Abstract
Introduction
Diabetic cardiomyopathy (DCM) is a complication of diabetes mellitus (DM) that can lead to heart failure and increase the risk of mortality. Pedunculoside (PE), a novel triterpenoid saponin, exhibits anti-inflammatory and anti-oxidative stress (OS) properties. However, its role in DCM remains unexplored.
Methods
DCM models were established and treated with PE or the Nrf2 inhibitor (ML385). In vitro, cell function was evaluated using CCK-8, flow cytometry, qRT-PCR, and ELISA. In vivo, fasting blood glucose and insulin levels in rats were measured. The effects of PE on DCM were assessed using HE staining, TUNEL staining, and corresponding kits. Additionally, Nrf2/HO-1 pathway proteins were analyzed by western blot.
Results
Low doses of PE (2.5, 5, 10, and 20 μM) did not affect the viability of H9c2 cells. PE (10 and 20 μM) improved cell viability and prevented apoptosis, inflammation, and OS in high glucose (HG)-stimulated H9c2 cells. PE also upregulated Nrf2 in the nucleus and enhanced HO-1 and NQO1 expression in HG-treated H9c2 cells. Furthermore, the Nrf2 inhibitor (ML385) reversed PE’s protective effects on HG-induced cell injury. In vivo, PE reduced blood glucose, increased insulin, alleviated myocardial injury, inhibited apoptosis, decreased levels of inflammatory factors and OS, and upregulated Nrf2, HO-1, and NQO1 in DCM model rats.
Discussion
PE alleviates DCM injury by activating the Nrf2/HO-1 pathway. These findings support the potential therapeutic application of PE in DCM.
Introduction
With changes in dietary structure and living habits, the incidence of diabetes mellitus (DM) continues to rise globally. 1 Diabetic cardiomyopathy (DCM), a significant cardiovascular complication of DM, 2 affects approximately 15–20% of DM patients. 2 DCM is characterized by left ventricular dysfunction, reduced diastolic and systolic function, decreased ejection fraction, heart failure, hypertension, and valvular heart disease. 3 Current research has identified metabolic abnormalities, oxidative stress (OS), vascular injury, ventricular remodeling, and apoptosis as key mechanisms contributing to the progression of DCM.4,5 However, there is currently no effective drug that can completely cure DCM.
The antioxidant effects mediated by the NF-E2-related factor 2 (Nrf2)/heme oxygenase-1 (HO-1) axis play a critical role in cellular defense against OS and in maintaining homeostasis.6,7 Nrf2, a basic leucine zipper transcription factor, is sequestered in the cytoplasm under normal physiological conditions by binding to Keap1 to form the inactive Nrf2-Keap1 complex.8,9 Upon stimulation by OS, the Nrf2-Keap1 complex dissociates, allowing Nrf2 to translocate to the nucleus. There, it binds to the antioxidant response element (ARE) and induces the expression of downstream antioxidant and phase II detoxifying enzymes,10,11 including superoxide dismutase (SOD), NAD(P)H quinone oxidoreductase 1 (NQO1), HO-1, and catalase. 12 Studies have demonstrated that activating the Nrf2 pathway can alleviate OS,13,14 inflammation, 15 and fibrosis 16 in DCM, suggesting that pharmacological interventions targeting the Nrf2/HO-1 pathway could provide a promising therapeutic strategy for DCM.
Pedunculoside (PE), a novel triterpenoid saponin isolated from the Ilex genus, exhibits various pharmacological effects, including anti-inflammatory, anti-cancer, and anti-fibrotic properties.17–20 For instance, PE has been shown to alleviate mastitis by suppressing inflammation and maintaining the integrity of the blood-milk barrier. 19 Additionally, PE reduces acute lung injury, inflammation, and pulmonary fibrosis in septic mice by inhibiting the AKT/NF-κB pathway. 21 It has also been found to prevent malignant progression and overcome gefitinib resistance in non-small cell lung cancer (NSCLC) by regulating the MAPK and Nrf2 pathways. 18 Despite these promising findings, whether PE can ameliorate DCM through the Nrf2/HO-1 pathway remains unclear.
This study aimed to investigate the protective effects of PE on DCM by using cellular and rat models of DCM. Moreover, we sought to determine whether the beneficial effects of PE on DCM were mediated via the Nrf2/HO-1 pathway, particularly about inflammation, OS, and apoptosis. These findings may provide valuable insights for the clinical development of targeted therapies for DCM.
