Abstract
Objective
To evaluate the prevalence of headache at onset and its association with 1-month mortality in stroke patients.
Methods
All patients with stroke in Dijon, France (2006–2011), were prospectively identified using a population-based registry. Cox regression models were used to evaluate the association between headache and 30-day all-cause mortality.
Results
Among 1411 stroke patients, data about headache were obtained for 1391 (98.6%) of whom 1185 had an ischemic stroke (IS), 201 had an intracerebral hemorrhage (ICH) and five had a stroke of undetermined etiology. Headache was found in 253 (18.2%) patients and was more frequent in those with ICH than in those with IS (46.3% vs 13.5%, p < 0.001). Overall 30-day mortality was 11.7%, and was greater for patients with than those without headache (17.0% vs 10.5%, unadjusted HR 1.70; 95% CI 1.20–2.41, p = 0.003). In multivariable analysis, an association between headache and 30-day mortality was observed (HR 1.51; 95% CI 1.02–2.25, p = 0.042). In stratified analyses, headache was associated with 30-day mortality in ICH (HR 2.09; 95% CI 1.18–3.71, p = 0.011) but not in IS (HR 1.01; 95% CI 0.53–1.92, p = 0.97).
Conclusion
Headache at stroke onset is associated with a higher risk of early mortality in patients with ICH.
Introduction
Although headache is considered a classical clinical feature of stroke, the exact prevalence of this symptom is controversial. Hence, the reported frequency of stroke-related headache ranges from 7% to 65% according to various studies (1–8). Several factors, such as the hospital-based setting of studies, which resulted in large differences in recruited patients, and the absence of differentiation between stroke subtypes probably contributed to the divergent findings. In addition, the clinical relevance of headache in terms of prognosis in stroke patients has not been fully investigated (5–8). Therefore, this population-based study aimed to evaluate the prevalence of headache at onset and its association with 1-month mortality in stroke patients.
Methods
Study population and case-ascertainment procedures
The study population included all patients with stroke that occurred between 1 January 2006 and 31 December 2011, who were prospectively identified among residents of the city of Dijon, France (2007 census: 151,543 inhabitants) from the Dijon Stroke Registry. This population-based registry complies with the criteria recommended for the running of high-quality stroke incidence studies (9,10), and case-collection procedures have been described elsewhere (11,12). Briefly, multiple overlapping sources of information were used to identify fatal and non-fatal stroke in hospitalized and non-hospitalized patients: (a) a review of medical records from the emergency rooms, and all the clinical and radiological departments of Dijon University Hospital, with a diagnosis of stroke made by one of the neurologists of the department of neurology, where the Stroke Registry is located; (b) a review of medical records from the emergency rooms and all of the clinical departments of the three private hospitals of the city and its suburbs, with diagnosis made by neurologists working in these establishments; (c) a review of computerized hospital diagnostic codes of Dijon University Hospital. The International Classification of Diseases, tenth revision (ICD-10) was used. The following codes are initially searched for: I60 (subarachnoid hemorrhage), I61 (intracerebral hemorrhage), I62 (non-traumatic intracranial hemorrhage), I63 (ischemic stroke), I64 (non-determined stroke), G45 (vascular syndromes), G46 (transient ischemic attack) G81 (hemiplegia). Study investigators then consulted the medical records of identified patients to confirm or not the reported diagnosis or to reclassify the patients if a misclassification was noted; (d) a review of computerized hospital diagnostic codes of the private hospitals with the same procedure as described above; (e) collaboration with the general practitioners to identify stroke patients managed at home or in nursing homes, with the diagnosis assessed by public or private neurologists from outpatient clinics; (f) a review of the medical records of patients identified from a computer-generated list of all requests for imaging to the private radiological and Doppler ultrasound centers of the city and its suburbs; (g) and regular checking of death certificates obtained from the local social security bureau that is responsible for the registration of deaths in the community particularly fatal strokes outside hospital. The quality and the completeness of the registry are certified every 4 years by an audit from the National Institute for Health and Medical Research and the National Public Health Institute.
