Abstract
Background
Until now, no definite standardized method has been used to promptly assess the severity and outcome of acute aluminum phosphide (ALP) poisoning. The current study aimed to evaluate the performance of the new Poisoning Mortality Score (PMS) and PGI score for predicting mortality in acute ALP-poisoned patients, highlighting the accuracy of new PMS components.
Patients and Methods
A 2-year cross-sectional study was conducted on ALP-poisoned patients admitted to Tanta University Poison Control Centre from April 2021 to March 2023. Socio-demographics, poisoning data, and initial vital signs were recorded. Additionally, new PMS and PGI scores were calculated on admission. Patients were categorized according to the mortality outcome into survivors and nonsurvivors.
Results
Out of 160 included ALP poisoned patients, mortality was recorded in 112 (70%) patients. The nonsurvivors had significantly higher median PGI and new PMS values than survivors. New PMS, vital signs component of new PMS, and PGI conveyed good discriminatory power for predicting mortality (AUC = 0.883, 0.873, and 0.817, respectively). Although the new PMS outperformed PGI in all predictive metrics, no significant difference in AUCs was observed between the new PMS and its vital signs component.
Conclusion
The new PMS vital signs component is closely aligned with the new PMS. Thus, it can be used as a valid, comprehensive, and practical tool to substitute the whole score calculation for rapid ALP-poisoned patient assessment to enhance emergency clinical decision-making.
Introduction
Aluminum phosphide (ALP), known as rice tablets or wheat pills, is a popular over-the-counter insecticide and rodenticide in some developing countries.1,2 Although it effectively protects crops from all stages of insects without leaving toxic residues on grains, it accounts for many suicidal deaths.3,4
ALP toxicity exists when ALP tablets are exposed to moisture or gastric acidity, releasing the rapidly absorbed phosphine gas. 5 Mechanistically, phosphine targets mitochondria, disrupting cytochrome C oxidase activity and cellular respiration. Additionally, it enhances the production of reactive oxygen species and impairs the endogenous antioxidant scavenging activities, resulting in lipid peroxidation or even cellular death.6,7
Clinically, ALP-poisoned patients often present initially with sudden onset of abdominal pain, vomiting, refractory hypotension, metabolic acidosis, and dysrhythmias. However, cases may rapidly deteriorate to multiorgan failure.8,9 Owing to the absence of a convenient antidote, the ALP mortality rates range from 37% to 100% within the first 24 hours due to cardiogenic shock and inevitable dysrhythmias. 10 Accordingly, recent statistical scales in various poison centers pointed to a tremendously increased incidence of ALP-poisoned cases with considerably alarming morbidities and mortalities.11–13
Currently, the mortality prediction in ALP poisoning depends on various baseline clinical and laboratory factors such as blood pressure, metabolic acidosis, abnormal electrocardiogram, presence of low Glasgow Coma Scale (GCS), and alteration in blood glucose levels and white blood cell counts.14,15 Additionally, other studies suggested scoring systems to provide more accurate assessment by incorporating more than one parameter, including Poisoning Severity Score (PSS), Acute Physiology and Chronic Health Evaluation II (APACHE II), Sequential Organ Failure Assessment (SOFA), and Simplified Acute Physiology Score II (SAPS II).16–18 However, most of them are not routinely used in emergencies due to time-consuming calculations resulting from including many laboratory parameters that impede the rapid decision-making of high-risk ALP-poisoned patients. 19
As a result of previous scores shortcomings, Pannu et al. (2020) 20 have suggested a simplified clinical-laboratory prognostic “PGI score” including low pH (P), low Glasgow Coma Scale score (G), and impaired systolic blood pressure (I) for assessing poisoning severity and predicting mortality. Previous studies have demonstrated its effectiveness in evaluating ALP poisoning severity as well as predicting mortality.21,22
Han et al. (2021) 23 have also constructed a new Poisoning Mortality Score (new PMS) by considering demographic factors, poisoning-related characteristics, and the patient’s physiological conditions to be more applicable to all age groups of acutely poisoned patients. Although it was validated for predicting mortality in acute organophosphate poisoning, it has not been evaluated in acute ALP poisoning.
Hence, the current study aimed to assess the performance of PGI and new PMS scores for predicting mortality in acute ALP poisoning. Owing to the wide range of new PMS (0–137 points), we further evaluate the predictive ability of each new PMS component to simplify the assessment process and assist the early treatment allocation for high-risk ALP-poisoned patients.
Patients and methods
Study design and setting
This cross-sectional study was conducted on ALP-poisoned patients admitted to Tanta University Poison Control Centre (TUPCC), Tanta Emergency Hospitals, Egypt, from April 2021 to March 2023.
Ethical consideration
This study was performed after the agreement of the institutional research ethics committee of the Faculty of Medicine - Tanta University (Approval number: 34577/3/21) and according to the Declaration of Helsinki guidelines. Written informed consent was attained from each patient or the legal guardian of the incompetent patients. Coding of the patients was considered to maintain their confidentiality and ensure anonymous data analysis.
