Abstract
Carbon tetrachloride (CCl4) is a toxic chemical that causes liver injury. CCl4 triggers endoplasmic reticulum (ER) stress and unfolded protein response (UPR). UPR triggers autophagy to deal with the damage. The aim of this study was to investigate the effect of baicalein, derived from
Introduction
Hepatic fibrosis is an inappropriate healing response to chronic liver damage caused by viruses, toxins, alcohol, and various factors, and is characterized by excessively secreted extracellular matrix components. If left untreated, it develops into liver dysfunction, cirrhosis, and hepatocellular carcinoma (HCC). Fibrosis occurs by the conversion of hepatic stellate cells into myofibroblast cells secreting extracellular matrix components. 1 Induction with CCl4 is the most commonly used model for the development of hepatic fibrosis. Profibrotic cytokines such as transforming growth factor beta1 (TGF-β1), tumor necrosis factor-alpha (TNF-α), and interleukin-6 (IL-6) have been reported to play an important role in CCl4-induced hepatic fibrosis. 2
CCl4 is converted by the cytochrome P450 2E1 into the trichloromethyl (CCl3−) free radical, which is reactive in the liver.3,4 The free radicals attack the unsaturated fatty acids in the membranes, resulting in lipid peroxidation, which leads to disruption of the membrane structure and protein synthesis. 3 For P450 enzymes to be activated, NADPH-P450 must transfer electrons from the reductase enzyme, during which some electrons leak from the system, which in return causes reactive oxygen species (ROS) to form.
The ER is an important organelle for cell survival. In ER, proteins are folded into functional three-dimensional states via chaperones. When the amount of unfolded proteins in the ER is greater than the existing chaperones, the unfolded proteins cause ER stress. This stress in the ER leads to a signal called UPR. UPR is triggered by GRP78 by detecting misfolded protein accumulation in the ER. While GRP78 stimulates chaperone BIP production which in turn activates ER stress-related proteins IRE1, PERK, and ATF6, 5 another ER stress-related protein ATF4 induces translation of numerous mRNAs including antioxidant enzymes, chaperones, and apoptosis-related genes including CHOP. Activated IRE1 excises an intron from XBP1 mRNA resulting in the formation of s-XBP1. The balance between XBP1/s-XBP1 is important for the expression of cytokines and protein folding capacity of the ER. If the cell cannot overcome ER stress, it stimulates autophagy. UPR, by stimulating autophagic destruction, tries to reduce the burden of unfolded proteins and damaged organelles to the cell. 6 If autophagy-mediated UPR fails to provide the necessary homeostasis, apoptosis is triggered in the cell. 7 Therefore, the fate of a cell under ER stress depends on the balance between UPR and autophagy. 8
Autophagy is an evolutionarily highly conserved process that leads to homeostasis by degrading molecules and organelles that are damaged in the cell via directing them to the lysosome, surrounded by double membranes called autophagosomes.9–11 Autophagosome formation is initiated by phosphoinositide 3 kinase (PtdIns3K), vacuolar protein sorting 34 (Vps34), and Beclin-1, which is controlled by ULK1, Atg13, and Atg17. PtdIns3P, generated by Beclin1/Vps34, is required for the docking of other Atg proteins to the sites. Atg proteins maintain the autophagosome structure. 12 The initial phase is followed by the formation of the Atg5 (autophagy-related 5)-Atg12-Atg16 complex, which converts LC3I to LC3II, and brings LC3II into the membrane to bind Atg7 (autophagy-related 7) and Atg3. Atg proteins are then cleaved on the cytosol for reuse, and LC3II remains in the membrane until the autophagosome merges with the lysosome.
Flavonoids, which are abundant in the structure of vegetables, fruits, and various plants, can prevent cellular damage caused by free radicals due to their antioxidant activity. Flavonoids can reduce ROS levels either by scavenging them or by activating enzymes that reduce ROS indirectly.
13
Baicalein is an active phytochemical found in
In summary, although there are several studies in the literature showing that baicalein induces autophagy through ER stress, there is no study showing the effect of baicalein on ER stress and autophagy combined with TEM analysis in CCl4-induced liver injury. This study will enlighten the role of baicalein on both ER stress and autophagy mechanisms.
