Abstract
Hypercoagulability in ST-segment elevation myocardial infarction (STEMI) as related to long-term clinical outcome is not clarified. We aimed to investigate whether prothrombin fragment 1+2 (F1+2),
Introduction
The most important underlying mechanism of coronary artery disease is a progressive atherosclerotic process, which, along with intracoronary thrombus formation, may lead to an acute myocardial infarction (AMI).
1
–4
During thrombus formation, both platelet activation and thrombin generation with fibrin formation play important roles.
2,5,6
Thrombin generation in vivo is indicated by the conversion of prothrombin to thrombin with formation of fragments 1 + 2 (F1+2) and the formation of fibrin. Activation of the fibrinolytic system with conversion of plasminogen to plasmin degrades fibrin, resulting in degradation products like
Studies have shown increased levels of prothrombotic markers in patients with myocardial infarction, being associated with myocardial injury and function
8,9
as well as clinical outcome.
10
–13
There is, however, limited knowledge on the association between the acute prothrombotic state and long-term clinical outcome in patients with ST-segment elevation myocardial infarction (STEMI). The aim of the present study was therefore to investigate whether plasma levels of the prothrombotic markers F1+2,
Material and methods
Study Population and Design
In a cross-sectional cohort study performed at Oslo University Hospital, Ullevål, a total of 987 patients with STEMI were included between 2007 and 2011, as previously described. 8,14 When admitted to the hospital, they were treated with primary percutaneous coronary intervention (PCI) according to the standard operating procedures. Patients below the age of 18 years and patients unwilling or unable to give written informed consent were not included, and patients on warfarin treatment were excluded from the present investigation. Of the patients included, 43% presented with an anterior infarction and significant stenosis was observed in 1, 2, and 3 vessels in 54%, 29%, and 17%, respectively.
Blood samples were collected between 8 and 10
Laboratory Methods
Citrated blood (0.129 M trisodium citrate in dilution 1:10) was centrifuged within 30 minutes at 2500g at 4°C and plasma kept frozen at −80°C until analyzed. Both F1+2 and
Electrochemiluminescence technology for quantitative measurement was used for repeated measures of TnT (third-generation cTroponinT, Elecsys 2010; Roche, Mannheim, Germany), and NT-proBNP was measured by Elecsys proBNP sandwich immunoassay on Elecsys 2010 (Roche Diagnostics, Indianapolis, Indiana), with interassay CV of 7% for both. Left ventricular ejection fraction (LVEF) was measured by echocardiography within 3 months after the AMI (n = 767).
Clinical Outcome
The primary outcome was a composite of all-cause mortality, reinfarction, stroke, unscheduled revascularization, or rehospitalization for heart failure, whichever occurred first. Secondary outcome was total mortality. All patients were followed from date of inclusion until December 2013, giving a median follow-up time of 4.6 years. End points were recorded by telephone call and cross-checked with hospital records. The hospital records were evaluated and adjudicated by an end point committee for each end point, before analyzing the results. Mortality end points were obtained from the Cause of Death Registry, administered by the Norwegian Institute of Public Health.
Statistics
Continuous variables are presented as median values with 25, 75 percentiles and categorical variables as number or proportions. As most of the variables were skewed, nonparametric methods were used. Differences between groups were tested by Mann-Whitney U test for continuous variables and by Pearson χ2 test for categorical variables. Multivariate analyses were performed by logistic regression, adjusting for relevant covariates as appears from Table 1 and variables associated with the coagulation markers, as well as troponin T, previously reported to be associated with the coagulation markers.