Inclusion criteria
The current study enrolled all adult symptomatic acute ALP patients above 18 years of both sexes. The diagnosis was based on the history of exposure, the availability of the pesticide containers, and suggestive toxidrome features of metabolic acidosis, hypotension, and garlic breath odor, as well as detecting phosphine gas by applying a silver nitrate test on the gastric aspirate. A silver nitrate test was performed by mixing 5 mL of gastric aspirate with 15 mL of water in a flask and covering its mouth with 0.1 N silver nitrate-soaked filter paper. The positive result is the blackening of filter paper when heating this mixture at 50o for 15 to 20 minutes, indicating the existence of phosphine gas. 24
Exclusion criteria
Patients with co-ingestion of other agents and comorbidities such as cardiovascular, hepatic, or renal disorders were excluded from this study. Additionally, our study excluded dead patients on admission, patients with unknown poisoning, incomplete data, persistent asymptomatic patients, transferred patients, and those who received any medical intervention pre-hospital admission.
Patient data collection
A standardized sheet was conducted to gather data from the enrolled patients’ medical records. Data included socio-demographics (age, gender, and residence) and poisoning data (alleged route, mode, amount, and delay time). Initial vital data including heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), respiratory rate (RR), and temperature, as well as level of consciousness using GCS, and central venous pressure (CVP) were recorded.
Additionally, the results of laboratory investigations were documented on admission and before receiving any treatment. Substantially, arterial blood gas analysis (ABG), including pH, partial arterial oxygen pressure (PaO2), partial arterial carbon dioxide pressure (PaCO2), and serum bicarbonate (HCO3), were retrieved. Additionally, the first serum electrolytes (sodium and potassium levels), liver enzymes (aspartate aminotransferase and alanine aminotransferase), kidney function tests (urea and creatinine), blood indices including complete blood count, and random blood sugar were recorded. Furthermore, electrocardiography was done for each patient.
Scoring calculations
On patient admission, in addition to PSS, two studied scores were calculated for every admitted patient as follows:
Poison severity score (PSS)
The Poisoning Severity Score is the most popular conventional scoring for grading the degrees of poisoning severity depending on the most severe patient’s symptoms or signs as follows: grade 0; asymptomatic, grade 1; mild transient spontaneous resolving symptoms, grade 2; pronounced or prolonged symptoms, grade 3; severe or life-threatening symptoms, grade 4; death 25 .
PGI score
The PGI score uses three characteristics of three systems: metabolic status, blood pressure, and consciousness level, including blood pH < 7.25, GCS score < 13, and SBP < 87 mmHg. Each variable gives one point, and the total score ranges from 0 (best) to 3 (worst). 20 This score can stratify the risk of ALP patients into three categories: zero points (low risk), one point (intermediated risk), and two to three points (high risk). 21
New poisoning mortality score
The new PMS consists of 10 predictors by incorporating demographic factors (age and sex), poisoning-related factors (intent of poisoning, route of exposure, and poison category), initial vital signs (SBP, HR, RR, temperature), and consciousness level assessment using AVPU scale (A-alert, V-verbal, P-painful, and U-unresponsive). The total points of new PMS range from 0 to 137, where 0 is normal, and 137 is the worst. Additionally, the new PMS categorizes the patient’s mortality risk into four groups: very low (0 – 27), low (28 – 40), intermediate (41 – 55), and high risk (≥ 56). 23
Treatment strategy and patient grouping
All patients received the standard treatment according to the TUPCC protocol, including ensuring a patent airway with supporting breathing and circulation. Additionally, hypotensive patients were treated by continuous intravenous fluid infusion and vasopressors (norepinephrine). Although some patients initially have stable hemodynamic status, they have rapidly deteriorated due to ALP-induced cardiotoxicity. 26 Accordingly, upon detecting hypotension through vital sign monitoring, we inserted a central venous catheter for further hemodynamic monitoring and administered intravenous fluids, potentially in combination with vasopressor infusion based on CVP recording, to sustain the SBP over 80 mm Hg as well as to prevent fluid overloading and pulmonary edema development.27–29 Furthermore, metabolic acidosis was corrected according to its degree and treatment of other expected manifestations.
Paraffin oil was used to perform gastric lavage according to recent treatment guidelines. The gastric lavage technique was performed by placing the nasogastric tube from the nose to the stomach after positioning the patients in the left lateral decubitus to reduce the risk of gastric content aspiration. Upon confirming the proper position of the nasogastric tube, the gastric contents were first aspirated for medicolegal preservation and silver nitrate test performance. Subsequently, 50 mL of paraffin oil was administered through the nasogastric tube with frequent aspiration after 3 to 5 min till the aspirate was clear. Finally, another 50 mL of paraffin oil was administered and left in the stomach. 30 Although the usefulness of gastric lavage with paraffin oil in cases of ALP poisoning has been documented in current literature, 31 there are certain situations in which it should not be used. Substantially, paraffin oil’s petroleum-based composition renders it unsuitable for disturbed-conscious patients and those with respiratory disorders to prevent the development of aspiration pneumonitis. Alternatively, coconut oil has been recommended for those patients as it is more safe and tolerable. 32
According to the primary outcome (mortality), patients were categorized into survivors and nonsurvivors.
