Abstract
We investigated the effects of seabuckthorn (SBT) oil in isoproterenol (ISO)-induced cardiotoxicity with reference to hemodynamic, antioxidant, histopathological, and ultrastructural parameters. Rats were administered SBT oil (5, 10, and 20 mL/kg per d) or vehicle orally for 30 days along with ISO (85 mg/kg, subcutaneously, at 24-hour interval) on 29th and 30th day. On 31st day, ISO control rats showed cardiac dysfunction, increased lipid peroxidation, depletion of cardiac injury marker enzymes, and antioxidant activities. Myocardial necrosis, edema, and inflammation were evident from the light microscopic and ultrastructural changes. Seabuckthorn oil at the dose of 20 mL/kg per d significantly modulates hemodynamic and antioxidant derangements. The preventive role of SBT oil on ISO-induced cardiotoxicity was reconfirmed by histopathological and ultrastructural examinations. Thus, the present study reveals that SBT oil mitigates myocardial damage in ISO-induced cardiac injury in rats by maintaining hemodynamic, biochemical, histopathological, and ultrastructural perturbations owing to its free radical scavenging and antioxidant activities.
Isoproterenol (ISO), a synthetic catecholamine and nonselective β-adrenoceptor agonist, at higher dose, has been reported to produce weakening of endogenous antioxidant system, contractile dysfunction, cardiotoxicity, and cardiac damage (infarct-like lesions) due to positive chronotropic and inotropic effects. 1 Various mechanisms have been proposed to explain the mechanism of ISO-induced myocardial injury including imbalance between oxygen supply and demand, hypotension due to reduced coronary perfusion pressure, and excessive generation of free radicals. 2 It has been reported that catecholamines undergo autoxidation that leads to the production of free radicals. 3 Experimental studies have shown that increased generation of free radicals or decreased level of antioxidants in the myocardium is involved in the pathogenesis of cardiovascular diseases such as ischemia–reperfusion injury, congestive heart failure, and cardiomyopathy. 4
In recent years, herbal drugs have received much attention as an antioxidant and preferred over synthetic drugs owing to their safety and efficacy in degenerative disease like myocardial infarction (MI). 5 Therefore, attempts are now being focused on to look for efficacy of antioxidant-rich herbs that can prevent ISO-induced myocardial damage and maintain redox homeostasis of the cell.
Seabuckthorn (SBT) oil (Hippophae rhamnoides L.), belonging to family Elagenaceae, is widely used in traditional system of medicine owing to its multifarious biological and medicinal properties. 6 All parts of plant are considered to be a rich source of vitamins, minerals, flavonoids, and carotenoids. 7 The potential biological and medicinal properties of SBT are attributable to the high content of antioxidant substances, mainly flavonoids and vitamins. 7,8 The alcoholic leaf extract of SBT has been documented to exert marked antioxidant and immunomodulatory activity against chromium-induced oxidative stress in rats. 9 Very few reports are available on the effect of SBT oil in experimental models of MI. Therefore, putting together the role of oxidative stress in MI and the afforded cardioprotection by antioxidants, the present study was aimed to investigate the effect of SBT oil on hemodynamic, biochemical, histopathological, and ultrastructural changes in ISO-induced cardiotoxicity in rats to delineate the underlying mechanism of its cardioprotective effect.
Materials and Methods
Animals
Male Wistar albino rats (170-200 g) were obtained from the Central Animal House Facility of All India Institute of Medical Sciences, New Delhi, India. The study protocol was reviewed and approved by the Institutional Animal Ethics Committee (IAEC No. 466/08) and all study-related activities conformed to the Indian National Science Academy Guidelines for the use and care of experimental animals in research. Animals were maintained in polypropylene cages, each containing a maximum of 4 animals. Animals were housed in the departmental animal house under controlled conditions of temperature at 25°C ± 2°C, relative humidity of 60% ± 5% and light–dark cycle of 12:12 hours. Standard rat chow and tap water were available ad libitum.
Drugs and Chemicals
Isoproterenol hemisulfate was dissolved in normal saline and used within 20 minutes of preparation. Creatine kinase-MB (CK-MB) isoenzyme detection kit was purchased from Spinreact (Spain). All the other chemicals used were of analytical grade and purchased from Sigma Chemicals (St Louis, Missouri). Seabuckthorn oil was obtained from Mantra Ayurveda (Ghaziabad, Uttar Pradesh, India). It contained vitamin E (171 mg/100 gm), vitamin K (54-59 mg/100 gm), carotenoids (300-870 mg/100 gm), total acids (38 mg/100 gm), total flavonoids (100-1000 mg/100 gm), total sterols (721 mg/100 gm), unsaturated fatty acids (67%), and saturated fatty acids (33%).
Experimental Groups
The rats were randomly assigned into 6 groups, each containing 10 rats. Group 1 (vehicle treated): Rats were administered normal saline orally (3 mL/kg per d) using intragastric tube for 30 days and on the 29th and 30th days, 0.3 mL saline was injected subcutaneously (sc) at an interval of 24 hours. Group 2 (SBT oil per se): Animals were treated with SBT oil (20 mL/kg per d) orally for a period of 30 days and on the 29th and 30th days, 0.3 mL saline was injected sc at an interval of 24 hours. Group 3 (vehicle + ISO): Rats were administered normal saline orally (3 mL/kg per d) for 30 days along with concurrent administration of ISO (85 mg/kg, sc at 24-hour interval) on the 29th and 30th days. Groups 4 to 6 (SBT oil + ISO): Animals were treated with SBT oil (5, 10, and 20 mL/kg per d) orally for a period of 30 days along with concurrent administration of ISO (85 mg/kg, sc at 24-hour interval) on the 29th and 30th days.
Surgery for Recording Hemodynamic Parameters
The detailed surgical procedure for recording of hemodynamic parameters has been described in our previous study.
10 Briefly, rats were anesthetized with sodium pentobarbitone (60 mg/kg, intraperitoneally [ip]). Atropine (4 mg/kg, ip) was administered along with the anesthetic to reduce tracheobronchial secretions. Throughout the experimental protocol, body temperature of the animals was maintained at 37°C. The neck was opened with a ventral midline incision to perform tracheostomy, and the animals were ventilated with room air from a positive pressure ventilator (Inco Pvt Ltd, Ambala, India) using compressed air at a rate of 90 strokes/min and a tidal volume of 10 mL/kg. Ventilator setting and P
Biochemical Parameters
A 10% homogenate of myocardial tissue was prepared in ice-chilled phosphate buffer (50 mmol/L, pH 7.4), and an aliquot was used to estimate thiobarbituric acid reactive substance (TBARS) level 11 and reduced form of glutathione (GSH) content. 12 Remaining homogenate was centrifuged at 5000 rpm for 20 minutes at 4°C, and the obtained supernatant was used for estimation of activities of enzymes such as CK-MB isoenzyme, lactate dehydrogenase (LDH), 13 catalase, 14 and superoxide dismutase (SOD), 15 and protein. 16 Creatine kinase-MB isoenzyme was estimated spectrophotometrically using a kit from Spinreact.
