid abundance at different time points in the healthy model to identify age-dependent changes in lipid–NP interactions. The number of shared lipids remained relatively consistent across all the given time points for the healthy BC whereas unique lipids increased at 8 weeks, declined at intermediate time points and peaked again at 24 weeks (Figure 8). Lipids typically interact with surfaces through hydrophobic interactions.
82
Once a stable layer of lipids forms, it tends to be relatively fixed.
82
Thus, lipids may demonstrate lower exchange rates compared to proteins which may somewhat decrease variability of interactions. The commonly identified lipids in the healthy BC across all ages included glycerophosphocholines (PC: phosphatidylcholines, PE: phosphatidylethanolamines), sphingolipids (SM: Sphingomyelins), glycerolipids (DG: Diacylglycerols), Fatty Acids and Conjugates (FA: Fatty Acyls) and some ceramides (CE) (Supplemental Table 6). Certain lipids such as CE (19:0) NH4, DG (36:7),DG (35:0)_C18:0; and DG (36:6)_C18:0, were found to associate with the NP surface at early time points in the healthy BC (Supplement Figure 4a), in contrast to lipids like FA (14:5), and LPC(18:2),LPC(P-19:1) appeared predominantly at 12 to 24 weeks on the healthy diet (Supplement Figure 4b). The presence of these lipids in healthy BCs suggests age-dependent changes in BC formation which may impact NP-cellular interactions in the lung contributing to susceptibility. Comparison of BC composition as age (healthy) progresses. Lipids identified as present in healthy BC at a given time point (week) on a healthy diet were compared to lipids identified on the BCs in subsequent week. Venn diagrams were used to illustrate the number of unique and common lipids.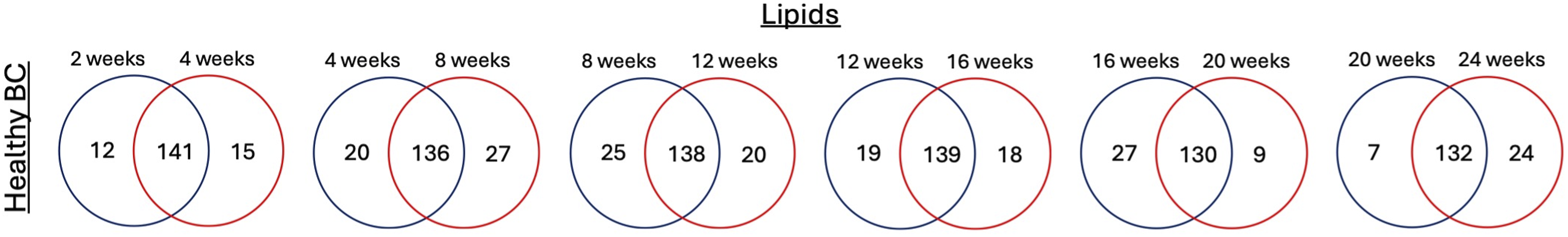
On studying the lipid abundances of healthy BCs as mice age, there was a stepwise increase in the lipid abundance on NPs from 2 to 24 weeks of some lipids. For example, these included CE (18:0) NA and CE (17:0)K and CAR(22:5)_QUAL within the healthy BC. Similarly, certain lipids showed decreases in abundance as age progresses which included CE (18:2) NH4, SM (d18:0/15:0) and (PC(37:5),PC(O-38:5),PC(P-38:4)) within the BC (Figures 9a and b). Literature suggests significant alterations in sphingolipid metabolism with aging modifying ceramide levels.
83
Multiple studies report that elderly individuals exhibit elevated ceramide levels, which are associated with increased risk of developing age-related pathologies like Alzheimer’s and cardiovascular diseases.84–86 Therefore, modifications observed in ceramides within the BCs are expected throughout aging as their content is known to change due to age. Lysophosphatidylcholine (LPC) levels can be modified due to aging processes and low LPC levels are predictive of numerous age-related impairments such as neurodegenerative diseases, mitochondrial dysfunction, myocardial infarction and others.87–89 Association within the BC on the NP surface may further impact biological processes LPCs are involved in and could modify the predictive value of this biomarker. Lipid changes overtime of selected lipids found in healthy BCs. Representative lipids identified were found to be increased and decreased overtime in healthy BC (A & B). All selected lipids depicted in the figure are determined by being more than 30% above the blank sample intensity.