Sample size
The sample size was calculated based on the mortality outcome (Pannu et al., 2022) 21 according to the methods described by Buderer (1996) 33 and Negida et al. (2019). 34 We calculated the number (N) required for sensitivity and that required for specificity using the following equations: N required for sensitivity = TP + FN/P, and N required for specificity = TN + FP/1-P, where TP, FN, TN, FP, P stand for true positive, false negative, true negative, false positive, and prevalence, respectively. Accordingly, the minimum required number was 140 patients, which increased to 160 to increase the predictive scores’ performance accuracy.
Statistical analysis
Analyses were conducted using the R Statistical language (version 4.4.0; R Core Team, 2024), using the packages pROC (version 1.18.5; Robin X et al., 2011 35 ), gtsummary (version 1.7.2; Sjoberg et al., 2021 36 ), ggplot2 (version 3.5.1; Wickham H, 2016 37 ). The distribution of numerical data was tested using the Shapiro-Wilk test for normality and the Q-Q plots. Normally distributed data were summarized using the mean and standard deviation (SD), and comparisons were performed using the unpaired t-test. Abnormally distributed data were summarized using the median and interquartile range (IQR, expressed as 25th – 75th percentiles), and between-group comparisons were done using the Mann-Whitney test (Wilcox rank sum test). Data were expressed as numbers and percentages for categorical variables and compared using the Cochran-Armitage test for trend and the Chi-squared Test for comparative analysis. The predictive performance of the studied scores was analyzed through receiver operating characteristic (ROC) curves. The area under the ROC curve and its 95% confidence interval (CI) were calculated. The optimal cutoff point was identified using the Youden J index, and the associated sensitivity, specificity, predictive values, and accuracy were computed. Pairwise testing of the AUCs of each two score was done using the DeLong method. A p-value <0.05 was the significance level of the statistical tests.
