Abstract
Objective
Precancerous lesions of gastric cancer (PLGC) are key pathological stages in the transformation of gastric “inflammation-cancer”, and timely and effective intervention at this stage is of great importance in the prevention and treatment of gastric cancer. Zhiwei Fuwei Pills (ZWFW), as a traditional Chinese medicine formulation, has been proven to have good clinical efficacy in the treatment of PLGC, but its specific mechanism of action has not been fully explained. Thus, this study validated the efficacy and explored the potential mechanisms of ZWFW in treating PLGC by integrating network pharmacology analyses and experimental verification.
Methods
The TCMSP database was used to obtain the active ingredients of ZWFW and their corresponding targets, and the GeneCards database was used to retrieve PLGC-related targets. The intersecting targets between ZWFW and PLGC were obtained through mapping, and protein-protein interaction (PPI) networks and “drug-active ingredient-target” networks were constructed by using Cytoscape software. The DAVID database was used for GO functional enrichment analysis and KEGG pathway enrichment analysis. AutoDockTools software was used for molecular docking of key active ingredients and key targets. In order to verify the analysis results of network pharmacology, TEM and H&E were used to observe the effects of different dosage groups of ZWFW on gastric mucosal microvasculature in PLGC rats. Subsequently, the ELISA, IF, IHC, RT-PCR and western blot were used to detected the expression levels of relevant targets in the tissues, so as to verify the potential mechanism of ZWFW in intervening PLGC.
Results
After the screening, 258 effective active ingredients and 325 targets were obtained, and 1294 disease-related targets were determined, resulting in 139 intersection targets through mapping. The KEGG enrichment results showed that PI3K/Akt and HIF-1 signaling pathway might play important roles in the treatment mechanism of PLGC. The molecular docking results showed that active ingredients of ZWFW all had a strong affinity and stable structure with key targets, including AKT1 and VEGF. In vivo experiments confirmed that ZWFW could improve gastric mucosal microvascular abnormalities in PLGC, effectively intervene in gastric mucosal pathological grading. Meanwhile, compared with the model group, this formulation could reduce the expression levels of PI3K, Akt, mTOR, HIF-1α, and VEGF in gastric mucosa, showing a dose-effect relationship.
Conclusion
ZWFW can intervene in the neovascularization and pathological evolution of PLGC, and this mechanism of action may be achieved by inhibiting abnormal activation of the PI3K/Akt/mTOR/HIF-1α/VEGF signaling pathway.
Keywords
Introduction
Gastric cancer is a malignant tumor that originates from the epithelial cells of the gastric mucosa. It has a high incidence rate, low overall survival rate, and poor treatment prognosis. It has become the third leading cause of cancer-related deaths, causing serious impacts and threats to human life and health. 1 According to the latest data from the International Agency for Research on Cancer, there were more than one million new cases of gastric cancer worldwide in 2020, with nearly 769,000 deaths. However, China accounted for about 44.0% of the global new cases and 48.6% of the deaths. 2 The Correa cascade reaction represents the evolution of gastric cancer as follows: normal gastric mucosa → chronic gastritis → chronic atrophic gastritis → intestinal metaplasia → dysplasia → intestinal-type gastric cancer, which has been universally recognized by domestic and foreign scholars. 3 The World Health Organization defines chronic atrophic gastritis, intestinal metaplasia, and dysplasia as precancerous lesions of gastric cancer (PLGC), considering them important stages and independent high-risk factors for the development of gastric cancer. 4 Therefore, timely and effective intervention in PLGC is an important measure to reduce the incidence of gastric cancer and a key focus of current research on secondary prevention of gastric cancer.
In the 1970s, Folkman J discovered angiogenesis in the early stages of tumors, a finding that garnered significant interest among researchers. 5 Angiogenesis is the biological process characterized by the activation of angiogenic signals or the inhibition of anti-angiogenic signals in response to various pathogenic factors. This process results in the sprouting, remodeling, and expansion of pre-existing blood vessels, ultimately leading to the formation of abnormal neovascularization. 6 This process not only serves as a continuous supplier of nutrients and a transporter of metabolic products essential for the growth and proliferation of tumors but also facilitates a route for tumor metastasis.7,8 Gastric cancer is characterized as a highly angiogenic malignant tumor, where the formation of new blood vessels plays a crucial role as a fundamental step for the accelerated advancement, migration, and invasion of gastric cancer. 9 Increasing evidence indicates that there is a presence of gastric mucosal neovascularization in the initial phase of PLGC. This neovascularization tends to escalate as gastric mucosal lesions advance, with the hypoxic microenvironment playing a crucial role as a triggering factor. 10 Hence, implementing targeted interventions in neovascular formation proves to be an efficacious treatment approach to impede the advancement of PLGC and forestall the onset of gastric cancer.
Current Western medicine treatment of PLGC mainly focuses on relieving clinical symptoms and lacks ideal treatment options. 11 Traditional Chinese medicine has always emphasized the diagnosis and treatment theory of “prevention before disease and prevention of changes after disease”. Moreover, traditional Chinese medicine has the advantages and value of multiple components, multiple targets, and multiple links, making it more advantageous in the treatment of this disease. 12 Many studies have confirmed that traditional Chinese medicine can interrupt or even reverse the pathological grading of gastric mucosa in PLGC, reduce the incidence of gastric cancer, and effectively improve the symptoms of patients, with low recurrence rates, providing beneficial treatment options and choices for this disease.13–15 Zhiwei Fuwei Pills (ZWFW) is further developed on the basis of traditional Chinese medicine classic prescriptions such as Shashen Maidong Decoction, Danshenyin, Baohe Pill and so on. It is composed of 14 kinds of traditional Chinese medicines, such as Pseudostellaria heterophylla (Miq.) Pax, Dioscorea oppositifolia L., Glycyrrhiza uralensis Fisch., Panax notoginseng (Burkill) F.H.Chen, Corydalis yanhusuo (Y.H.Chou & Chun C. Hsu) W.T.Wang ex Z.Y.Su & C.Y.Wu, Salvia miltiorrhiza Bunge, Forsythia suspensa (Thunb.) Vahl, Prunus mume (Siebold) Siebold & Zucc., Crataegus pinnatifida Bunge, Paeonia lactiflora Pall., Hordeum vulgare L., Raphanus raphanistrum subsp. Sativus (L.) Domin, Citrus × aurantium f. Deliciosa (Ten.) M. Hiroe (The above plant names have been checked on the Web site https://mpns.kew.org). It has been used to treat PLGC patients for more than 40 years. Clinical studies have shown that ZWFW has achieved satisfactory efficacy in improving the overall symptoms of patients, intervening in the pathological grade of gastric mucosa, and reducing the recurrence rate of disease. 16 Meanwhile, previous animal experiments have found that ZWFW can inhibit gastric mucosal inflammation in PLGC rats, improve mucosal secretion function, combat oxidative stress, regulate gastrointestinal hormone levels, enhance mucosal blood flow, repair epithelial cells, and maintain the stability of mucosal barrier function, which may be one of the mechanisms of this preparation in the treatment of PLGC.17–19 Modern pharmacological studies have shown that ZWFW has a variety of traditional Chinese medicines that can play an exact anti-angiogenic role.20–22 However, the mechanism of ZWFW in anti-angiogenesis in the treatment of PLGC is still unclear.