Lipidomic evaluation of the variations in BC formation with age and disease
A major component of MetS is lipid dysregulation and the presence of MetS has been shown to alter markers of aging in a manner suggestive of accelerated biological aging. This relationship dictates that aging and MetS be examined as comorbidities that may result in distinct BC formation. Like proteins, unique lipids were identified on Fe3O4 NPs following incubation in MetS BALF collected at different time points (2 weeks to 24 weeks on the high-fat diet). The presence of differential lipids and variations in lipid abundance were identified. In the MetS BCs there was a gradual increase in shared lipids over time, peaking at 20-24 weeks on the high-fat diet, whereas unique lipids followed a fluctuating pattern with increases at 20 and 24 weeks on the high-fat diet (Figure 10). This suggests an increase in the association of lipids as age and disease progresses. For example, shared lipids included DG (34:1)_C16:0, PC(34:2),PC(O-35:2),PC(P-35:1), PE (37:7),PE (P-38:6),PE (36:0),PE (O-37:0), FA (30:1), SM(d18:0/24:1), CE (18:0)Na and others (Supplemental Table 7). Evaluating lipid abundances over time for the MetS BCs, there was a stepwise increase in the lipid abundance on NP surfaces from 2 to 24 weeks on high-fat diet for some lipids. For example, these included SM(d16:1/17:0), DG (38:8), DG (dO-40:8), DG (37:1)_C18:0, and FA (22:1) within MetS BC (Figure 11a). Similarly certain lipids demonstrated a decrease in abundance as age and disease progressed including DG (34:2)_C18:2, PC(29:0), PC(O-30:0), and PE (36:3),PE (P-37:2) within the MetS BCs (Figure 11(B)). Lipids such as LPC (24:0) and DG (38:6),DG (dO-40:6)_C16:0 appeared in both the healthy BCs and MetS BCs, with higher abundance in MetS BC at time points, demonstrating NP-BC changes overtime with both age and disease progression (Supplement Figure 5a). Furthermore, the specific association of lipids such as FA (5:2) and FA (14:1) within healthy BC demonstrates binding pattern of lipids influenced by age while lipid such as DG (O-38:9),DG (36:2)_C18:0 and DG (28:2)_C18:1 within the MetS BC indicates a distinct pattern of BC development influenced by disease progression (Supplemental Figures 5b and 5c). Due to the uniqueness of the BC formed due to age in the MetS group it is possible it could be utilized to assess MetS development in terms of age and individual risks associated with the presence of comorbidities. Comparison of BC composition as disease (MetS) progresses. Lipids identified as present in MetS BC at the given time point (week) on high-fat diet were compared to lipids identified on the BCs in subsequent week. Venn diagrams were used to illustrate the number of unique and common proteins. Lipid changes overtime of selected lipids found in MetS BCs. Representative lipids identified were found to be increased and decreased overtime in MetS BC (A & B). All selected lipids depicted in the figure are determined by being more than 30% above the blank sample intensity.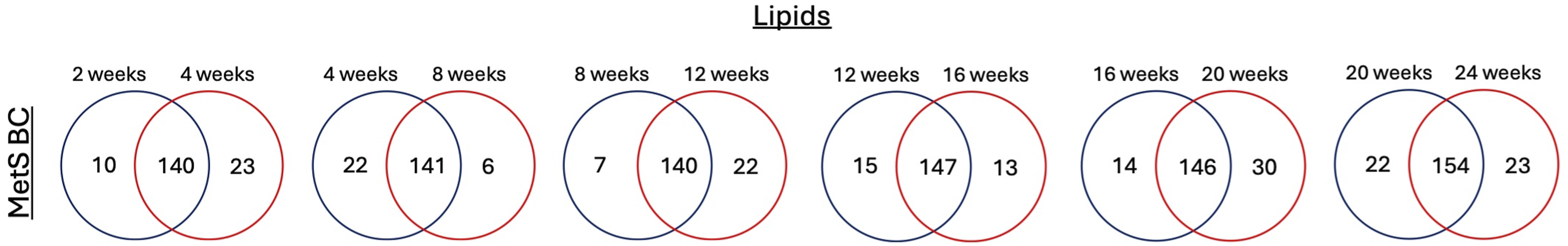

Comparison of variations in lipid content of the BC due to disease
Lipid components of the healthy BC and MetS BC were compared to understand disease-related BC variations that were not dependent on age. On comparison, the number of shared lipids peaked at 12 weeks on high fat diet with a slight decrease at intermediate time points and increased at 24 weeks on high fat diet (Figure 12). These included most of PE (37:5),PE (O-38:5),PE (P-38:4), PE (34:2),PE (O-35:2),PE (P-35:1), PC(44:12),PC(O-44:5), PC(40:7),PC(39:0),PC(O-40:0), FA (46:0), DG (34:1)_C18:1 and others (Supplemental Table 8). Overall, more unique lipids were identified