Materials and methods
Cell culture
Human normal cardiomyocytes (H9c2) were obtained from the Shanghai Institute of Biological Sciences (Shanghai, China) and cultured in DMEM (Gibco, USA; Cat. No. 11995-065) supplemented with 10% FBS (Hyclone, USA; Cat. No. SH30071.03) at 37°C in a 5% CO2 atmosphere.
Cell grouping
H9c2 cells were treated with different concentrations of PE and divided into the following treatment groups: 0 μM PE, 2.5 μM PE, 5 μM PE, 10 μM PE, 20 μM PE, and 40 μM PE. 18 The DCM cell model was constructed by inducing H9c2 cells with 33 mmol/L glucose (Sigma-Aldrich, USA; Cat. No. G7021).5,22 The cells were divided into the control (con) group (5.5 mmol/L glucose), high-glucose (HG) group (35 mmol/L glucose), HG + 5 μM PE group, HG + 10 μM PE group, HG + 20 μM PE group, and HG + 20 μM PE + ML385 group. Nrf2 inhibitor ML385 (IM1020, Sigma-Aldrich, USA) specifically downregulates Nrf2 expression.
CCK-8
H9c2 cells (1 × 10⁴ cells/well) were seeded into 96-well plates and treated as described above. After 48 h, 10 μL CCK-8 reagent (HANBIO, China; Cat. No. C0009) was added to each well. Absorbance at 450 nm was measured using a microplate reader (BioTek, USA; Cat. No. ELx808).
Flow cytometry
Cells (1 × 10⁵) from each group were collected, seeded into 6-well plates, and stained with 5 μL Annexin V-FITC (BestBio, Shanghai, China; Cat. No. BB-4109-2) and 5 μL PI dye (BestBio, Shanghai, China; Cat. No. BB-4104) in the dark for 20 min. Apoptosis was analyzed using a BD FACS Calibur flow cytometer (BD Biosciences, CA, USA; Cat. No. 655014).
Qualitative real-time PCR (qRT-PCR)
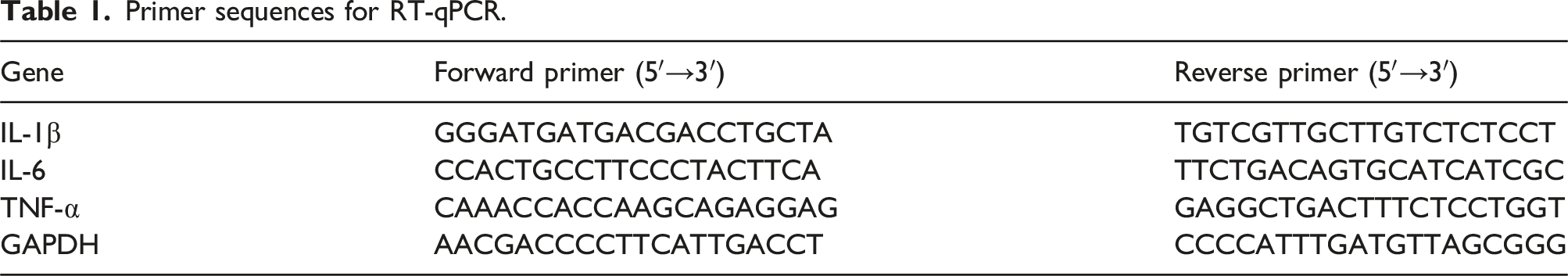
Primer sequences for RT-qPCR.
Western blot
Proteins from H9c2 cells and myocardial tissues were extracted using RIPA lysis buffer (Beyotime, Shanghai, China; Cat. No. P0013 C) and quantified using a BCA kit (Beyotime, Shanghai, China; Cat. No. P0012S). Proteins (30 μg per well) were separated via 10% SDS-PAGE, transferred to PVDF membranes (Thermo Fisher Scientific, USA; Cat. No. IPVH00010), and blocked with 5% skim milk (BioMed, China; Cat. No. B100) for 2 h. Membranes were incubated with primary antibodies at 4°C overnight, followed by secondary antibody incubation (1:10,000, Abcam, UK; Cat. No. ab6721). Bands were developed using ECL reagent (Proteinbio, Nanjing, China; Cat. No. P1010), and densitometry analysis was performed using ImageJ software (NIH, USA). Antibodies used included anti-Nrf2 (1:1000, Proteintech, USA; Cat. No. 16396-1-AP), anti-Lamin B1 (1:1000, Proteintech, USA; Cat. No. 12987-1-AP), anti-HO-1 (1:1000, Proteintech, USA; Cat. No. 10701-1-AP), anti-NQO1 (1:2000, Invitrogen, USA; Cat. No. PA5-85516), and anti-β-actin (1:5000, Sigma-Aldrich, USA; Cat. No. A5441).