Histopathological Examinations
After formalin fixation, the heart tissues were embedded in liquid paraffin and cross sections (4 µm thick) were cut using microtome (Leica RM 2125, Germany). Sections were stained with hematoxylin and eosin stain (H&E). Degree of myocardial necrosis was determined using light microscope and photographs were taken. The pathologist performing histopathological evaluation was masked to the treatment allocation in different study groups. The degree of necrosis was graded and scored as follows: Score (−): absence of any inflammation, edema, and necrosis. Score (+): focal areas of inflammation, edema, and necrosis. Score (++): patchy areas of inflammation, edema, and necrosis. Score (+++): confluent areas of inflammation, edema, and necrosis. Score (++++): massive areas of inflammation, edema, and necrosis.
Ultrastructural Analysis by Transmission Electron Microscopy
After fixation with Karnovsky reagent, the heart tissues were washed with ice-chilled phosphate buffer (pH 7.4) and postfixed for 2 hours in 1% osmium tetraoxide in the phosphate buffer at 4°C. The tissues were further washed in phosphate buffer, dehydrated with graded acetone, and then embedded in araldite CY212 to make tissue blocks. Semithin (1 µm) as well as ultrathin sections (70-80 nm) was cut by ultramicrotome (Ultracut E, Reichert, Austria). The tissue sections were stained with uranyl acetate and lead acetate and examined under transmission electron microscopy ([TEM] Morgagni 268D; Fei Co., the Netherlands) operated at 60 kV by an anatomist masked to the treatment allocations in different experimental groups. The degree of injury was graded and scored as follows: severe (+++), moderate (++), mild (+), and nil (–).
Statistical Analysis
Data were expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by Scheffe post hoc test was used for analysis of hemodynamic and biochemical data of different groups. A value of P < .05 was considered as significant.
Results
Per Se Effect of Drug
Seabuckthorn oil per se treatment (20 mL/kg) daily for a period of 30 days to normal control rats did not show any significant change in hemodynamic and biochemical parameters as compared to vehicle-treated group.
Effect of SBT Oil on Hemodynamic Parameters
Figure 1 shows the effect of SBT oil on arterial pressures. A significant decrease in systolic, diastolic, and mean arterial pressures were observed in ISO control animals as compared to normal control group (P < .0001). Pretreatment with SBT oil (5-20 mL/kg per d) preserved systolic, diastolic, and mean arterial pressures in a dose-dependent manner after ISO challenge. However, a significant restoration was observed at 10 and 20 mL/kg doses (P < .001). We did not observe any significant change in heart rate for all experimental groups (data not shown).
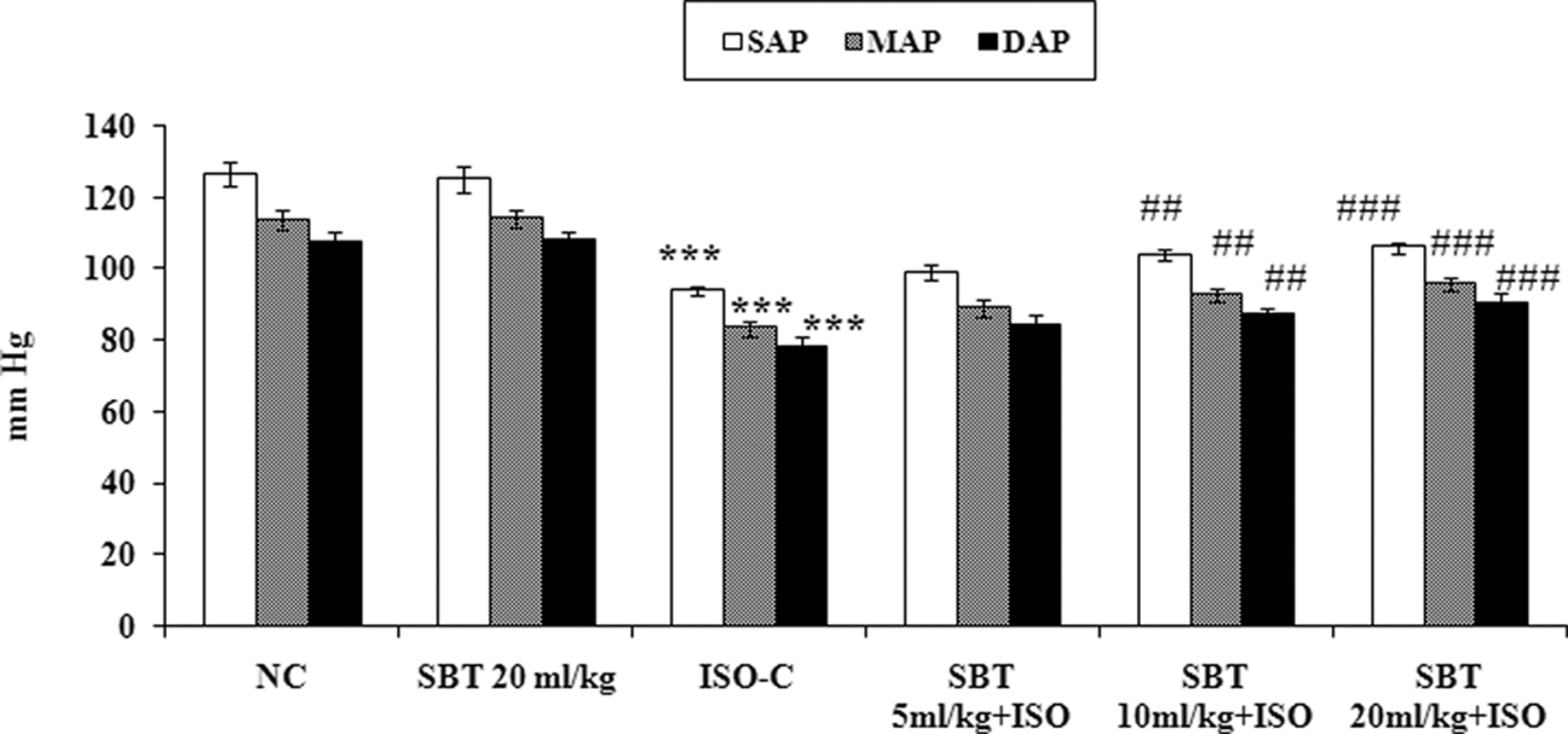
Effect of SBT oil on arterial pressure in different experimental groups. NC indicates normal control; SAP, systolic arterial pressure; MAP, mean arterial pressure; DAP, diastolic arterial pressure; ISO, isoproterenol; SBT, seabuckthorn. All values are expressed as mean ± standard deviation (SD). Significance was determined by 1-way analysis of variance (ANOVA) followed by Scheffe post hoc test. ## P < .01, ### P < .001 as compared to ISO group; ***P < .0001 as compared to normal control group.