Stroke was defined according to World Health Organization recommendations (13). We distinguished between ischemic stroke (IS), spontaneous intracerebral hemorrhage (ICH) and undermined stroke. For this study, subarachnoid hemorrhage was excluded.
Data collected
The following vascular risk factors were systematically collected (11,12): hypertension (high blood pressure noted in a patient’s medical history or patients on antihypertensive treatment), diabetes mellitus (glucose level ≥7.8 mmol/l reported in the medical record or patients on insulin or oral hypoglycemic agents), hypercholesterolemia (total cholesterol level ≥5.7 mmol/l reported in the medical history or patients treated with lipid-lowering therapy), atrial fibrillation, history of coronary heart disease, heart failure, peripheral artery disease (PAD), alcohol intake (≥3 units a day in men and ≥2 in women), smoking, cancer, and previous stroke or TIA. Treatments prior to the stroke, including antiplatelet agents, anticoagulants, antihypertensive treatments and statins were recorded. We also collected information on pre-stroke residence in a nursing home as a proxy of dependency and thrombolysis for IS patients.
Headache at stroke onset was defined as cephalic pain appearing just before or after other stroke symptoms. We assessed headache in all patients with a routine neurological questionnaire that was completed by the emergency medical dispatchers at the call center for emergencies and/or at admission for patients hospitalized or during clinical consultations for outpatients. Relatives were asked about the history of headache at stroke onset either by the emergency medical dispatchers or at the time of the first examination if the patients were unconscious, confused or aphasic. Stroke severity was quantified by the means of the National Institutes of Health Stroke Scale (NIHSS) score either obtained at the first clinical examination or estimated on the basis of the review of medical records and charts when patients were identified retrospectively, as previously validated in the literature (14). The etiological classification of IS patients was derived from the TOAST classification (15) as follows: large artery IS, cardioembolic IS, lacunar IS, IS from other identified cause, IS from undetermined cause and IS from multiple possible causes. The ICH locations were reported according to the findings of CT scans except for one patient in whom only MRI was performed. For this study, all brain imaging data were blindly reviewed by three investigators (VA, YB, MG) to confirm the location of the ICH. We considered the following locations, as previously reported: lobar, deep infratentorial (11). Intraventricular extension of bleeding was reported. In cases of uncertainty about the ICH location, especially for patients with large ICH, the most probable origin was discussed by the investigators. ICH location was classified as undetermined either when the origin could not be reliably identified, including cases of hemorrhage that overlapped two territories, or unreported data or lost files.
All-cause mortality at 30 days was systematically assessed using death certificates.
Statistical analysis
Proportions and mean values of baseline characteristics were compared between groups using the chi-square test, and the Wilcoxon–Mann–Whitney test when appropriate. Person-days were calculated from the date of onset until death, the last contact date and the end of follow-up at 1 month. Survival curves were obtained using Kaplan–Meier analysis, and the log-rank test was used for comparisons between groups. Cox regression models were used to estimate hazard ratios (HRs) of 1-month mortality and their 95% confidence intervals (CIs). In the first series of multivariable analyses concerning all stroke patients, we introduced into the models headache, age, gender, stroke subtype, and baseline characteristics with p < 0.20 in unadjusted models. Backward selection was done using the likelihood-ratio test to obtain the final model, which included the significant confounders. Stratifications by stroke subtypes (IS and ICH) were then done. In a second series of multivariable analyses concerning IS patients, we generated new models into which we introduced headache, age, gender, IS subtypes and thrombolysis, and baseline characteristics with p < 0.20 in unadjusted models. In a third series of multivariable analyses concerning ICH patients only, we generated new models into which we introduced headache, age, gender, ICH location, intraventricular extension of bleeding, and baseline characteristics with p < 0.20 in unadjusted models. For each analysis, interaction terms were added to the models to test the modifying effect of confounding variables on the association between headache and 1-month all-cause mortality, using the likelihood-ratio test. We used a dummy indicator for smoking status to prevent the deletion of data for 3.8% of the patients with missing values. The corresponding proportion for other variables was less than 1%. A p value <0.05 was considered statistically significant. STATA 10.0 software (StataCorp LP, College Station, TX, USA) was used for statistical analyses.
