Abstract
Peripheral neurotoxicity injury caused by local anesthetics is a common complication of clinical anesthesia. The study of its mechanism is helpful to prevent and treat the neurotoxic injury of local anesthetics. Previous studies on peripheral neurotoxicity injury caused by local anesthetics have mainly focused on in vitro cell experiments. Due to the lack of an animal model of peripheral neurotoxicity damage caused by local anesthetics, there are few in vivo experimental studies regarding this topic. Herein, 1% ropivacaine hydrochloride was injected into the sciatic nerve by direct incision and exposure of the sciatic nerve to create a local anesthetic neurotoxic injury model. The results showed that 1% ropivacaine hydrochloride could reduce the lower limb motor score and mechanical paw withdrawal threshold in mice 48 hours after injection. Pathological sections showed that 48 hours after treatment with 1% ropivacaine hydrochloride, the sciatic nerve showed increased axonal edema and degeneration, edema between nerve fiber bundles, increased degeneration of axon and myelin sheath vacuoles, edema of nerve bundle membrane and local degeneration and necrosis, and a large number of inflammatory cells around the nerve adventitia were soaked. The above results show that under open vision, 1% ropivacaine hydrochloride can cause injury to the sciatic nerve after 48 h of treatment, which can simulate the neurotoxic damage of local anesthetics. This animal model provides a research tool for studying the mechanism of neurotoxic injury caused by local anesthetics.
Introduction
Regional block is a common method for clinical anesthesia and treatment of acute and chronic pain.1–3 Regional block has the advantages of rapid onset, accurate clinical effect and small systemic impact. However, nerve injury caused by regional block is often reported.4–6 Local sensory abnormalities, pain and discomfort, and even abnormal motor function caused by regional blocks adversely affect patients' work and lives. With the development of visualization techniques such as ultrasound,7,8 the incidence of mechanical injury in the process of regional block has gradually decreased, and the neurotoxic injury of local anesthetics has become a common cause of nerve complications in regional block. A multicenter study showed that the incidence of sensory abnormalities caused by local anesthetic peripheral nerve block was 1.5%, and the incidence of nerve injury was 0.21%. Another study showed that the incidence of transient neurological syndrome (TNS) caused by local anesthetics was as high as 7.5%.9,10 Therefore, nerve injury caused by local anesthetics is an urgent problem to be solved in clinical anesthesia and pain treatment. Research on the mechanism of nerve injury caused by local anesthetics has theoretical and clinical significance.
In the past, research on the neurotoxicity of local anesthetics has mainly focused on in vitro cell experiments.11,12 Many studies are also based on in vitro cell culture models, and their research results cannot be fully applied to whole animals. In the past, the neurotoxicity experiment of local anesthetics in the whole animal experiment was usually based on the method of intrathecal injection of local anesthetics,13,14 and its main research sites were in the spinal cord and dorsal root ganglion. At present, there is no ideal animal model of peripheral neurotoxicity injury caused by local anesthetics. Therefore, this study aims to provide an animal model for the study of peripheral neurotoxicity injury caused by local anesthetics by establishing and evaluating a model of sciatic nerve injury caused by local anesthetics.
Materials and methods
Experimental animals
Male C57BL/6 mice were selected, with a body weight of 18–20 g, feeding conditions of room temperature (23 ± 2) °C, light control for 12 h (7:00 am to 7:00 pm), and free access to drinking water. The animals were provided by Guangdong Medical Experimental Animal Center. The experiment was divided into two groups: the normal group and the ropivacaine group (n = 6). This experimental study conformed to the standard requirements of the Animal Protection Committee. Animals were given anesthesia during the experiment to minimize pain.
Preparation before operation
The motor nerve function of mice in each group was scored before the operation, and those with limb dysfunction were excluded. The von Frey method was used to measure the mechanical withdrawal threshold of mice for postoperative control.
