Abstract
Introduction
The incidence of cerebral ischemia-reperfusion injury (I/R) is complex which seriously threatens the life safety of patients. Neither its prevention nor its treatment has been successful so far. Proteins that bind to DNA and belong to the C2/H2 zinc finger family are known as Krüppel-like factors (KLFs). Among them, KLF6 plays a vital role in proliferation, metabolism, inflammation, and damage responses, although its function in I/R remains largely unexplored.
Methods
In this study, we induced cerebral ischemia in rats using the middle cerebral artery occlusion (MCAO) model. Neural function, cerebral infarction volume, cognitive function, cortical pathological lesions, ferroptosis, and oxidative stress were measured.
Results
Our findings indicated that the MCAO model exhibited signs of ferroptosis and a concurrent increase in KLF6 levels. Inhibition of KLF6 resulted in a significant decrease in the escape latency during swimming tests (p < .05), an increase in the frequency of platform crossings, and prolonged duration in the target quadrant compared to the control group. Additionally, silencing KLF6 mitigated MCAO-induced brain injury and reduced oxidative stress and ferroptosis, as evidenced by altered levels of Nrf2/HO-1 signaling proteins.
Discussion
In conclusion, our results suggest that silencing KLF6 may protect against MCAO-induced pyroptosis, oxidative stress, and neurological dysfunction by inactivating the Nrf2/HO-1 signaling pathway. This study offers new perspectives on the molecular mechanisms related to MCAO and emphasizes the significance of targeting KLF6 for future therapeutic approaches.
Introduction
Ischemic stroke (ICH) is a potentially fatal condition in which brain tissue dies due to blocked or narrowed blood vessels that carry blood to the brain. 1 Perioperative brain injury is a potential risk. When the blood supply to the brain is interrupted, irreversible damage of neurons in key areas of the brain, including the hippocampus, cerebral cortex, and striatum, will occur, resulting in post-stroke depression.2,3 After injury, disability, memory deficit, and other cognitive dysfunction, and during blood reperfusion, will produce ischemia-reperfusion (I/R) injury. 4 This occurs due to a combination of oxidative stress, inflammation, and the activation of cell death pathways, compounding the initial injury. 5 The resulting damage can lead to long-term disabilities, including mobility issues, memory deficits, and a decline in overall quality of life. 6 The current treatments aim to inhibit several paths of nerve injury, which is a common occurrence in I/R. 7 Researching the causes of IR and identifying potential new treatment targets is crucial since there are now no viable medications for treating this condition.
KLF6 is a member of the Kruppel-like factor (KLF) family and can be involved in regulating various cellular functions such as cell metabolism, proliferation, and differentiation. 8 The role of KLF6 in ischemia-reperfusion damage in liver transplant patients has been elucidated using bioinformatics omics research. 9 KLF6 exacerbates inflammation and oxidative stress because it controls macrophage HIF-1 expression. 10 However, further research into the molecular processes by which KLF6 aids neuron regeneration after cerebral ischemia is required. Thus, we set out to conduct this research to learn how KLF6 influences cerebral I/R damage in an in vivo ischemic stroke model.
Materials and methods
Animals model
A total of 24 SPF-grade clean and healthy SD male rats aged 6 to 8 weeks and weighing between 150 and 200 g were randomly divided into four groups, including sham, MCAO, MCAO + sh-NC, and MCAO + sh-KLF6 groups. Each group consisted of 6. 5 μL lentiviral sh-NC or sh-KLF6 (1 × 109 TU/mL) were injected into the left ventricle of rats 7 days before MCAO, and brain tissue was taken 24 h later for testing.
MCAO model was constructed using the thread embolization method, as described previously. 11 The rats in the sham operation group underwent the same operation and inserted the thread plug without ligature.