in MetS BCs compared to healthy BCs from 12 to 24 weeks. For example, at 20 weeks, 8 unique lipids were identified on healthy BC while 45 unique lipids were adsorbed on MetS BC. For example, unique lipids in MetS BC included FA (37:0), PC(37:3),PC(O-38:3),PC(P-38:2), and PE (40:5) in contrast to unique lipids such as FA (9:0), FA (16:1), and DG (29:2)_C16:0 in healthy BC. This suggests that distinct lipids formulate the BC in the MetS condition due to differences in biofluid composition. Further we determined the differential abundance of statistically significant lipids (p value <0.05) between healthy and MetS BCs at each age. For example lipids that were significantly abundant in MetS BC are glycerophosphocholines (PC(34:2),PC(O-35:2),PC(P-35:1)), sphingomyelins (SM(d18:1/17:0)), fatty acids and conjugates (FA (16:0)), and diacylglycerols (DG (34:2)_C16:0) while lipids such as ceramides (CE (18:0)K) and fatty acyl carnitines (CAR(22:5)_QUAL) were significantly lower in abundance in MetS BC compared to the healthy (Figure 13). Lipids classes such as phospholipids and sphingolipids represent 25-30% of HDL components.
90
Interestingly, variations in concentrations of these molecules within HDL are associated with occurrence and severity of ischemic heart disease.91–93 Further, individuals with MetS show qualitative and quantitative changes in HDL, impairing their anti-atherogenic potential.90,94,95 For example, a study quantified different species of main phospholipids and sphingolipids in HDL within MetS subjects.
90
Different species of PCs and PEs were determined to be at altered levels in HDL of MetS patients, potentially reflecting differences in fatty acid composition (C18 and C20 chains) and their implications on metabolic pathways.
90
While our data indicate a significant association of these lipid classes in MetS BC, future studies are needed to elucidate their detailed structural characteristics and biological implications. However, our assessment demonstrated an association of these components within the BC which is consistent with their presence due to MetS. Comparison of BC composition between healthy and MetS conditions. Lipids identified as present in healthy BC were compared to lipids within the BC formed from MetS BALF for given time points on diets. Venn diagrams were used to illustrate the number of unique and common lipids. Relative abundance comparison of selected lipids found in healthy and MetS BCs. A value larger than 1 denotes a higher abundance in MetS BC compared to the healthy BC whereas a value less than 1 denotes lower abundance in the MetS BC compared to the healthy BC. All lipids depicted in the figure are significantly different between BCs based on p < 0.05 (n = 4/group). Blue bars = Healthy BC, Red bars = MetS BC.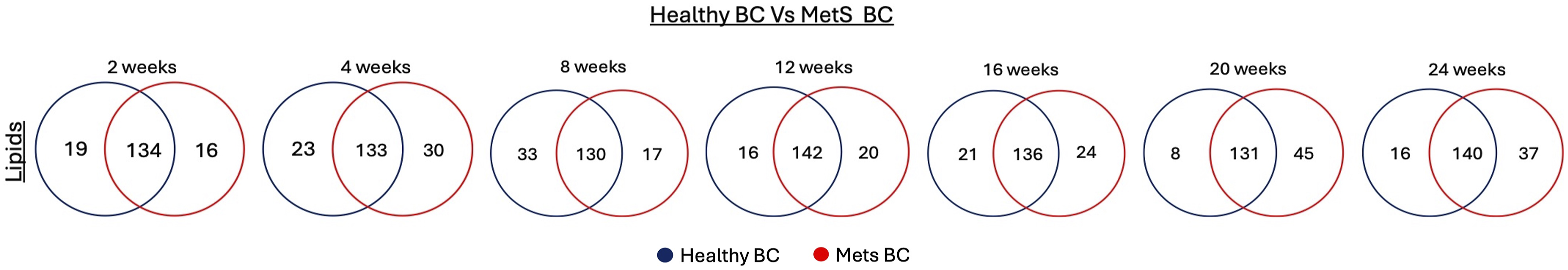

Compared to protein biocorona, lipid biocorona remains less explored, and available information is limited due to fewer studies in this area. 96 Overall, most of the lipid classes are conserved between healthy and MetS BC. However, a significant alteration was observed at the lipid species level as age and disease progresses. This is indicated by selective enrichment or depletion of specific lipid species within the same class. Aging significantly affects lipid composition, specifically increased sphingolipid turnover. 