8
The relatively low number of deaths restricted the number of variables included. Receiver operating characteristic (ROC) curve analysis with the corresponding area under the curve (AUC) with 95% confidence interval (CI) was performed to determine the overall performance of
Characteristics of the Total Study Population.a
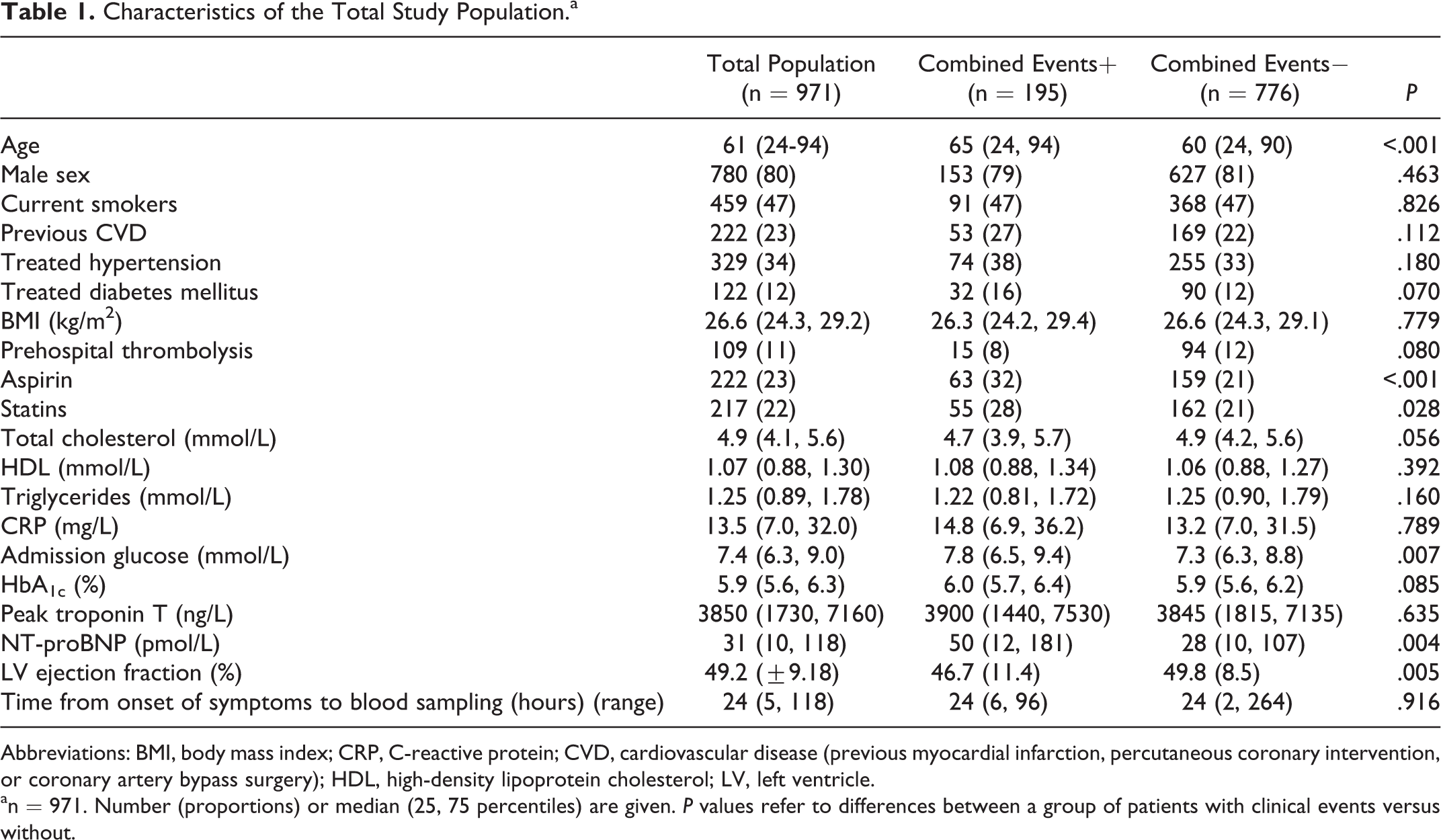
Abbreviations: BMI, body mass index; CRP, C-reactive protein; CVD, cardiovascular disease (previous myocardial infarction, percutaneous coronary intervention, or coronary artery bypass surgery); HDL, high-density lipoprotein cholesterol; LV, left ventricle.
an = 971. Number (proportions) or median (25, 75 percentiles) are given. P values refer to differences between a group of patients with clinical events versus without.
Results
A total of 987 patients were included and clinical end points were obtained from 971 (98.4%) patients. Sixteen patients were not reached by telephone or mail. The number of combined end points was 195, consisting of 62 deaths, 61 reinfarctions, 6 strokes, 52 unscheduled revascularizations, and 14 hospitalizations for heart failure. The number of total mortality was 79, of which 17 died after first having experienced a nonfatal event.