Morris water maze
The positioning navigation time and space exploration track of rats in each group were recorded, respectively. (1) Measurement of positioning navigation time: Rats in each group were taken, put into water from four different starting points (different image limits), forced to swim, and the rise time (incubation period) of the positioning navigation of rats in each group was recorded respectively. The average latency of the four tests was taken as the learning performance of the rats on that day, for a total of 5 days (2) Determination of space exploration trajectory: 24 h after the end of the positioning navigation experiment, the platform was removed, and the rats were put into the water at any one of the same entry points. The swimming path of the rats within 60 s was recorded, as well as the residence time of the rats in the target quadrant and the number of times they crossed the target quadrant. 12
Cerebral infarction volume percentage measurement
The rats in each group were euthanized 24 h after reperfusion, the brain tissues were taken and frozen, continuous coronal sections with a thickness of about 2 mm were prepared, and triphenyl tetrazolium chloride (TTC) staining was performed, and the infarct volume fraction of the brain tissues was determined by Image 6.0 software. 13
Tunel detected neuronal apoptosis
The brain tissue was permeated with 0.5% Triton X-100 for 30 min, stained with TUNEL solution for 1 h, and the cell nucleus was re-stained with DAPI. Neuron apoptosis was observed and calculated by fluorescence microscopy. 14
Nissl staining
After dewaxing and hydration treatment, the paraffin sections of the brain tissue of rats in each group were stained by 0.1% cresyl violet (Nissl stain) and sealed with neutral gum. The sections were observed and mapped with a microscope.
Evaluation of oxidative stress and iron concentration
To study ROS generation, we used 2′,7′-Dichlorofluorescein diacetate (Sigma) and evaluated the fluorescence using an Olympus microscope. An iron assay kit was used to measure the concentration of iron. Results for SOD, MDA, and GSH were obtained using assay kits from Solarbio, which allowed for the relative determination of these three compounds. 15
Immunohistochemistry
After antigen retrieval, the brain tissue sections were blocked in 3% BSA, incubated with a primary antibody against KLF6, Nrf2, and HO-1, respectively, and the addition of fluorescently labeled IgG secondary antibodies. Next, the sections were stained with DAB and hematoxylin, sealed with neutral gum, and observed under a microscope. 16
Real-time quantified PCR (qRT-PCR)
Trizol isolated total RNA and reversely transcribed using the PrimeScript™ RT Master Mix. SYBR Green Master Mix kit was used for qRT-PCR to detect the expression of KLF6, GPX4, ACSL4, FTH1, and TFR1. The sequences of the primers used are as follows: KLF6 forward, 5′-TTTGACAACCAGGAAGACC-3′ and reverse, 5′-GACATCAGAGTTCAGGCTATT-3′; GPX4 forward, 5′ ATTCGCAGCCAAGGACAT-3′ and reverse, 5′-GGCCAGGATTCGTAAACCAC-3′; ACSL4 forward, 5′- ATCGCTCTGTCACGCACTT-3′ and reverse, 5′- CCAGGCTGTCCTTCTTCC-3'; FTH1 forward, 5′-TGCCAAATACTTTCTCCA-3′ and reverse, 5′- CACGGTCAGGTTTCTTTAT-3′; TFR1 forward, 5′- CTGCTTTGCGACTATTGC-3′ and reverse, 5′-CTGACTTGTCCGCCTCTT-3'; and GAPDH forward, 5′-GCAAGTTCAACGGCACAG-3′ and reverse, 5′- GCCAGTAGACTCCACGACAT-3′
Western blot assay
After homogenizing the cerebral tissue in the ischemic penumbra of rats in each group, the total protein was extracted and subjected to electrophoresis, membrane transfer and closure, and the KLF6 (1: 500, Proteintech, No.14,716-1-AP), SOD1 (1:000, Proteintech, No.10,269-1-AP), NOX2 (1:000, Proteintech, No.19,013-1-AP), GPX4 (1:000, Proteintech, No.67,763-1-Ig), ACSL4 (1:000, Proteintech, No.22,401-1-AP), FTH1 (1:000, Proteintech, No.11,682-1-AP), TFR1 (1:000, Proteintech, No.65,236-1-Ig) and β-actin (1:20,000, Proteintech, No.60,008-1-Ig) primary antibody dilution were added, respectively, incubated at 4°C overnight. The second antibody was added after membrane washing and incubated at room temperature for 1 h. Using β-actin as the internal reference, Image J software analyzed the relative expression of each protein.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 9.0. Data were presented as mean ± standard error of the mean. The Student's t test was used to compare two groups, and the one-way ANOVA or two-way ANOVA was used to analyze the data among three or four groups. The statistical significance was assessed at p < .05.