Ethics
The Dijon Stroke Registry was approved by the Comité National des Registres (French National Committee of Registers) and the InVS (French Institute for Public Health Surveillance). Authorization of the CNIL (‘Commission Nationale de l'Informatique et des Libertés’; National Commission for the Protection of the Privacy of Electronic Data) was obtained.
Results
Baseline characteristics of stroke patients according to headache.

CI, confidence interval; SD, standard deviation; TIA, transient ischemic attack; PAD, peripheral artery disease; NIHSS, National Institutes of Health Stroke Scale; IS, ischemic stroke; ICH, spontaneous intracerebral hemorrhage.
Baseline characteristics of ischemic stroke patients (n = 1185) according to headache.

CI, confidence interval; SD, standard deviation; TIA, transient ischemic attack; PAD, peripheral artery disease; NIHSS, National Institutes of Health Stroke Scale.
Adjusted for age and sex.
Baseline characteristics of ICH patients (n = 201) according to headache.

CI, confidence interval; SD, standard deviation; TIA, transient ischemic attack; PAD, peripheral artery disease; NIHSS, National Institutes of Health Stroke Scale.
Adjusted for age and sex.
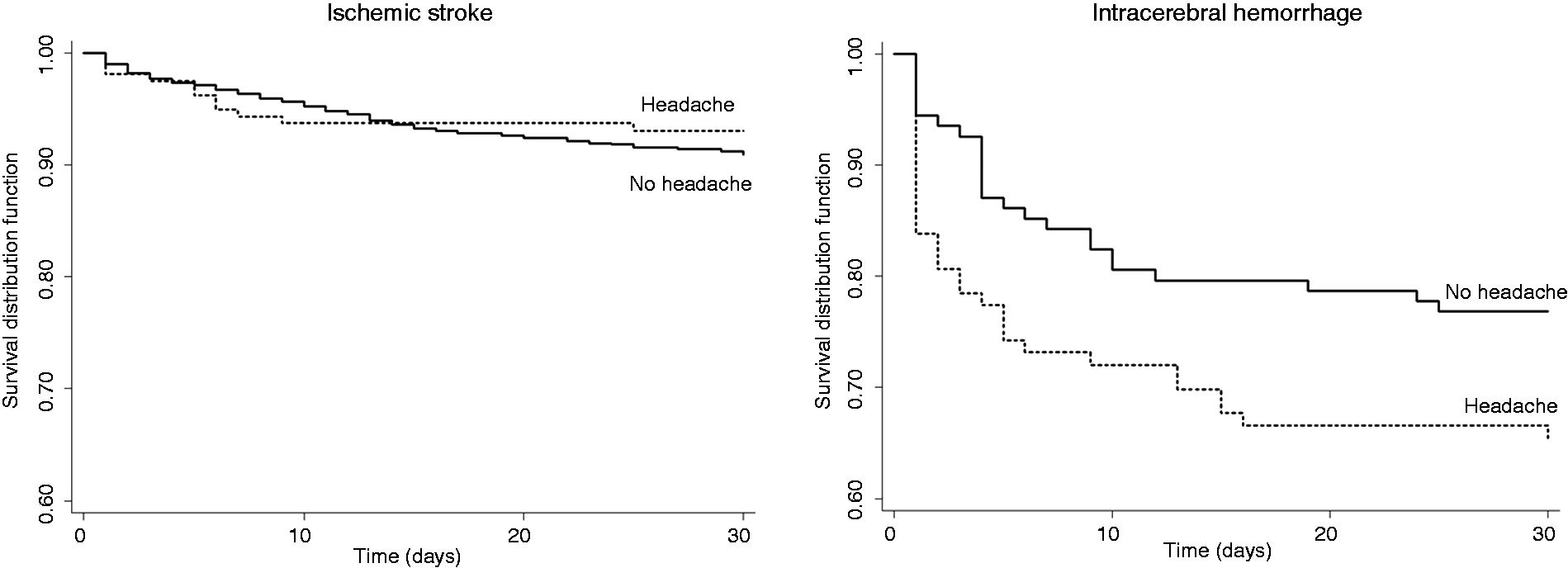
Over the whole study period, overall 30-day mortality was 11.7%. It was greater for patients with than for those without headache (17.0% vs 10.5%, unadjusted HR 1.70; 95% CI 1.20–2.41, p = 0.003) (Figure 1). When considering the stroke subtype, lower survival was observed in patients with headache for ICH (log-rank test = 0.05) but not for IS (log-rank test = 0.41) (Figure 2).