Table 1 shows the baseline characteristics of the study population (n = 971) and according to groupings of with or without combined end point. Patients with combined end points were older, they were more frequent users of aspirin and statins at hospital admission, and they had higher levels of admission glucose and NT-proBNP and lower LVEF. In the Supplemental Table a, results on groupings according to total mortality are shown. We have previously reported on significant associations between the measured biomarkers and levels of NT-proBNP and troponin T. 8
Prothrombotic Markers and Clinical Outcome
The prothrombotic markers as related to the clinical end points are shown in Table 2. The levels of F1+2 and
Levels of F1+2,
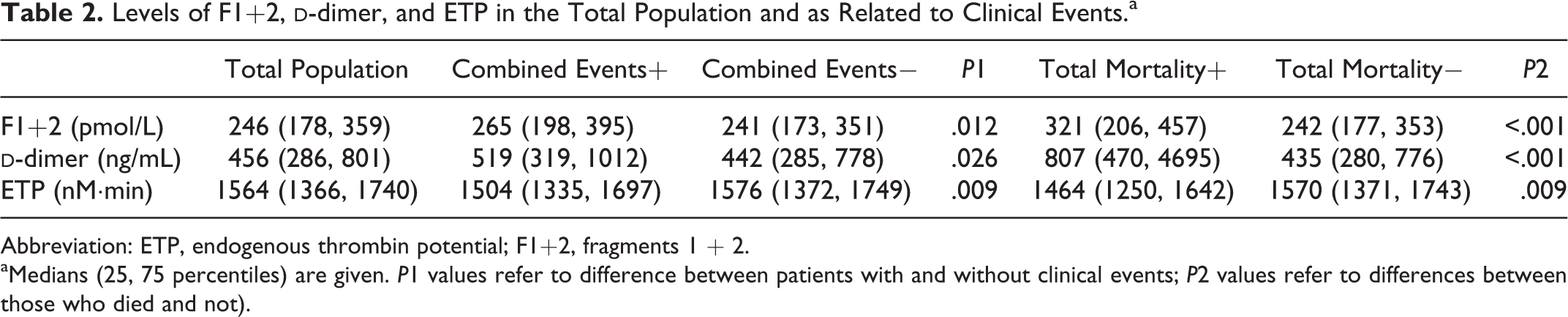
Abbreviation: ETP, endogenous thrombin potential; F1+2, fragments 1 + 2.
aMedians (25, 75 percentiles) are given. P1 values refer to difference between patients with and without clinical events; P2 values refer to differences between those who died and not).
When dividing the levels of F1+2,
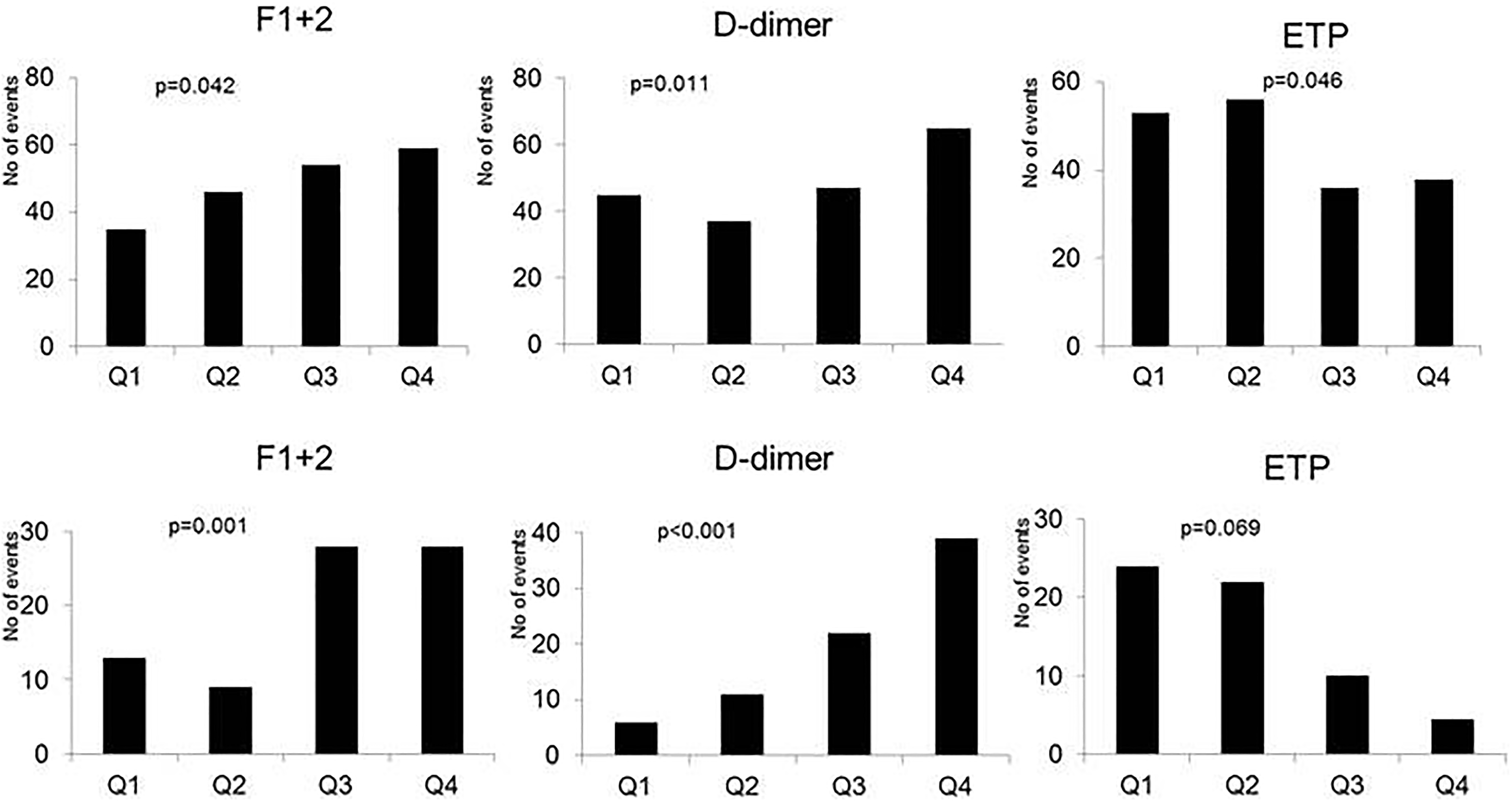
Levels of fragments 1 + 2 (F1+2),
Crude and Multivariate Associations Between the Markers of Hypercoagulability and Clinical Events (A) or Total Mortality (B).
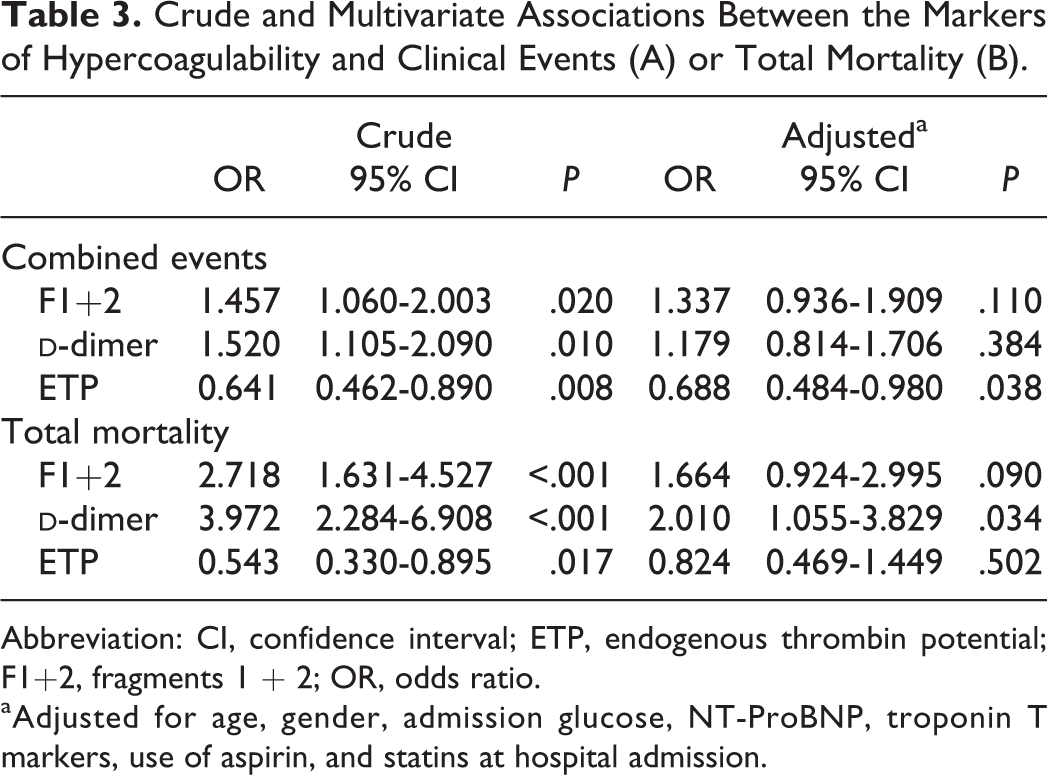
Abbreviation: CI, confidence interval; ETP, endogenous thrombin potential; F1+2, fragments 1 + 2; OR, odds ratio.