Results
Ferroptosis was presented in MCAO-rat, accompanied by an increase in KLF6
The TTC analysis after MCAO induction showed an increase in lesion volume compared to the sham group (Figure 1(a)). Reports have shown a possible connection between cerebral I/R damage and excessive ferroptosis. Researchers examined ferroptosis-related genes and ferroptosis iron content in MCAO-treated rats to understand the role of ferroptosis. The findings demonstrated that MCAO had a significant impact on iron levels and ACSL4 expression, whereas GPX4 and FTH1 levels were downregulated (Figure 1(b) and (c)). Moreover, KLF6 expression in MCAO was significantly increased compared with the Sham group (Figure 1(d)–(f)). Ferroptosis was presented in MCAO-rat, accompanied by an increase in KLF6. (A) Quantification of infarction volumes with TTC staining after MCAO injury in vivo. (B) Iron accumulation in MCAO. (C) WB detected the relative expression level of GPX4, ACSL4 and FTH1. (D) qRT-PCR and (E) WB measured the KLF6 expression. (F) IHC examined the expression level of KLF6.
KLF6 silencing ameliorates cognitive deficits in MCAO model rats
We investigated the effects of KLF6 knockdown in MCAO, and the rats were intracerebroventricularly injected with control shRNA or sh-KLF6. Figure 2(a) shows that after the removal of the platform in the MWM test (day 6), the typical swimming pathways of the rats migrated across various quadrants. In terms of cognitive performance, including latency to locate the submerged platform, frequency of crossing the platform, and time in the target quadrant, the findings showed that rats who received MCAO surgery had severe impairments, while rats that had KLF6 knockdown showed considerable recovery (Figure 2(b)–(c)). KLF6 Silencing ameliorates cognitive deficits in MACO model rats. (A) Representative swimming traces on Day 7. (B) Latency of the rats (C) Frequency of entering the target quadrant, (D) Duration of time spent in the target quadrant of different groups of mice in the MWM test.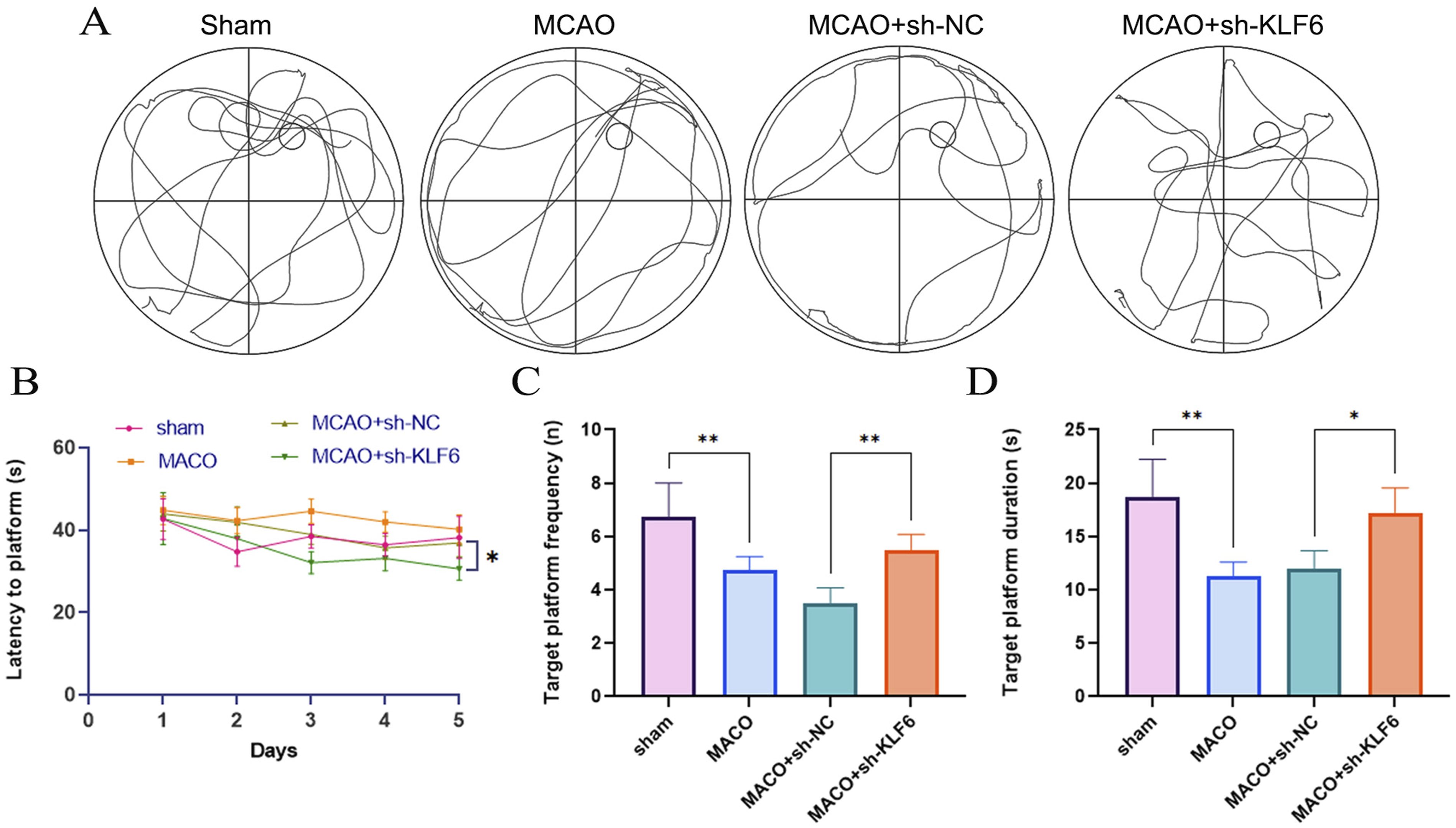