Network pharmacology is a new method that combines bioinformatics, network biology, chemoinformatics, and pharmacology. It can systematically reveal the active ingredients and potential mechanisms of complex traditional Chinese medicines, providing ideas and a basis for the research of traditional Chinese medicine.
23
In this research, a combination of network pharmacology and molecular docking technology was employed to analyze and predict the active components, potential targets, and molecular mechanisms of ZWFW in the treatment of PLGC. The study utilized the TCMSP database to identify the active ingredients of ZWFW and their corresponding targets, while the GeneCards database was utilized to retrieve PLGC-related targets. The overlapping targets between ZWFW and PLGC were determined through mapping, and protein-protein interaction (PPI) networks as well as networks were constructed using Cytoscape software. The DAVID database was utilized for GO functional enrichment analysis and KEGG pathway enrichment analysis. Molecular docking of key active ingredients and key targets was conducted using AutoDockTools software. To validate the findings of network pharmacology analysis, a PLGC rat model was established through a five-factor combination modeling method primarily involving N-methyl-N′-nitro-N-nitrosoguanidine (MNNG). Subsequently, the effects of different dosage groups of ZWFW on gastric mucosal microvasculature in PLGC rats were observed using transmission electron microscopy (TEM) and hematoxylin-eosin (H&E) staining. Additionally, enzyme-linked immunosorbent assay (ELISA), immunofluorescence (IF), immunohistochemistry (IHC), real-time fluorescence quantitative polymerase chain reaction (RT-PCR), and western blot were employed to assess the expression levels of relevant targets in the tissues, aiming to validate the potential mechanism of ZWFW in intervening in PLGC. Overall, these methodologies were utilized to confirm the anti-angiogenic properties of ZWFW and the potential molecular mechanisms influencing the expression of angiogenesis-related factors, thereby offering evidence and robust support for its clinical application. The working flow of this study was depicted in Figure 1. Workflow of the investigation strategy of ZWFW in the treatment of PLGC.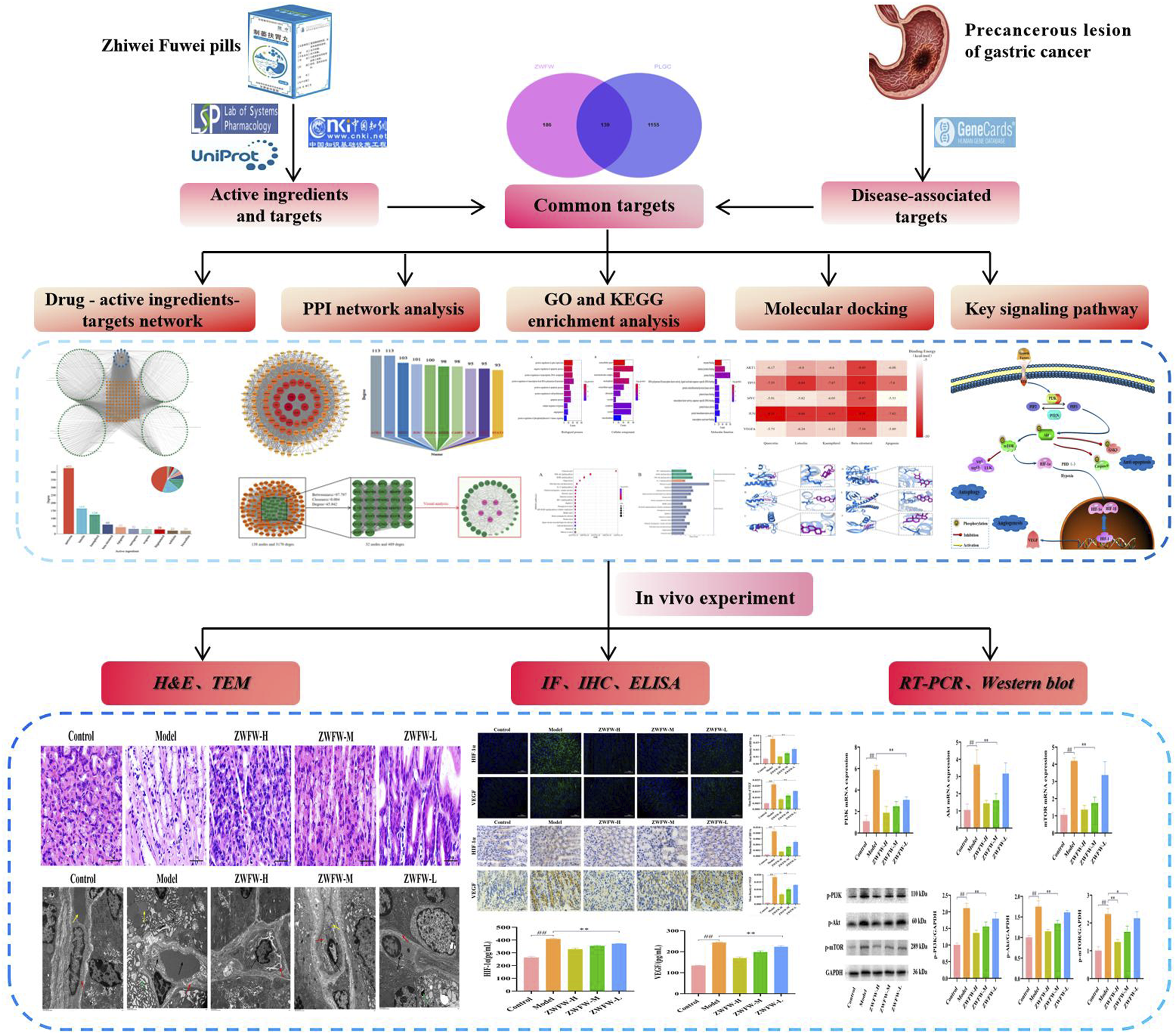
Materials and methods
Network pharmacology analysis
Screening of active ingredients and targets of ZWFW
The active ingredients of the above-mentioned drugs were retrieved through the TCMSP database (https://tcmspw.com/tcmsp.php). 24 A standard condition was set with an oral bioavailability ≥30% and a drug similarity ≥0.18 to obtain the active ingredients and their corresponding target proteins. 25 For Chinese medicines that were not found in the TCMSP database, information about the drugs was obtained from the China National Knowledge Infrastructure (https://www.cnki.net/) through relevant literature reports. 26 The screened target proteins were standardized using the UniProt database (https://www.uniprot.org/). 27
Screening of PLGC-related targets and intersection targets
Using “precancerous lesion of gastric cancer” as the keyword, retrieve PLGC-related targets from the GeneCards database (https://www.genecards.org/), 28 and filter the targets with a Relevance score ≥10.474. Take the intersection of the filtered disease-related targets and drug targets, and visualize the intersected targets using a Venn diagr-am (https://www.bioinformatics.com.cn). 29
Construction of the “drug-active ingredient-target” network
Creating relevant documents for drug-active ingredients, intersected targets, and using Cytoscape 3.9.1 software to construct a “drug-active ingredient-target” network diagram. 30 At the same time, use the “Network Analyzer” tool to perform topological analysis on the network and select the top five key active ingredients based on the degree value in descending order.
