Abstract
Background
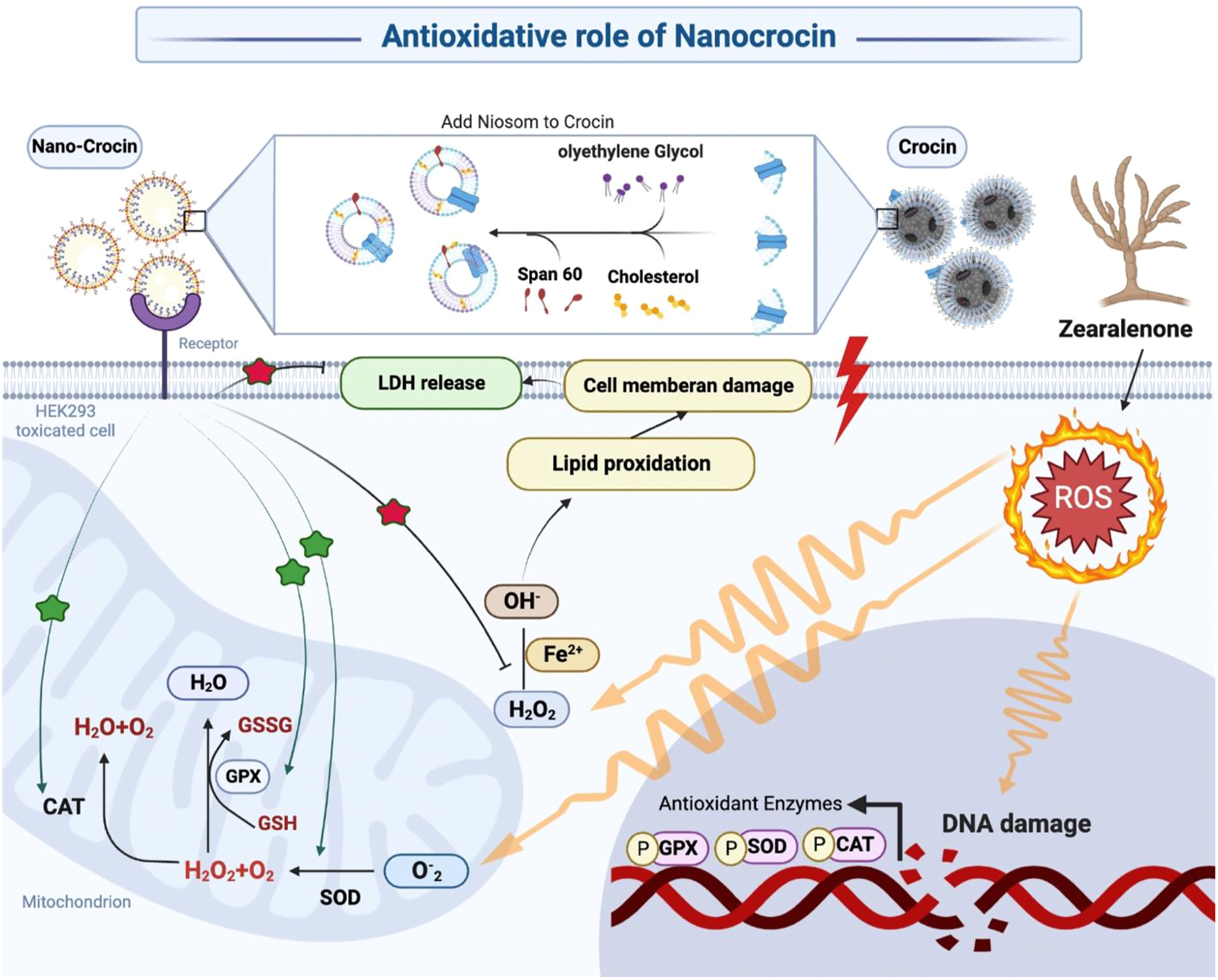
Zearalenone (ZEA) is a mycotoxin produced by fungi and induces cytotoxicity by the generation of reactive oxygen species. The aim of this study was to evaluate and compare the nephroprotective effects of crocin and nano-crocin against ZEA-induced toxicity in HEK293 cell line via modulation of oxidative stress and special formulation to make nano-crocin.