Figures 2 and 3 represent the effect of SBT oil on left ventricular functions in different experimental groups. Compared with normal control group, ISO control rats showed left ventricular dysfunction as indicated by a significant increase in left ventricular end-diastolic pressure (LVEDP) and fall in the values of ±LVdp/dtmax (P < .0001). Pretreatment with SBT oil at 20 mL/kg per d significantly (P < .001) improved left ventricular functions following ISO administration when compared with ISO control animals.
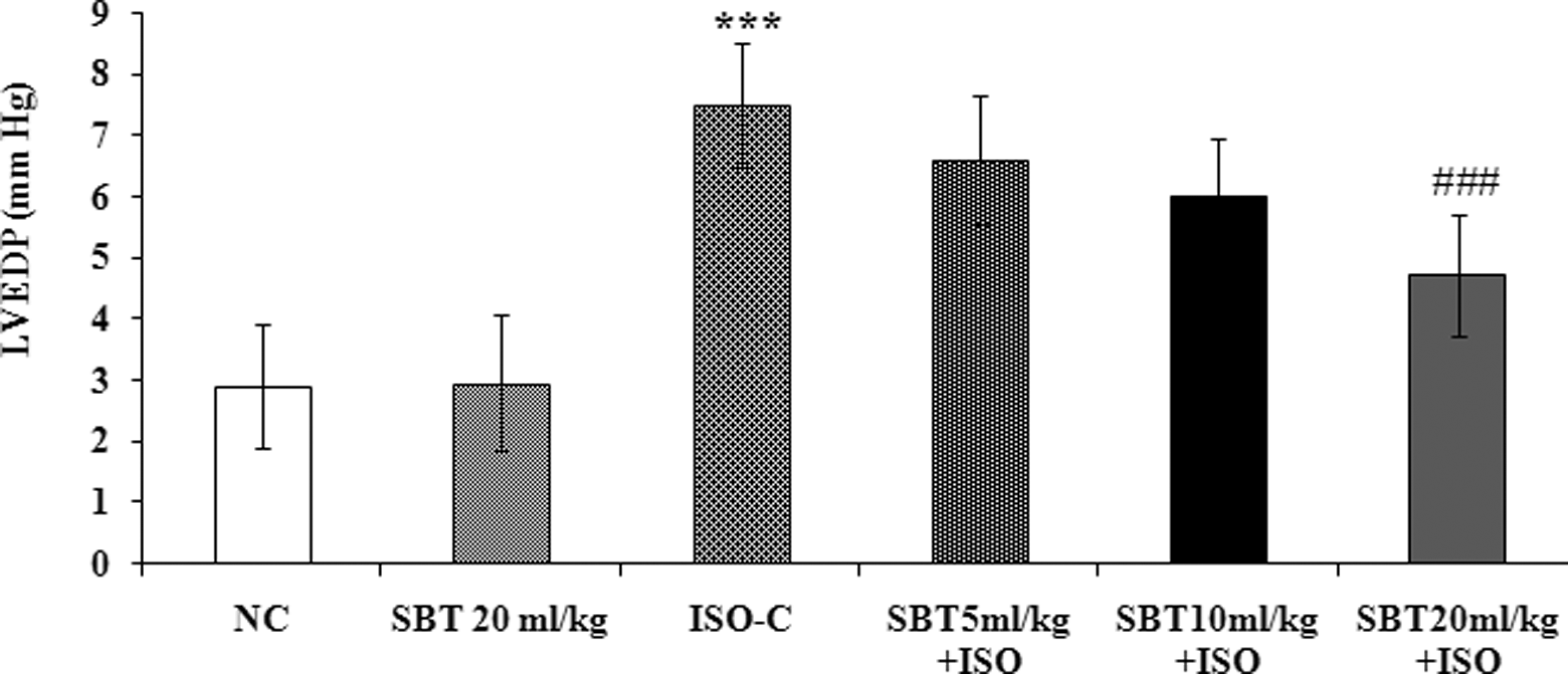
Effect of SBT oil on left ventricular end-diastolic pressure in different experimental groups. NC indicates normal control; LVEDP, left ventricular end-diastolic pressure; ISO, isoproterenol; SBT, seabuckthorn. All values are expressed as mean ± standard deviation (SD). Significance was determined by 1-way analysis of variance (ANOVA) followed by Scheffe post hoc test. ### P < .001 as compared to ISO group; ***P < .0001 as compared to normal control group.
