Abstract
Introduction
This study examined the cardioprotective effects of Pelargonidin-3-O-glucoside (Pg3G) against myocardial infarction induced by isoproterenol (ISO) in male Wistar rats.
Methods
Animals were divided into four groups each groups contain six animals. Group 1 control; Group 2 Pg3G treated control; Group 3 ISO-control; Group 4 Pg3G + ISO treated rats. At the end of the experiment period the animals were sacrificed and collected the serum, heart tissue used for the experimental work.
Results
According to the network pharmacology analysis, Prostaglandin-endoperoxide Synthase 2 (PTGS2), Matrix metallo proteins −9 (MMP-9), and tumour necrosis factor-alpha (TNF-α) were identified as potential targets among the 97 common targets between Pg3G and myocardial injury. Further, we investigated that prominent cardiac indicator such creatine kinase (CK), CK-MB, cardiac troponin T (cTnT), and cardiac troponin I (cTnI) were not elevated by ISO in the presence of Pg3G administration. Additionally, Pg3G administration decreased the pro-inflammatory cytokines generated by ISO, including as interleukin-6 (IL-6), interleukin-1 beta (IL-1β), and TNF-α, suggesting its anti-inflammatory qualities. Additionally, Pg3G increased levels of reduced glutathione (GSH) and restored the activity of important antioxidant enzymes that were depleted by ISO-induced oxidative stress, including glutathione peroxidase (GPx), catalase (CAT), and superoxide dismutase (SOD).
Discussion
Pg3G capacity to reduce ISO-induced inflammatory, fibrotic, and cardiac toxicity markers in myocardial tissue was demonstrated by gene expression investigations. Therefore, Pg3G may be considered for ISO-induced cardiac injury since it provides significant cardioprotection by reducing oxidative stress, inflammation, and fibrosis.
➢ This study shows that Pelargonidin-3-O-glucoside (Pg3G) prevents isoproterenol-induced myocardial infarction. The importance of Pg3G is found in its many functions as an effective anti-inflammatory, anti-fibrotic, and antioxidant. ➢ By reducing oxidative stress, Pg3G protects cardiac tissues by preventing lipid peroxidation, maintaining cardiac biomarkers, and raising antioxidant enzyme levels. ➢ It also reduces inflammatory cytokines, preventing fibrosis and chronic inflammation. ➢ This study suggests that Pg3G may improve cardiovascular outcomes and lay the groundwork for future heart disease treatments.Significance of the study
Introduction
Myocardial infarction (MI) is a most prevalent cause of cardiovascular morbidity and mortality across the world. Cardiovascular diseases (CVDs) are a significant public health issue, responsible for approximately 17.9 million deaths per year, constitutes for 31% of global mortality. 1 Myocardial infarction is induced by coronary artery obstruction, which diminishes the myocardium’s supply of oxygen and nutrients, resulting in irreversible damage to the cardiac tissues. Cardiac dysfunction is exacerbated by oxidative stress, inflammation, and fibrosis, which contribute to the pathophysiology of MI. 2 Despite recent advancements in the management of MI patients, the safer, non-toxic and effective treatment remains warranted.
Natural medicinal bioactive compounds gained significant interest for their cardioprotective activities. Anthocyanins, a major class of flavonoids found in various fruits and vegetables, demonstrate significant cardioprotective properties. 3 Pg3G is a predominant anthocyanin mainly present in berries. The cardioprotective properties of anthocyanins are mainly due to their capacity to scavenge reactive oxygen species (ROS), prevents lipid peroxidation, and modulates redox-sensitive signalling pathways. 4 Furthermore, anthocyanins showed the ability to modulate the expression of inflammation, apoptosis, and fibrosis related genes thereby prevents cardiac injury. 5 The Pg3G is a anthocyanin endowed with antioxidant properties and prevents oxidative damages.6,7 Although extensive studies carried out for its pharmacological properties, the preventive effect against cardiac injury has not been investigated. Therefore, a systematic investigation is necessary to elucidate the specific impacts of Pg3G on ISO-induced MI. The ISO-induced MI model is a widely utilised experimental approach for investigating the processes of myocardial injury and evaluating the cardioprotective properties of various drugs. 8 ISO, a synthetic catecholamine, induces cardiac hypertrophy, oxidative stress, fibrosis, and myocardial necrosis to mimic human MI. 9 The ISO causes MI by stimulating β-adrenergic receptors, increasing ROS, and causing mitochondrial dysfunction in cardiac cells. 10 These alterations caused heart injury and fibrosis via molecular signalling. 11 ISO-treated rats have increased CK and cardiac troponin levels, indicating myocardial damage. ISO injection causes extracellular matrix breakdown and cardiac fibrosis by activating MMPs and pro-inflammatory cytokines. 12 MI is caused and advanced by oxidative stress. 13 Ischemia-reperfusion injury overproduces ROS, surpassing the heart’s antioxidant defences and damaging proteins, lipids, and DNA. 14 The pathophysiology of MI involves inflammation, which supports tissue repair and damage. TNF-α and IL-6, pro-inflammatory cytokines, increase after MI and encourage immune cell migration. TNF-α and IL-6 are pro-inflammatory cytokines that increase after MI. 15 This results in the recruitment of immune cells to the injured myocardium. Persistent inflammation can lead to detrimental cardiac remodelling, fibrosis, and heart failure. 16 The MMPs, such as MMP-2 and MMP-9, induces cardiac remodelling and fibrosis by degrading the extracellular matrix. 17 Natural pharmaceutical substances with anti-inflammatory and antioxidant properties are actively investigated for preventing ISO-induced MI. In many disease models, 18 Pg3G has been shown to neutralise ROS, reduce lipid peroxidation, and alter the synthesis of inflammatory mediators. 19 Pg3G also controls MMPs and TIMPs, which modulate cardiac fibrosis signaling. 20 Pg3G modulates inflammation, fibrosis, and oxidative stress molecular pathways. 21 The heart’s ability to attenuate ROS and oxidative damage is improved by Pg3G therapy, which increases antioxidant enzymes such glutathione peroxidase, catalase, and SOD. 22 Additionally, Pg3G suppresses pro-inflammatory transcription factors, like NF-κB, which generate inflammatory cytokines. 23 Pg3G impacts the expression of key fibrosis regulators, such as TIMP-1 and PPAR-γ, which are essential for maintaining the extracellular matrix. 24 This study shows that Pg3G prevents ISO-induced myocardial infarction in Wistar male rats.