Method
Nano-crocin physicochemical properties, such as size, load, appearance, and drug release profile were determined. Also, the viability of intoxicated HEK293 cells was evaluated by MTT assay. Furthermore, lactate dehydrogenase lipid Peroxidation (LPO), and oxidative stress biomarkers were measured.
Result
The best nano-crocin formulation with superior entrapment effectiveness (54.66 ± 6.02), more significant drug loading (1.89 ± 0.01), better zeta potential (−23.4 ± 2.844), and smaller particle size (140.3 ± 18.0 nm) was chosen. This study showed that treatment with crocin and nano-crocin in ZEA-induced cells, significantly decreased LDH and LPO levels and increased superoxide dismutase (SOD), catalase (CAT) activities, and total antioxidant capacity (TAC) levels compared to the control group. Moreover, nano-crocin had a more curative effect against oxidative stress than crocin.
Conclusion
Niosomal structure of crocin, when administered with the special formulation, may be more beneficial in reducing ZEA-induced in vitro toxicity than conventional crocin.
Introduction
Mycotoxins are secondary metabolites made by fungi and when they are consumed, inhaled, or in touch with the skin, can be harmful. Zearalenone (ZEA) is a kind of mycotoxin that is produced by some Fusarium fungi, including F. graminearum, F. culmorum, F. equiseti, and F. cerealis. 1 ZEA has a relatively low acute toxicity, with an oral LD50 range of 2000–20,000 mg/kg in mice, rats, and guinea pigs, 2 and because it usually binds to estrogenic receptors, this mycotoxin is frequently referred to as a mycoestrogen. 3 At low dosages, it may have a variety of estrogen-disrupting effects, such as infertility, decreased serum testosterone concentrations and sperm counts, enlargement of the ovaries and uterus, decreased pregnancy rates, and changes in progesterone levels in animals.4,5
Also, it has been reported that some of the cytotoxic and genotoxic effects of ZEA are unrelated to its binding affinity to the estrogen receptor. For example, it has been shown that ZEA is, hepatotoxic, 6 hematotoxic, 7 nephrotoxic organs, 8 and also hazardous to the intestinal tract. 9 Numerous studies have demonstrated that ZEA induces cytotoxicity by the generation of reactive oxygen species (ROS), which causes lipid peroxidation (LPO), DNA damage, and death via the mitochondrial pathway.6,10 Moreover, ZEA is able to induce endoplasmic reticulum (ER) stress-mediated apoptosis in leukemic cells. 11
Reducing mycotoxin levels in food sources and improving dietary intake of nutrients such as vitamins and antioxidants are two strategies for preventing ZEA poisoning. It has been demonstrated that vitamin E as antioxidant prevents practically all ZEA toxic effects both in vitro and in vivo.10,12 Moreover, it has been shown that extracts from the cactus cladodes, 13 radish, 14 or garlic 15 provide strong protection against ZEA-induced toxicity. Thus, research on the impact of antioxidants, especially those found in food, seems to be very important to prevent ZEA-induced toxicity insight ROS damage.
One of the oldest medicinal crops in Iran is saffron (Crocus sativus L.), and farmers have played a major part in its development. 16 About 3.5% of the dry stigma weight of saffron is crocin. 17 This molecule has a saffron color and is one of the few water-soluble carotenoids in nature. 18 Crocin traps ROS because of its unique carotenoid structure and may function as an antioxidant.19,20 The pharmacological benefits of crocin have been the subject of numerous investigations, including their anti-depressant, 21 hypolipidemic, 22 anti-inflammatory, 23 anti-cancer, 24 and antioxidant actions. 25 Both in vitro and in vivo studies have shown that crocin has a strong antioxidant capability. 26 For instance, crocin can reduce LPO in skeletal muscle and kidney during ischemia-reperfusion-induced oxidative damage in rats, 27 but crocin is a highly water-soluble, unsaturated carotenoid that is sensitive to oxygen, low pH, light, and trans-cis isomerization, all of which alter its taste, nutritional value, and color.
