Abstract
Background
Nanoparticles are potential luminescent probes; among them, upconversion nanoparticles (UCNP) are currently being developed as fluorescent probes for biomedical applications. However, the molecular mechanisms of UCNP in human gastric cell lines remain poorly understood. Here, we aimed to examine UCNP cytotoxicity to SGC-7901 cells and explore its underlying mechanisms.
Methods
The effects of 50–400 μg/mL UCNP on human gastric adenocarcinoma (SGC-7901) cells were investigated. Flow cytometry was used to evaluate reactive oxygen species (ROS), mitochondrial membrane potential (ΔΨm), intracellular Ca2+ levels, and apoptosis. Activated caspase-3 and nine activities were measured; meanwhile, cytochrome C (Cyt C) in the cytosol and B-cell lymphoma 2 (Bcl-2), Bcl-2 associated X protein (Bax), protein kinase B (Akt), phosphorylated-Akt (p-Akt), 78 kDa glucose-regulated protein (GRP78), 94 kDa glucose-regulated protein (GRP94), calpain-1, and calpain-2 protein levels were also detected.
Results
UCNP inhibited the viability of SGC-7901 cells in a concentration- and time-dependent manner and increased the proportion of cell apoptosis. Exposure to UCNP enhanced the ratio of Bax/Bcl-2, elevated the level of ROS, decreased ΔΨm, increased intracellular Ca2+ and Cyt C protein levels, decreased the levels of phosphorylated Akt, increased the activity of caspase-3 and caspase-9, and upregulated the protein expression of GRP-78, GRP-94, calpain-1 and calpain-2 in SGC-7901 cells.
Conclusion
UCNP induced SGC-7901 cell apoptosis by promoting mitochondrial dysfunction and ROS-mediated endoplasmic reticulum (ER) stress, initiating the caspase-9/caspase-3 cascade.
Keywords
Introduction
The mortality rate associated with gastric cancer is one of the highest among all gastrointestinal cancers. 1 Endoscopic techniques are key to the early diagnosis and treatment of gastric cancer. 2 Currently, the fluorescent probe material used in the clinic is generally Proflavine (a nuclear dye with an absorption wavelength of 445 nm and an emission wavelength of 515 nm). This dye has the disadvantages of biological toxicity, limited penetration depth, and general accuracy and sensitivity. 3 Nanotechnology has led to a growing interest in combining nanomaterials with biomedicine. Among them, rare earth upconversion nanoparticles (UCNP) attract special attention. The near-infrared light excitation source has the advantages of a large nanoparticle penetration depth while exerting minimal damage on biological tissue. 4 NaYF4 fluoride is the best known matrix material. In this study, it is proposed to use a NaYF4 fluoride matrix doped with one or several rare earth ions (generally Yb3+ is the sensitizer ion, and Er3+ is an activator). Then, polyvinyl pyrrolidone (PVP) is used to modify the surface of the upconversion nanocrystals. The nanoparticles can be evenly distributed in water and exist stably, which also prepares for the loading of functional groups of the nanoparticles in the later stage. 5 Nanoparticles have been tested for physical properties, in vitro imaging, and luminescent properties. Gao reported PEG-modified NaGdF4:Yb, Er@NaGdF4 and then connected the antibody MGb2 specifically expressed in gastric cancer with PEG to image lymph nodes and gastric cancer. 6 Wang et al. used EDC/NHS-conjugated antibody rlgG on the surface of aminated UCNP to specifically combine with carcinoembryonic antigen CEA8 on the surface of HeLa cells for smart light imaging . 7 Li Fuyou’s research group used Ye Jun molecule (FA)-functionalized UCNP (FA UCNP) to perform confocal imaging on HeLa cells, 8 and the underlying molecular mechanisms of UCNP in human gastric cell lines have yet to be investigated.
Tumor cells continue to experience stress factors such as ischemia, hypoxia and nutrient deficiency during the growth process, which stimulate the body to produce many ROS. ROS are involved in the occurrence and development of tumors, such as thyroid cancer, 9 lung cancer, 10 colon cancer, 11 bladder cancer, 12 breast cancer, 13 cervical cancer, 14 and liver cancer. 15 Tumor cells have elevated reactive oxygen species (ROS) levels, redox imbalance, and increased oxidative stress. 16 In various tumor cells, elevated ROS levels mediate endoplasmic reticulum (ER) stress. 17 ER stress regulates several signaling cascades, including ER-associated protein degradation and glucose-regulated proteins (GRP). 18 In vitro, overexpression of antisense transcripts and ribozyme-based approaches demonstrated that 78 kDa glucose-regulated protein (GRP78) and 94 kDa glucose-regulated protein (GRP94) may protect against cell death.19–22
The molecular mechanisms of UCNP in human gastric cell lines remain poorly understood. In this study, we investigated UCNP cytotoxicity to SGC-7901 cells and explore its underlying mechanisms.