Methodology
Target retrieval and identification of intersecting genes
Pelargonidin-3-O-glucoside (Pg3G) chemical structure and SMILES were obtained from PubChem database. The SuperPred and SwissTargetPrediction was used to predict Pg3G’s molecular targets. Both NCBI-Gene and GeneCards databases were used for finding myocardial injury targets. After assembling target lists, duplicate entries were carefully removed for accuracy. Finally, using Venny 2.1.0, Pg3G’s shared targets with myocardial injury were intersected and visualised, suggesting plausible pathways of action.
Building of protein-protein interaction (PPI) network & HUB target prediction
Pelargonidin-3-O-glucoside (Pg3G) and heart injury targets were linked in a Homosapiens-based PPI network. The network was built by importing the common targets into the STRING database, which has great protein interaction data. STRING created a detailed PPI network showing target interactions and connections. To enhance analysis, PPI network data was merged with Cytoscape 3.7.2, a powerful molecular interaction network visualisation tool. Cytoscape’s cytoHubba plug-in identified network targets. CytoHubba ranks nodes using several topological methods; this study focused on connectivity. The top 10 targets with the highest node-connect degrees were critical hub proteins are expected to play important roles in the interaction network and may be considered as therapeutic targets for myocardial injury.
Gene ontology (GO) and kyoto encyclopedia of genes and genomes (KEGG) enrichment analysis
Furthermore, GO enrichment analysis was conducted to understand the biological processes (BP), cellular components (CC), and molecular functions (MF). The outcomes of the KEGG pathway and GO enrichment analyses were illustrated using SRplot, a tool that produces clear and informative visualisations of the enriched functional words, enhancing comprehension of the biological relationships within the dataset.
Molecular Docking Analysis
The three-dimensional chemical structures of Pg3G (PubChem CID: 3080714) were obtained from the PubChem database maintained by NCBI. The crystal structures of proteins such as NF-κB, MMP9, HIF1A, ESR1, EGFR, CXCR4, AKT1, TNF-α, SRC, and PTGS2, were downloaded from the PDB. The protein structures and ligand molecules were then converted to pdbqt files for molecular docking studies. Molecular docking simulations were carried out using Autodock Vina v1.5.6 (https://vina.scripps.edu). The findings of docking studies were analyzed using BIOVIA Discovery Studio Visualiser for understanding molecular interactions.
Chemicals
Pelargonidin-3-O-glucoside (Pg3G) was purchased from Sigma-Aldrich (USA). Before oral delivery, the chemical was solubilised in a 0.5% dimethyl sulfoxide (DMSO) solution. Isoproterenol (ISO) was procured from Tocris Bioscience (UK). Serum biomarkers suggestive of myocardial damage, such as CK and CK-MB were quantified utilising kits obtained from Randox Laboratories (UK). Cardiac Troponin I and Troponin T (cTnI and T) ELISA kits were purchased from Abcam (UK). RNA isolation kit (TRIzol), reverse transcription, and quantitative real-time PCR kit were procured from Invitrogen (USA).
Animals maintenance
The experimental animals were acclimatized in male albino Wistar rats (weighing 180–220 g) which were obtained from The Third People’s Hospital of Gansu Province, China. The animals were maintained at 25 ± 3°C with a light/dark cycle. Animal handling and experimental procedures were approved by the Institutional Animal Ethics Committee, The Third People’s Hospital of Gansu Province, China. The animals were cared in accordance with the “Guide for the Care and Use of Laboratory Animals” and “Committee for the Purpose of Control and Supervision on Experimental Animals”. The study plan and methods were approved by the Institutional Animal Ethics Committee of the Third People’s Hospital of Gansu Province, China (Approval number 20250281).
Myocardial infarction induction and dose dependent study
Myocardial infarction was induced in rats by subcutaneous administration of ISO at a dose of 85 mg/kg body weight) in the right thigh of the rat, dissolved in physiological saline, for two consecutive days (20th and 21st days). The 1st injection was given at 10 a.m. and the 2nd injection 10 a.m. the next day. Isoproterenol, a β-adrenergic agonist, induces myocardial necrosis, thereby mimicking myocardial infarction in experimental models. 25 All animals were monitored twice daily for signs of pain or distress (e.g., lethargy, proliferation, decreased food/water intake). Humane endpoints were predefined in accordance with institutional guidelines, including criteria for early euthanasia. No analgesics were administered, as the ISO dose and duration did not induce observable pain or distress, consistent with previous studies and approved protocols.
A preliminary study was conducted to determine the optimal dose of Pg3G by evaluating serum enzyme activities in ISO-treated rats. Pg3G was administered orally at doses of 0.5, 1, and 2 mg/kg body weight, once daily for 14 days. Among three doses, 1 mg/kg body weight showed the most significant effect and was thus selected for use in subsequent experiments (Figures S1A and S1B).
Experimental protocol
The rats were divided into four groups (n = 6). Group I (Control) animals were administered normal saline (0.9%). Group II (Pg3G Control Group) rats received Pg3G at a dosage of 1 mg/kg body weight for a duration of 21 days without ISO treatment. The Group III (ISO Group) animals were given ISO at a dosage of 85 mg/kg body weight over two consecutive days to induce MI. The Group IV (Pg3G + ISO Group) rats received pretreatment with Pg3G at a dosage of Pg3G at a dosage of 1 mg/kg body weight for a duration of 21 days. ISO was given on days 20 and 21 to induce MI, while Pg3G therapy persisted until the experimental period.
After the experimental period (21st day), the rats were subjected to overnight fasting and euthanised by anaesthesia. Blood samples were obtained using retro-orbital puncture, and the serum was isolated by centrifugation at 2000 rpm for 10 min. The separated serum was used for the analysis of cardiac marker enzymes and inflammatory markers. The heart tissue was excised immediately after cervical dislocation and used for further studies.
Biochemical estimations
The activities of CK and CK-MB were quantified using kits from Randox Laboratories (https://www.randox.com). cTnT and I levels were measured using ELISA kits from Abcam. 26 Lipid peroxidation (LPO) in cardiac tissue was quantified by assessing malondialdehyde (MDA) concentrations using the TBARS method. Antioxidant enzymes such as SOD, CAT, GPx and GSH were assessed using commercial kits from Abcam to measure the oxidative stress response.
Estimation of Serum Cytokines
Serum levels of TNF-α, IL-6, IL-1β and NF-κB were measured using ELISA kits as per manufacturer’s instructions (Invitrogen, USA). In the protocol, 50 μL of the sample and 50 μL of an antibody cocktail were added to the experimental wells and incubated at 37°C for 1 hr. The wells were then washed with 100 μL of 3,3′,5,5′-Tetramethylbenzidine substrates and incubated for 10 min. Finally, 100 μL of stop solution was added, and the color development was read at 450 nm using a microplate reader.
Molecular investigations
Primer sequences.