Animal
The animal experiments were approved by the Ethics Committee of Shaoxing Central Hospital. Model rats were fed a HG and high-fat diet for 4 weeks. After fasting for 12 hours, a single intraperitoneal injection of 30 mg/kg streptozotocin (STZ) dissolved in citrate buffer (0.1 mol/L, pH 4.5) was administered, with the buffer maintained on ice until injection. 23 Rats in the normal control group were fed a standard diet and injected intraperitoneally with the same volume of citrate buffer. After 72 hours, fasting blood glucose levels were measured. Rats with fasting blood glucose levels ≥16.7 mmol/L for three consecutive measurements were considered successfully diabetic. Experimental groups included the control (sham) group, PE treatment group (30 mg/kg PE, based on previously published studies 24 ), DCM group, and DCM + PE group (30 mg/kg PE). The DCM and DCM + PE groups continued on the HG and high-fat diet, while the sham and PE groups received a standard diet. PE or an equal volume of normal saline was administered intraperitoneally to the respective groups for 8 weeks. At the end of the feeding period, fasting blood glucose and insulin levels were measured. Following these assessments, rats were euthanized under anesthesia, and cardiac function was evaluated using ultrasonography.
Echocardiography
Rats were anesthetized with 1% pentobarbital sodium (Sigma-Aldrich, USA; Cat. No. P3761) prior to M-mode echocardiography, which was performed to assess left ventricular ejection fraction (EF) and left ventricular shortening fraction (FS). Electrocardiogram (ECG) measurements were recorded using the ECG-1000 system (Biopac Systems, Inc., USA).
H and E staining
Myocardial tissues were excised, fixed in formalin, dehydrated using graded ethanol, embedded in paraffin, and sectioned into 5 μm slices. The sections were dewaxed at 60°C and stained with hematoxylin and eosin. Pathological alterations in myocardial tissues were examined under a light microscope (Nikon, Japan).
TUNEL staining
The TUNEL assay kit (Atagenix, China; Cat. No. UNEL-KIT) was used to detect apoptosis in myocardial tissue sections. Sections were treated with 20 µg/mL protease K for 5 minutes, followed by incubation with TUNEL reaction mixture at 37°C for 1 hour. Nuclei were counterstained with DAPI, and apoptotic cells were visualized under a fluorescence microscope (Olympus, Japan). The percentage of TUNEL-positive cells was calculated.
Detection of SOD and MDA
Superoxide dismutase (SOD) and malondialdehyde (MDA) levels were quantified using the SOD activity assay kit (Jiancheng, Nanjing, China; Cat. No. A001-1) and the MDA assay kit (Jiancheng, Nanjing, China; Cat. No. A003-1), respectively. Assays were conducted on homogenized samples from H9c2 cells and rat myocardial tissues.
Detection of cardiac biomarkers
Serum levels of cardiac troponin I (cTnI), creatine kinase-MB (CK-MB), and brain natriuretic peptide (BNP) were measured using commercially available kits: the cTnI assay kit (Enzyme-Linked Biotechnology, China; Cat. No. ELISA-CTnI), the CK-MB assay kit (Senbeijia, Nanjing, China; Cat. No. CKMB-ELISA), and the BNP assay kit (TSZ Biosciences, USA; Cat. No. TSZ-BNP), following the manufacturer’s protocols.
ELISA
Myocardial tissue homogenates from each group were analyzed for pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6, using ELISA kits (Nanjing Jiancheng, China; Cat. Nos. A005-1 for TNF-α, A056-1 for IL-1β, and A056-2 for IL-6) based on the manufacturer’s instructions.
Statistical analysis
Data were presented as mean ± standard deviation (SD). Statistical analysis was performed using GraphPad Prism 7.0 software. Multiple group comparisons were conducted using one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test. A p-value of <0.05 was considered statistically significant.