Methods
Animals and study design
This study was performed under the rule of Eskisehir Osmangazi University Faculty of Medicine Ethics Committee (approval no: 111, meeting no: 595 on May 23, 2017) and during all stages of the experiment recommendation of Guide for the Care and Use of Laboratory Animals of the National Institutes of Health were followed. 250–300 g 42 female Wistar albino rats were obtained from Eskisehir Osmangazi University Medical Faculty Medical Surgery Research Center (TICAM) and placed in a room where the temperature is around 22 ± 2°C, humidity is 45–50% with 12/12 h light/dark cycles during the whole experiment. Rats were fed with standard laboratory chow and tap water ad libitum. 42 rats were divided into six groups randomly, each having seven rats, after a week of acclimatization (Table 1). To form random groups, first, rats were divided into two blocks according to body weight (250–275.5 and 275.6–300 g), and then within each block rats were placed in groups by drawing lots. Experimenters who carried out the histological analysis, TEM analysis, and ELISA marker measurements were blinded to the groups.
Experimental Groups and applied substances.

i.p., intraperitoneal; i.g., intragastric; DMSO, dimethyl sulfoxide.
CCl4 (Sigma-Aldrich; Product code: 289116, density: 1.594 g/mL) was dissolved in olive oil (1:1) and administered intraperitoneally as 0.2 ml/kg (i.p.) twice a day for 10 days. To the treatment group, baicalein (abcam, AB179832) was dissolved in dimethyl sulfoxide (DMSO) as 33 mg/ml and was administered intragastrically by gavage as 100 mg/kg for subsequent 14 days (day 11–24) after 10 days of CCl4 treatment was finished. DMSO (3.3 ml/kg), olive oil (0.2 ml/kg), and baicalein (100 mg/kg) alone treated groups were used as sham control groups. After the administration of substances, animals were sacrificed under anesthesia with ketamine/xylazine (90/10 mg/kg, intramuscular [i.m.]), and liver tissue samples were obtained.
Histopathological examination
The liver tissue samples were fixed with 10% formalin for 2 days and followed by dehydration by placing them respectively in 70%, 80%, 90%, and 96% alcohol series for 45 min. Then tissue samples were placed in xylene for 20 min and later embedded in paraffin wax at 65°C. 5 µm sections were cut by microtome from paraffin blocks, washed with xylol for 1 h to remove paraffin, and stained with hematoxylin-eosin. After that, stained samples were dehydrated with alcohol again and placed in xylol for 30 min, twice. Finally, these samples were coated with entellan for histological examinations under the Olympus BH-2 light microscope.
Transmission electron microscopy
Samples were fixed primarily with 2.5% glutaraldehyde containing 0.1 M phosphate buffer at 4°C for 24 h and washed with phosphate buffer for 15 min 3 times. For secondary fixation, samples were treated with 1% osmium tetroxide containing 0.1 M phosphate buffer at room temperature in rotator for 2 h, which were followed by washing again as indicated above. Later, dehydration was performed with increasing ethyl alcohol series (30%, 50%, 70%, 90%, 96%, and 100%) at 4°C twice, and propylene oxide was administered twice for 30 min. Samples were kept in 1:1 propylene oxide-araldite for 2 h in rotator and then placed in pure araldite overnight in rotator. After overnight incubation, samples were embedded into araldite and polymerized at 60°C for 2 days. 60 nm thickness sections were cut by ultramicrotome (Leica Ultracut R) and placed in 300 mesh copper grids to stain with uranyl acetate-lead citrate. These samples were examined by TEM (JEOL JEM 1220).
Determination of autophagy and endoplasmic stress markers
0.3 g liver tissues were weighed and placed in 0.3 ml phosphate-buffered saline (pH 7.4), and homogenized by ultrasonic homogenizer according to ELISA marker instructions (Shanghai YL Biotech Co., Ltd.). Total protein levels were determined by Sigma, Total protein kit (Product code: TP0100). The markers of ER stress: ATF4 (Cat no: YLA1404RA), ATF6 (Cat no: YLA1403RA), CHOP (Cat no: YLA1471RA), IRE1 (Cat no: YLA1405RA), GRP78 (Cat no: YLA0246RA), PERK (Cat no: YLA1406RA), and XBP1s (Cat no: YLA1407RA) and autophagy markers Atg5 (YLA1418RA), Atg7 (YLA1419RA), Beclin1 (Cat no: YLA0731RA), LC3-II (YLA1417RA), and Vps34 (YLA1420RA) were measured according to Shanghai YL Biotech Co., Ltd. ELISA kit protocols.