Statistical evaluation
Data were presented as mean ± standard error of the mean (SEM) for each group. Statistical comparisons were conducted using one-way analysis of variance (ANOVA) accompanied by post hoc analysis and Duncan’s Multiple Range Test (DMRT) using SPSS (version 21.0). A p-value below 0.05 was considered as statistically significant.
Results
Intersection of Pg3G and myocardial injury-related gene targets
A total of 5317 targets related to myocardial injury and 218 targets associated with Pg3G were identified after removing duplicate entries. Among these, 97 therapeutic targets were found to be common between Pg3G and myocardial injury, as shown in the Venn diagram (Figure 1(a)). To further explore the interactions between these shared targets, a protein-protein interaction (PPI) analysis was performed using STRING, and the resulting network was visualized in Cytoscape. The PPI network consisted of 95 nodes and 730 edges (Figure 1(b)). Topological analysis of the network revealed the top 10 most significant targets: AKT1, SRC, TNF, ESR1, EGFR, PTGS2, HIF1A, NFKB1, MMP9, and CXCR4, as shown in Figure 1(c). (a). Identification of Common Targets Between Pg3G and Myocardial Injury. The Venn diagram shows the overlap of targets for Pg3G and myocardial injury, identified using SuperPred, SwissTarget Prediction, NCBI-Gene, and Gene Cards, with intersection analysis by Venny 2.1.0. (b) PPI Network of Common Targets. The PPI network, constructed in the STRING database for Homo sapiens, maps interactions among the common targets, highlighting key proteins potentially involved in Pg3G’s therapeutic effects. (c) Identification of Top 10 Hub Proteins. The cytoHubba plug-in in Cytoscape 3.7.2 ranked targets by connectivity within the PPI network. The top 10 hub proteins, with the highest connect