As a result, several strategies, including the use of nanocarriers for medication administration, can be employed to resolve these problems.28,29 Technology that encapsulates the medication in a vesicle that gets its name from the fact that the vesicle is made up of a bilayer of non-ionic surface-active substances. Also, they are minuscule in size and are measured by the nanometer scale. 30 Niosomes have recently been proven to significantly improve transdermal drug delivery and can also be used for targeted drug delivery. Niosomes’ non-ionicity form can reduce toxicity properties, and their targetability raises the drug’s therapeutic index, this structure allows for the regulated delivery of drugs to their intended locations. 23 In the current investigation, the human embryonic kidney cell line (HEK293) was used as an in vitro model to evaluate the cytotoxicity of ZEA and the potential for kidney damage. This cell line’s applicability to human toxicity models has been well described.31,32 To explain the feasible mechanisms of cytotoxicity, a variety of surrogate parameters including cell viability, mitochondrial function (MTT assay), cell membrane damage (lactate dehydrogenase assay), LPO levels, and oxidative stress biomarkers were evaluated and compared with controls and groups were treated by Crocin and Nano-crocin.
Material and method
Reagents and chemicals
All the following reagents and chemicals were purchased from Sigma‐Aldrich (St Louis, MA): acetate buffer, ferric chloridetetraethoxypropane (TEP), 2,4,6‐tripyridyl‐S‐triazine (TPTZ), 2‐thiobarbituric acid (TBA), trichloroacetic acid (TCA), TPTZ, n‐butanol, Tris base, hydrochloric acid (HCl), ethylenediaminetetraacetic acid (EDTA), and crocin.
Preparation and characterization of the Nano-crocin
The amount of the ingredients in different formulations.

*CCN: Crocin; PEG: polyethylene glycol.
The samples were sonicated in the buffer for 5 min after being evenly distributed. It should be mentioned that the control samples were made in the same way but did not include any medicine. The Hamadan University of Medical Sciences Ethics Committee approved the study’s design (Res: IR. UMSHA.REC.1400.140). Therefore, in this experimental study Zetasizer used (Nano ZS3600, Malvern Instruments, UK) to use the dynamic light scattering (DLS) technique to measure the size and zeta potential of the niosomes. Then, the unloaded crocin was separated from the niosomes using an ultracentrifuge (Beckman Coulter, USA) at 18,000 RPM for 10 min at 4°C.
An ultraviolet spectroscopic device (Shimadzu, Japan) operating at 441 nm was then used to estimate the amount of crocin. Finally, the following equation was used to calculate the drug encapsulation and drug loading efficiency
34
:

Drug release study
The drug release analysis in this investigation was conducted by using the dialysis membrane method. 35 Briefly, 1 mL of niosomes were added into a dialysis bag (Sigma, cut off 12,000) and immersed into 100 mL of Phosphate-buffered saline (PBS) (pH 7.4). The resultant solution was then agitated for 48 h at 37°C using 110 rpm. Finally, the drug level was calculated using the spectrophotometer method using 2 mL of samples that were taken at the predetermined intervals.
Cell culture
The Iranian Pasteur Institute provided the HEK293 cells, which were grown in DMEM media with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin (Pen/Strep). They were grown in a 37°C incubator with 5% CO2. Six cell groups were used in this study for all of the experiments: (I) Untreated cells were named as controls. (II) crocin-treated cells (III) nano-crocin-treated cells. (IV) Cells treated with ZEA (IC50 concentration). (V) Cells were treated with ZEA (IC50 concentration) and were simultaneously treated with crocin. (VII) Cells were treated with ZEA (IC50 concentration) and simultaneously treated with nano-crocin.
Toxicity assessment of ZEA, crocin, and nano-crocin using MTT assay
The MTT test was applied as follows to assess the toxicity of ZEA, crocin, and nano-crocin and establish the inhibitory concentration of 50% (IC50): First, a complete medium was used to culture HEK293 cells in a 96-well plate (8000-6000 cells per well).
After 24–48 h cells were adhesion and grow, and reached a density of about 70–75%. Then the cells were individually exposed to ZEA, crocin, and nano-crocin.
Following the treatment period, 20 μL of culture medium including MTT (5 mg/mL) was added to each well and then re-incubated at 37°C, for 4 h. Three to 4 hours later, we drained the cell culture medium, then added Dimethyl sulfoxide (DMSO) to each well. Next, the absorbance of solutions in each well was noted by using a plate reader at 570 nm.
The ratio of living cells in each treatment to untreated cells served as the basis for calculating the treatment survival rates with 100% survival.
Treatment of HEK293 with crocin, and nano-crocin
The measurement was performed using the MTT method by first HEK293 cells, in this way the HEK293 cells were placed in plate 96 well that each well containing 8000-6000 cells, and then cultured in a complete medium.