aAdjusted for age, gender, admission glucose, NT-ProBNP, troponin T markers, use of aspirin, and statins at hospital admission.
The ROC curve for
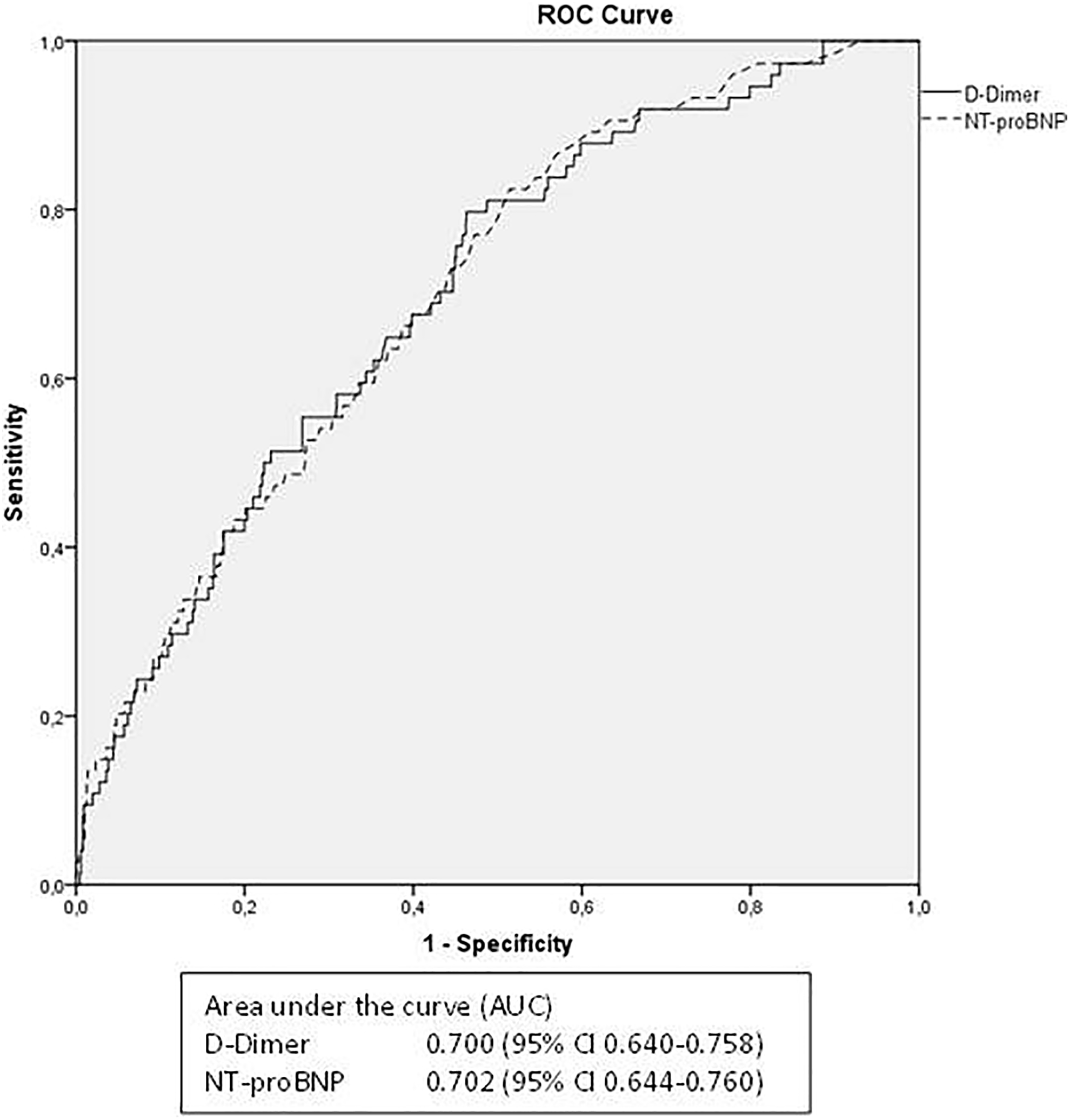
Receiver operating characteristic curves for
Discussion
The main findings in this study on patients with STEMI were that biomarkers of hypercoagulability, that is, F1+2,
The relationship between
The mechanism behind the association between
As for F1+2 which more directly reflects thrombin generation in vivo, there are limited data with respect to clinical events. In our study, F1+2 was strongly associated with both the primary end point and total mortality; however, after adjusting for covariates, of which age seemed to be the most important (data not shown), the significance was lost. In a small study of 68 patients, F1+2 measured 24 hours after an index STEMI, similar to ours, was shown to predict clinical outcome after 6 weeks. 19
In our study, ex vivo thrombin generation, assessed by ETP, was shown to be inversely associated with the primary end point, also after adjustments for covariates. We have previously shown an inverse relation between in vivo and ex vivo thrombin generation in the present population,
8
that is, as also reflected in the present findings. In accordance with our results, the large LURIC study on patients with AMI showed ETP to be inversely associated with cardiovascular events after a median of 10-year follow-up.