97 Degradation of sphingolipid is largely driven by sphingomyelinase activity, which hydrolyzes sphingomyelin into ceramide. Elevated ceramide has been implicated in multiple age-related cellular dysfunctions.98,99 A study reported increase in ceramide and ceramide/SM ratio in blood, serum and tissues such as heart, liver of 24-month old rats compared to 3-month-old rats. 100 Another, study reported elevated levels of Cer (18:1, 20:0, 22:0, and 24:1) in brown adipose tissue (BAT) and Cer (18:1 and 20:0) levels in white adipose tissue (WAT) of 15-month-old mice compared to 2-month-old mice. 101 This aligns with our observed data of the selective enrichment of ceramides within healthy BC, which might be associated with age-related changes. Furthermore, the most abundant lipids in BALF are phospholipids, particularly, phosphocholines, accounting for ∼75% of all phospholipids. 102 They are integral to maintain the air-liquid interface and decrease surface tension. 103 However, their composition varies due to differences in pulmonary physiology, especially on exogenous lipid supply. A study conducted in mice fed different diets (starch-rich diet, sucrose-rich diet, or fat-rich diet) for 30 weeks affected surfactant homeostasis and composition. The data demonstrated characteristic modifications of phospholipid species such as an increase in PC16:0/16:1 in response to carbohydrates while decrease in PC14:0/14:0; PC16:0/14:0 and increase in PG16:0/18:1 and PG16:0/18:2 species on excess dietary fat intake compared to mice fed control diet. 104 Another study reported association of several different phospholipid species such as PE 34:3, PE 36:0, PG 40:6 and PG 40:1 in BALF with increasing BMI of subjects (mean BMI of 34.9 ± 7.5 kg/m2), demonstrating that different PC, PE species can be associated with diseases such as obesity or MetS. 105 This aligns with our data having selective lipid species, particularly higher PCs and PEs within MetS BC can be associated with disease progression. Additionally, it is indicated in literature that lipids such as phosphatidylcholine and sphingomyelin species are found to be associated with premature coronary heart disease (CHD) or MetS. A study conducted on subjects has reported that the individuals with CHD or MetS has higher PC 32:1, PC 38:3 and PC 38:4 levels. 106 Proteins are the large molecules tend to easily bind and adsorb the NP surface via electrostatic interactions, hydrophobic bonding, and entropy changes in conformation interactions.107–109 Moreover, highly abundant proteins in mouse BALF such as albumin and immunoglobulins may cover the NP surface, potentially hindering the lipid association with NPs.110,111 In contrast, lipids mostly interact through hydrophobic interactions, hence, can form a stable biocorona and with less variations in enriched lipids and functional versatility. 112 However, hydrophobicity alone does not drive the lipid association. Although fatty acids differ in hydrophobicity based on chain length and degree of saturation or branching, for example, C20:2 > C20:1 > C18:3 > C18:2 > C18:1 > C16:1 > C16:0, studies have shown no clear correlation between hydrophobicity and adsorption percentage. 82 This suggests that while hydrophobic interactions are essential for initial binding, other factors such as lipid availability, steric constraints, and competitive adsorption with proteins play a more dominant role in determining the final lipid corona composition. An investigation was conducted with different sized polystyrene NP incubated with mouse serum at different time points. 113 The report reveals a dynamic association of lipids BC over time compared to proteins potentially demonstrating the possible interactions of lipids with NP surface. Additionally, the study also demonstrates the binding pattern of apolipoproteins, cholesterol and triglycerides, emphasizing the protein and lipid relationship for BC analysis. 113 This aligns with our current data that depicted the specific dynamic nature of lipids competing against other biomolecules for the surface of nanoparticles.