After 48 h, cells were adhesion and growth and cell density of about 70–75%. The cells were treated as follows: (I) Untreated cells were named as controls. (II) crocin-treated cells. (III) nanocrocin-treated cells. (IV) Cells treated with ZEA (IC50 concentration). (V) Cells were treated with ZEA (IC50 concentration) and were simultaneously treated with crocin and (VII) Cells were treated with ZEA (IC50 concentration), were simultaneously treated with nanocrocin.
After the treatment time, about 100 μL of culture medium containing MTT (5 mg/mL), was added to each well and transferred to the incubator again at 37°C for 4 h. After 3–4 h, the cell culture medium was drained, and 100 μL of DMSO was added to each well. Then, Elisa Reader was used to recording the absorption of solutions in each well at 570 nm. Moreover, therapeutic concentrations of crocin and nano-crocin were assessed.
Measurement of LDH
The cytosolic enzyme LDH, which is stable in healthy cells, can only leak into the extracellular fluid as a result of membrane damage. In our work, the LDH leakage test was utilized to assess the harm ZEA did to cell membranes. The growth media was aspirated and centrifuged at 3000 rpm for 5 min to produce a cell-free supernatant after exposure to the ZEA. Using a commercially available kit from Sigma Diagnostics, the activity of LDH in the medium was assessed (LD-L50). The test is based on the parallel decrease of NAD and the conversion of lactate to pyruvate in the presence of LDH. The aforementioned process produces NADH, which alters the absorbance at 340 nm. Aliquots of media and warm reagent were mixed in a 96-well plate (Fisher) and absorbance was recorded using a microplate spectrophotometer system (Spectra max190-Molecular Devices).
Measurement of LPO
LPO is the most common result of oxidative stress. Amount of LPO was determined by quantification of a thiobarbituric acid reagent.36,37 LPO production react with thiobarbituric acid to produce a pink color with maximum absorption at 532 nm.
Determination of total antioxidant capacity (TAC)
TAC was manually quantified using the Ferric reducing ability of plasma (FRAP) method, which is based on the conversion of Fe3+ to Fe2+ in the presence of Tripyridyl-s-triazine (TPTZ). A blue-colored complex is created as a result of the interaction between TPTZ and Fe2+. Additionally, the maximum optical density at a wavelength of 593 nm was assessed and determined. 38
Measurement of superoxide dismutase (SOD) activity
Superoxide radicals are produced by the reaction of xanthine and xanthine oxidase, and the quantity of these radicals was measured using 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyl tetrasodium chloride. In order to create a red formazan dye, 2-(4-iodophenyl)-3-(4-nitrophenol)-5-phenyl tetrasodium chloride combines with superoxide radicals. The level of the reaction’s inhibition was then assessed by measuring its absorbance at 550 nm in order to quantify the SOD activity. To determine the total SOD activity, The standard calibration curve of inhibition percentage was prepared by plotting absorbance versus the concentrations (U/mL). 39
Measurement of catalase (CAT) activity
The level of CAT activity was determined at an absorbance reduction of 240 nm in a mixture containing sodium phosphate buffer (50 m
Statistical analysis
Graph Pad Prism software was used to perform one-way analysis of variance (ANOVA) and Tukey’s test for multiple comparisons on the collected data, which were reported as the mean ± SEM (version 9.0). p-values under .05 were regarded as statistically significant.
Results
Characteristics of nano-crocin
Characteristics of nano-crocin formulation. (EE: entrapment efficiency, DL: drug loading, PDI: polydispersity index, NA: not applicable).

The statistics acquired by examining the particle size for the best formulation were consistent with the electron microscopic image of the formulation that was chosen. Figure 1 illustrates the nanoparticles’ morphology (Figure 1). Scanning electron microscopy (SEM) image of crocin-loaded Niosome with Scale bar = 1 μm.
Drug release study
Based on the standard crocin curve, the amount of released crocin into the phosphate buffer (pH = 7.4) was calculated. The pattern of drug release is seen in Figure 2. The release curve in both mediums included an initial burst followed by a zero-degree release pattern (Figure 2). The release profile of the optimum crocin-loaded nanoparticles in phosphate buffer, pH = 7.4 (n = 3).