20
In that study, however, blood sampling was performed between 1 day and 4 weeks after AMI. Also in line with our result, Smid et al showed low ETP levels alone to be insignificantly associated with clinical end points; however, in combination with high
Nevertheless, the hypercoagulable state shown to be present in the acute phase of STEMI might indicate a role for long-term anticoagulation as shown in earlier studies with warfarin
24,25
and the recent study with rivaroxaban in addition to aspirin.
26
However, confirmatory studies are needed before any cutoff value of
Limitations
Single blood sampling prevented us from studying the initial time course of the measured markers. We do not know whether we have captured the real peak values of the variables or transient changes due to the variability in the time frame from onset of symptoms to blood sampling. However, the results did not change when time from symptom onset was included into the multivariate models. Our cohort of patients with STEMI was a low-risk population with few complications, and this fact may have influenced the results.
All patients in our study received heparin during the PCI procedure approximately 16 to 20 hours before blood sampling. Although any influence of heparin seems unlikely, we cannot completely exclude that small amounts of heparin were remaining.
Conclusion
The hypercoagulable state in the acute phase of a STEMI seems to be of significant importance for future clinical outcome, especially for mortality. Thus, anticoagulant treatment after AMI might still be discussed.
Supplemental Material
Supplemental Material, Revised_Supplementary_File_FINAL - Markers of Thrombin Generation Are Associated With Long-Term Clinical Outcome in Patients With ST-Segment Elevation Myocardial Infarction
Supplemental Material, Revised_Supplementary_File_FINAL for Markers of Thrombin Generation Are Associated With Long-Term Clinical Outcome in Patients With ST-Segment Elevation Myocardial Infarction by Charlotte Holst Hansen, Vibeke Ritschel, Geir Øystein Andersen, Sigrun Halvorsen, Jan Eritsland, Harald Arnesen, and Ingebjørg Seljeflot in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
The results were presented as a moderated poster at the ESC, August 2016, Roma, Italy. The study was a part of the Biobanking in Myocardial Infarction (BAMI) project at Oslo University Hospital, Ullevål, which is led by a Steering Committee including A. Mangschau, R. Bjørnerheim, and the following authors: Arnesen (Chair), Eritsland, Halvorsen, Andersen, and Seljeflot. Charlotte Holst Hansen participated in the coordination of the study, contributed to acquisition of data, performed analyses and interpretation of the data, and drafted the manuscript. V. Ritschel contributed to acquisition of data. Harald Arnesen participated in the design of the study and drafting the manuscript. Sigrun Halvorsen, Geir Øystein Andersen, and Jan Eritsland participated in the design of the study and reviewed the manuscript. Ingebjørg Seljeflot participated in the design of the study, statistical analyses, and drafting and reviewing the manuscript. All co-authors approved the final manuscript.
Acknowledgments
The authors thank the study nurses and the staff at the Coronary Intensive Care Unit and Center for Clinical Heart Research for excellent assistance and medical technologist Beate Vestad for laboratory analyses.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Stein Erik Hagen Foundation for Clinical Heart Research, Oslo, Norway.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