KLF6 silencing attenuates MCAO-induced brain injury
Figure 3(a) and (b) show that after sh-KLF6 transfection, the MCAO model’s expression level of KLF6 protein was significantly reduced. In addition, blocking KLF6 clearly decreased the enlarged infarcted size of brain tissues in the MCAO group (Figure 3(c)). In the MCAO group, Nissl staining revealed disorganized neuronal organization and a dearth of Nissl bodies; however, sh-KLF6 had the opposite impact (Figure 3(d)). Figure 3(e) shows that inhibiting KLF6 also reduced neural cell death in the MCAO group. Evidence from these experiments demonstrated that KLF6 restraint reduced brain damage caused by MCAO. KLF6 Silencing attenuates MCAO-induced brain injury. (A) WB analyzed the levels of KLF6. (B) The immunohistochemical assay of KLF6 in different rats. (C) TTC staining detected the infarct size of brain tissues. (D) CNissl staining was performed to evaluate neuronal damage. (E) Neuron apoptosis in each group (TUNEL staining).
Knockdown of KLF6 ameliorated oxidative stress in MCAO rats
The results showed that MCAO induces ROS and malondialdehyde (MDA) elevations, whereas sh-KLF6 treatment attenuates ROS (Figure 4(a)) and MDA levels in MCAO. By contrast, other biomarkers of antioxidant activity, that is, superoxide dismutase (SOD) and glutathione (GSH), increased after MCAO (Figure 4(c) and (d)). Notably, these elevations could be reversed by sh-KLF6. Moreover, sh-KLF6 also significantly restored the decreased SOD1 level and suppressed increased NOX2 expression in brain tissues of MCAO rats. Knockdown of KLF6 ameliorated oxidative stress in MCAO rats. (A) The levels of ROS using DCFH-DA staining. (B–D) The levels of MDA, SOD, and GSH were assessed using relevant test kits. (E) WB measured the SOD1 and NOX2 expression.