Cytotoxicity
In this study, the effect of ZEA, crocin, and nano-crocin on HEK293 cell lines was investigated using the MTT method for 48 h. The HEK293 cell viability was significantly reduced by treatment with different concentrations of ZEA. As shown in Figure 3 this change was dose-dependent, and the significant effect of this toxin on cell death was quite evident. The inhibitory concentration of 50% (IC50) of ZEA in 48 h in this cell line was equal to 86.5 m HEK293 cell viability at different concentrations of ZEA using MTT assay. The results were reported as the mean percentage of cell viability (n = 3). ***p < .001, *p < .05 compared with the control groups.
HEK293 cell viability due to treatment with different concentrations (in the range 0.31–160 μM) crocin decreased significantly. As shown in Figure 4, this change was dose-dependent, and the significant effect of this drug on cell growth was quite evident. The inhibitory crocin concentration of 50% (IC50) in 48 h in this cell line was 5 μM (Figure 4). Also, HEK293 cell viability due to treatment with different concentrations (in the range 0.31–160 μM) nano-crocin decreased significantly. This change was dose-dependent, and the significant effect of this drug on cell growth was quite evident. The inhibitory nano-crocin concentration of 50% (IC50) in 48 h in this cell line was obtained to be 5 μM (Figure 5). HEK293 cell viability at different concentrations of crocin using MTT assay. The results were reported as the mean percentage of cell viability (n = 3). HEK293 cell viability at different concentrations of nanocrocin using MTT assay. The results were reported as the mean percentage of cell viability (n = 3).

The effect of crocin and nano-crocin on the survival HEK293 cell
The percentage of cell survival in the ZEA-poisoned group was significantly lower than in the control group (p < .0001). However, the survival rate in the control group treated with nano-crocin was significantly lower than the control group (p < .05). Also, the survival rate in the ZEA poisoned groups that treated with crocin (p < .05) and nano-crocin (p < .01) was significantly higher than the ZEA group (Figure 6). Cell viability rate in different groups of our study. The data are shown as mean ± SEM. It should be noted that ANOVA, the Tukey test was used to compare the data. ****p < .0001, **p < .01, *p < .05 compared with the other groups. (Cnt: control, ZEA: zearalenone, CCN: crocin, NCCN: nanocrocin).
The effect of crocin and nano-crocin on the LDH
According to our findings, ZEA group caused a significant increase in LDH compared to the control group (p < .01). In the ZEA + nano-crocin group, LDH decreased significantly (p < .05) compared to the ZEA group. Additionally, the LDH level in ZEA + nano-crocin group was superior than ZEA + crocin group significantly (p < .05) (Figure 7). Effect of crocin and nanocrocin on LDH in Hek293 cells. The data are shown as mean ± SEM, n = 3. It should be noted that ANOVA, the Tukey test was used to compare the data. **p < .01, *p < .05 compared with the other groups. (Cnt: control, ZEA: zearalenone, CCN: crocin, NCCN: nanocrocin).
The effect of crocin and nano-crocin on the LPO
According to the findings, ZEA caused a significant increase in LPO compared to the control group (p < .05). In the ZEA + crocin and ZEA + nano-crocin groups, LPO decreased significantly (p < .01) compared to the ZEA group. Nano-crocin was found to be superior to crocin in the recovery of LPO but it wasn’t statistically significant (Figure 8). Effect of crocin and nanocrocin on lipid peroxidation in Hek293 cells. The data are shown as mean ± SEM, n = 3. It should be noted that ANOVA, the Tukey test was used to compare the data. **p < .01, *p < .05 compared with the other groups. (Cnt: control, ZEA: zearalenone, CCN: crocin, NCCN: nanocrocin).
The effect of crocin and nano-crocin on the TAC
As shown in Figure 9, the TAC level was observed to decrease significantly in the ZEA group compared to the control group (p < .001). Also, in the ZEA + crocin (p < .01) and ZEA + nano-crocin (p < .001) groups, TAC increased significantly compared to the ZEA group. It is interesting that nano-crocin showed a further induction in the progression of TAC than crocin (Figure 9). Effect of crocin and nanocrocin on total antioxidant capacity (TAC) in Hek293 cell. The data are shown as mean ± SEM, n = 3. It should be noted that ANOVA, the Tukey test was used to compare the data. ***p < .001, **p < .01 compared with the other groups. (Cnt: control, ZEA: zearalenone, CCN: crocin, NCCN: nanocrocin).
