Abstract
Introduction:
ST segment elevation myocardial infarction (STEMI) due to coronary artery occlusion caused by intracoronary thrombosis in the setting of acute carbon monoxide (CO) poisoning is a very rare presentation. We present a case of intracoronary large and mobile thrombus formation after CO poisoning.
Case presentation:
A previously healthy 50-year-old woman was referred for CO poisoning. She had chest pain after exposure to CO. Her initial mental status was preoccupied with chest pain. Her initial CO fraction was 28.1%, and initial laboratory data showed creatine kinase–myocardial isoenzyme of 134 U/L (upper limit 25 U/L) and troponin I of >50 ng/mL (upper limit 0.06 ng/mL). Electrocardiography was carried out on admission, revealing an ST segment elevation in the inferolateral leads. After initial evaluation, coronary angiography was performed and an intracoronary large mobile thrombus was seen in the proximal left anterior descending (LAD) artery with no significant stenosis. We administered tenecteplase with heparin. After the thrombolytic therapy, ST elevation in the inferolateral leads resolved. Repeat angiography was performed after 24 h; the thrombus in LAD had resolved. The patient was discharged after 5 days, with persistent Q wave in the inferior leads and mild hypokinesia of the inferoposterior wall suggesting myocardial injury.
Conclusion:
We describe intracoronary thrombus formation induced by CO poisoning. Because intracoronary thrombus can result in myocardial infarction, its consideration following CO poisoning is important. Patients with CO poisoning who have symptoms of STEMI should be carefully evaluated with serial electrocardiograms, cardiac biomarkers, and an echocardiogram. When there is evidence of acute myocardial injury, a primer in coronary angiography can determine which patients could benefit from intervention.
Keywords
Introduction
Carbon monoxide (CO) is a colorless, odorless, and nonirritant gas that is lighter than air. 1 CO intoxications can be accidental, suicidal, or homicidal. Although the exact toxic mechanism following exposure to CO remains unclear, the major cause of death is multiorgan failure and encephalopathic brain death by tissue hypoxia. 2 Acute CO intoxication may be fatal. 3 Moreover, frequent neurologic and cardiovascular consequences have been defined. 4 The neurologic manifestations of CO intoxications include headache, dizziness, nausea, weakness, and confusion. Cardiac consequences of CO intoxications include arrhythmias and electrocardiographic (ECG) alterations, acute myocardial infarction, pulmonary edema, and cardiogenic shock. 5 Myocardial injury is common in patients with moderate to severe CO poisoning manifested as elevated cardiac biomarkers and the changes of regional wall motion abnormality on echocardiography. 6
Case
A 50-year-old woman with no preexisting disease was brought to the emergency unit with chest pain after the exposure to CO. The duration of the exposure to CO was unclear. During first clinical evaluation, she showed a good general appearance, the level of consciousness was alert, and Glasgow Coma Scale was measured up to 15. Vital signs such as blood pressure, pulse rate, respiration rate, and body temperature were in the range of 147/91 mmHg, 102 pulses/min, 21 breaths/min, and 36.3°C, respectively. Oxygen saturation measured using pulse oximetry was 98% when 5 L/min oxygen was applied through a reservoir bag mask. Peripheral arterial and venous examinations showed normal circulation. No pathological neurologic signs were detected after detailed system examination.
The chest radiography was normal. Laboratory analyses revealed cardiac enzymes were elevated with a troponin I of >50 ng/mL and creatine kinase–myocardial isoenzyme of 134 U/L and arterial blood gas revealed pH 7.37, partial pressure of carbon dioxide 46.3 mmHg, partial pressure of oxygen 62 mmHg, bicarbonate 23 mmol/L, saturation level of oxygen 86%, and carboxyhemoglobin 28.1% (reference range <2.5%). Also, white blood cells, 11,600 cells/μL; blood urea nitrogen, 18 mg/dL; creatinine, 0.81 mg/dL; and blood glucose, 109 mg/dL (Table 1).
Serum biomarkers of the patient before and after therapy.
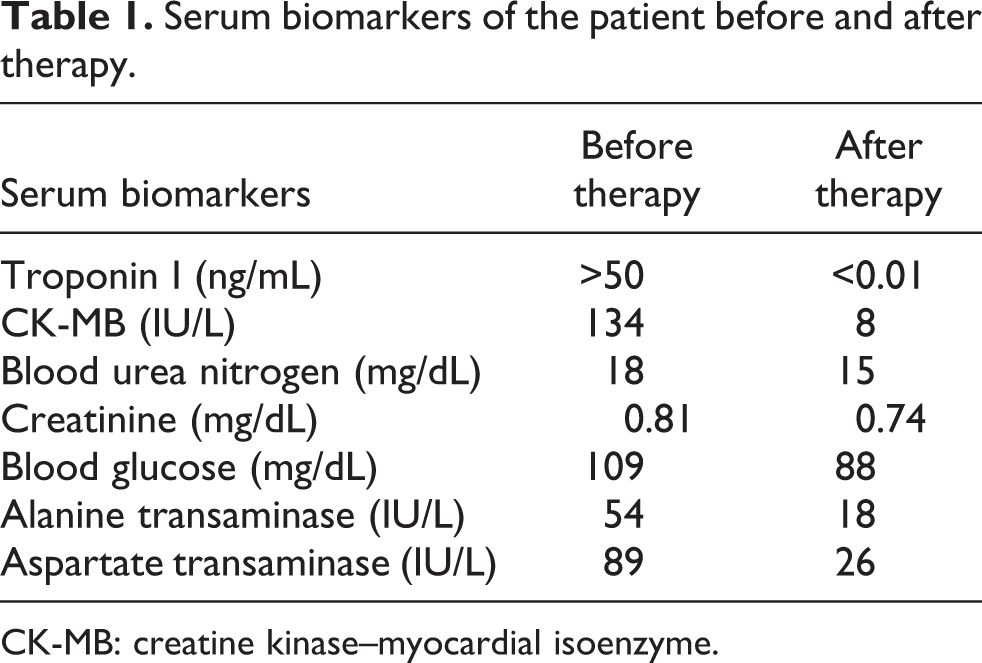
CK-MB: creatine kinase–myocardial isoenzyme.