Model preparation
After anesthesia, the mice in the ropivacaine group underwent incision of the skin and subcutaneous tissue at the middle and lower 1/3 of the right thigh of the mice, the muscle space was separated, and 1 μL/g of 1% ropivacaine hydrochloride (AstraZeneca AB, Sweden) was injected under the condition of exposing the sciatic nerve; the incisions were then sutured layer by layer after injection. The mice in the normal group were not treated.
Observation indicators
At 24 h and 48 h after the injection of 1% ropivacaine hydrochloride, the lower limb motor function of the mice was evaluated by the Basso mouse scale (BMS) score, and the mechanical paw withdrawal threshold (MWT) was detected by the von Frey method. At 48 h after the end of the detection, the mice were decapitated and killed. The affected side of the sciatic nerve of the experimental mice was taken and fixed with 10% formalin, and pathological sections were made. Pathological changes in the sciatic nerve were observed by hematoxylin/eosin (HE) staining.
Motor function evaluation
After the mouse moved freely for 5 minutes, the motor function of the mouse's hind limb movement was scored from 0–9 points according to the BMS (0 points indicating complete paralysis, 9 points indicating normal function). The BMS scores of the right hind limbs were calculated and averaged to obtain a single value for each test of each mouse. 15
Mechanical paw withdrawal threshold
The mechanical withdrawal threshold (MWT) of mice was measured with a von Frey filament. 16 The MWT was measured before and 48 h after the operation, and the threshold value of 50% foot retraction was calculated by the up-down method with a von Frey filament. A glass box of 5808 cm3 (22 × 12 × 22 cm), with the bottom open, was placed on a metal screen, and after the mouse was acclimated to the glass box for 15 min, the middle part of the hind foot of the mouse was vertically stimulated with von Frey fiber for a duration of ≤4 s. The mouse’s foot lifting or licking behavior was considered a positive reaction; any other reaction was considered negative. When the stimulus did not cause a positive reaction, the adjacent stimulus with a higher intensity was given; if there is a positive reaction, the next lower level of stimulation was given.
Statistical analysis
SPSS 22.0 statistical software was used for statistical analysis. The data are expressed as the mean ± standard deviation. A paired t test was used for comparisons between groups. p < 0.05 was statistically significant.
Results
Hindlimb movement scores (BMS)
There was no significant difference in the preoperative BMS score between the two groups, but the BMS score of the mice in the ropivacaine group was significantly reduced 48 h after the operation, which was statistically significant compared with the normal group (see Figure 1). The motor function score of the mouse’s hind limb movement (BMS) (mean ± standard deviation, n = 6). Normal: Normal group. Ropivacaine: Ropivacaine group. 48 h: 48 h after operation. (
*
p < 0.01 vs. Normal preoperation,
#
p < 0.01 vs. Normal 48 h).
Mechanical paw withdrawal threshold
There was no significant difference in the mechanical paw withdrawal threshold between the two groups before the operation. The 48-hour mechanical paw withdrawal threshold in the ropivacaine group was significantly lower than that in the normal group, and the difference was statistically significant.