Construction and topological analysis of PPI network
The intersection targets were imported into the “multiple proteins” template in the STRING 11.5 database (https://string-db.org/). The species was selected as “Homo sapiens” and the confidence score was ≥0.4 to obtain the PPI network.31,32 Then import the data into Cytoscape 3.9.1 software to complete the visual analysis. Finally, the centiscape 2.2 plug-in tool is used for topological analysis of the PPI network, 33 and targets that are greater than the threshold value are selected as the key targets. According to the Degree value, the top five key targets are selected from the largest to the smallest.
GO and KEGG enrichment analysis
The intersection targets were imported into the DAVID database (https://david.ncifcrf.gov/), and the identifier was set as “OFFICIAL GENE SYMBOL” and the species as “H. sapiens”. GO function enrichment analysis and KEGG pathway enrichment analysis were performed on the intersection targets, where GO function enrichment analysis included molecular function (MF), biological process (BP), and cellular component (CC). At the same time, with p < .05 as the condition, the top 10 GO analysis and the top 20 KEGG pathway analysis were screened according to the order from small to large,34,35 and the data were visualized.
Molecular docking
The top five key targets and key active components with the highest degree value were verified by molecular docking. The mol2 format files of key active components were obtained from TCMSP database and PubChem database (https://pubchem.ncbi.nlm.nih.gov/) as docking ligands. Meanwhile, the pdb format files of key targets were obtained from RSCB PDB database (https://www.rcsb.org/) as docking receptors. 36 The molecular docking was performed after hydrogenation and dehydration of target proteins and active components by using AutoDockTools-1.5.7 software, and then the stability between receptor and ligand was evaluated according to the value of binding energy. Finally, the selected results were visualized by Pymol software. 37
Experimental verification
Experimental animals
Obtained from Beijing Sipeifu Biotechnology Co., Ltd., male sprague dawley (SD) specific pathogen-free (SPF) rats at 5 weeks old with a weight of (130 ± 20 g) were used in this study. The animal production license number is SCXK (Beijing) 2019-0010. All animals were bred in the SPF-level Experimental Animal Center of Gansu University of Chinese Medicine. The use of these animals and experimental procedures were approved by the Animal Ethics Review Committee of Gansu University of Chinese Medicine, with an ethics review number of 2021-020.
Drugs and reagents
ZWFW (batch number: Z09001923, 40 g/bottle) was provided by the Pharmaceutical Preparation Center of Gansu Provincial Hospital of Traditional Chinese Medicine. N-methyl-N′-nitro-N-nitrosoguanidine (batch number: R030453-25g) was purchased from Kebao Biotechnology Co., Ltd. (Lanzhou, China). 25% ammonia solution (batch number: 202152) was purchased from Beichen Fangzheng Reagent Factory (Tianjin, China). Anhydrous ethanol (batch number: 20201015) was purchased from Huihang Chemical Technology Co., Ltd. (Tianjin, China). 0.03% ranitidine feed was purchased from Maohua Biotechnology Co., Ltd. (Shenyang, China). Primary antibodies against PI3Kinase p110 beta (USA, CST Company, batch number: 4249), Akt (USA, Affinity Company, batch number: AF0836), mTOR, HIF-1α, VEGF (UK, Abcam Company, batch numbers: ab32028, ab179483, ab32152), GAPDH (USA, Immunoway Company, batch number: YM3215), and goat anti-rabbit IgG (H + L) HRP antibody (USA, Immunoway Company, Catalog number: RS0002) were used. An ECL chemiluminescence detection kit (catalog number: P10100) was purchased from NCM Biotechnology Co., Ltd. (Suzhou, China). The summer-erythrosin staining solution (catalog number: B1003) was purchased from Aidisheng Biotechnology Co., Ltd. (Jiangsu, China). Animal total RNA extraction kit, FastKing one-step genomic DNA removal cDNA synthesis premix, and SuperReal fluorescent quantitative premix enhanced version (SYBR Green) kit (catalog numbers: DP451, KR118, FP205) were all purchased from Tiangen Biotech Co., Ltd. (Beijing, China).
Animal modeling, grouping, and sampling
After 1 week of adaptive feeding, 70 SPF-grade male SD rats were randomly divided into a control group of 10 rats, ZWFW high-dose (ZWFW-H), medium-dose (ZWFW-M), low-dose (ZWFW-L) groups, and a model group, with 15 rats in each group. The control group was fed with regular feed daily, while the other groups were modeled using the five-factor composite modeling method of MNNG combined with the hunger-satiation disorder, ethanol gavage, ammonia water-free drinking, and 0.03% ranitidine feed. A 100 μg/mL MNNG solution (brown bottle, avoid light) was freely available for drinking daily. After normal feeding for 2 days, the rats were fasted for 1 day and then given 0.1% ammonia water for free drinking during the fasting period. They were also given 2 mL/each of 20% anhydrous ethanol by gavage (with water restriction 1 h before and after gavage). 0.03% ranitidine feed was provided for free consumption. After continuous modeling for 26 weeks, 2 SD rats were randomly euthanized for gastric mucosal pathological examination. Based on the results of previous research,38,39 the prescribed weight of ZWFW (40 g/bottle, approximately 0.22 g per pill) was weighed, and the ZWFW was grinded into water. From the 27th week, high, medium, and low doses of ZWFW were prepared as 1.67 g/kg, 0.84 g/kg, and 0.42 g/kg solutions, respectively, and administered by gavage at a dosage of 1 mL/100g. The control and model groups were given an equal volume of physiological saline by gavage once daily for 4 weeks. At the end of the 30th week, the rats were fasted for 18 h and then euthanized by intraperitoneal injection of 2% pentobarbital sodium (2 mL/kg). The whole gastric antrum tissue was removed for analysis. Some gastric antrum tissue was fixed with 4% paraformaldehyde for H&E staining, and the remaining gastric tissue was frozen at −80°C for other tests.