degrees, are shown, indicating their potential role in modulating myocardial injury through Pg3G d) Gene Ontology (GO) Analysis of Common Targets. This figure presents the Gene Ontology (GO) analysis of the common targets between Pg3G and myocardial injury. The analysis categorizes the targets based on their involvement in biological processes, molecular functions, and cellular components. Key enriched GO terms are highlighted, offering insights into the potential mechanisms through which Pg3G may influence pathways relevant to myocardial injury. e) Top five significant key signalling by KEGG pathway.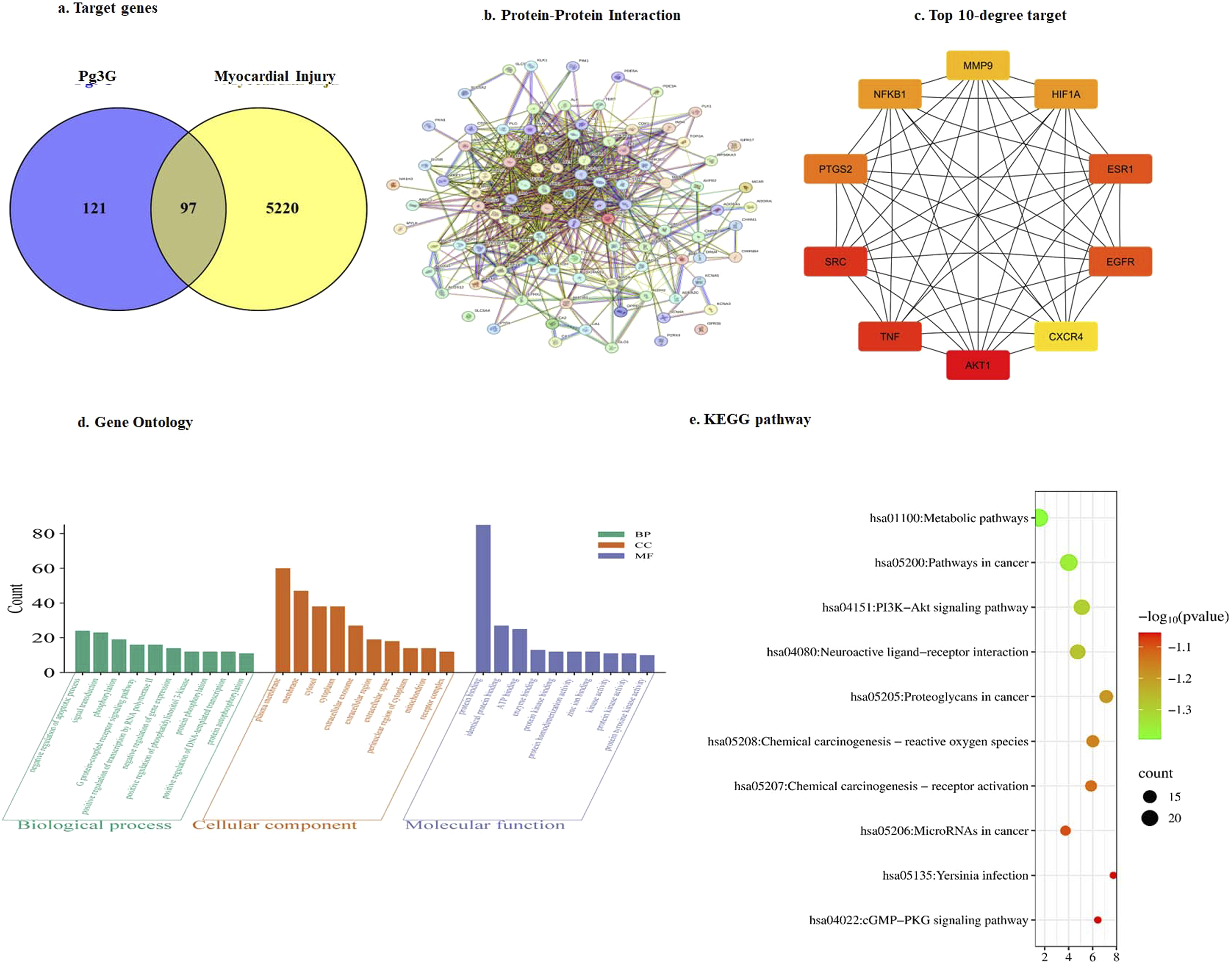
GO and KEGG pathway enrichment analysis with intersected targets
The 97 common targets associated with Pg3G and Myocardial Injury were subjected to GO and KEGG pathway enrichment analysis at a significance level of P < 0.05. In the GO enrichment analysis, a total of 287 biological processes, 43 cellular components, and 87 molecular functions were identified, with the top 10 terms selected for further analysis
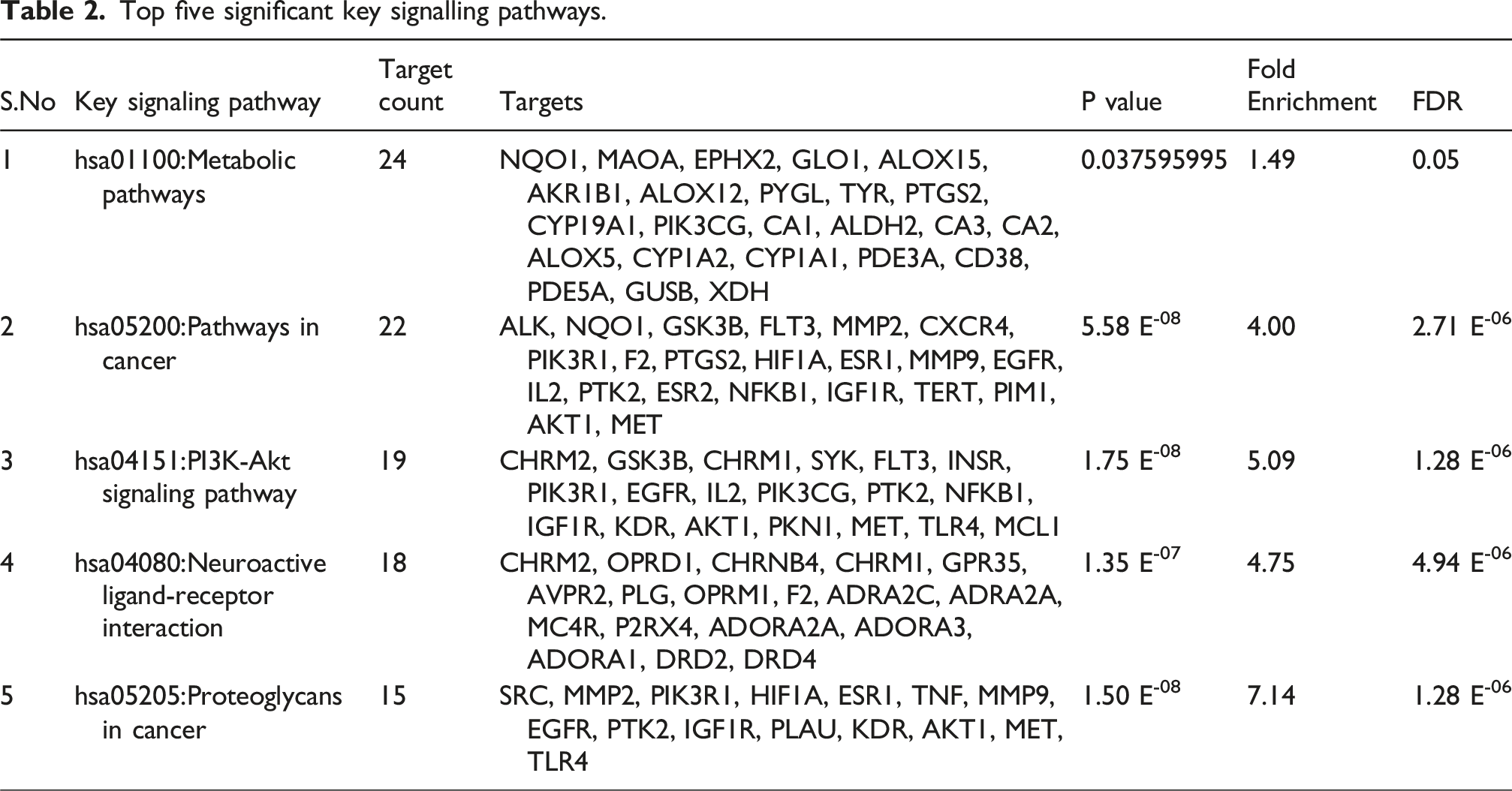
Top five significant key signalling pathways.
Binding interaction of Pg3G with significant targets
Binding interaction of Pg3G with significant targets.
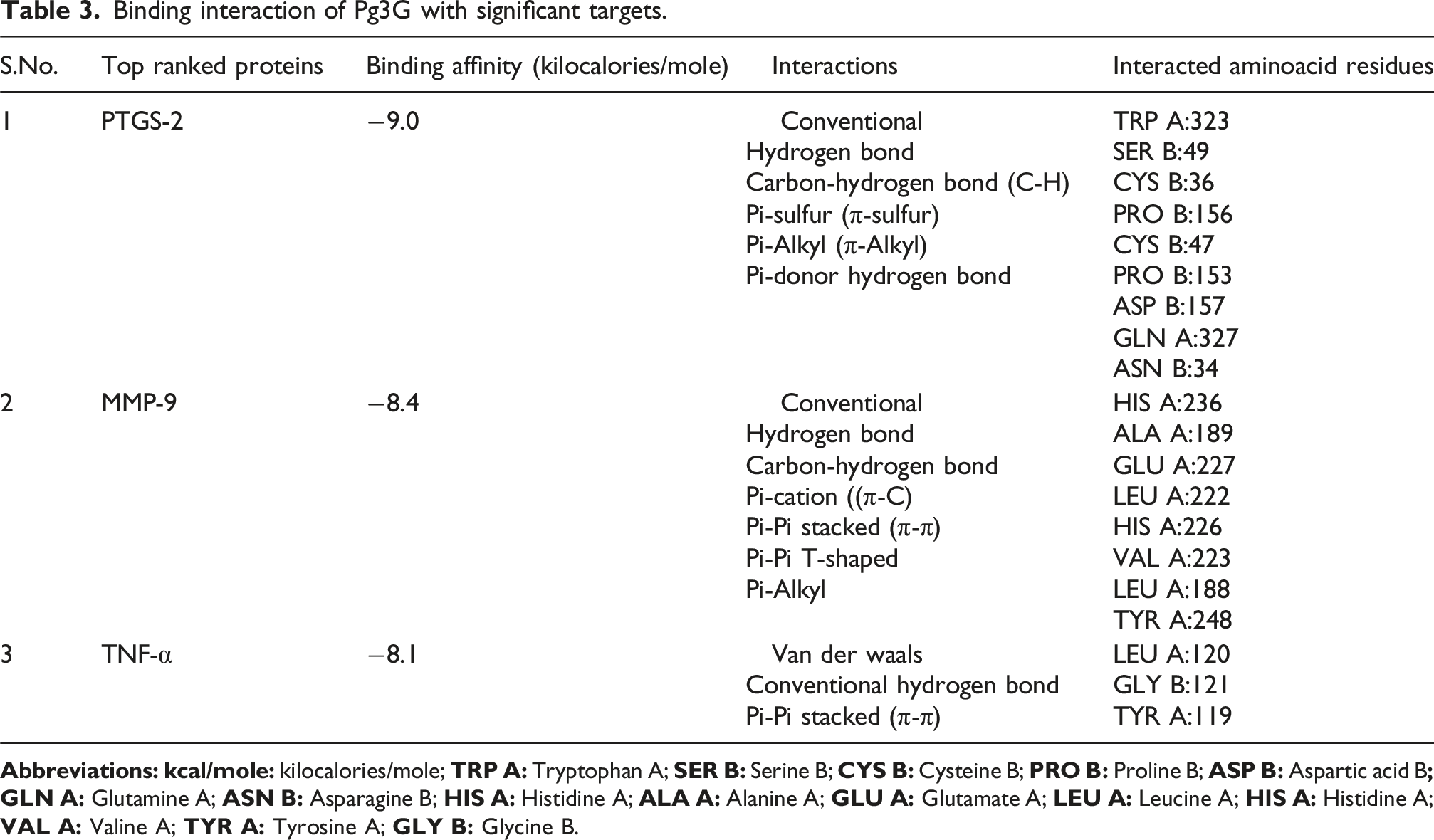

Molecular Docking Analysis of Pg3G with Key Target Proteins. The figure presents the molecular docking results of Pg3G with key target proteins identified from the PPI network. Docking scores reflect the binding affinity between Pg3G and its target proteins, with lower scores indicating stronger interactions. Pg3G showed binding affinities of −9.0 kcal/mol for PTGS2, –8.4 kcal/mol for MMP9, and –8.1 kcal/mol for TNF, suggesting its potential to interact with these proteins and modulate pathways involved in myocardial injury.