Results
PE promotes viability and prevents apoptosis of HG-treated H9c2 cells
To investigate the effect of PE on DCM, the structure of PE was first analyzed. The molecular formula of PE was C36H58O10, with a molecular weight of 650.84 (Figure 1(A)). CCK-8 data indicated that PE at concentrations of 2.5, 5, 10, and 20 μM did not affect the viability of healthy H9c2 cells, but 40 μM PE decreased cell viability (Figure 1(B)). Based on these results, 5, 10, and 20 μM PE were selected for further experiments. H9c2 cells were treated with high glucose (HG) to establish an in vitro diabetes model. HG-treated H9c2 cells were then treated with PE at concentrations of 5, 10, and 20 μM. CCK-8 results showed that HG treatment reduced the viability of H9c2 cells. The viability of HG-treated H9c2 cells was increased by 10 and 20 μM PE, with the most prominent effect observed at 20 μM PE (Figure 1(C)). Similarly, HG treatment increased the apoptosis rate in HG-treated H9c2 cells, which was reduced by 10 and 20 μM PE, particularly at 20 μM PE (Figure 1(D) and (E)). PE reverses HG-induced viability inhibition and apoptosis promotion in H9c2 cells. (A) The chemical molecular structure of PE. (B) CCK-8 exhibited the change of cell viability in H9c2 cells treated with 0, 2.5, 5, 10, 20, and 40 μM PE, *p < 0.05. Then HG-treated H9c2 cells were administrated with 5, 10, and 20 μM PE. (C) Cell viability was assessed by CCK-8. (D and E) Verification of changes in apoptosis using flow cytometry. Con, control. **p < 0.01, ***p < 0.001 versus the con group; #p < 0.05, ##p < 0.01 versus the HG group.
PE alleviates HG-induced inflammation and OS in H9c2 cells
Next, the effect of PE on inflammation and oxidative stress (OS) was further investigated. Inflammatory factors were assessed using qRT-PCR. The results showed that the expression levels of IL-1β, IL-6, and TNF-α in H9c2 cells were increased in the HG group, but PE treatment at 10 and 20 μM reversed the HG-induced elevation of these inflammatory markers in H9c2 cells (Figure 2(A)–(C)). Moreover, HG treatment decreased SOD levels and increased MDA concentrations in H9c2 cells, both of which were notably reduced by 10 and 20 μM PE treatment, especially at 20 μM PE (Figure 2(D) and (E)). PE