Kaplan–Meier estimates of 1-month survival stratified by headache (Log-rank test = 0.003). Kaplan–Meier estimates of 1-month survival stratified by headache in patients with ischemic stroke (Log-rank test = 0.41) and those with intracerebral hemorrhage (Log-rank test = 0.05).
In multivariable analysis, patients with headache were 51% more likely to die than were those without headache (adjusted HR 1.51; 95% CI 1.02–2.25, p = 0.042) (Figure 3). In stratified analyses by stroke subtype, headache was associated with 30-day mortality in ICH (adjusted HR 2.09; 95% CI 1.18–3.71, p = 0.011) but not in IS (adjusted HR 1.01; 95% CI 0.53–1.92, p = 0.97) (Figure 3).
Association between headache and 1-month all-cause mortality in multivariable models. CI, confidence interval; HR, hazard ratio.
Restricted analyses to IS patients only were performed. Headache at onset was noted in 11.6% (30/238) of patients with large artery IS, 15.6% (50/320) of patients with cardioembolic IS, 10.1% (23/228) of patients with lacunar IS, 29.7% (19/64) of patients with IS from other identified cause, 11.8% (32/272) of patients with IS from undetermined cause and 13.9% (6/43) of patients with IS from multiple possible causes. In the multivariable model including IS subtypes and thrombolysis, there was no association between headache and 30-day mortality (adjusted HR 0.87; 95% CI 0.44–1.72, p = 0.69). No statistical interaction was noted.
Additional analyses were performed on ICH patients in whom data about location were available, which represented 194/201 (96.5%) of overall ICH patients. Among these patients, 97 (50%) had lobar ICH, 75 (38.7%) had deep ICH and 22 (11.3%) had infratentorial ICH. Headache was more common in infratentorial (68.2%) and lobar (51.6%) locations than in deep locations (34.7%). In the multivariable model, after adjustment for confounding variables including age, gender, ICH location, intraventricular extension of bleeding, and baseline characteristics with p < 0.20 in unadjusted models, the association between headache and 30-day mortality was still observed (adjusted HR 2.02; 95% CI 1.08–3.77, p = 0.027). No significant interaction between headache and the variable included in the final model, especially ICH location, was noted. When excluding 17/195 ICH patients (8.7%) for whom information about headache was collected from relatives because of severely impaired consciousness and/or severe aphasia, the association was marginally significant (adjusted HR 2.01; 95% CI 0.99–4.45, p = 0.051).
Discussion
This population-based study demonstrates that headache is particularly frequent in patients with ICH in whom this symptom is independently associated with a higher risk of early death.
The prevalence of headache in our study was consistent with that reported in previous works (1–8), even though direct comparisons are difficult as all of the previous studies were conducted in hospital-based cohorts, thus leading to a potential selection bias of patients. One illustration is the higher mean age of patients at stroke onset than that observed in hospital-based registries. As previously reported (1,3), we found a clearly higher frequency of headache in patients with ICH. Indeed, one-third to one-half of ICH patients presented with a headache. A massive increase in intracranial pressure as well as the direct irritation of the trigeminovascular system at the base of the skull, especially in patients with infratentorial ICH, may account for this high frequency of headache in the clinical presentation of ICH.