Impact of Pg3G on serum cardiac marker levels (CK and CK-MB) in control and ISO-Induced MI rats
Figure 3 illustrates the effects of Pg3G on serum levels of cardiac markers, specifically CK and CK-MB, across various experimental groups. The control group serves as a baseline, showing normal CK and CK-MB levels, whereas the ISO-induced group (MI model) exhibits significantly elevated levels due to cardiac damage. The group treated with Pg3G prior to ISO administration shows a noticeable reduction in CK and CK-MB levels compared to the ISO-only group, highlighting the cardioprotective effect of Pg3G. The reduction of these markers suggests that Pg3G helps mitigate the myocardial injury typically induced by isoproterenol. Impact of Pg3G on CK and CK-MB levels in the serum of control and ISO-induced Rats. Data are presented as the mean ± SEM based on six independent experiments per group. Groups that do not share the same superscript letters (a, b, c, etc.) exhibit statistically significant differences at p ≤ 0.05, as determined by Duncan’s Multiple Range Test (DMRT).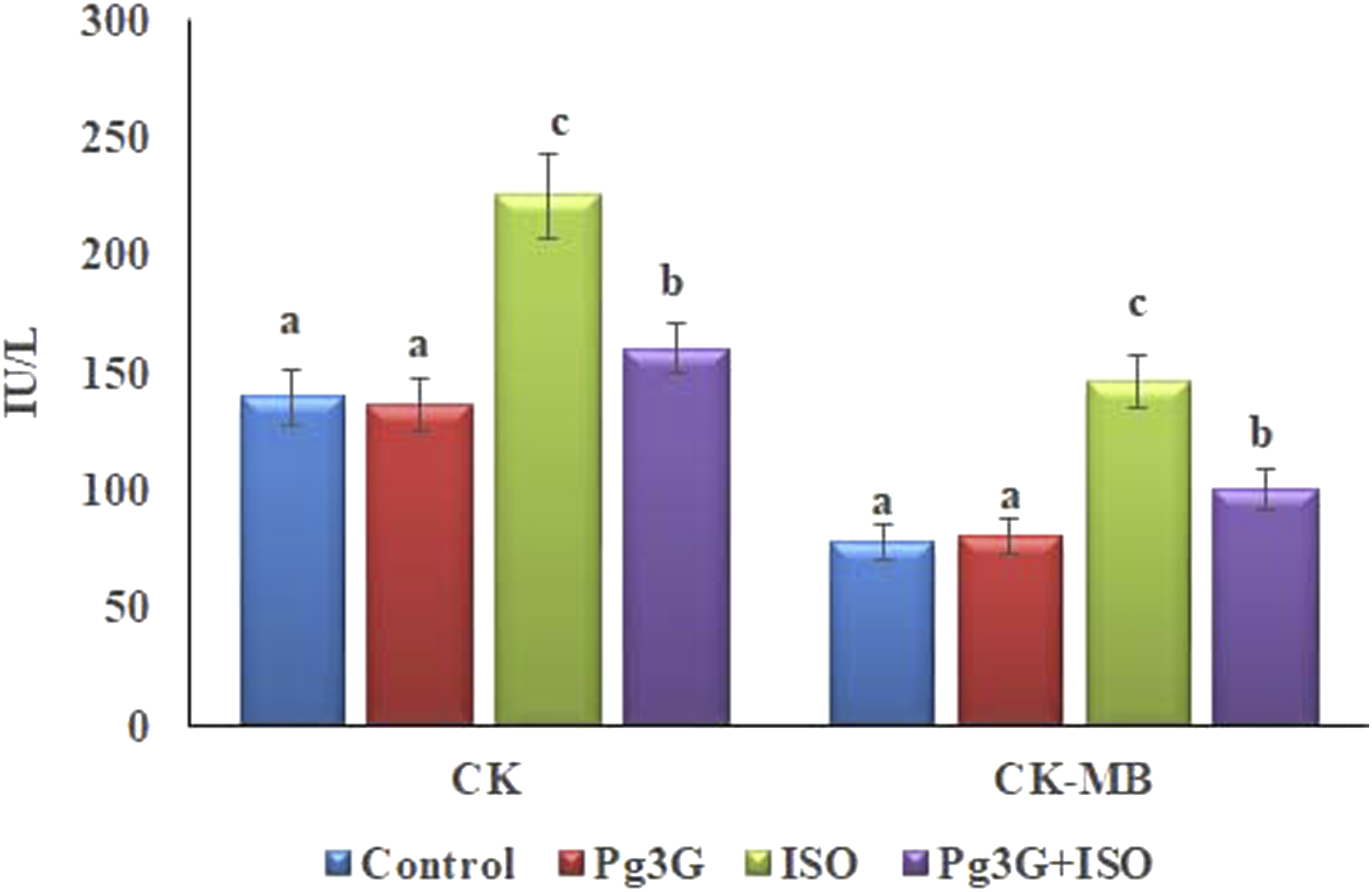
Effect of Pg3G on serum cTnT and cTnI levels
The serum level of cTnT was significantly elevated in the ISO-treated group (2.99 ± 1.22 ng/mL) compared to control (0.62 ± 0.55 ng/mL. Treatment with Pg3G in the ISO-induced MI group led to a reduction in cTnT level to 1.23 ± 0.04 ng/mL, indicating a protective effect against myocardial injury. Similarly, cTnI level was notably higher in the ISO group (1.23 ± 1.00 ng/mL) than in the control (0.55 ± 0.24 ng/mL). In the Pg3G + ISO group, cTnI level returned to 0.55 ± 0.66 ng/mL, further supporting the cardioprotective effects of Pg3G (Figure 4). The effect of Pg3G on cTnT and cTnI in the serum of control and ISO-induced rats. Values represent means ± SEM from six independent experiments in each group. Groups that do not share a common superscript (a, b, c) differ significantly at p ≤ 0.05, as determined by the Duncan’s Multiple Range Test (DMRT).