The effect of crocin and nano-crocin on the SOD activity
The results showed that the level of SOD activity in the ZEA group was lower than in the control group (p < .05). Furthermore, treatment with crocin and nano-crocin in ZEA-toxicated cells was able to increase the level of SOD more than the ZEA group, but there wasn’t any statistically significant comparison between them (Figure 10). Effect of crocin and nanocrocin on the SOD activity in Hek293 cell. The data are shown as mean ± SEM, n = 3. It should be noted that ANOVA, the Tukey test was used to compare the data. *p < .05, compared with the control group. (Cnt: control, ZEA: zearalenon, CCN: crocin, NCCN: nanocrocin).
The effect of crocin and nano-crocin on the CAT activity
As shown in Figure 11, the level of CAT activity in the ZEA group was lower than in the control group (p < .01). Moreover, our findings demonstrated that treatment with crocin and nano-crocin increase the rate of CAT activity compared to the ZEA group but there were no significant differences between them (Figure 11). Effect of crocin and nanocrocin on total antioxidant capacity (TAC) in Hek293 cell. The data are shown as mean ± SEM, n = 3. It should be noted that ANOVA, the Tukey test was used to compare the data. **p < .01, compared with the control group. (Cnt: control, ZEA: Zearalenon, CCN: crocin, NCCN: nanocrocin).
Discussion
ZEA’s ability to cause oxidative stress has garnered a lot of attention throughout time. ROS, which is made up of many active molecules produced from O2 or hydrogen peroxide (H2O2), is often a significant by-product of cellular metabolism. 41 When ROS present at a moderate level can function as a second messenger in the control of cell signaling pathways. 42
In oxidative stress status, ROS are created in excess, which damages cells and tissues and ultimately leads to cell death. 43 Numerous pathological conditions, including aging, carcinogenesis, atherosclerosis, ischemia-reperfusion injury, tissue damage, and acute or chronic inflammatory disorders, have been linked to oxidative stress (38). Numerous studies have shown that low concentrations of ZEA can enhance cells’ ability to neutralize ROS whereas overly high concentrations of ZEA can directly impair the antioxidant enzyme’s function, causing ROS to build up and eventually resulting in oxidative stress.42,44 It has been recommended that the use of antioxidants and radical scavengers in mycotoxins toxicity help in the recovery of cell and animal damage.45,46 In this vein, Forough et al. examined how broccoli hydroalcholic extract affected the cellular alterations in testicular tissue and how diazinon controlled oxidative stress. This study’s findings demonstrate that broccoli extract has a beneficial dose response impact on the damage diazinon produces to male rats’ spermatogenesis. 47
Many in vitro and in vivo studies have demonstrated the preventive properties of crocin, a carotenoid derived from stress-induced damage.48,49 In this investigation, we showed that crocin therapy provided significant protection against oxidative stress-induced by ZEA in kidney cells. Because crocin is a water-soluble carotenoid, it is sensitive to environmental conditions. Therefore, different approaches can be used to solve these problems, such as using nanocarriers to deliver crocin. 28
Drugs are protected from outside effects by niosomes’ capacity to encapsulate lipophilic and hydrophilic substances inside their structure. 50 The present study was performed to appraise to compare the effects of crocin-loaded niosomes and crocin in ZEA-intoxicated HEK293 cells similar to lycopene niosoms. The previous studies showed that niosomes can promote cellular absorption, which in turn boosts pharmacological activity while also improving drug stability. 51 Also, niosome is a type of medication formulation that lowers drug doses and, as a result, lowers drug toxicity. 52 The loading rate of niosomal formulations is one of the most crucial metrics to consider. Highly effective nano-drugs have high load effectiveness and retain their qualities in the body for a long period. 53
In this study, the percentage of drug entrapment efficiency in the optimal formulation was obtained to be 54%. Notably, the only change between the first, second, and third formulations is the variation in the proportion of cholesterol to surfactant. According to other earlier investigations, the proportion of drug loading within the niosomal structure would fluctuate significantly as the quantity of cholesterol compared to the surfactant in various formulations changes. 54 Varshosaz et al. examined the effectiveness of loading ascorbic acid and α-tocopherol into the niosomal structure and the findings demonstrated that compared to α-tocopherol, ascorbic acid loading efficiency was substantially lower. Therefore, these results could be a new formulation of the hydrophobicity of the molecule of α-tocopherol. Due to the hydrophobic nature of the cholesterol surfactant bilayer membrane internal part, the drug is stored to a greater extent within these parts of the niosomes structure.