Pathological changes
Compared with the normal group, the pathological manifestations of the mouse sciatic nerve in the ropivacaine group were increased axonal edema and degeneration, edema between nerve fiber bundles, increased degeneration of axon and myelin sheath vacuoles, edema of nerve bundle membrane and local degeneration and necrosis, and infiltration of a large number of inflammatory cells around the nerve adventitia (Figure 2). Figure 3 The mechanical paw withdrawal threshold (MWT, g, mean ± standard deviation, n = 6). Normal: Normal group. Ropivacaine: Ropivacaine group. 48 h: 48 h after operation. (*p < 0.01 vs. Normal preoperation,
#
p < 0.01 vs. Normal 48 h, ∆p < 0.01 vs. Ropivacaine preoperation). (a) and (c) show the HE staining of the long-axis and short-axis sections of the mouse sciatic nerve in the normal group, (b) and (d) show the HE staining of the long-axis and short-axis sections of the mouse sciatic nerve in the ropivacaine group (HE, ×40).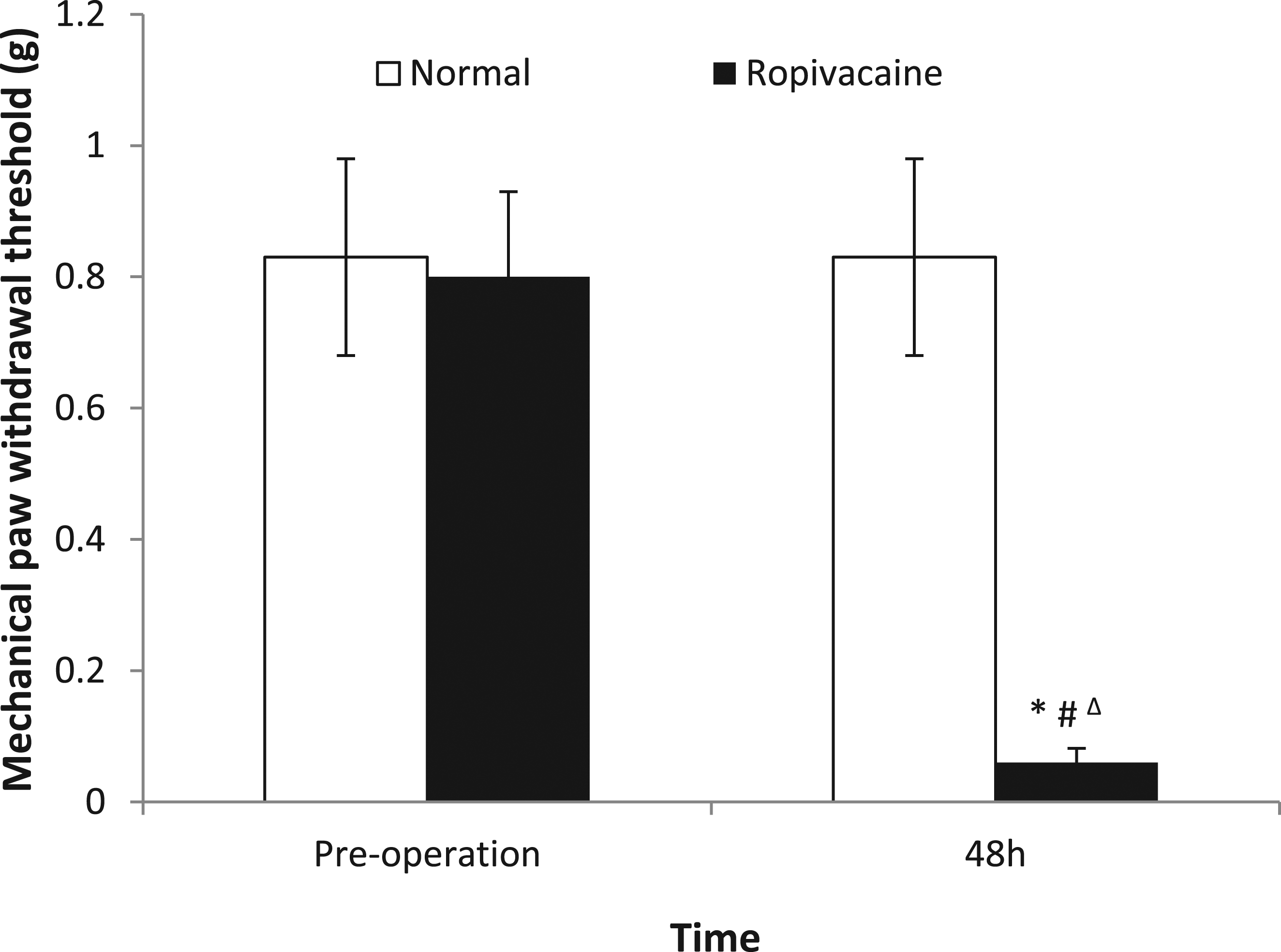

Discussion
This study observed pathological changes in the sciatic nerve after injecting 1% ropivacaine around the sciatic nerve of C57BL/6 mice. In the past, few animal models have been used to study the peripheral neurotoxicity of local anesthetics. Some studies have used intrathecal injection of local anesthetics to observe their neurotoxicity.13,14,17 This study injected local anesthetics around the sciatic nerve, which is similar to the clinical peripheral nerve block method, directly simulating the occurrence of peripheral neurotoxicity of local anesthetics in clinical practice. Ropivacaine hydrochloride is the most widely used local anesthetic for nerve block in the clinic at present, and the concentration used in the clinic varies from 0.1 to 1%.18–20 Because the sciatic nerve of C57BL/6 mice is thinner than the peripheral nerve of humans and direct incision injection is more intuitive than puncture injection, it is not easy to cause mechanical damage to the peripheral nerve. A previous study found that local anesthetics cause significant neurotoxic damage to developing neurons, and the concentration and exposure time of local anesthetics are important factors for nerve damage. 21 In general, developing neural neurons may be susceptible to the neurotoxicity of local anesthetics. Therefore, in this study, we selected C57BL/6 mice at the age of 6 weeks which had reached physical maturity. The concentration of local anesthetics and the time of neuronal exposure are the main factors causing neurotoxic damage by local anesthetics. The sciatic nerve of the mouse is very thin, and we injected 1% ropivacaine 1 μL/g to completely cover the sciatic nerve. Therefore, C57BL/6 mice aged 6 weeks were injected with 1% ropivacaine (1 μL/g) around the sciatic nerve under direct vision to prepare a local anesthetic neurotoxic injury model.