Knockdown of KLF6 ameliorated ferroptosis in MCAO rats
The levels of Fe2+ were found to be greatly raised by MCAO when compared to the sham, but they were dramatically lowered by KLF6 knockdown (Figure 5(a)). In addition, when comparing the MCAO group to the sham group, we found that ACSL4 and TFR1 had significantly higher levels, whereas FTH1 and GPX4 had significantly lower levels. Figure 5(b) and (c) show that after MCAO damage, the expression levels of these genes were considerably restored in the brain tissue of rats when KLF6 was inhibited. Knockdown of KLF6 ameliorated ferroptosis in MCAO rats. (A) The changes in iron were measured. (B) Changes of ferroptosis-related proteins after KLF6 inhibition. (C) The mRNA expression of ferroptosis-related indicators in MCAO rats.
Knockdown of KLF6 inhibited activation of Nrf2/HO-1 in MCAO rats
We investigated Nrf2 and its downstream target marker HO-1 expression levels in MCAO rats subjected to KLF6 interference in light of Nrf2’s role in oxidative stress and neuroprotection. After KLF6 knockout, MCAO rats showed markedly restored elevated levels of Nrf2 and HO-1 in brain tissues of MCAO rats (Figure 6(a) and (b)). Knockdown of KLF6 inhibited activation of Nrf2/HO-1 in MCAO rats. (A) Protein expression levels were analyzed using WB for Nrf2 and HO-1. (B) The immunohistochemical assay of Nrf2 and HO-1 in different rats.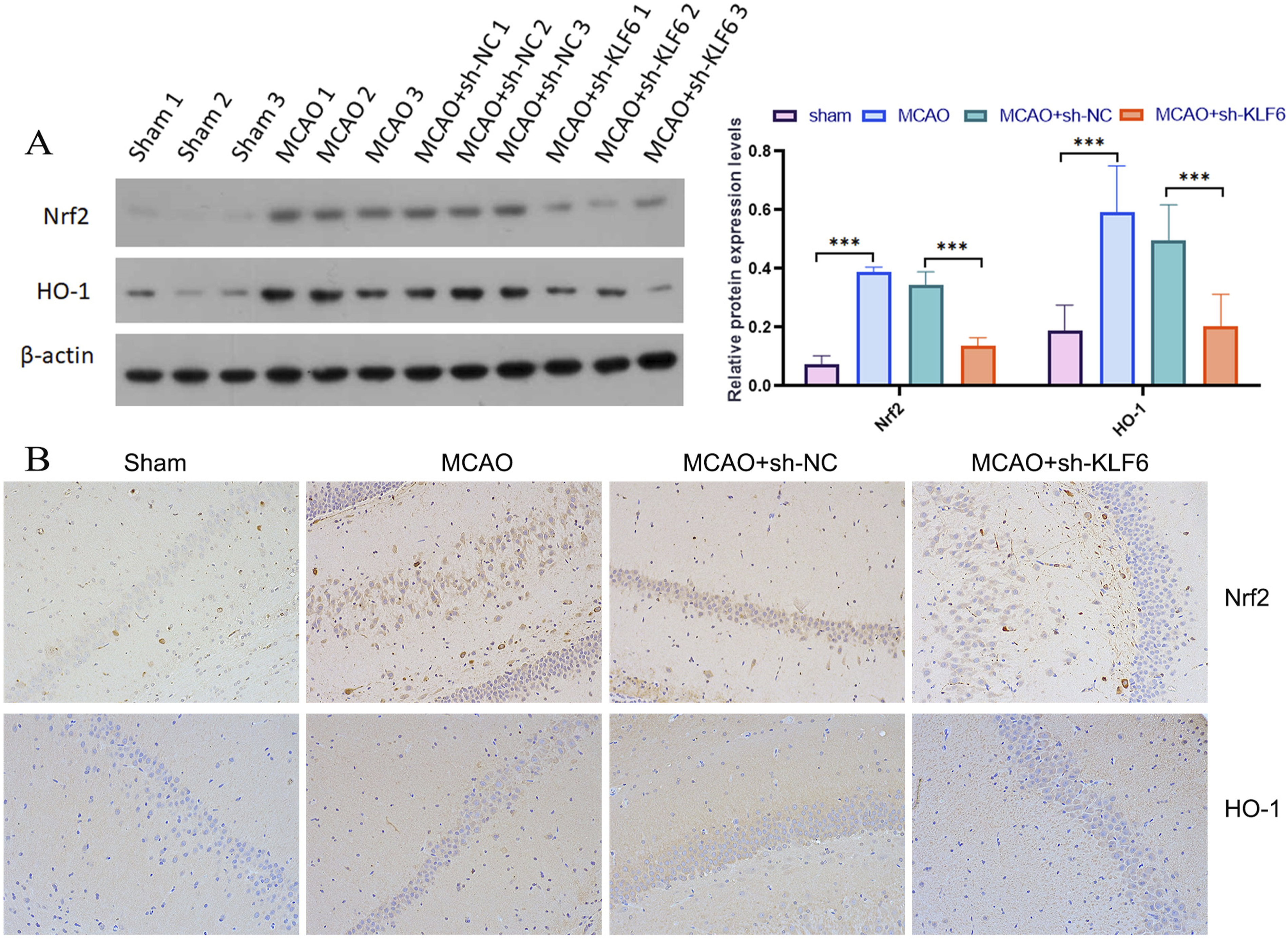
Discussion
Cerebral IR injury involves such pathophysiological processes as oxidative stress reaction, inflammatory reaction, and apoptosis, among which the brain tissue injury in the central area of the lesion is irreversible, increasing the risk of disability.5,17 Therefore, timely restoration of cerebral perfusion is crucial to reduce brain tissue ischemia-reperfusion injury. Recent evidence has highlighted the pivotal role of KLF6 in mediating myelination within the central nervous system. 18 Therefore, the purpose of this work was to find out how KLF6 contributes to cerebral IR and what processes are involved in the process. Through stimulation of the Nrf2/HO-1 signaling pathway, the experimental findings revealed that KLF6 promotes neurological impairment after MACO.