Statistical analysis
All data were analyzed with IBM SPSS 21 software. Descriptive statistics of the variables were shown as mean ± standard deviation. Normality was checked by the Shapiro–Wilk test. Comparisons of the groups were done with one-way analysis of variance (ANOVA) analysis. Statistically significant differences between groups were evaluated by Bonferroni test for multiple comparisons, and
Result
Histological examinations
In CCl4 administered group, dense vacuolization in especially pericentral regions was observed in addition to cellular degeneration, central vein congestion, and partial cellular infiltration. In the treatment group, damage at pericentral regions was still present at a reduced rate. Cellular degeneration and vacuolization were lowered compared to the CCl4 group. There was no sign of congestion of the central vein. Sham and baicalein treated groups were found to have normal liver histology (Figure 1).

Histological examination of liver tissues. Control (
TEM analysis
In the control group, hepatocytes were observed as a normal appearance with healthy cytoplasm and nucleus (Figure 2). Hepatocyte nucleus was round or oval-shaped, and chromatin was distributed homogenously. Intact healthy mitochondria, abundant rough endoplasmic reticulum (ER) were observed. In CCl4 treated groups, dense vacuolization was observed especially near mitochondria. Lowered ER quantity, rupture and swellings of some ER were determined. Electron density of mitochondria was found very high, and some mitochondria have been shown to have atypical appearance and disrupted double membrane. Chromatin condensation at the periphery of nucleus was also detected. In the baicalein group, atypical damaged mitochondria, irregularities in cristae, breaks, swelling, and ruptures in the outer membrane were observed. Autophagosomes containing some digested material were traced. Chromatin condensation at the periphery of nucleus was also detected. In the baicalein treatment group, autophagosome and vacuole numbers were highly increased. Lesser mitochondrial damage was observed compared to CCl4 group. Dense glycogen and locally autophagosomes were observed in the cytoplasm. In addition, lipid particles and apoptotic nuclei were observed in the cytoplasm.

Transmission electron microscope examination of hepatocytes. Normally shaped, euchromatic nucleus (N), healthy mitochondrion (M), and normal structured Endoplasmic reticulum (ER) were observed in control group
Endoplasmic reticulum stress and autophagy markers
There were no significant differences regarding endoplasmic reticulum stress markers between sham groups (control, olive oil, and DMSO groups) (
Effects of baicalein on endoplasmic reticulum markers.
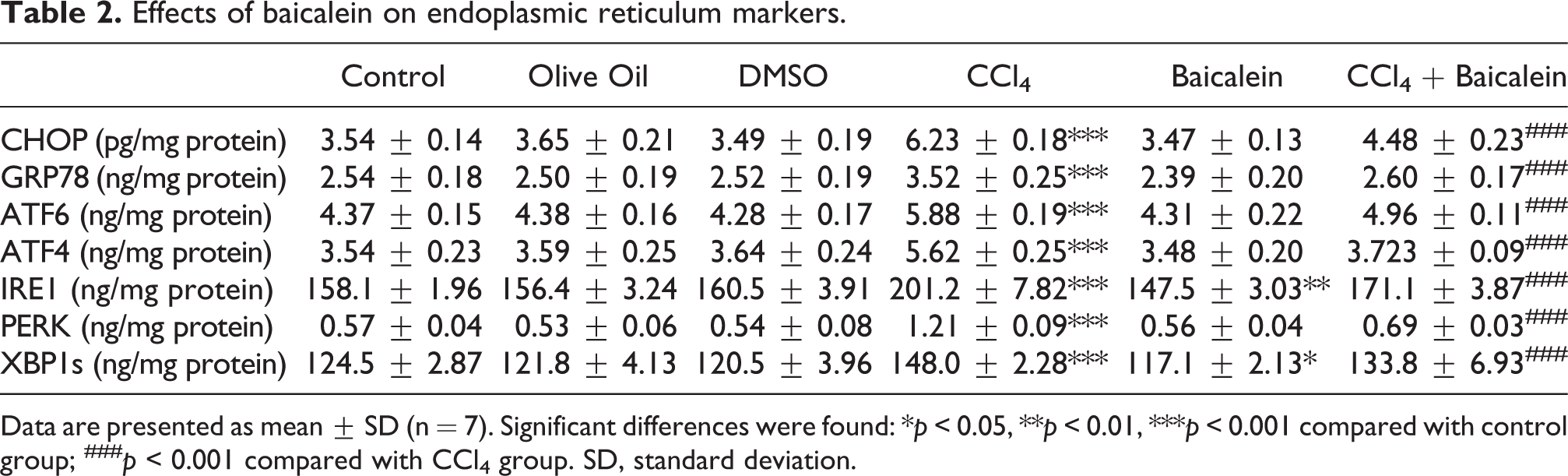
Data are presented as mean ± SD (n = 7). Significant differences were found: *
Regarding autophagy markers, there were no significant differences between control, olive oil, DMSO, and CCl4 treated groups (
Effects of baicalein on autophagy markers.