Materials and methods
Cell culture
The human gastric adenocarcinoma cell line SGC-7901 and the human gastric epithelial cell line GES-1 were obtained from the Key Laboratory of Blood Cancer Center, Ministry of Education, at Jilin University (Jilin, China). Cells were maintained in DMEM (Beijing Dingguo Changsheng Biotechnology, Co., Ltd., Beijing, China) supplemented with 10% FBS, penicillin (100 U/ml, Beyotime Institute of Biotechnology), and streptomycin (100 mg/mL, Beyotime Institute of Biotechnology) at 37°C in 5% CO2.
MTT assay
An MTT assay (Beyotime Institute of Biotechnology) was used to assess GES-1 and SGC-7901 cell viability following exposure to UCNP. 23 Cells were treated with control, 50, 100, 150, 200 or 400 μg/ml UCNP for 24, 48 or 72 h. Subsequently, MTT medium was removed, and 150 μl DMSO (Beijing Dingguo Changsheng Biotechnology, Co., Ltd) was added. Absorbance at 490 nm was recorded with a Synergy™ four Microplate reader (BioTek Instruments, Inc., Winooski, VT, USA).
Flow cytometry (FCM)
FCM with the Annexin V-FITC/propidium iodide (PI) double-labeling method was used to measure the apoptotic rate induced by UCNP. 24 SGC-7901 cells were treated with control, 50, 100, 150, 200 or 400 μg/ml UCNP for 24 h. Cells were incubated with Annexin V-FITC (5 μg/ml, 2019.05-2020.05) (Beyotime Institute of Biotechnology) at room temperature for 15 min and incubated with PI (5 μg/ml, 2019.05-2020.05) at 5°C for 5 min. Samples were analyzed on a BD FACSCalibur flow cytometer (BD Biosciences, San Jose, CA, USA) with CELL-Quest software version 2.7 (Microsoft Corporation, Redmond, WA, USA).
Determination of ROS
SGC-7901 cells (2 × 105 cells/ml) were cultured in 6-well plates for 24 h and exposed to control, 50, 100, 150, 200 or 400 μg/ml UCNP for an additional 24 h. ROS were studied by incubating cells with 10 μ
Mitochondrial membrane potential (ΔΨm) measurement
SGC-7901 cells were maintained in DMEM supplemented with 10% FBS at 37°C in an atmosphere containing 5% CO2 for 24 h. SGC-7901 cells (2 × 105 cells/ml) were seeded in 6-well plates, and 24 h later, they were exposed to UCNP (control, 50, 100, 150, 200 or 400 μg/mL) for 24 h. Rh123 (10 μM) was then added to the cell cultures for 30 min at 37°C. 26 Cells were washed twice with PBS and subsequently analyzed using a BD FACSCalibur flow cytometer (BD Biosciences) and CELL-Quest software version 2.7 (Microsoft Corporation).
Measurement of intracellular calcium
SGC-7901 cells (2 × 105 cells/ml) were seeded in 6-well plates for 24 h and exposed to UCNP (control, 50, 100, 150, 200 or 400 μg/mL) for another 24 h. Cells were harvested, and 1 × 106 cells per sample were loaded with Fluo-3/AM (8 μmol/l) at 37°C for 45 min. 27 Cells were subsequently analyzed using BD FACSCalibur flow cytometry (BD Biosciences) and CELL-Quest software version 2.7 (Microsoft Corporation).
Terminal deoxyuridine triphosphate nick-end labeling (TUNEL) assay
SGC-7901 cells (2 × 105 cells/ml) were seeded in 6-well plates and maintained in DMEM supplemented with 10% FBS at 37°C in an atmosphere containing 5% CO2 for 24 h and exposed to UCNP (control, 50, 100, 150, 200 or 400 μg/mL) for an additional 24 h. The cells were fixed with 4% paraformaldehyde at 30°C for 60 min. Cells were harvested, and 1 × 106 cells per sample were loaded with TUNEL detection solution at 37°C for 60 min. 28 Cells were subsequently analyzed using a BD FACSCalibur flow cytometer (BD Biosciences) and CELL-Quest software version 2.7 (Microsoft Corporation, Redmond, WA, USA).
Determination of caspase activity
Treated cells were collected and washed with PBS and then incubated with cell lysis buffer on ice for 30 min. Supernatants were collected, and the protein concentration was measured. Cell lysates were contained in 96-well plates, and specific caspase substrates were added according to the manufacturer’s protocol. 29 Cell lysates were incubated at room temperature for 1 h, and the fluorescence intensity was determined by absorbance. Absorbance was measured at 405 nm using a Synergy™ four microplate reader (BioTek Instruments, Inc., Winooski, VT, USA).