Results
Comparison of basic characteristics and complications among survivors and nonsurvivors of acute aluminum phosphide-poisoned patients (n = 160).

Regarding vital signs, nonsurvivors had significantly lower mean values of HR, SBP, DBP, and mean arterial pressure (MAP) than survivors (p < 0.001 for each). Likewise, GCS was significantly lower in nonsurvivors than survivors (p = 0.002), where all survivors had normal GCS, and all patients with severe GCS did not survive. Conversely, the mean value of RR was significantly higher in nonsurvivors than in the survivors (26.68 versus 21.17 cycles/minute; p < 0.001). Similarly, in nonsurvivors, CVP was significantly higher than in survivors (24.22 mmHg versus 15.67 mmHg; p < 0.001). Furthermore, there was a significant difference regarding PSS grading (p < 0.001), where severe grading was observed in 92.9% of the nonsurvivors versus 29.2% of the survivors. Accordingly, the nonsurvivors had a significantly higher rate of admission to the ICU and mechanical ventilation than the survivors (p < 0.001). Cardiac arrest was significantly more prevalent in nonsurvivors than in survivors (p < 0.001), as shown in Table 1.
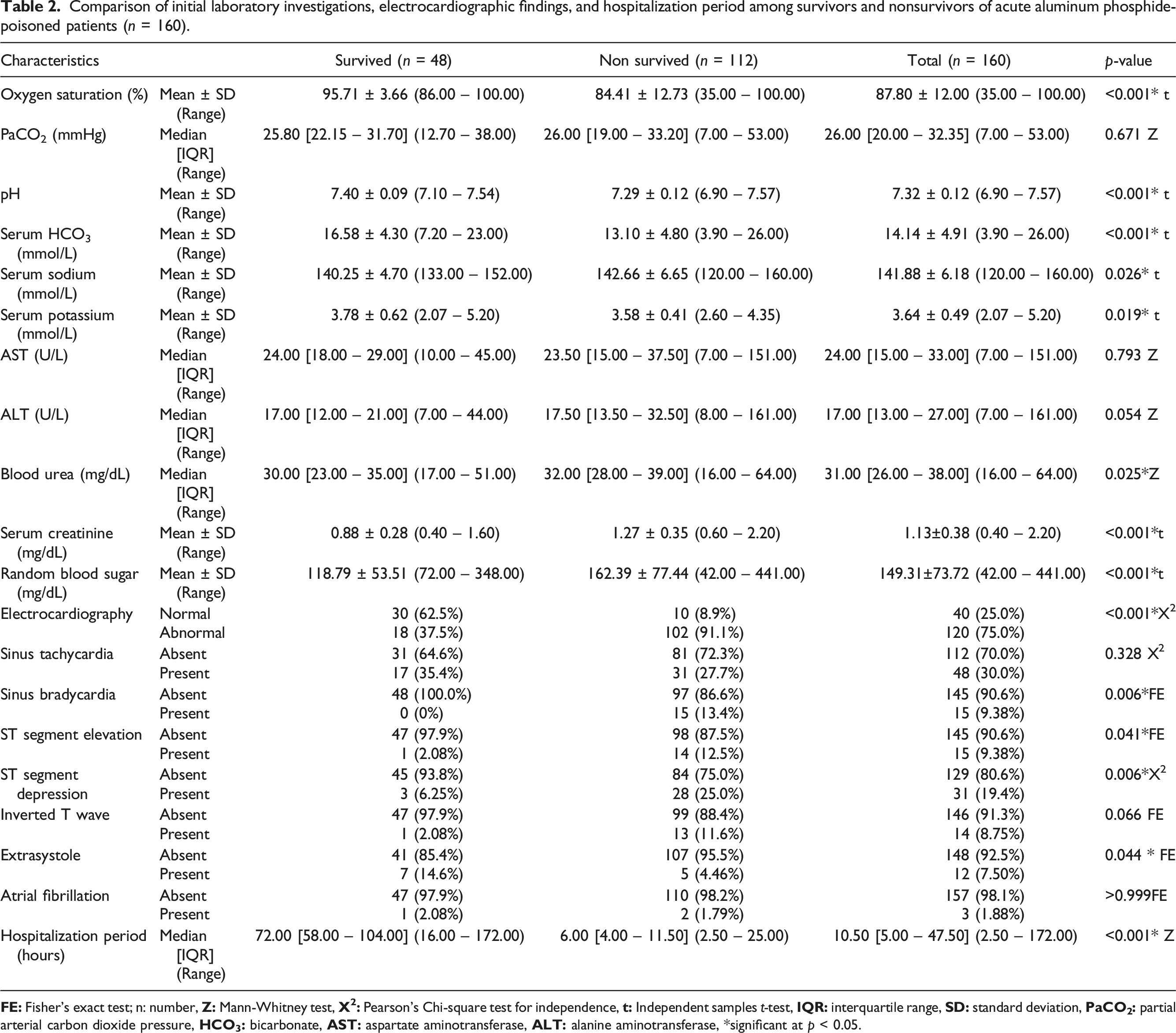
Comparison of initial laboratory investigations, electrocardiographic findings, and hospitalization period among survivors and nonsurvivors of acute aluminum phosphide-poisoned patients (n = 160).
Comparison of PGI score and new poisoning mortality score between survivors and nonsurvivors of acute aluminum phosphide-poisoned patients (n = 160).

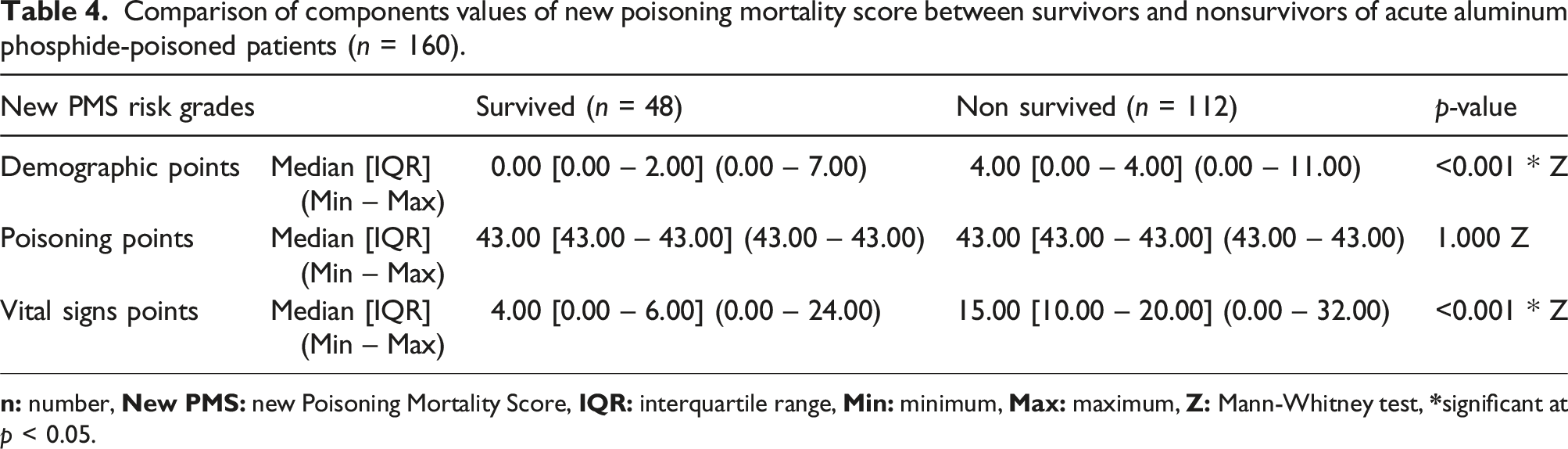
Comparison of components values of new poisoning mortality score between survivors and nonsurvivors of acute aluminum phosphide-poisoned patients (n = 160).