BC proteins and lipids and pathways analysis with age and disease progression
To determine the functional categories, molecular pathways and/or biological processes of identified proteins and lipids within BCs, we performed pathway analysis for a global interpretation of the interactions observed. Protein enrichment and pathway analysis using KEGG and Reactome databases were employed to assess the statistically significant proteins identified in healthy and MetS BCs at each timepoint. This enrichment analysis identifies biological pathways and processes potentially disrupted by NP-biomolecule interactions in BALF. Analysis with the KEGG database provided the top 10 enriched pathways emphasizing specific signaling and metabolic pathways. These included the carbon metabolism pathway, regulation of actin cytoskeleton, biosynthesis and metabolism of amino acid pathways at almost all given time points from 2 to 20 weeks suggesting age-related changes. However, pathways such as complement and coagulation cascade, cholesterol metabolism, mTOR signaling pathway, HIF−1 signaling pathway, phagosome pathway, and others were identified at later time points from 12 to 24 weeks reflecting disease-related association (Supplement Figure 6). Similar analysis was performed using the Reactome database to understand the molecular interactions and biological processes potentially perturbed by BC formation. Pathways identified such as hedgehog ligand biogenesis, GTP hydrolysis, cellular responses to stress were involved at earlier time points (2 to 8 weeks on diet) while regulation of complement cascade, chylomicron remodeling, plasma lipoprotein assembly, platelet activation, signaling and aggregation were identified at later time points (20 and 24 weeks on diet) of disease progression (Supplement Figure 7).
Lipid classification was performed using lipid ontology (LION enrichment analysis) for statistically significant lipids between healthy and MetS BC at each timepoint. Different lipids classes enriched on NP-BC over age and disease progression (2 to 20 weeks on diet) includes steryl esters, polyunsaturated fatty acids, fatty acid with 19−21 carbons, fatty acid with more than 3 double bonds, C18:3 etc. as opposed to lipid classes such as fatty acid with 16−18 carbons, fatty acid with less than 2 double bonds, lipid−mediated signalling, glycerolipids etc. were enriched only at 24 weeks on diet (Supplement Figure 8). This approach allows us to performtemporal assessment of shifts in class of lipids across time, suggesting of enrichment of lipids on NP-BC associated with age and disease progression. This analysis captures complementary insights into the biological pathways and processes involved in the formation of BC integrated with different lipid class enrichment potentially influenced by NP–biomolecule interactions, thereby enhancing the robustness and depth of our analysis.
Conclusion
Biofluid composition is a critical determinant governing BC identity, influencing how NPs interact with biological systems. In this study, BALF from healthy and MetS mice, collected at different time points on either healthy or high-fat diets were utilized to form BC following incubation in Fe3O4 NP. Our findings revealed distinct BC profiles between healthy mice across selected time points, suggesting age-related BC alterations. Moreover, disease-related changes in the biological milieu significantly impacted NP-biomolecular interactions as evidenced by specific proteins and lipids enriched on MetS BC. Comparison of BCs as the disease state progresses will aid in understanding NP-biomolecular interactions that may contribute to inflammation, toxicity, and susceptibility observed in our population. This may be increasingly significant as our population ages and the prevalence of chronic diseases rises. Formation of these unique BCs modifies biological interactions potentially influencing cellular uptake, immune responses, and functional outcomes. Our evaluation is considered comprehensive due to the inclusion of multiple factors including age and disease progression as well as our characterization of both protein and lipid BC components. Future assessments are required to elucidate how variations in BC composition influence susceptibility particularly in populations with underlying health conditions or age-related vulnerabilities.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Supplemental material
Supplemental material - Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression
Supplemental material for Pulmonary iron oxide (Fe3O4) nanoparticle-biomolecule interactions modified during aging and metabolic syndrome disease progression by Akshada Shinde, Li Xia, Venkatesh P. Thirumalaikumar, Christina R. Ferreira, Nadia Lanman, Jonathan Shannahan in Human & Experimental Toxicology.
Footnotes
Acknowledgments
The authors would like to acknowledge the support of the Purdue Metabolite Profiling Facility and Purdue Proteomics Facility for their technical support in generating the data presented within the manuscript as well as Purdue University Institute for Cancer for their bioinformatics analysis support from the Research (Grant P30CA023168) and the Walther Cancer Foundation.
Author contributions
Conceptualization, J.H.S. and A.S.; methodology, A.S., L.X., N.L., V.P.T.K., and C.R.F.; software validation, A.S.; formal analysis, A.S., N.L.; data curation, A.S., N.L., V.P.T.K. and C.R.F.; writing— original draft preparation, A.S.; writing—review and editing, A.S., J.H.S., V.P.T.K. and C.R.F.; funding acquisition, J.H.S. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study is from National Institute of Environmental Health Sciences (NIEHS) grant R01ES033173.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional review board statement
The animal study protocol was approved by the Purdue University Animal Care and Use Committee (Protocol Code: 2111002222 and Approved On: 12/16/21).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