The initial ECG findings revealed sinus tachycardia with a heart rate of approximately 105 beats/min, ST segment elevation in II, III, aVF, and V5-V6 and ST segment depression in V1-V3 and aVL (Figure 1(a)). A bedside transthoracic echocardiogram demonstrated an inferoposterior hypokinesia of the left ventricle, with an ejection fraction of 55%. Primary coronary angiography was performed; intracoronary large and mobile thrombus was viewed in proximal left anterior descending (LAD) artery with the remaining arteries being normal (Figure 2(a) and (c), Video 1). Localization of intracoronary thrombus was not compatible with ECG finding. The patient was transferred to the intensive care unit and given oxygen for treatment of CO poisoning. Thrombolytic therapy (tenecteplase 10 mg) was started at same time with heparin. ST elevation and chest pain resolved after the thrombolytic and anticoagulant therapy (Figure 1(b)). Control angiography was performed after 24 h; thrombus in LAD disappeared (Figure 2(b) and (d), Video 2). Serial cardiac biomarkers were performed and normalized after several days. The patient was discharged home after 5 days, with persistent Q wave in inferior leads and mild hypokinesia of the inferoposterior wall suggesting myocardial injury.

(a) Electrocardiograms with ST elevation suggesting inferolateral myocardial infarction on admission and (b) control electrocardiogram taken before discharge.

(a) Coronary angiography in the right anterior oblique view showing large thrombus in proximal LAD artery and no coronary stenosis, (b) right anterior oblique view showing disappeared thrombus in proximal LAD artery, (c) lateral view showing large thrombus in proximal LAD artery (arrow), and (d) lateral view showing disappeared thrombus in proximal LAD artery. LAD: left anterior descending.
Discussion
CO poisoning has particular impact on organs which are sensitive to oxygen deprivation such as the heart, brain, and kidney. CO poisoning (moderate to severe) can cause myocardial injury even in patients with normal coronary arteries, assessed by ECG and cardiac enzyme elevation. 4 Generally, myocardial dysfunction is transient and normalizes with conventional treatment including high concentration of oxygen.
Various mechanisms have been proposed like global myocardial dysfunction and resultant myocardial stunning. The affinity of hemoglobin for CO is 200 times greater than its affinity for oxygen, which leads to tissue hypoxia. 7 Furthermore, the release of oxygen free radicals, cellular apoptosis, and atherogenesis are important mechanisms of CO toxicity. 2 Although it is usually accepted that endogenous CO has antithrombotic activity, an environmental CO exposure can increase the tendency to intravascular thrombosis, presumably due to CO-induced enhancement of coagulation by binding to a fibrinogen-bound heme. 8
Acceptable explanation for thromboembolic events is the effect of CO on platelet aggregation and reduced oxygen supply to the myocardium that might induce coronary artery spasm with accompanying coronary thrombosis. Furthermore, CO poisoning leads to some changes in blood vessel and CO and nitric oxide (NO) on platelet. Troubled mitochondrial mechanisms by NO and its derivatives facilitate production of free oxygen radicals. 9 Resultant oxidative stress may lead to endothelial damage and subsequent platelet aggregation. 10
Two different clinical patterns of cardiovascular symptoms have been suggested in CO poisoning 11 First, younger patients with few cardiac risk factors, left ventricular global hypokinesia, in whom normal coronary arteries are frequently detected. Second, older patients with multiple cardiac risk factors and regional wall motion abnormalities as a result of preexisting coronary artery disease. We detected intracoronary thrombus in LAD with inferolateral ST elevation. Localization of intracoronary thrombus was not compatible with the ECG finding. As in our case, regional ventricular hypokinesia does not necessarily imply coronary occlusion as spontaneous lysis of an intracoronary thrombus or resolution of coronary vasospasm might occur before angiography. 12 Additionally, direct toxic effects of CO on mitochondria might produce a “stunned myocardium-like syndrome” with focal hypokinesia and completely normal coronary angiograms. 13
Single case reports on successful primary percutaneous intervention (PCI) in patients with ST segment elevation myocardial infarction (STEMI) in the setting of CO poisoning have been reported. 14 Although young age and at low risk for coronary artery disease, our patient had persistent ST segment elevation with chest pain and regional wall motion abnormalities when we performed primary coronary angiography. Primary coronary angiography was performed but not PCI because the coronary arteries were normal. We decided on thrombolysis and the ECG findings and intracoronary thrombus disappeared.
Conclusions
Patients presenting with CO poisoning and symptoms of STEMI should be carefully evaluated with serial electrocardiograms and cardiac necrosis marker measurements. When there is evidence of myocardial injury, primary coronary angiography can identify patients who could benefit from thrombolysis and PCI.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