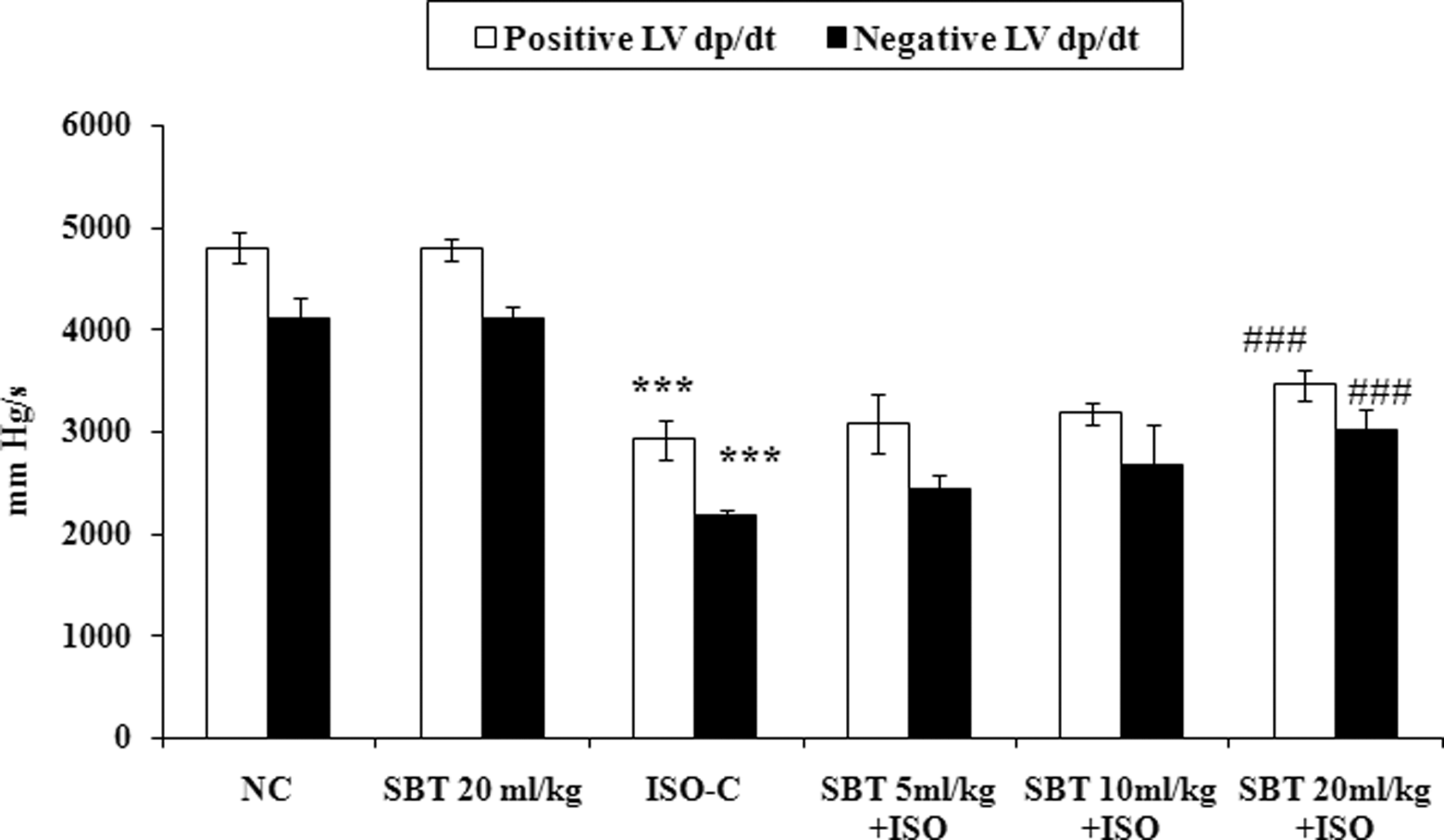
Effect of SBT oil on maximum positive and negative rate of change of left ventricular pressure in different experimental groups. NC indicates normal control; +LVdP/dtmax, left ventricular maximum rate of positive pressure development; −LVdP/dtmax, left ventricular maximum rate of negative pressure development; SBT, seabuckthorn. All values are expressed as mean ± standard deviation (SD). Significance was determined by 1-way analysis of variance (ANOVA) followed by Scheffe post hoc test. ### P < .001 as compared to ISO group; ***P < .0001 as compared to normal control group.
Effect of SBT Oil on TBARS Content and Activities of Myocardial Injury Markers
Table 1 depicts the activity of myocardial injury markers (CK-MB and LDH) and TBARS level in normal and treatment groups. The level of TBARS was significantly increased in ISO control animals (P < .0001), whereas the activities of CK-MB and LDH were significantly (P < .0001) decreased in heart tissue as compared to normal control group. Oral pretreatment with SBT oil (10 and 20 mL/kg per d) for a period of 30 days significantly decreased the level of TBARS (P < .001), while SBT oil 20 mL/kg per d significantly (P < .001) prevented the increase in activities of CK-MB and LDH enzymes when compared to ISO control animals. However, SBT oil (5 and 10 mL/kg per d) failed to significantly preserve the activities of CK-MB and LDH enzymes when compared to ISO control animals.
Effect of SBT Oil on TBARS and Myocardial Injury Markers in Different Experimental Groups a
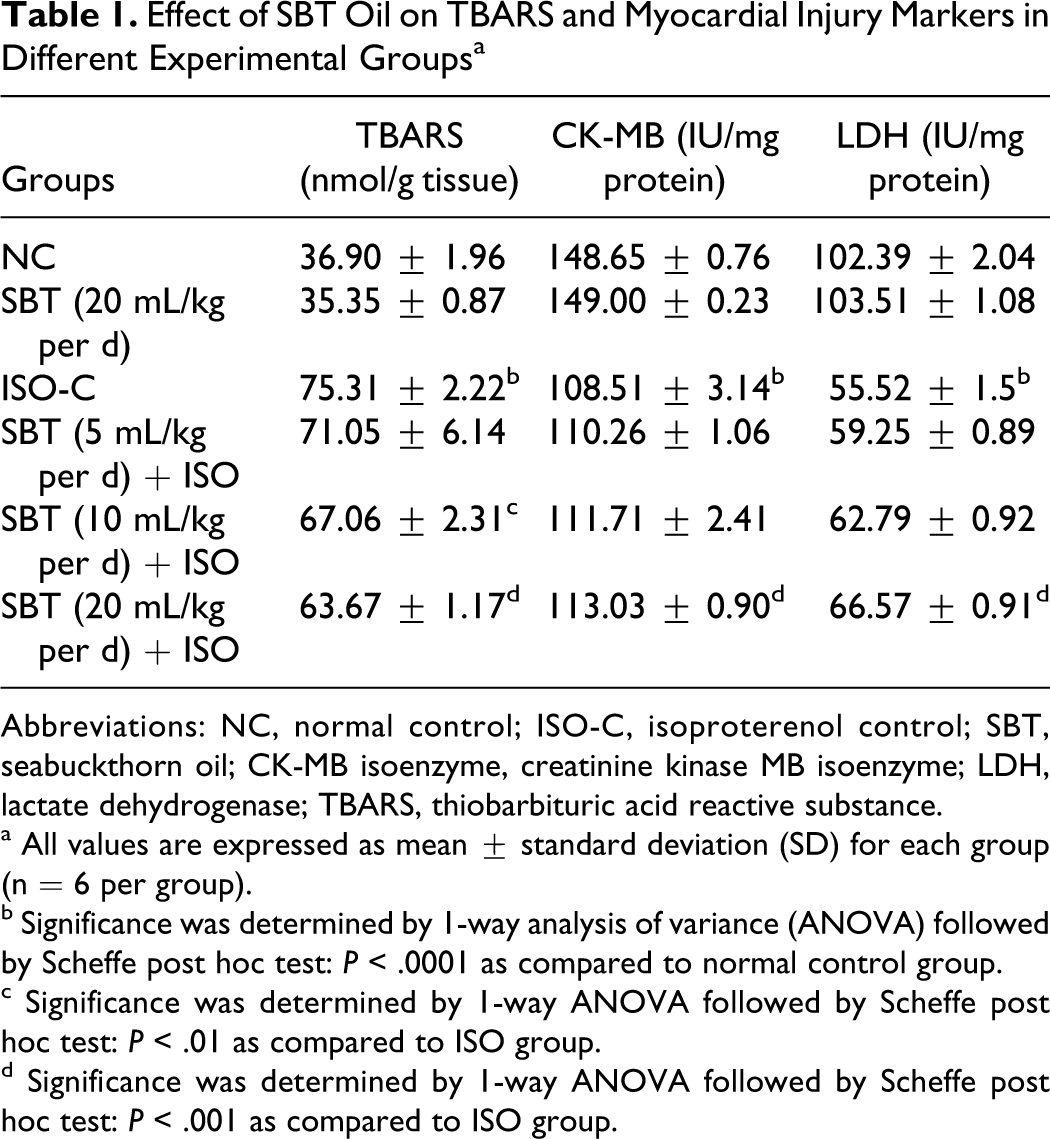
Abbreviations: NC, normal control; ISO-C, isoproterenol control; SBT, seabuckthorn oil; CK-MB isoenzyme, creatinine kinase MB isoenzyme; LDH, lactate dehydrogenase; TBARS, thiobarbituric acid reactive substance.