Data are presented as mean ± SD (n = 7). Significant differences were found: ***
Discussion
The liver is a very important organ that provides the homeostasis of our body, detoxification, and bioactivation of various toxins, drugs, and xenobiotics. Clearance of toxicity in the liver is very crucial for the maintenance of liver and body health.24,25 The liver initiates the hepatic fibrosis process to treat the damage caused by exposure to various agents, and if it fails to treat the damage or chronic exposure to the agent continues, fibrosis leads to cirrhosis and later HCC. 26 Exposure of liver to the toxins causes the increase of unfolded proteins and triggers ER stress. Unfolded proteins and damaged organelles due to ER stress are eliminated by the autophagy pathway, if it is not successful, it drives the cell to apoptosis. 27
It was shown that the bioactivated form of CCl4 causes ER stress.28,29 Fang and Lin 30 demonstrated that CCl4 caused lipid peroxidation and development of liver fibrosis on mice. Li et al. investigated the effect of resveratrol treatment on CCl4 treated HepG2 cells, and in this study, XBP-1s, CHOP, IRE1, ATF6, ATF4, eIF2a, PERK, GRP78, IRE1, and PERK protein levels were found higher in the CCl4 treated group, whereas resveratrol treatment decreased these protein levels. 31 In another study, PERK, ATF6, and IRE1 protein levels were significantly increased in the CCl4 treated group, and when treated with melatonin, these protein levels were decreased. 29
There are various studies in the literature showing that baicalein has hepatoprotective effects. Choi et al. 32 has shown that baicalein treatment reduced the level of ER stress-associated proteins like CHOP, GRP78, XBP1s on ER stress-induced HT22 mouse hippocampal neuronal cells. Moreover, reduction in apoptosis and ROS accumulation was also observed. Our results showed that CCl4 treatment significantly elevated the level of ER stress markers compared to the control group. This indicated that CCl4-induced ER stress and UPR signaling in hepatocytes. The administration of baicalein for treatment significantly reduced the CCl4-induced ER stress marker levels to control levels. The levels of IRE1 and XBP1s in the baicalein control group were also found significantly lower compared to the control group. These results suggested that the treatment of CCl4-induced rats with baicalein had an inhibitory effect on ER stress on liver cells.