Western blot
SGC-7901 cells exposed to control, 50, 100, 150, 200 or 400 μg/ml UCNP for 24 h were incubated with cell lysis buffer on ice for 30 min. For the extraction of Cyt C in cytoplasm, collect cells (1 × 106/ml) in ice bath precooled homogenate buffer (250 mmol/L Sucrosse, 20 mmol/L Hepes/KOH, pH 7.5, 10 mmol/L KCI, 1.5 mmol/L MgCl2, 1 mmol/L EDTA, 1 mmol/DTT, 0.1 mmol/L PMSF) 400 μL Resuspended, transferred into a glass homogenizer, homogenized in an ice bath for 5 min, the homogenized solution centrifuged at 4°C for 10 min, twice in total, and the supernatant was centrifuged at 4°C for 15 min. The collected supernatant was the cytoplasmic component without the nucleus and mitochondria. Protein concentration was measured in supernatants. Samples were separated by 8% SDS‒PAGE and transferred onto polyvinylidene fluoride membranes. Primary antibodies were rabbit anti-Cyt C (1:1000; Cell Signaling Technology, USA, 11,940), rabbit anti-Bcl-2 (1:500; Cell Signaling Technology, USA, 3498), rabbit anti-Bax (1:500, Cell Signaling Technology, USA, 2774), rabbit anti-Akt (1:500, Cell Signaling Technology, USA, 5373), rabbit anti-phospho-Akt (1:1000; Cell Signaling Technology, USA, 5012), rabbit anti-caspase-3 (19/17 KD) (1:1000; Cell Signaling Technology, USA, 9662), rabbit anti-caspase-9 (35 KD) (1:1000; Cell Signaling Technology, USA, 9502), rabbit anti-GRP-78 (1:1000; Cell Signaling Technology, USA, 89,947), rabbit anti-GRP-94 (1:1000; Cell Signaling Technology, USA, 20,292), rabbit anti-calpain-1 (1:1000; Cell Signaling Technology, USA, 2556) and rabbit anti-calpain-2 (1:1000; Cell Signaling Technology, USA, 70,655). The secondary antibodies were horseradish peroxidase (HRP)-conjugated goat anti-rabbit polyclonal secondary antibody (1:10,000; Beyotime Institute of Biotechnology, A0208). Detection was performed by enhanced chemiluminescence. Images were captured on Kodak radiographic film (Kodak, Rochester, NY, USA) in the dark. Grayscale analysis of the western blot images was conducted with Photoshop (Microsoft Corporation). 30
Statistical analysis
Statistical analysis was performed using SPSS v19.0 for Windows (IBM SPSS, Armonk, NY, USA). Each experiment was independently replicated at least 3 times, Data are expressed as the mean ± standard deviation. Student’st test was applied to the difference between the experimental groups and the control group. Differences with a p < 0.05 were considered statistically significant, p < 0.01 were considered significant statistical difference, p < 0.001 were considered very significant statistical difference. The resulting graph was plotted using GrapPad prism (version8.3.0).