a All values are expressed as mean ± standard deviation (SD) for each group (n = 6 per group).
b Significance was determined by 1-way analysis of variance (ANOVA) followed by Scheffe post hoc test: P < .0001 as compared to normal control group.
c Significance was determined by 1-way ANOVA followed by Scheffe post hoc test: P < .01 as compared to ISO group.
d Significance was determined by 1-way ANOVA followed by Scheffe post hoc test: P < .001 as compared to ISO group.
Effect of SBT Oil on Activities of Antioxidant Enzymes
Table 2 represents the effect of SBT oil on the activities of SOD, catalase, and GSH content in the heart. Administration of ISO showed a significant (P < .0001) decrease in the levels of these antioxidants in the heart compared with the normal control rats. Pretreatment with SBT oil (20 mL/kg per d) enhanced the content of these antioxidants significantly (P < .001) compared with ISO-alone-treated rats.
Effect of SBT Oil on Antioxidants Status in Different Experimental Groups a
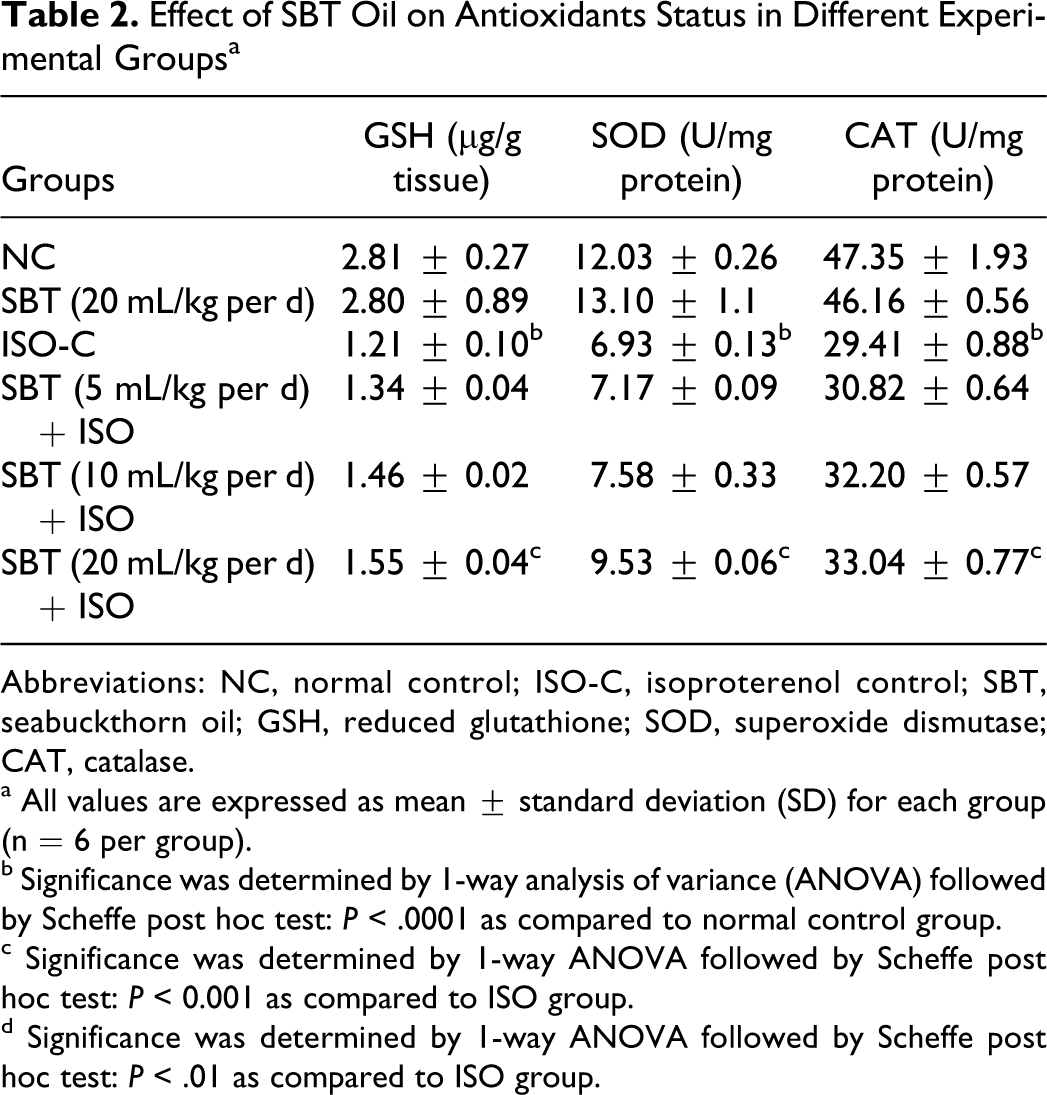
Abbreviations: NC, normal control; ISO-C, isoproterenol control; SBT, seabuckthorn oil; GSH, reduced glutathione; SOD, superoxide dismutase; CAT, catalase.
a All values are expressed as mean ± standard deviation (SD) for each group (n = 6 per group).
b Significance was determined by 1-way analysis of variance (ANOVA) followed by Scheffe post hoc test: P < .0001 as compared to normal control group.
c Significance was determined by 1-way ANOVA followed by Scheffe post hoc test: P < 0.001 as compared to ISO group.
d Significance was determined by 1-way ANOVA followed by Scheffe post hoc test: P < .01 as compared to ISO group.