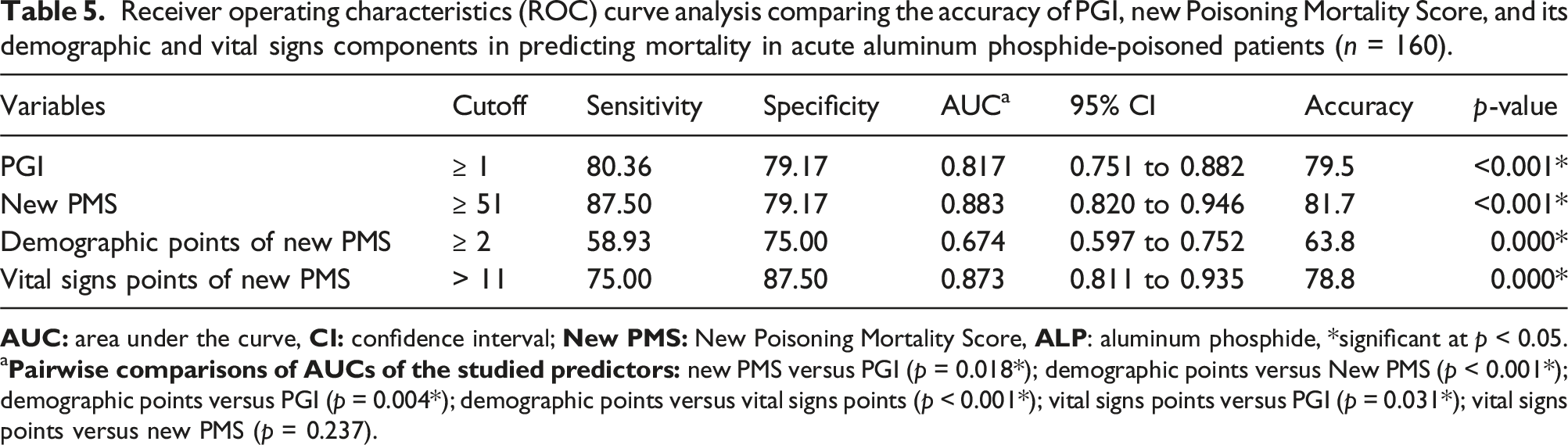
Receiver operating characteristics (ROC) curve analysis comparing the accuracy of PGI, new Poisoning Mortality Score, and its demographic and vital signs components in predicting mortality in acute aluminum phosphide-poisoned patients (n = 160).
a

Receiver operating characteristics (ROC) curve of PGI score for predicting mortality of acute aluminum phosphide poisoned patients.

Receiver operating characteristics (ROC) curve of new Poisoning Mortality Score for predicting mortality of acute aluminum phosphide poisoned patients.
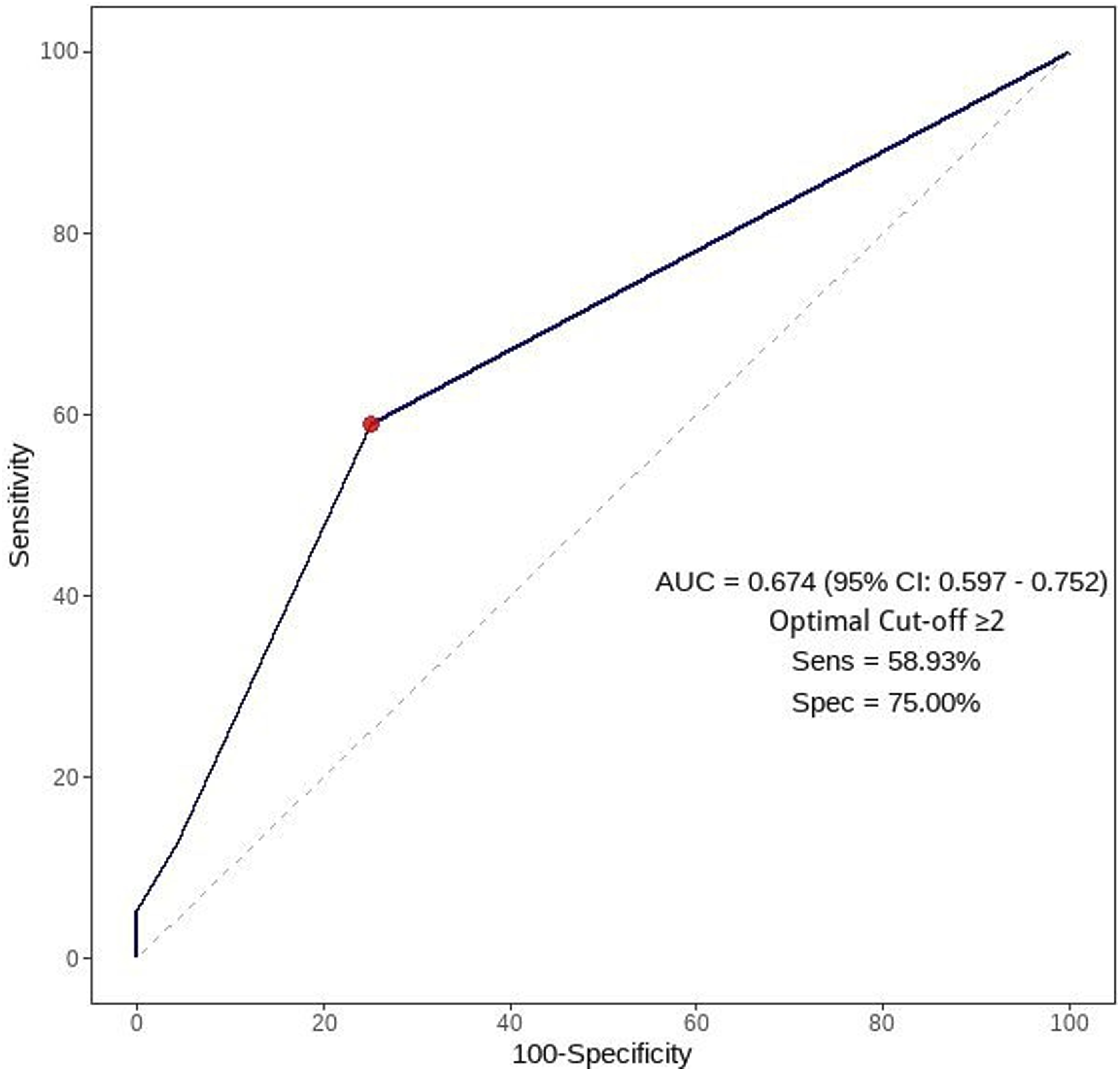
Receiver operating characteristics (ROC) curve of demographic component of new Poisoning Mortality Score for predicting mortality of acute aluminum phosphide poisoned patients.