reduces inflammation and OS in HG-induced H9c2 cells. HG-treated H9c2 cells were processed with 5, 10, and 20 μM PE. qRT-PCR analysis of IL-1β (A), IL-6 (B), and TNF-α (C) levels. The content of MDA (D) and SOD (E) was verified using the kits. Con, control. **p < 0.01, ***p < 0.001 versus the con group; #p < 0.05, ##p < 0.01 versus the HG group.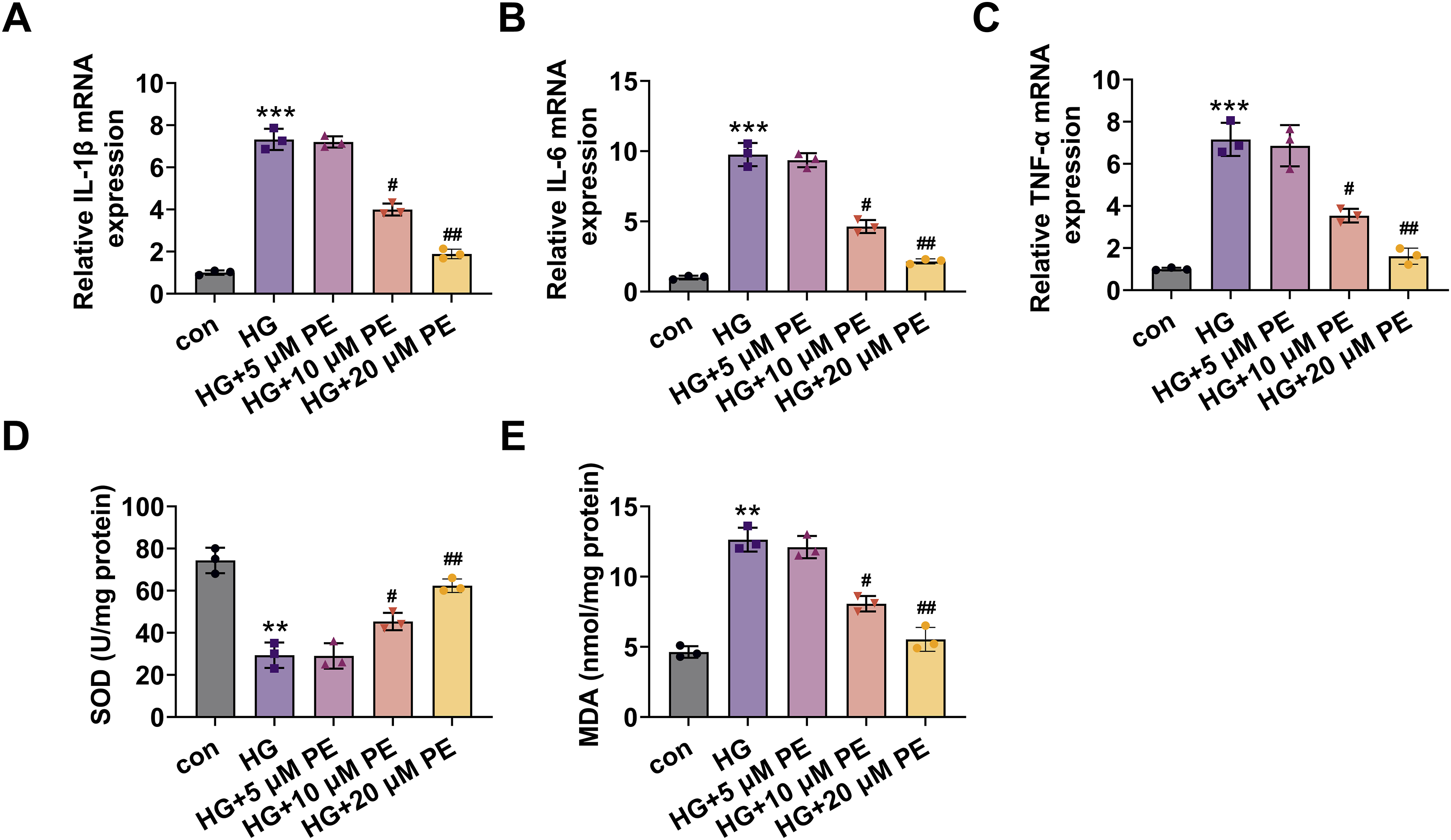
PE enhances cell viability and reduces apoptosis, inflammation, and OS via the Nrf2 pathway in HG-treated H9c2 cells
To explore the potential mechanism of PE in DCM, we examined the effects of PE on key regulatory proteins. As shown in Figure 3(A), HG treatment decreased Nrf2 expression in the nucleus and lowered the levels of HO-1 and NQO1 proteins. These effects were gradually reversed by 10 and 20 μM PE, with 20 μM PE showing the most substantial effect (Figure 3(A)). These data suggest that PE mitigates HG-induced injury in H9c2 cells through the Nrf2/HO-1 pathway. To further confirm the role of Nrf2, we applied the Nrf2 inhibitor ML385. Our results showed that 20 μM PE increased the viability of HG-treated H9c2 cells, but this effect was reversed by ML385 (Figure 3(B)). Additionally, PE treatment (20 μM) reduced apoptosis in HG-treated H9c2 cells, whereas ML385 treatment increased the apoptosis of HG-treated H9c2 cells, reversing PE’s protective effect (Figure 3(C) and (D)). Furthermore, PE (20 μM) reduced IL-1β expression and MDA concentrations, both of which were reversed by ML385 treatment in HG-treated H9c2 cells (Figure 3(E) and (F)). ML385 attenuates the promotion of cell viability, inhibition of apoptosis, inflammation, and OS mediated by PE in HG-treated H9c2 cells. (A) Western blotting analysis for the determination of Nrf2, Lamin B1, HO-1, and NQO1 expressions in HG-induced H9c2 cells after PE treatment. Then HG-induced H9c2 cells were processed with 20 μM PE in combination with ML385. (B) CCK-8 displayed a change in cell viability. (C and D) Cell apoptosis was monitored by flow cytometry, and the apoptosis rate was calculated. (E) qRT-PCR analysis of IL-1β. (F) The assessment of MDA content with the kit. Con, control. **p < 0.01, ***p < 0.001 versus the con group; #p < 0.05, ##p < 0.01 versus the HG group; &p < 0.05, &&p < 0.01 versus the HG + 20 μM PE group.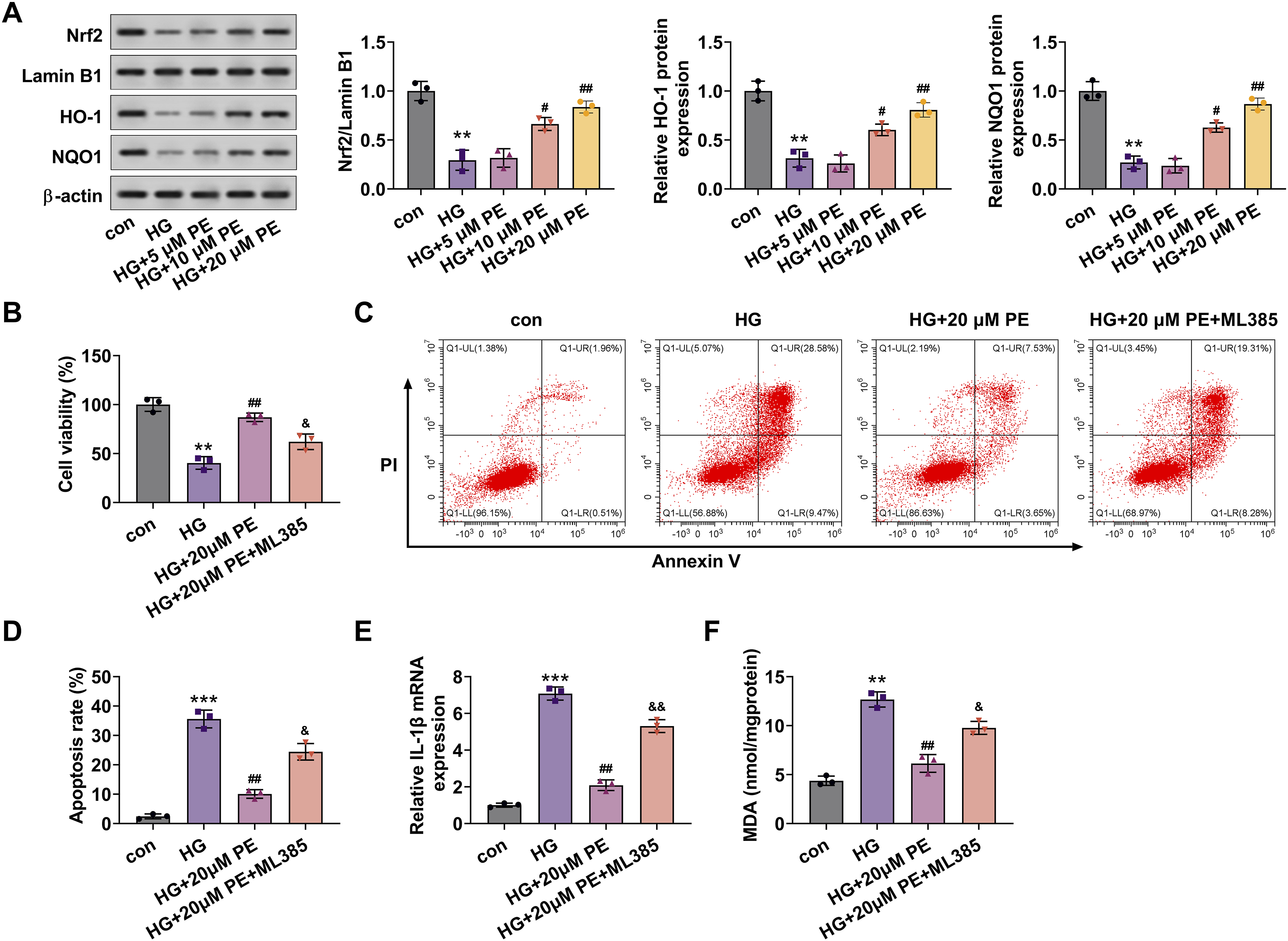