Effect of SBT Oil on Histopathological and Ultrastructural Alterations of Rat Myocardium
The histopathological changes were graded and are summarized in Table 3 . Light micrographs of myocytes of normal rat showed intact myocardial architecture, with no evidence of any infarct or lesions (Figure 4A). Normal rats treated with SBT oil (20 mL/kg) showed normal cardiac muscle bundles without any damage (Figure 4B). However, ISO-administered rats showed cardiac muscle separation, myonecrosis, edema, mononuclear infiltrate, and inflammation (Figure 4C). Seabuckthorn oil failed to protect the myocardium significantly at the dose of 5 mL/kg per d as evidenced by damage similar to ISO control group (Figure 4D). Nevertheless, SBT oil (10 mL/kg per d) showed patchy areas of necrosis and mild edema without inflammatory cells (Figure 4E). Rat hearts treated with SBT oil at 20 mL/kg per d showed mild edema with significant reduction in necrosis, showing near-normal myocardial architecture (Figure 4F).
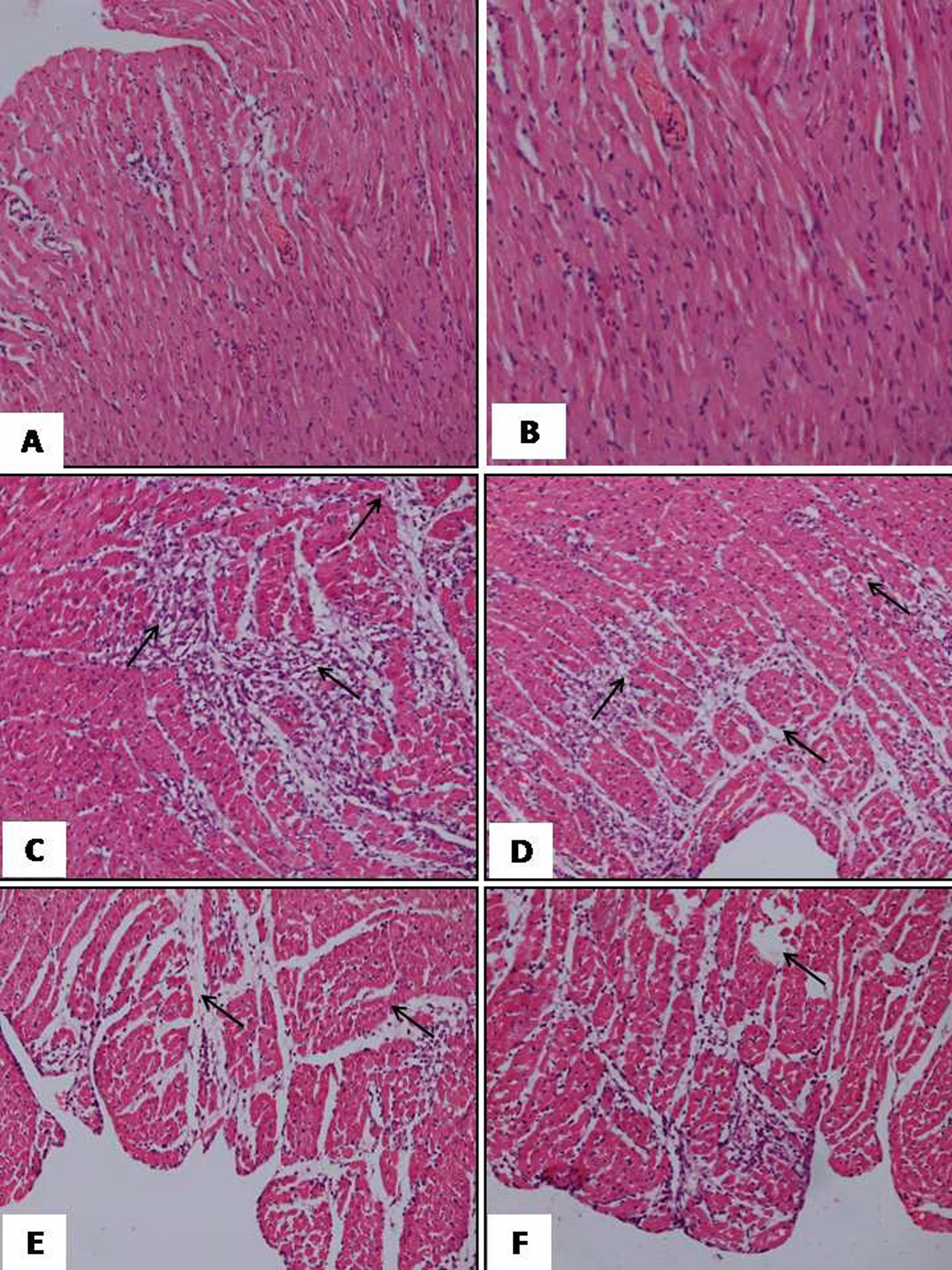
A, Light micrographs of normal control rat heart showing normal architecture of myocytes (H&E, ×200). B, Light micrographs of rat heart treated with SBT oil (20 mL/kg per d) alone, showing normal cardiac fibers without any fraying or infarction (H&E, ×200). C, Light micrographs of ISO control group heart, showing (→) myonecrosis, inflammatory cell infiltration, and edema with fibroblastic proliferation (H&E, ×200). D, Light micrograph of 5 mL/kg per d plus ISO, showing (→) marked myocardial necrosis, edema SBT oil with inflammatory cells infiltration (H&E, ×200). E, Light micrograph of SBT oil 10 mL/kg per d plus ISO, showing (→) less myocardial injury, less edema, and inflammatory cells (H&E, ×200). F, Light micrograph of SBT oil 20 mL/kg per d plus ISO-treated heart tissue showing (→) mild edema but no infarction. The myocardial fibers are normal in architecture (H&E ×200). SBT indicates seabuckthorn; H&E, hematoxylin and eosin.