Gastric mucosal tissue pathological analysis
The gastric antrum tissue fixed with 4% paraformaldehyde was dehydrated in alcohol and xylene, embedded in paraffin, and sliced (4 μm). H&E staining was performed, and the pathological changes of gastric antrum tissue in each group of rats were observed under a light microscope.
Observation of gastric mucosal microvascular ultrastructure
Pieces of gastric mucosal tissue (1 mm × 1 mm × 1 mm) from each group were quickly placed in 2.5% glutaraldehyde and stored overnight at 4°C. The next day, the tissues were rinsed three times with phosphate-buffered saline (PBS) and fixed with 1% osmium tetroxide solution for 1 h, followed by three washes with PBS. After dehydration and embedding, ultra-thin sections with a thickness of 60 nm were made. Double staining with 3% lead citrate and 0.1% uranyl acetate was performed, and the microvascular ultrastructure of gastric mucosa was observed and photographed under a TEM.
Detection of gastric mucosal tissue hypoxia status
The gastric mucosal tissue slides of each group were subjected to baking, dewaxing, dexylene, deethanol, and antigen repair, and incubated and closed at room temperature for 30 min. After 3 times of cleaning with PBS buffer, the reagent was added and incubated in 37°C water bath for 2 h. After 3 times of cleaning with PBS buffer, the tablets were nucleated with DAPI dyeing solution. After 3 times of cleaning with PBS buffer, the tablets were sealed with an anti-fluorescence quencher. After drying, the slides were placed under a fluorescence microscope for observation and sampling, and the results were analyzed with Image-Pro Plus 6.0 software.
IHC staining
The paraffin sections of gastric mucosa in each group were subjected to routine dewaxing and hydration. 3% H2O2 was used to block endogenous peroxidase and citrate buffer was used for high-temperature antigen retrieval. After being washed 3 times with PBS buffer, the primary antibodies VEGF (1:250) and HIF-1α (1:500) were added and incubated at 4°C overnight. After being washed 3 times with PBS buffer, the secondary antibodies coupled with HRP were added and incubated at room temperature for 50 min. Then, DAB staining, tap water washing, hematoxylin re-staining, gradient ethanol dehydration, xylene transparency, and sealing treatment were performed sequentially. Finally, the slides were observed under a microscope, and Image-Pro Plus 6.0 software was used to select brownish-yellow color as the positive criterion and analyze the positive values.
ELISA analysis
Strictly follow the ELISA kit instructions for correct operation. The ELISA kit and micropore reaction strip were placed at room temperature for 30 min, and three control holes were set: positive, negative, and blank. According to the reagent operation, 50 μL standard solution was injected into the corresponding micropore strip, and then 50 μL specific enzyme was injected into the corresponding micropore strip. The sealing plate was sealed and placed in a water bath at 37°C for 1 h. The liquid in the micropore reaction strip was removed and washed repeatedly 5 times. The reagent was added to the reaction strip and further sealed. The reagent was placed in a water bath at 37°C for 30 min, and then 50 μL termination solution was added and mixed. Finally, the levels of VEGF and HIF-1α in the serum of rats in each group were determined and recorded.
Real-time fluorescence quantitative polymerase chain reaction (RT-PCR)
Primer sequence summary table.

Western blot (WB) analysis
The total tissue protein was obtained after the gastric mucosal tissues of each group were lysed with high-efficiency RIPA lysate, and the concentration of total tissue protein was detected with a BCA protein quantitative kit. The protein loading buffer was added at the rate of 4:1 and denatured at 100°C for 10 min. Equal amounts of protein from each group were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and the proteins were transferred to an activated PVDF membrane using a rapid transfer buffer. After the 5% BSA sealing solution was closed at room temperature for 2 h, the corresponding primary antibodies p-PI3K (1:1000), p-Akt (1:1000), p-mTOR (1:1000) and GAPDH (1:1000) were added, incubated at 4°C overnight, and then washed with 1xTBST for 3 times. Then the corresponding fluorescent secondary antibody (1:5000) was added and incubated in a shaker at room temperature for 1 h. The enhanced chemiluminescence method was used for exposure and color development. The gray values of each protein band were obtained by Image J software and normalized for statistical analysis.
Statistical analysis
SPSS 26.0 and GraphPad Prism 8.0.2 were used for data analysis. All data were expressed as means ± standard deviation (SD). When the data were normally distributed and had equal variances, one-way analysis of variance was used for comparison between groups, and the LSD-t method was used for multiple comparisons. p < .05 indicates statistically significant differences between groups, while p < .01 indicates significant differences between groups.
Results
Prediction of active ingredients and targets of ZWFW
After filtering according to the conditions, active ingredients without corresponding targets were deleted. Among them, pseudostellaria heterophylla has seven active ingredients and 76 targets, Dioscorea polystachya has 12 active ingredients and 52 targets, G. uralensis has 88 active ingredients and 191 targets, P. notoginseng has seven active ingredients and 154 targets, C. yanhusuo has 49 active ingredients and 172 targets, S. miltiorrhiza has 59 active ingredients and 105 targets, F. suspensa has 19 active ingredients and 183 targets, Mume Fructushas eight active ingredients and 156 targets, Crataegus monogyna has 12 active ingredients and 237 targets, P. lactiflora Pall has eight active ingredients and 72 targets, Fructus Hordei Germinatus has 15 active ingredients and 85 targets, Semen Raphani has two active ingredients and three targets, and Pericarpium Citri Reticulate has five active ingredients and 53 targets. Massa Medicata Fermentata, as a fermented product composed of Vigna umbellata, Artemisia carvifolia, and flour, did not find specific active ingredients and targets. After merging and removing duplicates, a total of 258 active ingredients and 325 targets were obtained.