Receiver operating characteristics (ROC) curve of vital signs component of new Poisoning Mortality Score for predicting mortality of acute aluminum phosphide poisoned patients.
Although the AUC of new PMS was significantly higher than PGI and the new PMS components (vital and demographic), no significant difference in AUCs was observed between the new PMS and its vital component (p = 0.237) by pairwise comparison analysis of AUCs of the studied predictors as demonstrated in Table 5.
Discussion
To date, ALP has become a prevalent suicidal agent with an alarming increase in related morbidities and mortalities, especially in developing countries. 38 The great challenge is the wide availability of highly fatal poison without restricted purchasing rules. 39 Additionally, the rapidly deteriorated clinical course with the lack of specific antidotes augmented this issue’s complexity despite performing many trials of adjuvant therapies with standardized supportive strategies.32,40,41
As a result, our study recorded a high mortality rate (70%) out of enrolled ALP-poisoned patients. Consistent with our findings, Khalaf et al. (2023), 13 Ghonem et al. (2024), 38 and Dorooshi et al. (2022) 42 reported nearly ALP fatality incidences (72%, 72.3%, and 77.4 %, respectively). However, lower mortality rates (39%) were reported by Anbalagan et al. (2023). 43 They justify this decrease by consuming expired tablets, variation of the exposure formulation (tablet, pellet, and powder), multiple vomiting episodes postexposure, and rapid treatment initiation.
Accordingly, determining the high predictive power score containing readily available parameters is crucial to prioritizing risky patients for intensive management while allocating limited hospital resources. Hence, our study goal was to compare new PMS and PGI scores, focusing on their clinical applicability in predicting mortality to provide an immediate, accurate tool for decision-making in high-risk ALP-poisoned patients.
Our study recorded a significant increase in medians of new PMS and PGI scores among nonsurvivors than survivors (59 versus 47.50 and 1 versus 0, respectively; p < 0.001 for each). In accordance, Saeed and Elmorsy, 2024 44 and Lee et al. (2024) 45 demonstrated a significant increase of means and medians new PMS in nonsurvivors compared to survivors (59.32 ± 13.86 versus 47.29 ± 12.93; p < 0.001 and 66 [57-77] versus 31 [23-40]; p < 0.001, respectively) in acutely poisoned patients.
Substantially, Moorthy et al. (2023) 46 demonstrated a significant increase of new PMS in nonsurvivors compared to survivors among patients with acute organophosphate poisoning (70.12 ± 12 versus 51 ± 7, respectively; p < 0.001). Likewise, Pannu et al. (2022) 21 revealed that nonsurvivors had higher PGI medians than survivors (3 [2-3] versus 0 [0-0]; p < 0.001) in acute ALP-poisoned patients.
Accordingly, the higher severity grades of PSS, new PMS, and PGI scores on admission in the nonsurvivors could explain the increased median values of the studied scores in our mortality group. Noticeably, 92.9%, 67.9%, and 80% of nonsurvivors had severe PSS, high new PMS, and intermediate to high PGI score grading. Likewise, Pannu et al. (2022) 21 and Sakr et al. (2023) 22 recorded 82.60% and 95.38% of nonsurvivors had high PGI scores in patients with acute ALP poisoning, respectively. Although no previous studies assessed new PMS in acute ALP poisoning, Saeed and Elmorsy, 2024 44 recorded that 57.1% of nonsurvivors had high new PMS grading in acutely poisoned patients. Furthermore, Lee et al. (2024) 45 demonstrated a good calibration curve for new PMS for predicting in-hospital mortality, especially in high-risk groups of acutely poisoned patients. Accordingly, the risk for mortality is increased in acutely poisoned patients with intermediate to high scores of the new PMS. 46
Additionally, the new PMS demographic and vital components were significantly higher among nonsurvivors than survivors in the current study (4, 15 versus 0 and 4, respectively; p < 0.001 for each). This significant association was attributed to substantial differences in age, gender, vital signs, and consciousness level between the two groups. However, there was no significant difference between the two groups regarding poisoning variables, as all patients were exposed intentionally to the same substance category (ALP) by oral route.