Histopathological Assessment of Myocardium in Different Experimental Groups
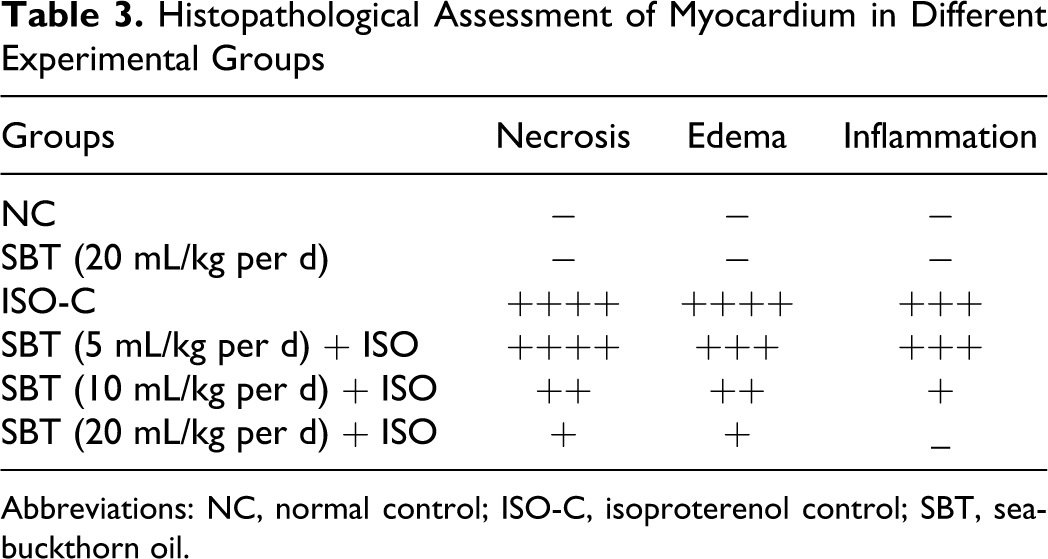
Abbreviations: NC, normal control; ISO-C, isoproterenol control; SBT, seabuckthorn oil.
Figure 5 and Table 4 depict the degree of ultramicroscopic changes in myocardial tissues of normal, ISO control, and SBT oil-treated rats. The TEM images of the heart showed normal architecture in vehicle-treated rats (Figure 5A). In normal rats treated with SBT oil (20 mL/kg), the heart myocyte showed normal architecture without any pathological changes (Figure 5B). However, distinct myonecrosis, swelling of mitochondria, and disruption of cristae with vacuolation was observed in ISO control rats (Figure 5C). Similar ultrastructural perturbations were observed with SBT oil at 5 and 10 mL/kg per d plus ISO groups, although the extent of changes were milder with 10 ml/kg per d dose of SBT oil (Figure 5D and E). Seabuckthorn oil at 20 mL/kg per d in ISO-treated group showed very mild separation of cristae with no swelling and vacuolation, an observation quite similar to normal ultramicroscopic structure (Figure 5F).
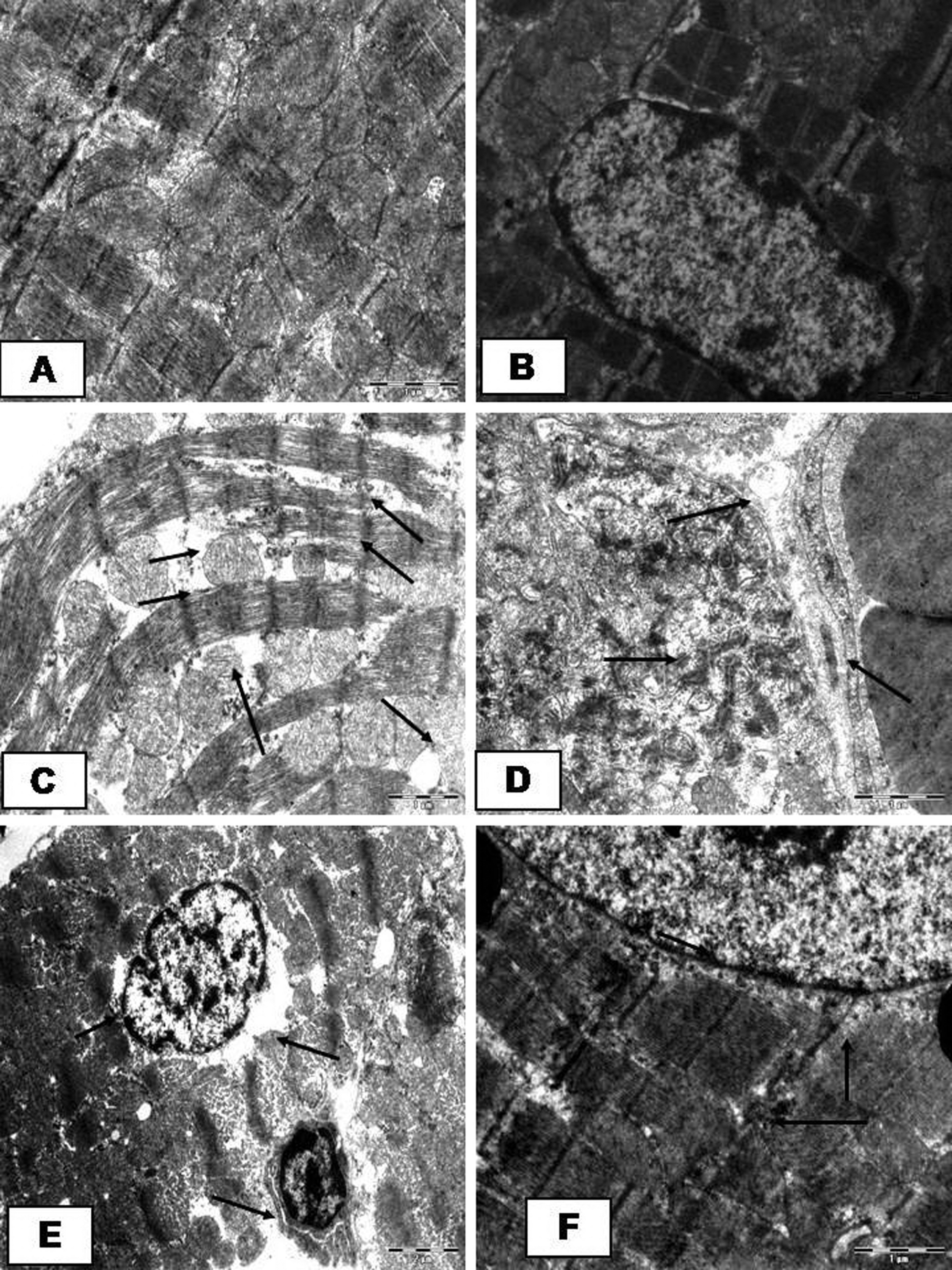
A, Electron micrograph (magnification ×4700) of normal myocardial tissue, showing normal architecture of myofibrils and mitochondria. B, Electron micrographs (magnification ×4800) of rat groups treated with SBT oil (20 mL/kg per d) alone, showing normal myocardial tissue, myofibrils, and mitochondria. C, Electron micrograph of myocardium of ISO control group (magnification ×4700) showing (→) lipid droplets, myocardial necrosis with marked disruption of Z bands, and myofilaments along with changes in mitochondria and vacuolization of myofibrils and mitochondrial damage. D, Electron micrograph of rat hearts treated with SBT oil 5 mL/kg per d plus ISO group, showing (→) severe myonecrosis and swollen nucleus and vacuoles. In mitochondria, loss of double membrane, disarrangement of cristae, and accumulation of electron dense material is seen (×4500). E, Electron micrograph of rat hearts treated with SBT oil 10 mL/kg per d plus ISO group, showing (→) swelling of nucleus, less myocardial necrosis, and edema along with vacuolization and mitochondrial changes (×4500). F, Electron micrograph of rat hearts treated with SBT oil 20 mL/kg per d plus ISO, showing (→) irregular disruption of myofilaments along with focal loss of mitochondrial double membrane and disarrangement of cristae. Almost normal structure is seen in most areas (×4500).SBT indicates seabuckthorn; ISO, isoproterenol.