Prediction of PLGC-related targets and acquisition of intersection targets
A total of 2645 disease-related targets were obtained from the GeneCards database, and the targets were filtered according to a Relevance score ≥10.474. Finally, 1294 PLGC-related targets were obtained. In addition, by combining disease-related targets and drug targets, a total of 139 intersection targets were obtained (Figure 2). These targets can be potential targets for ZWFW in the treatment of PLGC. Venn diagram of ZWFW active ingredient targets and PLGC-related targets.
“Drug-active ingredient-target” network analysis
A complete “drug-active ingredient-target” network was constructed using Cytoscape 3.9.1 software (Figure 3). The diagram shows 410 nodes and 4303 edges. The size of the nodes represents the Degree value, and the edges represent the interaction between them. Then, after topological analysis using the “Network Analyzer” tool, the top five key active ingredients in descending order of Degree value are quercetin, luteolin, kaempferol, beta-sitosterol, and apigenin (Figure 4). This indicates that these active ingredients may play a crucial role in the treatment of PLGC with ZWFW. Network diagram of drug-active ingredient-target (blue represents traditional Chinese medicine, green represents active ingredients, orange represents intersection targets). Bar chart of key active ingredients of the drug (top 10° values).

Construction and topological analysis of the PPI network
The intersection targets were used to generate a PPI network in the STRING 11.5 database, and the data were imported into Cytoscape 3.9.1 software for visualization analysis. The PPI network diagram contains 138 nodes and 3170 edges. The size of the nodes and the depth of the color represent the degree value, and each edge represents the interaction between proteins (Figure 5(a)). Then, after topological analysis using the centiscape 2.2 plugin, a total of 32 key targets were obtained based on the criteria of betweenness >97.797, Degree >45.942, and Closeness >0.004 (Figure 5(b)). The top five key targets are AKT1, TP53, MYC, JUN, and VEGFA (Figure 5(c)). This indicates that these targets may play an important role in the treatment of PLGC with ZWFW. Strategies for constructing PPI network diagrams. (a) PPI network diagram constructed by Cytoscape (node size and color depth represent the degree size). (b) Selection of key targets from PPI network topology analysis (green represents key targets, purple represents core targets with top 5° values). (c) Bar chart of key target convergence at the bottom (top 10° values).
GO and KEGG enrichment analysis
Enrichment analysis of the intersection targets was performed using the bioinformatics online platform. A total of 950 GO enrichment terms were obtained, including 735 BP, 78 CC, and 137 MF. The top 10 GO terms were sorted from smallest to largest according to p < .05, and then visualized. The results showed that BP mainly involves positive regulation of gene expression, positive and negative regulation of the apoptosis process, DNA template-dependent positive regulation of transcription, cell response to hypoxia, and angiogenesis (Figure 6(a)). CC mainly involves extracellular space, nucleus, nuclear plasma, chromatin, cytoplasm, and mitochondria (Figure 6(b)). MF mainly involves enzyme binding, protein binding, protein serine/threonine/tyrosine kinase activity, and protein homodimerization activity (Figure 6(c)). Bar chart of GO enrichment analysis. (a) Bar chart of BP enrichment analysis. (b) Bar chart of CC enrichment analysis. (c) Bar chart of MF enrichment analysis. Length of the bars represents the number of enriched genes, color depth represents the degree of enrichment.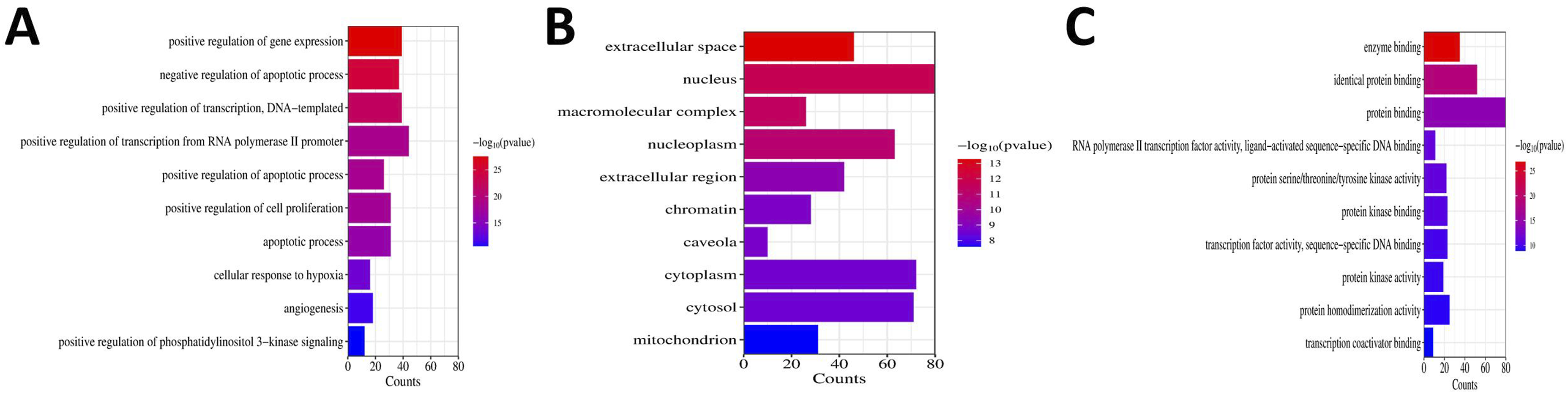
A total of 177 pathways were obtained through enrichment analysis of KEGG pathways. The first 20 pathways were visualized according to the same screening method (Figure 7(a)), and then sorted according to the pathway type (Figure 7(b)). The results showed that, except for disease-specific signaling pathways, the HIF-1 signaling pathway, MAPK signaling pathway, IL-17 signaling pathway, PI3K/Akt signaling pathway, lipids, and atherosclerosis, fluid shear stress, and atherosclerosis showed multi-target enrichment. PI3K/Akt signaling pathway ranks third in the enrichment classification of the KEGG pathway, and it and the HIF-1 signaling pathway play an important role in angiogenesis (Figure 8). Angiogenesis is closely related to the occurrence and development of PLGC, which can be used as a key pathway for the treatment of PLGC. Therefore, this study focused on and explored this signaling pathway. KEGG pathway enrichment analysis. (a) Bubble chart of KEGG pathway enrichment analysis (bubble size represents the number of enriched genes, color depth represents the degree of enrichment). (b) KEGG pathway classification chart. Mechanism diagram of PI3K/Akt signaling pathway promoting angiogenesis.