Our study recorded that the mean age of nonsurvivors was significantly higher than survivors (25.73 versus 20.52 years; p = 0.001), with male predominance (51.8% versus 48.2%; p < 0.001). Accordingly, Khodabandeh et al. (2014) 47 observed that the majority of the nonsurvivors of ALP poisoning (54.1%) were males aged 15–43 years. Consequently, a systematic review and meta-analysis by Bagherian et al. (2021) 48 in Iran concluded that the majority of nonsurvivors were males (62.3%) with significantly higher mean age than survivors (p < 0.01).
Although the gender proportion may vary among studies, numerous studies recorded a young age group with suicidal intentions, highlighting the importance of assessing the psychiatric status of those patients.49,50 Substantially, El-Farouny et al. (2020) 51 recorded that the predominant instances of intentional self-poisoning (38.4%) were caused mainly by pesticides and occurred in the youngest age group (10–20 years), with personality traits being a significant risk factor for suicidal attempts. Furthermore, the 5-year cross-sectional study by Majidi et al., (2021) 14 demonstrated that the highest incidence of suicidal ALP poisoning (67.9%) belonged to the age group (14–30) with a mean age of 28.6 years. However, Wang et al. (2020) 52 revealed that middle-aged 40-year-olds had the highest frequency of suicidal pesticide ingestion (3.43%). Furthermore, Song et al. (2020) 53 demonstrated increases in the percentage of intentional pesticide poisoning with age; hence, the mortality rate in the elderly patients was higher than in the younger patients.
The contradictory age-related findings among studies may be attributed to various stresses affecting distinct age groups. Noticeably, the youngest age group may subjected to psychic troubles in the family, as well as emotional and educational issues. 54 Alternatively, the higher age group may experience marital concerns, occupation obstacles, and financial difficulties. 51 Another justification may be related to geographical diversity, as well as variation in sample size and eligibility criteria.
In consistent with previous studies,21,38,55 all hemodynamic parameters (HR, SBP, DBP, and MAP) were significantly decreased in nonsurvivors compared to survivors (p < 0.001). Hypotension, bradycardia, and ECG changes are the most prominent presentations of ALP-induced cardiotoxicity, which is the leading cause of mortality during the first 24 h.32,56 The hypovolemic shock could occur due to dehydration, substantial intravascular fluid loss resulting from increased capillary permeability, and adrenal damage inducing peripheral vasodilatation, as well as direct myocardial tissue damage and life-threatening arrhythmias.38,57,58
However, a significant increase in RR was observed in nonsurvivors (26.68 versus 21.17 cycles/minute; p < 0.001). It may be explained by a compensatory increase in the respiratory rate in response to metabolic acidosis in the nonsurvivor group to wash the excess CO2 by exhalation. Correspondingly, the blood pH and serum HCO3 were significantly decreased in the fatal group compared to survivors. Consistent with these findings, Pannu et al. (2022) 21 observed the concomitant increase in RR and decrease of pH and HCO3 in the nonsurvivor group compared to survivors in acute ALP-poisoned patients (25.6 ± 3.6 versus 20.6 ± 5.2, 7.04 ± 0.23 versus 7.34 ± 0.10, and 9.7 ± 5.8 versus 16.7 ± 5.0, respectively, with p < 0.001 for each).
In agreement with prior studies,58,59 all survivors had normal GCS. However, all patients with severe GCS belonged to the non-survivors group (p < 0.001). Consciousness level impairment in nonsurvivors may be explained by phosphine gas-inducing cerebral hypoxia and hypoperfusion resulting from refractory shock.38,58
The current study demonstrated that new PMS had reasonable discriminatory power for mortality in acute ALP-poisoned patients (AUC = 0.883). Approximately similar, Moorthy et al. (2022) 46 recorded a valid discrimination of new PMS for mortality prediction in acute OP-poisoned patients (AUC = 0.917). However, Han et al. (2021) 23 recorded higher AUC of new PMS in derivation and validation groups for predicting mortality in acutely poisoned patients (AUC = 0.941 and 0.946, respectively). On the contrary, Saeed and Elmorsy, 2024 44 reported a lower predictive value of new PMS (AUC = 0.735) in patients with acute intoxication.
The higher predictive power in Han et al. (2021) 23 study is attributed to significant differences in all new PMS components (demographic, poisoning, and vital signs) among survivors and nonsurvivors (p < 0.001). However, Saeed and Elmorsy, 2024 44 did not observe significance in all new PMS components between survivors and nonsurvivors except for poisoning categories and heart rate parameters (p = 0.046 and 0.025, respectively).