Effect of Pg3G on serum pro-inflammatory cytokines
The level of TNF-α was significantly increased (78.81 ± 4.25 pg/mL) in the ISO-induced group when compared to control group (38.37 ± 1.50 pg/mL). However, Pg3G + ISO group demonstrated a significant decrease in the level of TNF-α
(44.36 ± 3.56 pg/mL) compared to the ISO-treated group. In the control group, The level of IL-6 was significantly increased (120.54 ± 8.65 pg/mL) in the ISO-treated group when compared to control group (71.9 ± 4.20 pg/mL). While treatment with Pg3G + ISO group suggested that a significant decrease in the level of IL-6 (81.32 ± 6.49 pg/mL) compared to the ISO-treated group. In the control group, IL-1β level was significantly increase in ISO-induced group (94.46 ± 6.49 pg/mL) (p ≤ 0.05) when comparted to control group (52.47 ± 2.50 pg/mL). While Pg3G + ISO treatment significantly decreased IL-1β level to 62.11 ± 3.52 pg/mL (p ≤ 0.05, suggesting a modulating effect of the combined treatment on inflammation. In, NF-κB level was 108. 26 ± 9.57 pg/mL significantly increased ISO-induced group when compared to control group 64.28 ± 4.12. pg/mL However, Pg3G + ISO treatment significantly reduced NF-κB level 78.51 ± 6.09 pg/mL when compared to ISO-treated group (p ≤ 0.05) (Figure 5). Effect of Pg3G on pro-inflammatory cytokines of TNF-α, IL-6, IL-1β and NF-κB in the serum of control and ISO-induced rats. Values are expressed as means ± SEM. Statistical significance is denoted by *p ≤ 0.05 and **p ≤ 0.01. TNF-α, IL-6, IL-1β and NF-κB levels were significantly altered in response to treatments, as shown in the corresponding bar graph.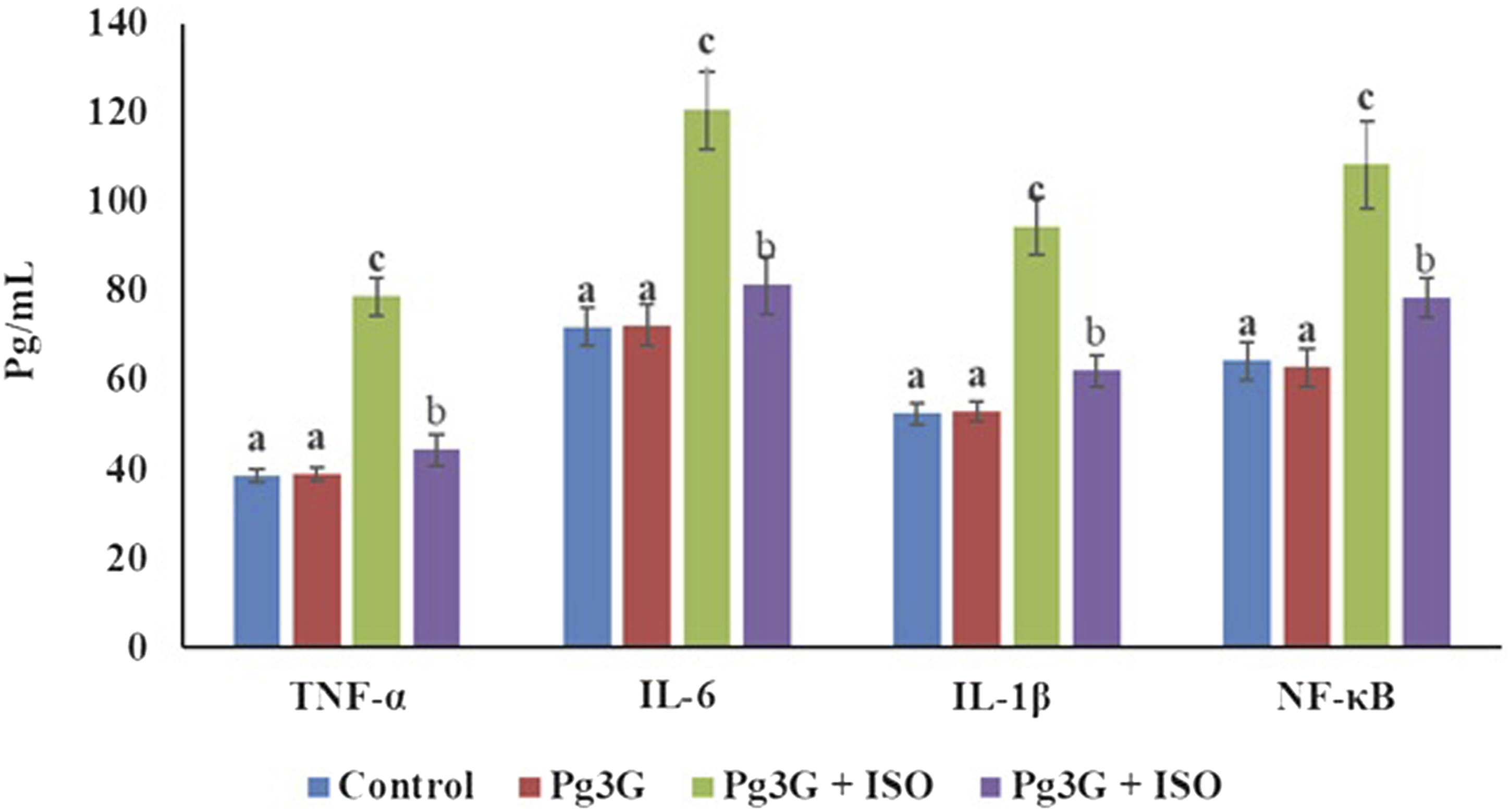
Effect of Pg3G on the activities of antioxidant enzymes in the rat heart tissue
Impact of Pg3G on TBARS and LHP levels in the heart tissues of control and ISO-induced rats.

Data are presented as mean ± SEM from six independent experiments (n = 6).