PE alleviates myocardial injury in DCM model rats
To investigate the effects of PE in vivo, we constructed a rat model of DCM. We found that blood glucose levels were elevated in the DCM group, but treatment with 30 mg/kg PE partially reversed this increase, while insulin levels exhibited an opposite trend (Figure 4(A)). Echocardiographic analysis revealed that the EF and FS were decreased in the DCM group compared to the sham group. PE treatment improved both EF and FS in DCM model rats (Figure 4(B)). Additionally, myocardial markers cTnI, CK-MB, and BNP levels were elevated in the DCM group but reduced by PE treatment (Figure 4(C)). H&E staining showed that in the sham group, the myocardial structure was organized and tightly arranged, with no fiber breaks. In the DCM model group, the myocardial structure was disorganized, and there was abundant infiltration of inflammatory cells, such as neutrophils and lymphocytes. Pathological changes, including fiber breaks and inflammatory cell infiltration, were reduced in the PE-treated group, alleviating myocardial injury compared to the model group (Figure 4(D)). Histological sections were prepared using transverse sectioning, and areas of interest for analysis were randomly selected to minimize bias. All sections were reviewed and interpreted in a blinded manner to ensure objective analysis. TUNEL staining indicated increased apoptosis of cardiomyocytes in the DCM group, while PE treatment inhibited myocardial apoptosis in DCM model rats (Figure 4(E)). PE alleviates the myocardial injury in DCM model rats. DCM rat model was established and the rats were intraperitoneally injected with 30 mg/kg PE. (A) Measurement of blood glucose and insulin levels in rats. (B) The levels of EF and FS were evaluated by echocardiography. (C) The levels of cTnI, CK-MB, and BNP were credited using the corresponding kits in serum samples of rats. (D and E) The pathological structure and apoptosis of myocardial tissue were evaluated by H&E and TUNEL staining, and the TUNEL-positive cells were also counted, magnification = 500×. *p < 0.05, **p < 0.01 versus the sham group; #p < 0.05, ##p < 0.01 versus the PE group.