Regarding the PGI score, our study demonstrated good discrimination in predicting mortality of ALP-poisoned patients (AUC = 0.817) at cutoff value ≥1 with a sensitivity of 80.36%, specificity of 79.17%, and overall accuracy of 79.5%. However, Sakr et al. (2023) 22 demonstrated that the PGI score had excellent power of mortality discrimination (AUC = 0.951) at cutoff point ≥ 2, with a sensitivity of 95.4%, specificity of 87.5%, and overall accuracy of 94.5% in ALP-poisoned patients. The increased number of patients with PGI grade 3 (high risk) in Sakr et al. (2023) 22 study could explain the higher predictive power of PGI in their research than ours. Likewise, Pannu et al. (2020) 20 recorded that PGI > 2 had the best sensitivity, negative predictive value, and diagnostic accuracy (93.9%, 93.5%, and 84.8%, respectively) for anticipating mortality in acute ALP-poisoned patients.
In our study, the new PMS significantly outperformed the PGI score in all ROC metrics (p = 0.018). The superiority of the new PMS over the PGI score could be attributed to incorporating demographic and poisoning variables with the vital assessment with a grading scale to stratify patients into four risk groups (very low, low, intermediate, and high risk). Additionally, it provides subgrouping analysis for different poison categories to reinforce its predictive performance in coingestions.
In contrast, the PGI relies on three particular variables with definite cutoff levels (blood pH < 7.25, GCS score < 13, and SBP < 87 mmHg). Accordingly, many previous studies have recorded mortality risk at higher blood pH and GCS than the abovementioned cutoff levels. Substantially, Wahdan and Khalifa, 2020, 58 Abdelkader et al. (2023), 60 and Sagah & Elhawary, 2022 15 revealed that pH ≤ 7.28, 7.30, and ≤ 7.33 had good predictive ability for mortality discrimination (AUC = 0.881, 0.752 and 0.938, respectively) in patients with ALP poisoning. Furthermore, El-Sarnagawy (2017) 55 and Majidi et al. (2021) 14 observed that 25.5% and 35% of nonsurvivors had normal GCS. Alternatively, Erfantalab et al. (2017) 61 did not find a significant difference in GCS between survived and non-survived ALP-poisoned patients, as consciousness may be maintained till late poisoning stages depending on hypotension and hypoxia severity. 62 Therefore, some ALP-poisoned patients with mortality risk may be unidentified by PGI score.
Unanticipatedly, the current study recorded comparable predictive power between the new PMS and its vital signs component (AUC = 0.883 versus 0.873; p = 0.237) by pairwise comparison analysis. This finding may be explained by the equality of poisoning-related factors between groups and the poor discriminatory power of demographic variables in forecasting ALP mortality (AUC = 0.674).
Strengths and limitations
This study’s strength is demonstrating the effectiveness of the new PMS and its vital signs-related variables over PGI score in predicting ALP mortality. Accordingly, the hemodynamic and AVPU variables can be calculated instead of the total score, especially in intentional acute ALP-poisoned cases by oral route. However, the drawback of this study is the inability to evaluate other poisoning circumstances, such as non-intentional or inhalational exposure. This limitation impeded assessing the association between poisoning-related factors and in-hospital mortality. Additionally, owing to the unavailability of gas chromatography in our institution, the silver nitrate test is a promising preliminary diagnostic tool for ALP poisoning, demonstrating 100% sensitivity to gastric content. However, false positive results may occur in the presence of sulfide derivatives due to the production of silver sulfide. To exclude the presence of sulphides, filter-soaked paper with one drop of 10% lead (II) acetate is used. If it is further blackened, sulfides may be present, leaving the results equivocal. As a result, the confirmatory ammonium molybdate test was carried out by dropping ammonium molybdate suspension over the dark filter paper, causing bluish discoloration in the presence of phosphine gas.63,64 Furthermore, this study is a single center with a relatively small sample size, limiting the generalization of our findings. Therefore, further multicenter studies should be conducted to assess the applicability and reliability of new PMS and its vital component in various ALP poisoning circumstances.
Conclusions
Although the new PMS and PGI scores performed well in predicting ALP mortality, the new PMS outperformed the PGI score in various metrics. The new PMS is a simple, valid score based on objective predictors reflecting the demographic and poisoning characteristics, as well as hemodynamic and mental status. However, the vital signs component of the new PMS provided a comparable result to the whole score. Therefore, determining the initial vital signs related to new PMS factors in intentional ALP poisoning offers rapid triaging for high-risk patients requiring intensive care management and excludes others not in need. Additionally, it is easily applicable as it depends on routine clinical parameters that all emergency clinicians can apply.
Footnotes
Acknowledgments
All authors express gratitude to the staff members of Tanat University Poison Control Center (TUPCC) for their support and help in this study.
Author contributions
Ghada N. El-Sarnagawy and Mona M. Ghonem formulated the study’s conception and design. Material preparation and data collection were accomplished by Ghada N. El-Sarnagawy, Amira A. Abdelnoor, and Mona M. Ghonem. The first draft was written by Ghada N. El-Sarnagawy, Amira A. Abdelnoor, and Mona M. Ghonem. All authors approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