Means with different superscript letters (a, b, c, etc.) indicate significant differences at p ≤ 0.05 (DMRT).
Impact of Pg3G on the activities of antioxidant enzymes in the heart tissues of control and ISO-induced rats.

Values are expressed as means ± S.D. for six rats in each group. Values not sharing a common superscript (a, b, c, etc.) differ significantly at p ≤ 0.05 (DMRT). U* - Enzyme concentration required to inhibit the chromogen produced by 50% in 1 min under standard conditions (SOD activity). U** - µmole of hydrogen peroxide decomposed per minute (CAT activity). U@ - µmole of GSH utilized per minute (GPx activity).
Effect of Pg3G on ISO-induced fibrotic gene expression profile in heart tissue
The hierarchical clustergram analysis of PCR array results revealed significant alterations in fibrotic gene expression profiles in heart tissues from Pg3G-treated rats compared to ISO-induced rats. As illustrated in Figure 6, the expression levels of CK was markedly reduced in the Pg3G-treated group, indicating a protective effect against cardiac injury. Additionally, levels of cTnT and cTnI demonstrated significant downregulation, suggesting that Pg3G helps maintain cardiomyocyte integrity following ISO-induced damage. Moreover, pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β, exhibited substantial decreases in expression in Pg3G-treated hearts, highlighting its potential in alleviating inflammation associated with myocardial injury. The expression of MMP-2 and -9 was also significantly modulated, with a decrease indicating that Pg3G may contribute to the regulation of extracellular matrix remodeling, essential for cardiac repair processes. Furthermore, Pg3G treatment resulted in notable downregulation of PTGS2, involved in pro-inflammatory prostaglandin synthesis, suggesting a role in mitigating the inflammatory response in cardiac tissues. Collectively, these results demonstrate that Pg3G significantly alters the expression of key fibrotic and inflammatory genes in ISO-induced rat heart tissue, suggesting its potential therapeutic role in protecting against cardiac fibrosis and injury. Effect of Pg3G on ISO-induced cardiac, inflammatory and fibrotic gene expression profile in heart tissue. This analysis presents a hierarchical clustergram of PCR array results of Pg3G and ISO-induced rats. In the clustergram, bright red indicates the highest normalized signal values, bright green represents the lowest signal values, and black denotes median signal values. Total mRNA was isolated from heart tissues and analyzed using a custom PCR array following the manufacturer’s instructions. The clustergram results of three independent experiments were analyzed using the SA Biosciences online tool. Key genes analyzed include CK, cTnT, cTnI, TNF-α, IL-6, IL-1β, MMP-2, -9, and PTGS2.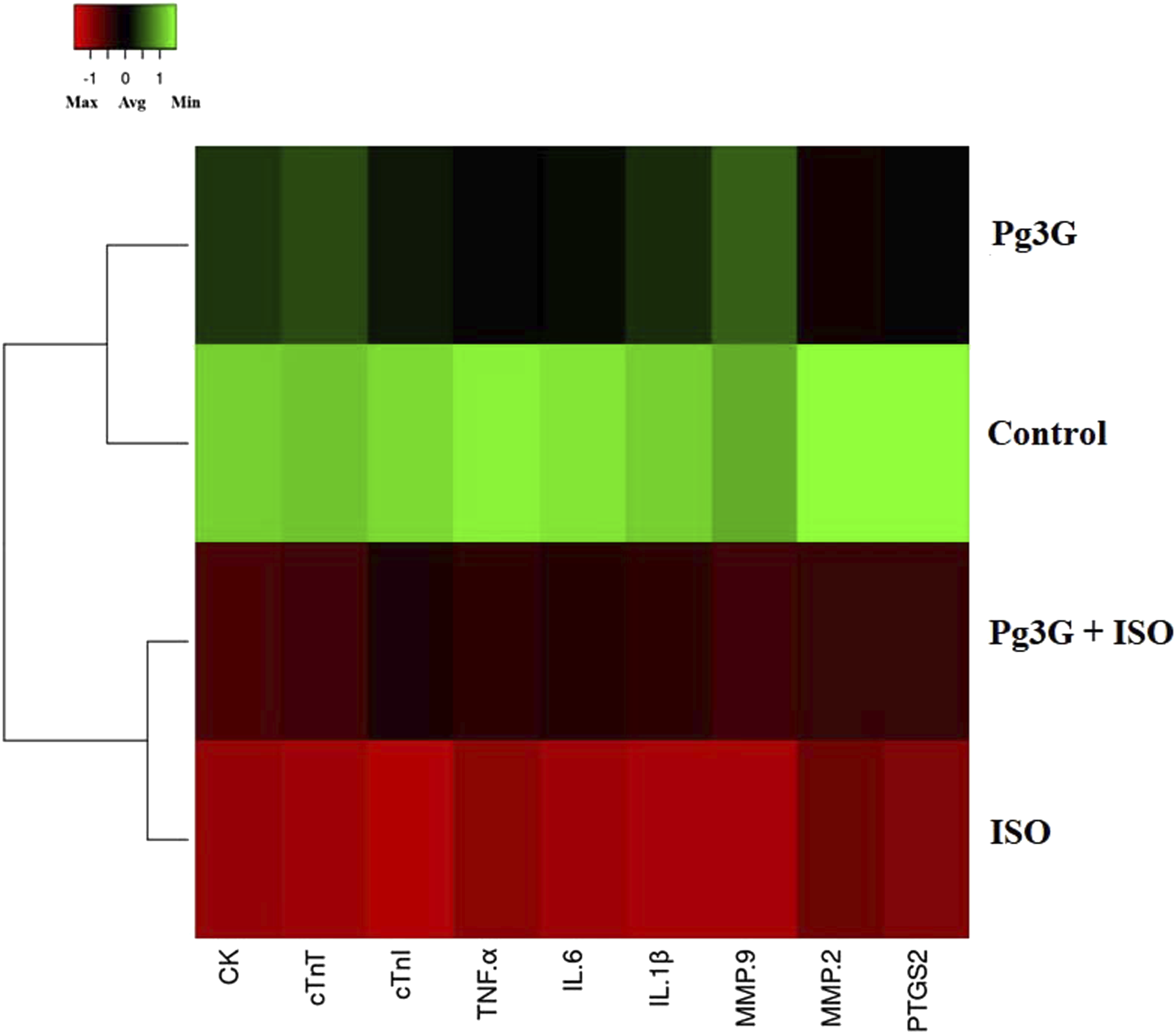
Discussion
The present study investigated the cardioprotective effects of Pg3G, an anthocyanin, on ISO-induced myocardial injury in a rat model. Our findings demonstrate that Pg3G effectively mitigates myocardial damage primarily through its antioxidant and anti-inflammatory properties. Myocardial injury induced by ISO is a widely accepted model that replicates the biochemical and physiological alterations observed in myocardial infarction. 26 ISO administration leads to oxidative stress and significant cardiac injury characterized by elevated levels of cardiac biomarkers such as CK, CK-MB, cTnT and cTnI. These markers serve as crucial indicators of myocardial damage, and their elevation post-ISO administration confirms the severity of cardiac injury.27,28 Our study’s observation of elevated CK, CK-MB, cTnT, and cTnI levels in ISO-treated rats corroborates findings from previous research that also highlighted the impact of ISO on myocardial integrity. Pg3G pre-treatment resulted in a marked prevention of these cardiac biomarkers expression, indicating its protective effect against ISO-induced myocardial injury. This result is particularly noteworthy, as anthocyanins are known for their potent free radical-scavenging abilities, which can help reduce oxidative stress. 29 The mechanisms underlying the cardioprotective effects of Pg3G can be attributed to its capacity to neutralize ROS and preserve cellular structures from oxidative damage. The relationship between oxidative stress and myocardial injury is well-documented, as excessive ROS production leads to lipid peroxidation, protein modification, and DNA damage. 30 Our study confirmed significant increases in lipid peroxidation markers, indicating oxidative damage in ISO-treated rats. Pre-treatment with Pg3G effectively restored antioxidant enzyme activities such as SOD, CAT and GPx, as well as increased level of GSH. These results are consistent with previous findings demonstrating the ability of anthocyanins to enhance endogenous antioxidant defenses, thus mitigating oxidative damage. 31
Furthermore, the role of inflammation in myocardial injury cannot be overlooked. ISO-induced myocardial damage is associated with an increase in pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1β. Our findings showed that ISO administration significantly elevated the levels of these cytokines, confirming the inflammatory component of ISO-induced myocardial injury. Inflammatory cytokines play a crucial role in exacerbating myocardial damage, promoting further cardiac remodeling and dysfunction. 32 Interestingly, Pg3G pre-treatment resulted in a significant reduction in the levels of TNF-α, IL-6, and IL-1β, suggesting that Pg3G exerts an anti-inflammatory effect. This observation is supported by studies indicating that anthocyanins can inhibit the activation of key inflammatory pathways.33,34 By inhibiting these pathways, Pg3G reduces the expression of pro-inflammatory cytokines, thus attenuating the inflammatory response and its detrimental effects on cardiac tissue.