Molecular docking results and visualization
Molecular docking was performed between the top five key targets AKT1 (PDB ID: 1UNQ), TP53 (PDB ID: 3DCY), MYC (PDB ID: 5I4Z), JUN (PDB ID: 1A02), VEGFA (PDB ID: 4KZN) and the top five key active ingredients quercetin, luteolin, kaempferol, beta-sitosterol, apigenin. The binding energy between the ligands and receptors was calculated, and a lower binding energy indicates a more stable binding. When the binding energy is less than −5 kcal/mol, it indicates good binding ability between the ligand and receptor. When the binding energy is less than −7 kcal/mol, it indicates strong binding ability. The docking heat map results (Figure 9) show that the binding energies of all groups are less than −5 kcal/mol, and the binding energies of most groups are less than −6 kcal/mol, indicating a strong affinity between the key active ingredients and key targets. Furthermore, the combination with the lowest binding energy between each key target and key active ingredient was selected for visualization analysis. The results show that the ligands and receptors can interact with each other through hydrogen bonding and form stable spatial structures. For example, AKT1 and beta-sitosterol form hydrogen bonds at GLU-9 and LYS-8. TP53 and beta-sitosterol form hydrogen bonds at ILE-21. MYC and beta-sitosterol form hydrogen bonds at ALA-43. JUN and quercetin form hydrogen bonds at ARG-537. JUN and beta-sitosterol form hydrogen bonds at LYS-664 and ARG-665. JUN and beta-sitosterol form hydrogen bonds at TYR-21 (Figure 10(A)–(F)). This further indicates that the above key active ingredients and key targets may play an important role in the treatment of PLGC with ZWFW. Molecular docking binding energy heat map (darker color indicates smaller binding energy and stronger binding ability). Visualization of combinations of key targets and key active ingredients. (a) AKT1 and beta-sitosterol. (b) TP53 and beta-sitosterol. (c) MYC and beta-sitosterol. (d) JUN and quercetin. (e) JUN and beta-sitosterol. (f) JUN and beta-sitosterol. Purple represents drug active molecules, blue represents target protein molecules, yellow dashed lines represent hydrogen bonds.

ZWFW improves gastric antrum histopathological changes
Gastric antrum histopathological changes were further evaluated by H&E staining (Figure 11). Compared with the control group, the model group showed a large number of goblet cells, moderate intestinal metaplasia, and partial severe dysplasia in the gastric mucosa, as well as disordered arrangement of some glandular cells, nuclear abnormalities, and increased nuclear division, with infiltration of inflammatory cells in the lower layer. This indicates the successful modeling of PLGC rats. Compared with the model group, the high-dose ZWFW group showed a significant improvement in the arrangement of glands and a significant reduction in the degree of damage. The medium-dose ZWFW group showed closely arranged glands with a few inflammatory cells and rare multilayer and back-to-back changes. The low-dose ZWFW group showed partial glandular dilation, epithelial cell atrophy, and a slight improvement in the degree of damage. These results indicate that ZWFW can effectively improve the histopathological grading of gastric antrum in PLGC rats. H&E staining of PLGC rat gastric antrum tissue, ×400.
ZWFW improves gastric mucosal microvascular abnormalities
The morphological changes of gastric mucosal microvessels in rats were further observed under TEM (Figure 12). Compared with the control group (black arrow indicates vascular endothelial cells, red arrow indicates vascular wall, yellow arrow indicates red blood cells), the gastric mucosal microvessel structure in the model group was abnormal, with obvious vascular wall damage, rough and thickened surface, loose structure (indicated by red arrow), red blood cells and a large number of platelets in the lumen (indicated by black arrow), edema in the surrounding tissues, and a large number of electron-lucent areas (indicated by green arrow); yellow arrow indicates a large number of lipid droplets. This indicates that the gastric mucosal microvessels in PLGC rats are in an abnormal state. Compared with the model group, the high-dose ZWFW group showed a significant improvement in microvascular damage (black arrow indicates vascular endothelial cells), with a dense vascular wall, no significant thickening, and an unsmooth surface (red arrow indicates vascular wall). The medium-dose ZWFW group showed a slight alleviation of vascular damage (black arrow indicates vascular endothelial cells), with significant thickening of the vascular wall, loose structure, and rough surface (red arrow indicates), and a few autophagosomes (yellow arrow indicates). The low-dose ZWFW group showed a slight alleviation of microvascular damage compared to the model group, with inflammatory cells seen in the lumen and autophagosomes in the cytoplasm (black arrow indicates), thickening of the vascular wall, loose structure (red arrow indicates), edema in some surrounding areas, and the presence of autophagosomes (green arrow indicates). These results indicate that ZWFW can transform the ultrastructure of gastric mucosal microvessels toward normal and show a good therapeutic effect on microvascular abnormalities. TEM image of microvessels in PLGC rat gastric mucosa, ×2000.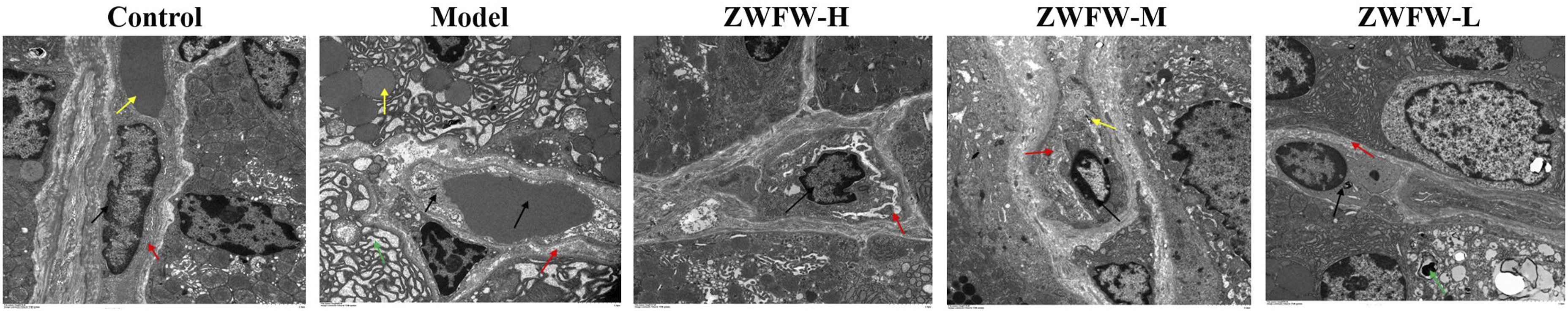
ZWFW improves gastric mucosal hypoxia and inhibits angiogenesis