In this study, we found that 48 h after injecting 1% ropivacaine hydrochloride around the sciatic nerve, the lower limb motor function score (BMS) and mechanical paw withdrawal threshold of mice were significantly reduced, suggesting that the motor function of the sciatic nerve might be damaged. Through pathological sections and HE staining, the following effects were observed in the ropivacaine group: the axon edema and degeneration of the sciatic nerve increased; edema between nerve fiber bundles; the degeneration of axon and myelin sheath vacuoles increased; the edema of nerve bundle membrane; local degeneration and necrosis; and a large number of inflammatory cells infiltrated around the nerve adventitia. This neuropathological change may be one of the mechanisms of pain and discomfort after neurotoxic injury caused by local anesthetics. 22 The above pathological results suggested that the sciatic nerve was injured after 1% ropivacaine hydrochloride treatment.
Although this animal model can simulate the peripheral neurotoxicity injury of local anesthetics in clinical practice, it still has certain limitations. For example, the incidence of neurotoxic injury from local anesthetics in clinical practice is relatively low, and most patients recover. The observation time point in this study was 48 h after administration. If the observation time is extended to 72 h or longer, the lower limb motor function score and mechanical retraction threshold may return to the preoperative level. Despite the above limitations, this model is still of great significance for the study of peripheral neurotoxicity of local anesthetics and provides an animal model for the study of neurotoxicity injury of local anesthetics.
Conclusion
In this study, an animal model of neurotoxic injury caused by ropivacaine was established, which can be used to study peripheral neurotoxic injury caused by local anesthetics.
Footnotes
Authors’ contributions
All authors participated in the design and review of the manuscript. WX designed and implemented the experiments and wrote the paper; EQ, WY, LX, PJ, ZZ and CM implemented the experiments and collected the data; LX performed the statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Project of Guangdong Administration of Traditional Chinese Medicine (No. 20231323), Guangdong Medical Science and Technology Research Foundation (No. A2022341) and Hospital level project of Foshan Second People's Hospital (No. 2022A01).