Ultrastructural Evaluation of Myocardium in Different Experimental Groups

Abbreviations: NC, normal control; ISO-C, isoproterenol control; SBT, seabuckthorn oil; +++, severe; ++, moderate; +, mild; –, nil.
Discussion
The present study demonstrated the cardioprotective potential of SBT oil in ISO-induced myocardial necrosis in rats. The results of the study showed that SBT oil at the dose of 20 mL/kg per d prevented myocardial damage induced by sc injection of ISO. Isoproterenol-induced myocardial necrosis is a well-authenticated model to study the beneficial effects of many drugs on cardiac function. 17 Subcutaneous injection of ISO has been shown to cause myocardial necrosis, particularly in the subendocardial regions of left ventricle and interventricular septum, which closely resembles subendocardial necrosis produced by myocardial ischemia in humans. Through spontaneous enzymatic reaction ISO leads to the formation of products like adrenochrome which is implicated in the generation of highly toxic oxygen-derived free radicals. 18 Isoproterenol-induced oxidative stress causes severe lipid peroxidation in heart tissue which results in the myocardial injury. 19
On functional aspects, ISO administration caused systolic and diastolic dysfunction, increased left ventricular end-diastolic volume, and ST-segment elevation. 20 Pretreatment with SBT oil following ISO administration exhibited a dose-dependent increase in systolic, diastolic, and mean arterial pressures as compared to ISO control group, though the restoration of arterial pressure indices was significant with 10 and 20 mL/kg per d doses. However, no significant alterations were observed in heart rate, among all groups. Along with altered arterial pressure, ISO has been observed to alter the left ventricular dynamics. Treatment with SBT oil also improved left ventricular end-diastolic function by increasing the inotropic (+LVdp/dtmax, marker of myocardial contraction) and lusitropic (−LVdp/dtmax, marker of myocardial relaxation) states of the heart. It also attenuated ISO-induced increase in LVEDP (a marker of preload) which further reflects an improvement in left ventricular function. Taken together, SBT oil offers protection to the myocardium by attenuation of the ventricular dysfunction through maintaining the hemodynamic parameters to near normal in ISO-treated rats. Our results are consistent with previous studies wherein the total flavones of SBT oil improved dilated systolic function, cardiac function, and decreased total peripheral resistance. 21
Lipid peroxidation plays a critical role in the pathogenesis of MI. A significant increase in the level of lipid peroxidation product, TBARS, in the heart obviously indicates severe oxidative stress in ISO-induced MI in rats, which is in consonance to previous studies. 22 Alterations in the metabolism of lipid peroxides are closely associated with oxidative damage due to free radicals produced by ISO in myocardium. 23 Pretreatment with SBT oil decreased the lipid peroxidation product, TBARS, in ISO-treated rats. We speculate that protection offered by SBT oil might be a result of flavonoid, a constituent of SBT oil, which has been known to possess anti–lipid peroxidative action. 24
The diagnostic marker enzymes of MI such as CK-MB and LDH were decreased in ISO-treated rats. Decrease in the level of these enzymes is an indication of ISO-induced necrotic damage to the myocardial membrane and confirms to previous studies demonstrating the deleterious effects of ISO on myocardium. 25 We observed that pretreatment with SBT oil significantly prevented the depletion of these myocardial marker enzymes following ISO administration. It is presumed that SBT oil might have maintained the cell integrity and stabilized the myocardial membrane which restricts the leakage of these marker enzymes from the heart into blood.
Isoproterenol is known to induce oxidative stress by generating free radical moieties via its quinine metabolites which react with oxygen to produce superoxide anions and other reactive oxygen species in rat myocardium. Free radical scavenging antioxidants such as SOD, catalase, and GSH are the first line of cellular defense against oxidative injury. 26 Role of ISO has been well documented in the reduction of myocardial SOD and catalase activities and GSH content. 26 In our study, the observed decrease in the values of these antioxidants in heart following ISO administration confirms the excessive generation of reactive oxygen species, such as superoxide and hydrogen peroxide, which in turn leads to consumption of these endogenous antioxidants. We observed that treatment with SBT oil increased the activities of SOD, catalase, and GSH level in ISO-injected rats by preventing the depletion of antioxidants and quenching of free radicals. Seabuckthorn oil is rich in vitamins, carotenoides, flavanoids, and nutrients. It is well known that vitamin C, E, carotenoides, and flavanoids have antioxidant and cardioprotective effects in animal models of MI. 27 –29 Moreover, other parts of the plant such as leaf extract have also been reported to exhibit cytoprotective and antioxidant properties against hypoxia-induced oxidative stress. 30 Therefore, these potential constituents present in SBT oil could have conferred cardioprotection against free radical–mediated tissue injury in ISO-induced myocardial damage.
Histopathology of the myocardium of ISO-administered rats showed extensive myonecrosis, edema, and myofiber disorganization. 31 However, SBT oil pretreatment in ISO-challenged heart showed reduced myonecrosis and cardiac damage as well as myocardial salvage. Myocardium of the group of rats treated with SBT oil per se showed normal cardiac fibres without any pathological changes. This indicates that SBT oil does not possess any adverse effects under normal conditions. Similar to histopathology, electron microscopy study also revealed the myocardial damaging effects of ISO administration. Pretreatment with SBT oil depicted protection of myocardium against ISO on myocardial subcellular and ultrastructural components. Thus, the histopathological and ultrastructural findings of the present study strongly support and confirm the hemodynamic and biochemical observations of this study.
On the basis of present study findings, it can be concluded that SBT oil protects the myocardium against ISO-induced cardiac damage by maintenance of hemodynamic functions and lipid peroxide metabolism. Furthermore, it also scavenges free radicals and improves antioxidant system by its antioxidant effect. The results also show that SBT oil is safe and an effective antioxidant in animals. Thus, a diet containing SBT oil may be beneficial in those who are at risk of myocardial injury.
Footnotes
The authors Salma Malik and Sameer Goyal contributed equally to this work.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported by the grant from the National Agricultural Innovation Project (NAIP) of Indian Council of Agricultural Research, New Delhi, India.