The hypoxia status and neovascularization in gastric mucosal tissue were observed by immunofluorescence staining (IF). The results showed that compared with the control group, the model group had significantly increased HIF-1α and VEGF positive values, indicating the presence of hypoxic microenvironment and angiogenesis in the gastric mucosa of PLGC rats, which is consistent with existing studies. Compared with the model group, the high, medium, and low-dose ZWFW groups showed a decrease in HIF-1α and VEGF positive density values, and showed a dose-effect relationship (Figure 13(a)). These results indicate that ZWFW may improve gastric mucosal hypoxia status and inhibit angiogenesis by reducing HIF-1α and VEGF expression. Subsequently, the expression levels of HIF-1α and VEGF in the gastric mucosa of rats were detected by IHC and ELISA, respectively. The IHC results showed that the expression of HIF-1α and VEGF proteins in the model group was significantly higher than that in the control group. After drug intervention, the expression of HIF-1α and VEGF proteins in the high, medium, and low-dose ZWFW groups decreased, showing a dose-effect relationship (Figure 13(b)). These data indicate that ZWFW can effectively inhibit the expression of HIF-1α and VEGF proteins. In addition, ELISA results confirmed that the levels of HIF-1α and VEGF in the serum of the model group rats were significantly higher than those in the control group. After ZWFW intervention, the levels of HIF-1α and VEGF in the serum of different dose groups decreased, and the effect was more significant with higher drug doses (Figure 13(c)). These results indicate that ZWFW can effectively inhibit the expression levels of HIF-1α and VEGF, and provide multiple validations for the conclusion that ZWFW improves gastric mucosal hypoxia status and inhibits angiogenesis in PLGC rats. Effects of ZWFW on gastric mucosal hypoxia and angiogenesis. (a) Immunofluorescence staining of HIF-1α and VEGF (magnification, ×100), quantification of HIF-1α and VEGF-positive cells. (b) Immunohistochemical staining of HIF-1α and VEGF (magnification, ×200), quantitative analysis of HIF-1α and VEGF immunohistochemical staining. (c) Analysis of HIF-1α and VEGF levels in serum. Data expressed as means ± S.D. (n = 3);
##
p < .01 compared with control group,
**
p < .01 compared with model group.
Effects of ZWFW on the expression of related genes and proteins in the PI3K/Akt/mTOR signaling pathway
The expression levels of proteins and mRNA in the PI3K/Akt/mTOR pathway in the gastric mucosa of PLGC rats were determined by WB and RT-PCR, respectively. The RT-PCR results showed that the expression of PI3K, Akt, and mTOR mRNA in the model group was significantly higher than that in the control group. After ZWFW intervention, the mRNA expression of PI3K in the different dose groups decreased significantly, while the mRNA expression of Akt and mTOR in the high and medium dose groups decreased to varying degrees (Figure 14(a)). In addition, WB results confirmed that the expression of p-PI3K, p-Akt, and p-mTOR proteins in the model group was significantly higher than that in the control group. After drug intervention, the expression of these proteins in the high and medium-dose groups decreased to varying degrees, while the expression in the low-dose group did not decrease significantly (p > .05) (Figure 14(b)). These results from different perspectives confirm that ZWFW can reduce the expression levels of proteins and genes in the PI3K/Akt/mTOR signaling pathway. HIF-1α is located downstream of mTOR and regulated by PI3K/Akt signaling pathway, and previously it has been verified that ZWFW can inhibit the expression of HIF-1α and VEGF. In summary, ZWFW inhibits the expression of HIF-1α/VEGF by blocking the activation of the PI3K/Akt/mTOR signaling pathway. Therefore, the PI3K/Akt/mTOR/HIF-1α/VEGF pathway may be the potential mechanism of ZWFW intervention in gastric mucosal angiogenesis of PLGC rats. Effects of ZWFW on protein and gene expression in PI3K/Akt/mTOR signaling pathway. (a) Quantitative analysis of PI3K, Akt and mTOR genes mRNA. (b) Western blot assay of p-PI3K, p-Akt, p-mTOR proteins, and the p-PI3K, p-Akt and p-mTOR were quantitatively analyzed. Data expressed as means ± S.D. (n = 3);
##
p < .01 compared with control group,
*
p < .05,
**
p < .01 compared with model group.
Discussion
PLGC, as a pathological concept, refers to intestinal metaplasia or dysplasia based on chronic atrophic gastritis. 40 Studies have shown that the incidence of gastric cancer in patients with extensive gastric mucosal atrophy and intestinal metaplasia is 1.9%-10.0% and 5.3%-9.8%, respectively, 41 while the incidence of gastric cancer in patients with low-grade dysplasia can reach 25.95%. 42 Therefore, early intervention of PLGC is of great significance for blocking the occurrence of gastric cancer and reducing the incidence rate. Western medicine treatment of PLGC mainly focuses on removing risk factors and relieving symptoms. Although related guidelines 43 recommend the use of cyclooxygenase inhibitors, antioxidant vitamins, and other drugs to reduce the risk of gastric cancer, the quality of evidence is low, and there is still a lack of precise treatment plans for this disease.
Traditional Chinese medicine, as an alternative therapy, has shown good clinical efficacy and unique advantages in the treatment of PLGC and is recommended for the treatment of this disease. 44 ZWFW, as a traditional Chinese medicine preparation, has been proven to have good therapeutic effects on PLGC and high oral safety. 45 Moreover, previous animal experiments have shown that ZWFW can intervene in gastric mucosal inflammatory injury, resist oxidative stress, regulate gastrointestinal hormone levels, and affect autophagy levels. As Shu J et al. 39 found that ZWFW could increase food intake and body mass in chronic atrophic gastritis (CAG) rats, improve gastric mucosal pathological status, and its mechanism of action might be related to the up-regulation of ghrelin levels in serum by ZWFW. In addition, Feng ZZ et al. 38 found that ZWFW could significantly improve the pathological manifestations of abnormal gastric mucosa in PLGC model rats. The mechanism might be related to down-regulating mTOR expression, up-regulating Beclin1 and LC3B expression, so as to promote autophagy. However, the mechanism of action of this preparation in inhibiting angiogenesis in PLGC is still not fully understood, which has a certain impact on its promotion and application. This study integrated network pharmacology, molecular docking, and in vivo experiments, and focused on the analysis and verification of the anti-angiogenesis mechanism of ZWFW.