Results
UCNP inhibited SGC-7901 cell viability in a concentration and time-dependent manner
Exposure to 50–400 μg/ml UCNP for 24 h, 48 h, or 72 h inhibited SGC 7901 cell viability in a concentration- and time-dependent manner compared to controls. Exposure to 100–400 μg/ml UCNP for 24 h or 48 h (p < 0.05) or 50–400 μg/ml UCNP for 72 h (p < 0.01) significantly inhibited the viability of SGC-7901 cells compared to control cells. Exposure to 400 μg/ml UCNP for 24 h or 150–400 μg/ml UCNP for 72 h significantly inhibited the viability of GES-1 cells compared with the control cells (p < 0.05) (Figure 1). UCNP significantly reduce the survival rate of SGC-7901 cells (A). Cell viability in SGC-7901 and GES-1 cells treated with control or 50–400 μg/ml UCNP for 24 h (B). Cell viability in SGC-7901 and GES-1 cells treated with control or 50–400 μg/ml UCNP for 48 h (C). Cell viability in SGC-7901 and GES-1 cells treated with control or 50–400 μg/ml UCNP for 72 h. *p < 0.05, **p < 0.01 and***p < 0.001 vs. control. UCNP, upconversion nanoparticles.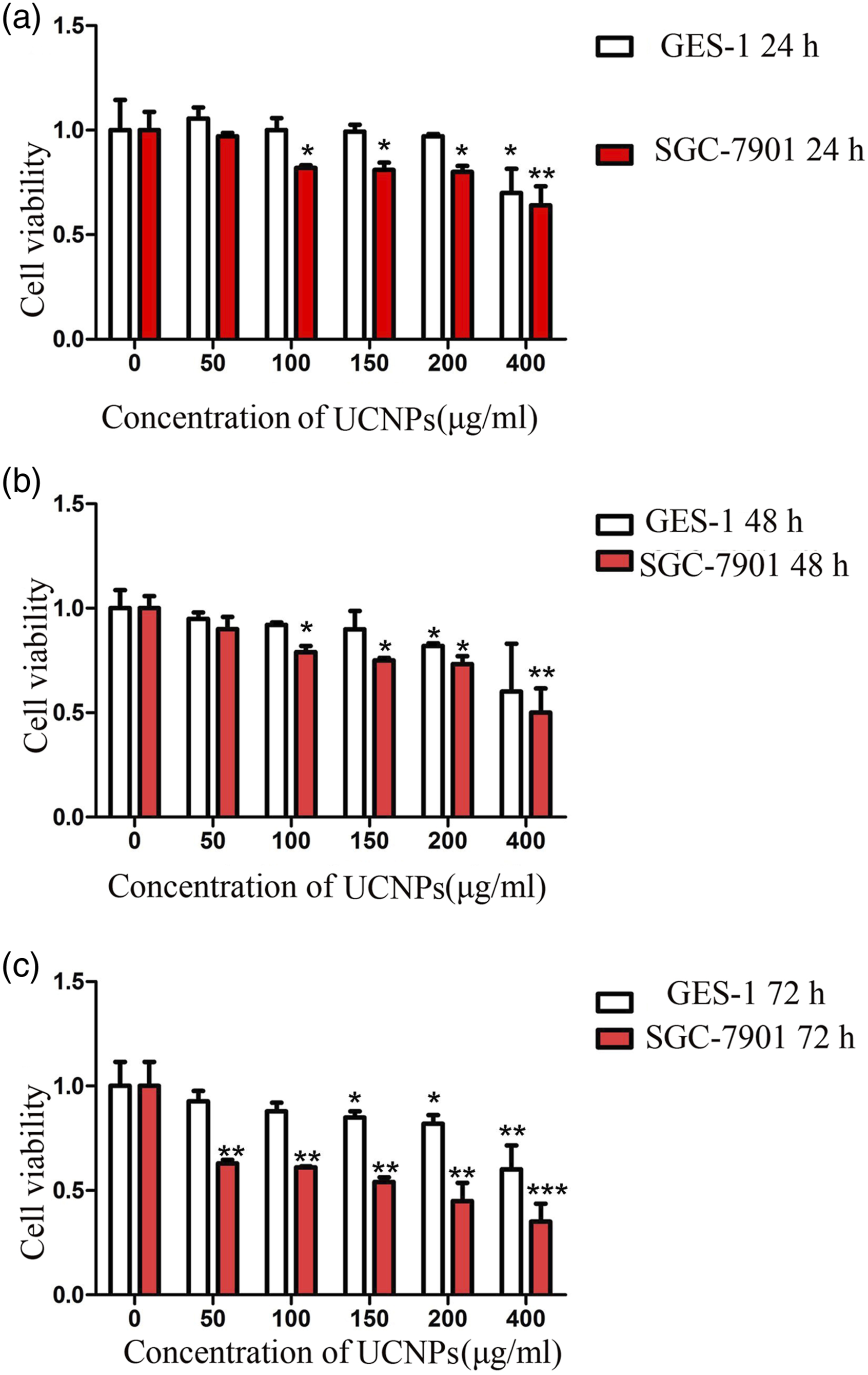
UCNP induced SGC-7901 cell death and upregulated BAX but downregulated BCL-2 protein expression
We selected GES-1 cells and SGC-7901 cells and observed the cell survival rates in different concentrations of UCNP for 24 h, 48 h and 72 h. As shown above, cell death occurred 24 h before the action of UCNP, and we further explored the possible mechanism regarding the cell death caused by UCNP acting on cells for 24 h. Flow cytometry revealed that exposure to 400 μg/ml UCNP for 24 h induced SGC-7901 cell death (Figures 2A and 2B). Western blot analysis demonstrated that exposure to 50–400 μg/ml UCNP for 24 h increased Bax and decreased Bcl-2 protein levels in SGC-7901 cells compared with control cells in a concentration-dependent manner. Exposure to 400 μg/ml UCNP for 24 h significantly increased Bax protein levels (p < 0.05), and exposure to 150–400 μg/ml UCNP for 24 h (p < 0.01) significantly decreased Bcl-2 protein levels in SGC-7901 cells compared to controls (Figure 2C). UCNP induced SGC-7901 cell death, upregulated BAX protein expression, and downregulated BCL-2 protein expression (A). Flow cytometry of SGC-7901 cells treated with control or 50–400 μg/ml UCNP for 24 h (B). Proportion of apoptotic SGC-7901 cells (C). Western blot of Bcl-2 and Bax protein *p < 0.05 and **p < 0.01 vs. control. UCNP, upconversion nanoparticles; Bcl-2, B-cell lymphoma-2; Bax, Bcl-2-associated X protein.