PE alleviates inflammation and OS in DCM model rats
Furthermore, we observed that IL-1β, IL-6, and TNF-α levels were elevated in the DCM group. However, PE treatment reduced the levels of these inflammatory cytokines in the myocardial tissues of DCM model rats (Figure 5(A)). PE treatment also decreased MDA levels and increased SOD content in the myocardial tissues of DCM rats (Figure 5(B)–(C)). Moreover, the expression levels of Nrf2, HO-1, and NQO1 proteins were decreased in the DCM group, but PE treatment partially restored these levels (Figure 5(D)). Taken together, these findings suggest that PE inhibits cardiomyocyte apoptosis, inflammation, and OS through the Nrf2/HO-1 pathway (Graphical abstract). PE inhibits inflammation and OS in DCM model rats. The sham and DCM model rats were treated with 30 mg/kg PE. (A) IL-1β, IL-6, and TNF-α levels were assessed using ELISA kits in myocardial tissues. (B and C) MDA and SOD levels were identified by applying the kits. (D) Nrf2, Lamin B1, HO-1, and NQO1 expressions were assessed using Western blot in myocardial tissues. **p < 0.01 versus the sham group; #p < 0.05 versus the PE group.
Discussion
Currently, there is no specific drug for DCM therapy in Western medicine, and the management of DCM risk mainly focuses on lowering blood glucose, reducing blood lipids, regulating blood pressure, and promoting insulin secretion in clinical practice.
25
In recent years, the efficacy of traditional Chinese medicine in DCM therapy has been increasingly recognized.26,27 The genus Ilex (Aquifoliaceae) is known for its triterpenes and their glycosides, flavonoids, and phenols, which possess anti-inflammatory, antibacterial, anti-thrombotic, and cardiovascular protective effects.
28
Among these, PE is one of the most abundant and representative active compounds.
29
Studies have shown that PE exhibits multiple pharmacological effects, including antibacterial, anticancer, anxiolytic, and local anesthetic properties.18,19,30 Additionally, PE has been found to protect against acute myocardial ischemia/reperfusion injury in rats by mechanisms involving increased SOD levels and decreased MDA and cTnI content.
17
These findings suggest that PE holds promise as a candidate drug or precursor for heart disease treatment. However, its therapeutic effects on DCM have not yet been reported. In this study, we further investigated the protective effects of PE in cellular and animal models of DCM and explored its underlying mechanisms to provide a basis for the development of therapeutic drugs for DCM (Figure 6). Graphical abstract. The action and mechanism diagram of PE in DCM. PE reduces cardiomyocyte apoptosis, OS, and inflammation by enhancing the Nrf2/HO-1 pathway in HG-treated H9c2 cells and DCM model rats.