While the current study provides strong evidence for the cardioprotective effects of Pg3G, it is important to consider the broader context of anthocyanin research. Numerous studies have demonstrated the cardiovascular and other health benefits of anthocyanin-rich foods 35 The consumption of these foods has been linked to reduced risk factors for cardiovascular diseases, including hypertension, hyperlipidemia, and inflammation. 36 The results of our study align with this growing body of evidence, suggesting that Pg3G may contribute to the protective effects observed with dietary anthocyanins. In addition to the antioxidant and anti-inflammatory properties, Pg3G may also influence other mechanisms related to cardiovascular health. For instance, recent research indicates that anthocyanins can improve endothelial function, enhance nitric oxide availability, and modulate lipid metabolism. 37 These mechanisms may further contribute to the overall cardioprotective effects of Pg3G, highlighting its multifaceted role in cardiovascular health.
The PCR array hierarchical clustergram analysis further confirms the effects of Pg3G on fibrotic gene expression in ISO-induced rat heart tissue. The modulation of key genes indicates Pg3G’s potential therapeutic role in mitigating cardiac fibrosis. The downregulation of CK suggests that Pg3G may protect myocardial integrity by reducing markers of cardiac injury. Additionally, decreases in cTnT and cTnI levels further imply preservation of cardiomyocyte viability. Pg3G also downregulated inflammatory cytokines, including TNF-α, IL-6, and IL-1β, indicating reduced inflammation, which is crucial for preventing chronic cardiac remodeling. Moreover, the modulation of MMP-2, -9 and PTGS2 by Pg3G highlights its role in restoring extracellular matrix balance and reducing pro-inflammatory prostaglandins. The ISO treatment induced overexpression of inflammatory cytokines and NF-kB p65 transcription factor in the experimental animals which resulted in heart failure. 38 The present study revealed that ISO-treated rats showed significantly upregulated expression of NF-κB and subsequent overexpression of pro-inflammatory cytokines in the heart tissue. Interestingly, Pg3G treatment prevented ISO-induced inflammatory responses in the myocardial tissue. The anti-inflammatory role of Pg3G has also been proved in other inflammatory experimental models Similarly, Pg3G exhibits protective effects in cardiomyoblasts through modulating several inflammation and oxidative stress related genes. 39 Overall, these findings suggest that Pg3G offers multifaceted cardioprotection, warranting further exploration of its mechanisms and clinical applications in cardiac fibrosis.
Conclusion
In conclusion, this study demonstrates that Pg3G provides significant cardioprotective effects against ISO-induced myocardial injury in rats. By reducing oxidative stress, modulating inflammatory responses, and preserving cardiac structure, Pg3G shows promise as a potential therapeutic agent for cardiovascular diseases. The findings are consistent with existing literature on the cardioprotective effects of anthocyanins and underscore the importance of dietary interventions in promoting heart health. Further research, particularly in clinical trials, is essential to validate the therapeutic potential of Pg3G and its application in cardiovascular disease management.
Supplemental Material
Supplemental Material - Pelargonidin-3-O-glucoside prevents isoproterenol-induced myocardial infarction via modulating cardiac and inflammatory markers expression in experimental rats
Supplemental Material for Pelargonidin-3-O-glucoside prevents isoproterenol-induced myocardial infarction via modulating cardiac and inflammatory markers expression in experimental rats by Jianmei Chang, Xiaoling Guo, Peng Hou in Human & Experimental Toxicology.
Footnotes
Ethical considerations
All animal experiments performed in this study were in accordance with the ethical standards of the institutional animal ethical committee of the Third People’s Hospital of Gansu Province, Gansu, China (Approval number 20250281).
Consent to participate
This study does not need informed consent as it does not involve human experiments.
Author’s contribution
Author contributions Jianmei Chang and Peng Hou confirmed the authenticity of all the raw data and edited the manuscript, Jianmei Chang and Xiaoling Guo collected data and processed the data. Jianmei Chang and Xiaoling Guo conducted the statistics. Peng Hou reviewed and revised the article. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported byClinical study of portable remote three-lead synchronous ECG monitoring system in the diagnosis of arrhythmia in remote areas of Gansu Province from the perspective of precise poverty alleviation. Longyuan Youth Innovative and Entrepreneurial Talents Project (2018)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data of this study were available with the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