Mitochondrial dysfunction was involved in UCNP-induced cell death
Disruption of mitochondrial integrity is one indicator of early apoptosis.
31
Exposure to 50–400 μg/ml UCNP for 24 h significantly decreased ΔΨm in SGC-7901 cells in a concentration-dependent manner compared with control cells (p < 0.05; Figure 3A). Exposure to 200 or 400 μg/ml UCNP for 24 h significantly increased intracellular Ca2+ in SGC-7901 cells compared to control cells (p < 0.05; Figure 3B). TUNEL results indicated that exposure to 200 or 400 μg/ml UCNP for 24 h significantly induced apoptosis in SGC-7901 cells compared to the controls (p < 0.05; Figure 3C). Release of CytC usually accompanies a decrease in ΔΨm.
32
Western blot revealed that exposure to 400 μg/ml UCNP for 24 h significantly increased Cyt C protein levels in SGC-7901 cells compared to control cells (p < 0.05; Figure 3D). The PI3K-Akt signaling pathway regulates tumor cell proliferation and resistance to chemotherapy.
33
Western blot revealed that exposure to 400 μg/ml UCNP for 24 h significantly decreased phosphorylated Akt protein levels in SGC-7901 cells compared to controls, which may have proapoptotic effects (p < 0.05; Figure 3E). These results imply that UCNP induced mitochondrial dysfunction and activated apoptosis-related pathways in SGC-7901 cells. UCNP induced cell death through mitochondrial inactivation (A). ΔΨm of SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h (B). intracellular Ca2+ of SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h (C). TUNEL staining of SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h (D). Western blot of CytC protein of SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h. E. Western blot of p-Akt and Akt proteins of SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h. *p < 0.05, **p < 0.01 and ***p < 0.001 vs. control. UCNP, upconversion nanoparticles; ΔΨm, mitochondrial membrane potential; TUNEL, terminal deoxynucleotidyl transferase dUTP nick-end labeling; Cyt C, cytochrome C; Akt, protein kinase B; p-Akt, phosphorylated-Akt.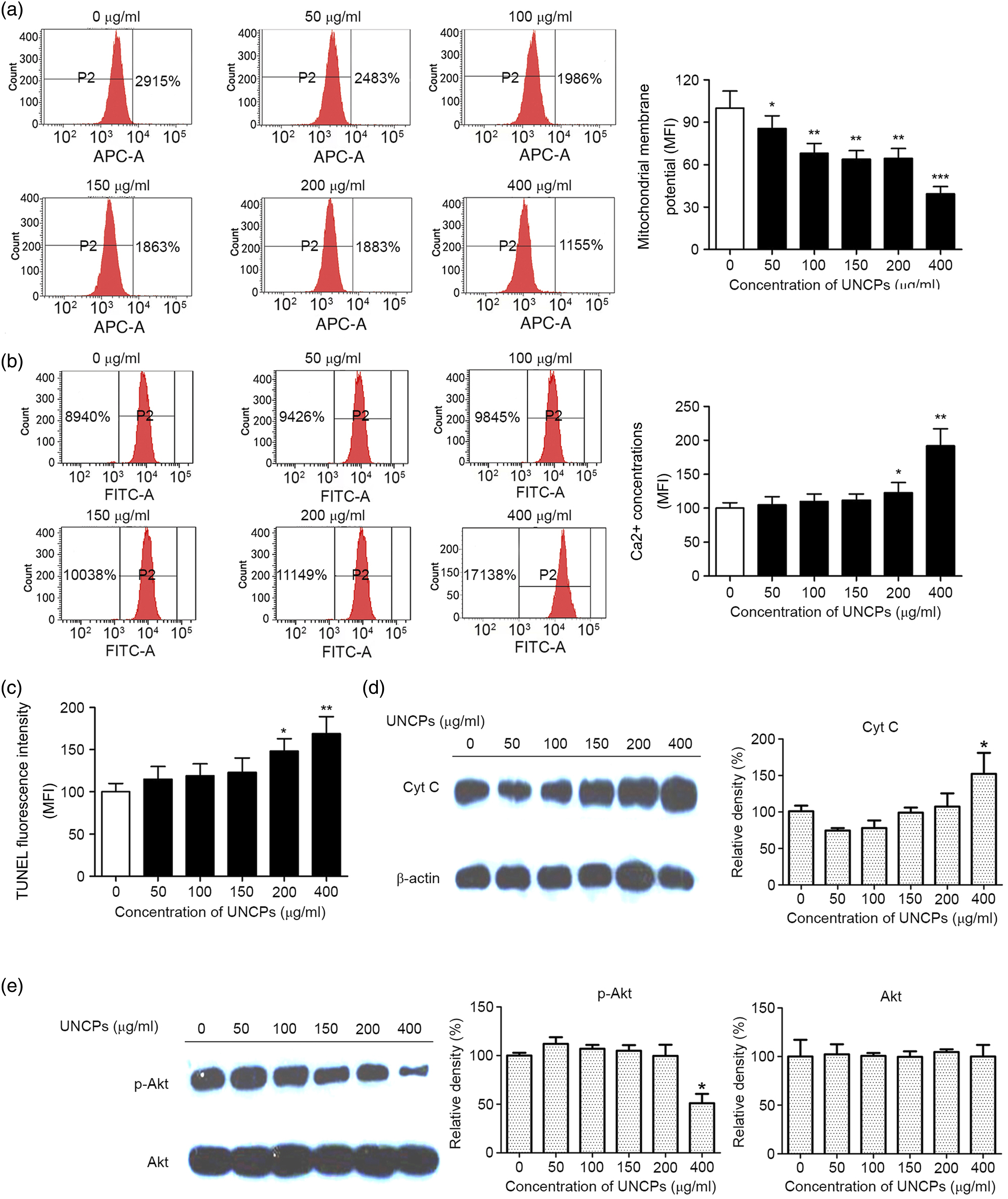
UCNP regulated cell apoptosis and death by triggering the mitochondrial pathway
Caspase-3 is a crucial mediator of apoptosis, and caspase-9 may trigger the mitochondrial apoptotic pathway.
34
Exposure to 50–400 μg/ml UCNP for 24 h significantly increased activated caspase-3 activity and protein levels in SGC-7901 cells in a concentration-dependent manner compared to controls (p < 0.05), with the greatest increase seen in SGC-7901 cells exposed to 400 μg/ml UCNP (p < 0.01). Exposure to 400 μg/ml UCNP for 24 h significantly increased activated caspase-9 activity and protein levels in SGC-7901 cells compared to control cells (p < 0.05, Figure 4A–D). These results imply that UCNP regulated apoptosis-related protein expression and induced SGC-7901 cell death by triggering the mitochondrial