H9c2 cells are widely used as a cardiomyocyte model for studying the pathogenesis and potential treatments of DCM. 31 For example, chuanxiongzine has been shown to ameliorate HG-induced injury in H9c2 cells by activating the AMPK pathway, 32 while berberine and its metabolites demonstrated protective effects against HG-induced H9c2 injury. 33 These studies highlight the utility of H9c2 cells in DCM research. Building upon prior studies,17,18 we treated H9c2 cells with PE at concentrations of 2.5, 5, 10, 20, and 40 μM. Our results showed that PE at concentrations up to 20 μM had no cytotoxic effects on H9c2 cells, whereas 40 μM PE reduced cell viability. Consequently, we selected 5, 10, and 20 μM PE for subsequent experiments. In this study, we also established a DCM cell model by exposing H9c2 cells to 35 mmol/L glucose. HG treatment reduced cell proliferation, indicating HG-induced injury in H9c2 cells. For in vivo validation, we constructed a rat model of DCM following previously established protocols.5,17 Blood glucose levels were elevated, while insulin levels, EF, and FS were decreased in the model group, along with elevated markers of cardiac injury such as cTnI, CK-MB, and BNP, confirming successful model establishment. Upon PE pretreatment, these markers were reversed, with high-dose PE demonstrating superior efficacy compared to low-dose PE, suggesting a dose-dependent therapeutic effect of PE on myocardial injury.
Apoptosis, OS, and inflammation are pivotal pathological processes in DCM development. 34 Apoptosis is a key mechanism of cardiomyocyte death in DCM, closely associated with metabolic dysregulation and OS. 35 Hyperglycemia-induced OS triggers the production of ROS, which damage cell membranes, proteins, and nucleic acids, ultimately leading to apoptosis. 36 Chronic inflammation exacerbates myocardial cell damage, promotes fibrosis, and facilitates cardiac remodeling by activating inflammatory cells and cytokines. 37 A previous study demonstrated that naringenin ameliorated myocardial injury through the Nrf2 and NF-κB pathways in diabetic mice. 38 Thus, targeting apoptosis, OS, and inflammation is a promising therapeutic strategy for DCM.
Nrf2 is a pivotal antioxidant transcription factor that combats OS and inflammation by promoting the expression of antioxidant genes. 39 Upon stimulation by exogenous OS, Nrf2 binds to antioxidant response elements, upregulating HO-1 and enhancing anti-inflammatory and antioxidant defenses. 40 In DCM, persistent hyperglycemia increases OS, posing a severe threat to cardiac cells. 35 Activation of the Nrf2/HO-1 pathway reduces OS and inflammation, thereby protecting cardiomyocyte structure and function and slowing DCM progression. 41 Recent studies have shown that PE can regulate Nrf2 to inhibit malignant progression and reduce gefitinib resistance in NSCLC. 18 However, its role in regulating the Nrf2 pathway in DCM remains unclear. Our study demonstrated that PE upregulated nuclear Nrf2 and cellular HO-1 and NQO1 levels in HG-induced H9c2 cells and DCM model rats, with the most significant effects observed at 10 and 20 μM doses. These findings suggest that the cardioprotective effects of PE in DCM may be mediated via the Nrf2/HO-1 pathway.
Our results showed that 10 and 20 μM PE improved cell viability and inhibited apoptosis, inflammation, and OS in HG-induced H9c2 cells, while 5 μM PE had no significant effects. Including 5 μM PE in our study provided a comprehensive evaluation of dose-dependent effects under pathological HG conditions. Although 5 μM PE showed no significant protective effects, its inclusion helped establish a baseline for comparison and highlighted the threshold effect of PE efficacy, where higher doses were necessary to trigger protective mechanisms. These findings emphasize the importance of exploring dose-response relationships to determine the minimum effective concentration of PE for DCM therapy. Future studies should investigate the effects of intermediate doses (e.g., 7.5 μM) to refine the therapeutic window for PE. In vivo experiments further validated PE’s ability to attenuate myocardial injury, inflammation, and OS in DCM model rats, reinforcing its potential as a therapeutic agent.
Conclusions
Our study revealed that PE mitigates cardiomyocyte apoptosis, oxidative stress, and inflammation while improving cell viability via the Nrf2/HO-1 pathway, suggesting its potential as a therapeutic option for DCM. However, this study is limited by the lack of investigation into autophagy, a critical factor in DCM pathology. Future research will explore the role of PE in autophagy and further clarify its regulatory mechanisms within the Nrf2/HO-1 pathway.
Footnotes
Author contributions
Yuanben Lu and Jianqiang Meng conceived and designed the study and provided administrative support. Dewen Zhu and Zhenhua Jiang performed the experiments and analyzed data. Hailiang Ma analyzed and interpreted the data. Yuanben Lu and Jianqiang Meng wrote the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Shaoxing Health Science and Technology Plan (2023SKY105).