The primary finding of this work was the increased expression of KLF6 in rat brain tissues after MACO modeling. Cognitive impairment is one of the complicated complications after cerebral IR. There is an urgent need to develop neuroprotective medications to prevent and restore it. 19 In this study, the role of KLF6 in cognitive deficits was demonstrated by the finding that KLF6 silencing ameliorates cognitive deficits in MACO model rats.
There is growing evidence that oxidative stress and ferroptosis are intimately associated with I/R damage in the brain.20,21 Through promoting Nrf2 nuclear translocation and inhibiting the GSNOR/GSTP1 axis, Duan et al. discovered that N6022 reduces microglial ferroptosis caused by cerebral I/R damage. 22 Our findings indicate that the knockdown of KLF6 significantly alleviates brain injury and reduces infarct volume following MCAO. In addition, following cerebral I/R damage, KLF6 silencing restored the levels of GSH, GPX4, SLC7A11, MDA, ROS, and iron in rat brain tissues. Ferroptosis sensitivity is regulated by ACSL4, a key contributor to this form of cell death. 23 Interestingly, KLF6 has been shown to exacerbate myocardial ischemia/reperfusion damage by activating ACSL4-mediated ferroptosis. 24 Furthermore, our research suggests that downregulating KLF6 expression and activating the STAT3 pathway might protect against cerebral I/R damage and improve neurological recovery after an ischemic stroke, respectively, by silencing HOTAIR. 25 One significant mechanism in resistance to ferroptosis is the Nrf2/HO-1 pathway. 26 According to previous research, normobaric oxygen activates the Nrf2/HO-1 antioxidative stress pathway, which has a neuroprotective effect after brain ischemia. 27 Additionally, we discovered that in MCAO rats, knocking down KLF6 prevented Nrf2/HO-1 activation.
A significant mechanism underlying resistance to ferroptosis involves the Nrf2/HO-1 pathway. By inhibiting KLF6, MCAO-induced brain injury was ameliorated, infarct size was reduced, cognitive impairments were ameliorated, neuronal damage was reversed, oxidative stress injury and cell ferroptosis were reduced via the Nrf2/HO-1 axis, and KLF6 plays an essential role in ferroptosis. Our research showed that KLF6 might be a therapeutic target for repairing brain I/R damage.
Footnotes
Authors’ contributions
HQ, SZ and CW designed and performed the research; HQ, SZ, RQ, QL, JZ and YD analyzed the data; HQ and SZ wrote the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