pathway. UCNP regulated apoptosis-related protein expression and induced SGC-7901 cell death by triggering the mitochondrial pathway (A). SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h activated caspase-3 activity (B). SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h activated caspase-9 activity (C and D). Western blot of activated caspase-3 (19/17KD) and activated caspase-9(35KD) proteinsof SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h. *p < 0.05 and **p < 0.01 vs. control. UCNP, upconversion nanoparticles.
UCNP induced apoptosis in SGC-7901 cells through ROS-mediated ER stress
ROS are essential for cell proliferation and cell death.
35
Exposure to 50–400 μg/ml UCNP for 24 h significantly increased ROS levels in SGC-7901 cells compared to control cells in a concentration-dependent manner (p < 0.05; Figure 5A), with the greatest increase seen in SGC-7901 cells exposed to 400 μg/ml UCNP (p < 0.001; Figure 5A). Numerous studies have indicated that ROS induce cell death through activation of ER stress. GRP mediates ER homeostasis.
36
Exposure to 400 μg/ml UCNP increased GRP-78 and GRP-94 protein levels in SGC-7901 cells compared to control cells in a time-dependent manner (Figure 5B). Exposure to tunicamycin (TM), an inducer of ER stress, increased GRP-78 and GRP-94 protein levels in SGC-7901 cells compared to control cells (Figure 5B). Exposure to 400 μg/ml UCNP increased calpain-1 and calpain-2 protein levels in SGC-7901 cells compared to control cells in a time-dependent manner (Figure 5(C)). These results imply that UCNP induced apoptosis in SGC-7901 cells through mitochondrial dysfunction and ROS-mediated ER stress, subsequently promoting the caspase-9/caspase-3 cascade (Figure 5D). Molecular mechanism of UCNP-induced SGC-7901 apoptosis (A). ROS levels of SGC-7901 cells treated with control, 50–400 μg/ml UCNP for 24 h (B and C). Western blot of GRP-78, GRP-94, calpain-1 and two proteins of SGC-7901 cells treated with 400 μg/ml UCNP for 6–24 h (D). Schematic of the underlying molecular mechanism of UCNP-induced SGC-7901 apoptosis. *p < 0.05, **p < 0.01 and***p < 0.001 vs. control. UCNP, upconversion nanoparticles; ROS, reactive oxygen species; ER, endoplasmic reticulum; GRP, glucose-regulated protein.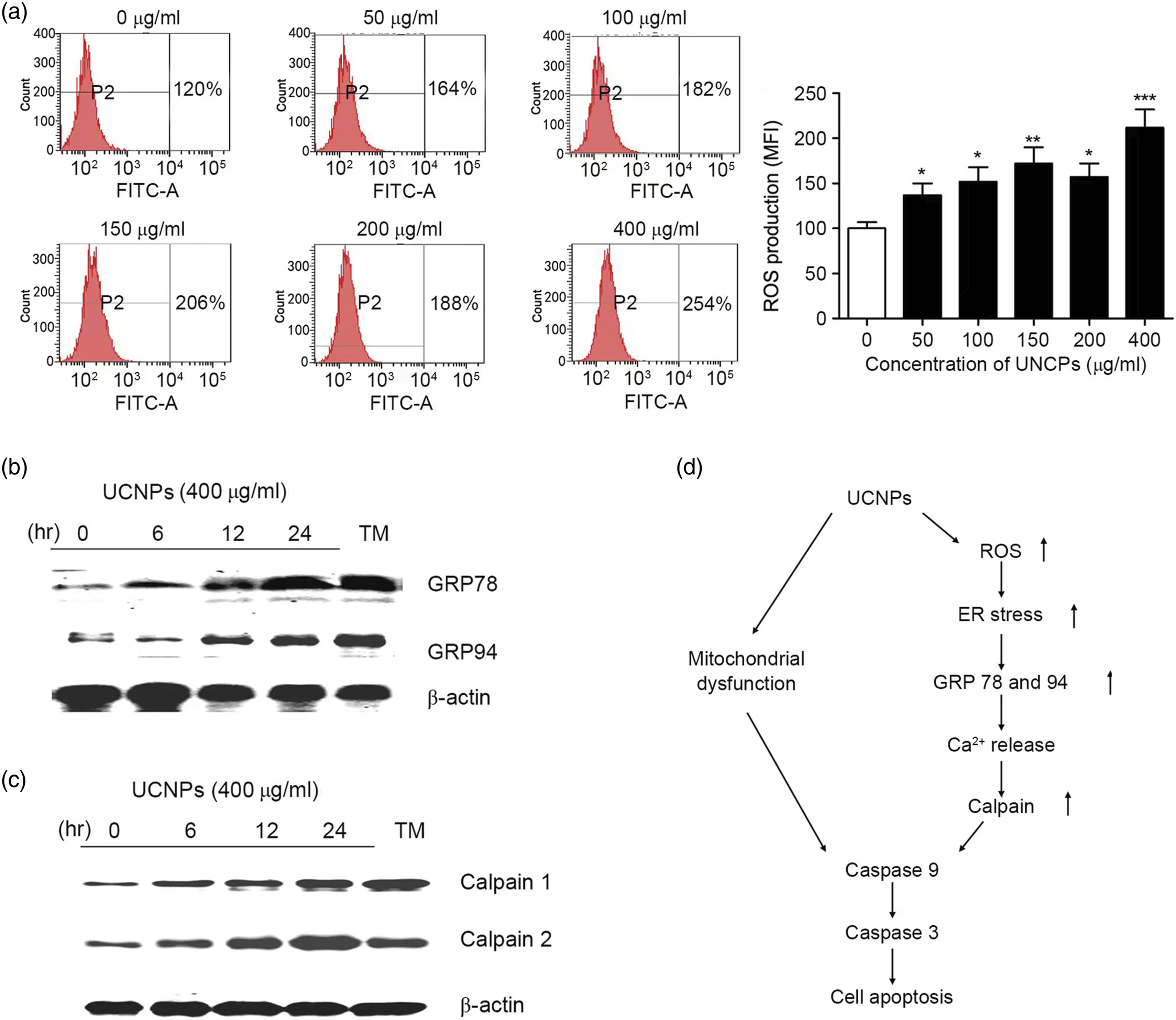
Discussion
UCNP are currently being developed as fluorescent probes for biomedical applications. In our early experiment, UCNP with different concentrations acted on GES-1 and SGC-7901 cells for 24 h, and UCNP with a capacity of 200 μg/ml had the best imaging ability. In further experiment, we observed that with 200 μg/ml UCNP, the survival rate of GES-1 cells was almost 100%, and the survival rate of SGC-7901 cells was more than 80%. Evidence suggests that 200 μg/ml UCNP have notable fluorescence imaging capabilities and biocompatibility, 37 and NaYF4:Yb3+/Er3+ (Tm3+) is the most efficient