The new finding of the present study is the association between headache at onset and 1-month mortality in patients with ICH but not in those with IS, even after considering the etiological subtype. In keeping with this observation, a study conducted on patients admitted for an IS or a transient ischemic attack reported the absence of any association between headache at onset and stroke severity measured at 1 week after the event (7). Moreover, a Danish cohort, which included hospitalized first-ever strokes, demonstrated that there was no correlation between headache and either functional prognosis or mortality (6). In contrast, headache was shown to be associated with early neurological deterioration in patients with cardioembolic stroke (5), whereas another recent study suggested that this symptom predicted a good outcome in IS patients (8). However, to the best of our knowledge, the association between headache and early mortality in ICH has never been assessed in population-based studies. Of note, the prognostic value of headache in our study was independent of several confounding factors including stroke severity, which is the main determinant of post-stroke outcome. In addition, as multivariable models were adjusted for intraventricular extension of bleeding, which is also a great predictor of a poor outcome in ICH patients (16–18), the hypothesis that the association between headache and early mortality simply mirrored the high frequency of headache in patients with intraventricular bleeding can be ruled out. Conversely, a higher volume of ICH or active bleeding at the time of management leading to hemorrhage growth may explain the poorer outcome in patients presenting with headache. However, as neither data about the ICH volume, nor serial brain imaging were available in the present study, we were not able to confirm this hypothesis. Interestingly, in a cohort of 189 patients with supratentorial ICH, Leira et al. demonstrated that headache at onset was associated with clinical and biochemical markers of inflammation (19). Conversely, only a mass effect not ICH volume, nor intraventricular bleeding, nor peripheral hypodensity was associated with this symptom. Contrary to our findings, the authors did not identify headache as an independent predictor of death or poor functional outcome at 3 months, but concluded that this symptom was associated with higher residual cavity volume (19). Finally, a study from a hospital-based stroke registry demonstrated that in patients presenting with a lacunar syndrome, headache was an independent predictor of hemorrhagic rather than ischemic lacunar stroke, suggesting that this symptom would be useful for the clinical distinction between these two entities (20).
Another limitation of our study was the absence of information about the headache type, which prevented us from determining whether the particular clinical features of a headache may be associated with the stroke subtype or outcome. In addition, for unconscious, confused or aphasic patients, information about headache at onset was obtained from their relatives, and this may have engendered an underestimation of this symptom. However, as these patients have more severe stroke and consequently a higher risk of death, this limitation may also have led to an underestimation of the true association observed between headache at onset and early mortality. Moreover, some confounding variables may not have been measured in our study, and could account for unexplained inter-group differences in the baseline characteristics of our patients. Finally, despite the population-based design, the sample size of the ICH subgroup was small.
Despite these limitations, our study has several strengths including its exhaustive, continuous and prospective ascertainment of stroke in a well-defined population without any selection of patients, and with clinically meaningful outcome measures. Thanks to prospective active monitoring, data about headache were missing in less than 2% of the population and follow-up of patients was complete.
To conclude, headache is particularly frequent in ICH and particular attention must be paid to patients presenting with this symptom because of its predictive value on early mortality. Further studies are required to better understand the underlying mechanisms that account for this association.
Clinical implications
In this population-based study, headache at onset was observed in 18.2% of stroke patients. Headache was more frequent in patients with intracerebral hemorrhage (46.3%) than in those with ischemic stroke (13.5%). Headache was independently associated with a higher risk of 30-day mortality in patients with intracerebral hemorrhage but not in those with ischemic stroke
Footnotes
Funding
The Dijon Stroke Registry is supported by the French Institute for Public Health Surveillance (InVS) and Inserm. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