UPR is a cellular response of the cell to alleviate the burden of unfolded proteins and restore ER homeostasis. When UPR is activated, PERK phosphorylates and activates eIF2α which leads to translation of several proteins to reduce protein load in ER. 33 Activated eIF2α also triggers formation of Atg5–Atg12–Atg16 complex and LC3 transformation. 34 Moreover, knockdown of BIP chaperone resulted in inhibition of autophagosome formation in mammalian cells. 35 In the study conducted by Liu et al., 36 it was shown that baicalein-induced autophagy through the heme oxygenase 1 (HO1) molecule in the case of hepatic ischemia. In another study importance of the March5 protein, a mitochondrial ubiquitin ligase, was indicated on autophagy. 37 In that study, the expression levels of Beclin, LC3-I, and LC3-II were increased by baicalein treatment which indicated that baicalein could regulate autophagy by inducing the expression of March5. 37 Wang et al. also determined a significant increase of conversion from LC3-I to LC3-II due to baicalein treatment. 23 In our study, similar to literature autophagy markers ATG5, Beclin1, and LC3-II proteins were significantly elevated in the baicalein control group compared to both control and CCl4 groups. Our results showed that all autophagy marker levels in the baicalein treatment group were significantly increased compared to the control group. All autophagy markers’ levels except for VPS34 were also significantly higher compared to CCl4 treated group. These results showed that baicalein-induced autophagy when given alone. Moreover, administration of baicalein after CCl4-induced damage increased even more autophagy marker levels. This might be due to increased ER stress and ROS formation caused by CCl4 treatment. This result indicated the importance of baicalein as a strong autophagy inducer with the presence of an ER stress.
Yi et al. showed that baicalein treatment increased vesicular structures in TEM images. 38 In our study, dense vacuolization, lowered ER quantity, rupture and swellings of some ER, and atypical appeared electron-dense mitochondria were revealed in the CCl4 treated group by TEM analysis. In the TEM images of the baicalein treatment group, deterioration in the cristae structure of the mitochondria, enlargements in the outer membrane, and increased number of autophagosomes were observed. This shows that baicalein stimulates autophagy in the cell via mitochondria possibly by the March5 pathway. 37
In the experimental liver damage model created with CCl4, microscopic examination of liver tissue samples taken from the CCl4 applied group revealed necrotic regions in the liver, cell infiltration, cytoplasmic swelling, and hepatocyte degeneration characterized by nuclear pyknosis. 39 Lima et al. 40 showed that experimental liver hepatotoxicity created by the administration of CCl4 leads to liver tissue degeneration, necrosis areas, and morphological changes. Our histology results have shown that baicalein treatment reduced cellular degeneration, vacuolization, and damage in pericentral regions. Congestion of the central vein was abolished.
Although there are numerous studies showing the hepatoprotective, anti-inflammatory, antioxidant effects of baicalein, there are also some studies showing hepatotoxic effect of
Baicalein effect should be considered as a complex intertwined network involving inflammatory response, oxidative stress, ER stress, autophagy, and apoptosis. 43 For example, Pang et al. 44 has shown that baicalein-induced Nrf2 activation, which is a transcription factor of several drug-metabolizing enzymes (DMEs). In another study, it was shown that baicalein significantly suppressed the levels of tumor necrosis factor-alpha (TNF-α), Interleukin-6 (IL-6), and monocyte chemotactic protein 1 (MCP-1), and increased the activities of superoxide dismutase (SOD) and glutathione peroxidases (GSH-Px) in the liver. 45 They also determined NF-κβ inhibition by baicalein. Further researches indicated that ER stress resulted in calcium efflux which in return followed by reactive oxygen intermediates (ROI) formation and NF-κβ activation. Regarding all these findings, although the exact mechanisms of baicalein on ER stress and autophagy are not well known, CCl4 triggers ROS formation and ER stress, which leads to the trigger of inflammatory mediators. Inflammatory mediators cause more ROS to form. All these loops might increase the damage to the liver. On the other hand, baicalein might inhibit ER stress by inhibiting inflammatory response, by inducing antioxidant enzyme expression, by directly suppressing ER stress markers, or by triggering autophagy.
In summary, our study showed that CCl4 administration caused increased ER stress and hepatic damage, which was abolished by the autophagy pathway upon treatment of baicalein. While alone baicalein treatment-induced autophagy, the application of baicalein after CCl4 administration induces even more autophagy to clear away ER stress, which in return resulted in decreased cellular and histological damage. In this respect, this study is important for the use of this active substance in the treatment of liver damage and to shed light on future studies.
Footnotes
Author’s note
Z Özdemir Köroğlu is now affiliated with the Department of Medical Laboratory, Vocational School of Health Services, Eskisehir Osmangazi University, Eskisehir, Turkey.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Commission of Scientific Research Project Support Bilimsel Araştırma Projeleri Komisyonu (BAP) Eskisehir Osmangazi University (Turkey).