upconversion nanomaterial. After 24 h of treatment with UCNPs, the survival rate of SGC-7901 cells decreased significantly in a concentration-dependent manner, and the survival rate was the lowest when treated with 400 μg/mL UCNPs. The survival rate of GES cells did not decrease with the increase of UCNPs concentration, so we concluded that the endogenous differences between GES cells and SGC-7901 cells resulted in their different survival rates against UCNPs. Exposure to 400 μg/ml UCNP for 24 h increased the proportion of SGC-7901 cells in early apoptosis, as well as late apoptosis. Early apoptosis means that cells undergo a series of biochemical and morphological changes before apoptosis occurs. 38 These changes include cell size reduction, cell morphological changes, nucleus fragmentation and chromatin aggregation, these changes are caused by intracellular enzyme systems, including cysteine protease, endoplasmic reticulum protease and ribonuclease. Late apoptosis means that cells undergo more obvious morphological and biochemical changes on the basis of early apoptosis. These changes include the rupture of the cell membrane, the release of enzyme systems within the cell, and the production of toxic substances by the cell, these changes lead to the complete death and breakdown of cells. Early apoptosis and late apoptosis are different stages of apoptosis, and early apoptosis is the early stage of apoptosis, which mainly shows the changes of cell morphology and biochemistry. Late apoptosis is the late stage of apoptosis, and the process of cell death and decomposition is more obvious. 39 Apoptosis is a type of programmed cell death that can be initiated by the death receptor-mediated extrinsic pathway and/or the mitochondria-mediated intrinsic pathway. 40 ROS may trigger mitochondrial-mediated apoptosis. 41 Exposure to 50–400 μg/ml UCNP for 24 h significantly increased ROS levels in SGC-7901 cells compared to controls in a concentration-dependent manner, with the greatest increase seen in SGC-7901 cells exposed to 400 μg/ml UCNP. Tumor cells have higher ROS levels and sensitivity than normal cells, which can result in tumor cell damage or death. 42 ROS levels in SGC-7901 cells exposed to 400 μg/ml UCNP were significantly higher than those in the control group, which corresponded with a decrease in ΔΨm, increase in intracellular Ca2+, elevated cytoplasmic expression of CytC, increased Bax and decreased Bcl-2 expression. These changes are markers of early apoptosis, and these findings imply that SGC-7901 cell death occurred through the mitochondrial-mediated pathway. Dissipation of ΔΨm across the mitochondrial membrane is considered an early apoptosis signal that leads to irreversible damage. 43 The BCL-2-protein family regulates Cyt C release. 44 Cyt C is a pro-apoptotic factor that binds with apoptotic protease activating factor-1 (APAF-1) to activate pro-caspase-9. 45 Caspase-3 is involved in a cascade of reactions that ultimately lead to apoptosis.