By constructing and analyzing the drug-active ingredient-target network, the top five key active ingredients were screened: quercetin, luteolin, kaempferol, beta-sitosterol, and apigenin. Quercetin is a widely distributed flavonoid compound in nature, with various biological functions such as anticancer, anti-inflammatory, and antioxidant activities.46,47 In vitro and in vivo studies have shown that quercetin can intervene in various cancers, including gastric cancer, by changing the cell cycle progression, promoting apoptosis, inhibiting angiogenesis, and regulating autophagy.48,49 Luteolin is a member of the flavonoid compound family derived from plants and has various pharmacological properties such as anti-inflammatory, anti-tumor, and immunomodulatory effects. It has attracted much attention from researchers in inhibiting tumor angiogenesis. 50 Studies have shown that luteolin can intervene in gastric cancer by inhibiting the activation of the PI3K/Akt signaling pathway and the Notch1-VEGF signaling pathway, interfering with the formation of neovascularization and angiogenesis models in cells, and thereby blocking the progression of gastric cancer.51,52 Kaempferol is a natural polyphenol whose anticancer effects have been demonstrated in in vitro and in vivo studies. 53 Research has shown that kaempferol exerts anticancer effects by activating tumor suppressor genes, 54 promoting apoptosis, 55 inducing cell cycle arrest, 56 inhibiting inflammatory responses, and angiogenesis. 57 The main mechanism of inhibiting tumor angiogenesis is the inactivation of the HIF-1α/VEGF signaling pathway. 58 Beta-sitosterol, as the most abundant beneficial plant sterol in plants, exhibits anticancer properties in various cancers, including gastric cancer. 59 Studies have shown that beta-sitosterol can intervene in various cell signaling pathways, such as angiogenesis, cell cycle, cell proliferation, and apoptosis. 60 Apigenin belongs to the flavonoid compound and has biological activities such as anti-inflammatory, antioxidant stress, and anticancer effects. 61 Studies have shown that apigenin can intervene in various tumors by regulating the cell cycle, promoting apoptosis and autophagy, inhibiting angiogenesis, and regulating cellular immune responses. The key signaling pathways targeted include PI3K/Akt/mTOR, NF-κB, MAPK, and so on.62,63 It can be seen that the above key active ingredients can intervene in tumors through various pathways, among which the inhibition of angiogenesis is an important pathway shared and exerted by the active ingredients. Meanwhile, the key active ingredients and the key targets (such as AKT1, VEGFA) obtained by PPI network topology analysis were docked molecularly, and the results showed that they showed strong affinity and stable structure between each other. These findings further illustrate the potential of ZWFW in inhibiting angiogenesis in the treatment of PLGC.
Angiogenesis is a complex process involving multiple factors, dynamic development, and hierarchical evolution, which involves the growth, migration, and differentiation of endothelial cells, and the formation of tubular structures. This process is regulated by multiple signaling pathways.64,65 In order to determine the molecular pathways through which ZWFW intervenes in PLGC angiogenesis, KEGG enrichment analysis was performed, and the results showed that in addition to specific disease signaling pathways, the pathways involved mainly include the HIF-1 signaling pathway, MAPK signaling pathway, IL-17 signaling pathway, PI3K/Akt signaling pathway, etc. Among them, the PI3K/Akt and HIF-1 signaling pathways, which are closely related to angiogenesis, showed multiple target enrichment states. 66 Therefore, this study also focused on exploring the relationship between this signaling pathway and angiogenesis in PLGC. Under the induction of various factors, receptor tyrosine kinases (RTKs) can activate PI3K, which further catalyzes the production of phosphatidylinositol 3,4,5-trisphosphate (PIP3) from phosphatidylinositol 4,5-bisphosphate (PIP2). PIP3, as a second messenger, further activates and phosphorylates Akt, 67 and activated Akt can directly or indirectly phosphorylate and activate mammalian rapamycin target protein (mTOR). 68 Studies have shown that the activation of the PI3K/Akt/mTOR signaling pathway can positively regulate the expression of HIF-1α at the protein level. 69 HIF-1α is an oxygen-dependent transcription activator, which is relatively stable in hypoxic microenvironment and is not easy to degrade. It often transfers to the nucleus and abnormally activates the downstream important target gene VEGF. As the strongest angiogenic factor, VEGF mainly binds to transmembrane receptors after activation, promoting intracellular signal transduction, and then induces neovascularization by regulating endothelial cell activation and increasing vascular permeability.70–72 The results of this study showed that the expression levels of the PI3K/Akt/mTOR/HIF-1α/VEGF pathway were significantly increased in the PLGC model group of rats. Additionally, abnormal blood vessel formation was observed in the gastric mucosa of rats, indicating that this signaling pathway is abnormally activated in the gastric mucosa of PLGC rats and plays an important role in promoting the formation of new blood vessels in the gastric mucosa.
In order to verify the analysis results of network pharmacology, we conducted in vivo experiments for further validation. In terms of in vivo research, this study confirmed the anti-angiogenic properties of ZWFW using various experimental methods. First, TEM and H&E results showed that ZWFW in different dosage groups could promote the transition of microvascular abnormalities in the gastric mucosa of PLGC rats toward normal and effectively intervene in the pathological grading of the gastric mucosa. Subsequently, to make the results more convincing, we quantitatively detected the expression levels of relevant targets in the tissues through IF, IHC, RT-PCR, WB, and ELISA. Interestingly, all these experimental results showed that the expression levels of PI3K, Akt, mTOR, HIF-1α, and VEGF in the model group were significantly higher than those in the control group, while compared with the model group, ZWFW high and medium-dose groups could effectively reduce the expression levels of PI3K, Akt, mTOR, HIF-1α, and VEGF. It can be seen that the above results not only effectively validate the mechanism of ZWFW intervention in PLGC angiogenesis predicted by network pharmacology analysis but also further indicate that inhibiting the activation of the PI3K/Akt/mTOR/HIF-1α/VEGF signaling pathway may be a potential mechanism by which ZWFW exerts its anti-angiogenic effects in PLGC. However, this study still has some limitations, such as the lack of techniques such as liquid chromatography and mass spectrometry to detect and analyze the exact active ingredients of ZWFW and the lack of corresponding in vitro experiments. Nevertheless, the results of this study provide important evidence and support for the mechanism of ZWFW in intervening in PLGC angiogenesis and also indicate the direction for further research by the research team.
Conclusion
In this investigation, it can be concluded from two aspects of network pharmacological analysis and in vivo experimental verification that ZWFW can inhibit PLGC gastric mucosal angiogenesis and improve microvascular abnormalities, and effectively intervene in the pathological evolution of gastric mucosa. Meanwhile, further studies found that the antiangiogenic properties of this preparation may be achieved by inhibiting the abnormal activation of the PI3K/Akt/mTOR/HIF-1α/VEGF signaling pathway.
Footnotes
Acknowledgments
Not applicable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 82060860), and the Gansu Provincial Natural Science Foundation (No. 21JR7RA588).