The PI3K-AKT signaling pathway regulates tumor cell proliferation and resistance to chemotherapy. 33 UCNP inhibit Akt phosphorylation at the Ser473 site, which promotes the mitochondrial apoptotic pathway and may underlie UCNP-induced SGC-7901 cell death. In the present study, exposure to 400 μg/ml UCNP increased GRP-78, GRP-94, calpain-1 and two protein levels in SGC-7901 cells in a time-dependent manner, and exposure to TM, an inducer of ER stress, increased GRP-78 and GRP-94 protein levels in SGC-7901 cells compared with control cells. Calpain-1 and two are two cysteine protease families that regulate pathological cell death. 46 These data imply that UCNP induced SGC-7901 cell death through the activation of ER stress. Briefly, UCNP induced SGC-7901 cell death by promoting mitochondrial dysfunction and ROS-mediated ER stress, initiating the caspase-9/caspase-3 cascade.
Endoscopy is a special clinical examination method for hollow organs that plays an important role in the diagnosis of digestive tract diseases (especially tumors) and has become the first choice for gastric cancer screening. UCNP are soluble in water and can enter cells and emit strong light. UCNP have the advantages of stable physical properties, strong tumor specificity and high affinity. UCNP (200 μg/ml) are a promising near-infrared imaging dye. The wavelength of near-infrared fluorescence is between 700 and 900 nm, with high tissue penetration (centimeter level). In the future, UCNP (200 μg/ml) will be chemically combined with gastric cancer-specific antibodies to construct a gastric cancer molecular imaging probe with a near-infrared bimodal imaging function. Photodynamic therapy (PDT) is a new and promising tumor treatment method. Its main mechanism is to stimulate the energy transfer between photosensitizers and adjacent oxygen molecules under laser irradiation and then generate a large number of ROS to inhibit tumors.
Conclusion
Collectively, UCNP can effectively accumulate at the tumor site through passive targeting, while UCNP (400 μg/ml) can induce more ROS generation. If UCNP are combined with photosensitizers, more ROS can be generated. UCNP can be designed and prepared for the integration of gastric cancer diagnosis and treatment. Through targeted imaging of gastric cancer combined with phototherapy, a new idea is provided to solve the current dilemma of gastric cancer diagnosis and treatment.
Footnotes
Author contributions
CL: Conceptualization, Methodology, Software, Investigation, Formal Analysis, Writing-Original Draft. SQS: Data Curation, Writing-Original Draft; JWM: Conceptualization, Funding Acquisition, Resources, Supervision, Writing-Review & Editing. CL and SQS confirm the authenticity of all the raw data. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