The rate of penetration into the target tissue rises as the decreases and then the drug’s efficacy will be rises. 49 Thus, the benefits of employing niosomes as drug transporters are engaged in the absorption and accumulation in the target tissue and inducing effective accumulation of the drug at the target location due to their tiny size. 55
In the current investigation, the first formulation’s (F1) size was much less than that of the second (F2) and third (F3) formulations. It was discovered after characterizing the resultant nanoparticles that the PEG surface decorating had a favorable impact on the nanoparticles’ characteristics and rate of encapsulation. These improvements in physicochemical qualities appear to have been significantly influenced by the steric stabilization of niosomes by PEGylation. In the current investigation, the F1’s zeta potential is noticeably higher than the third formulation’s (F3). As a result, F1’s electrical stability was larger than F3’s. The tendency of the drug placed in the niosome structure to release the medication slowly is another significant characteristic of niosomal nanomedicines. Nasser et al. synthesized the medication diclofenac sodium in a niosomal structure and evaluated its effectiveness in comparison to the market’s available conventional topical diclofenac gel. They demonstrated that the drug release pattern when loaded in a niosome structure was more uniform and sustained. The F1 in this investigation had a more consistent release pattern than the F2 and F3. The F1 was chosen as the best formulation following testing, and HEK293 cells were employed for cellular investigations. 56
As, carotenoids, an antioxidant molecule, may behave as a pre-oxidant in excessive concentrations and cause harm. 22 It was demonstrated that crocin dramatically reduced cell survival in a dose-dependent way, which is consistent with the findings of the present investigation. They also demonstrated that crocin had no harmful impact at low doses. It has been reported that other malignant cells’ programmed death at dosages over 3 mg/mL is the main mechanism for crocin. 24
Our study revealed that the administration of crocin and nano-crocin was able to normalize the LPO levels and antioxidant defense in damaged kidney cells, and nano-crocin provides better recovery than crocin in this condition. LPO, commonly used as a biomarker of oxidative toxic stress, 57 may destroy cell membrane structure, cause DNA fragmentation, rearrangement, cross-linking, and accelerate apoptosis. 58 In 2015 Salem et al. demonstrated have been demonstrated that crocin treatment decreased LPO levels, dose-dependently in the liver and kidney of ZEA-induced toxicity mice as compared with the ZEA-treated group. 59 Also, Altinoz et al. 60 2015, reported that LPO levels decreased significantly in Diabetic rats treated with crocin, compared to untreated Diabetic rats.
Also in our study, the LDH leakage test was used to track the ROS-induced cell membrane destruction since normal cells can only secrete LDH, a stable cytosolic enzyme after their membranes have been damaged. LDH release from HEK cells increased in response to ZEA exposure. According to the examination of particle exposure medium for LDH, nano-crocin reduces LDH leakage and safeguards cell viability.
Living tissues have built-in antioxidant defense mechanisms that include both enzymatic and nonenzymatic antioxidants. SOD and CAT are the initial line of defense against oxidative damage among the antioxidant enzymes. In addition, SOD is the first line of defense against oxidative stress by catalyzing the dismutation of superoxide radicals (O-2) into molecular oxygen (O2) and H2O2. 61 In this vein, Raeeszadeh et al. 62 examined at how rats’ kidney and liver damage from mercury was affected by an ethanolic extract of Medicago sativa L. (Alfalfa). They found that the mercury group had higher levels of malondialdehyde and lower levels of glutathione peroxidase (GPx), CAT, TAC, and SOD activity. As comparison to the mercury group, the levels of these metrics dramatically increased in the groups receiving the extract. Also, the histological analysis revealed that the mercury group had glomerular and tubular damage as well as liver necrosis, while these characteristics were improved in the treatment group.
Our study’s findings demonstrate that ZEA produces significant oxidative damage in the kidney because alterations in oxidative stress biomarkers and the antioxidant defense system were seen in kidney cells following exposure to ZEA. The results of this study showed that ZEA treatment with antioxidant significantly decreased LPO levels and increased SOD, CAT activities, and TAC levels compared to the control group. Also, crocin when delivered as a niosomal structure is potentially more effective than the standard crocin. Our results have been similar to another studies that confirms nanoparticle form of the drug is more effective in reducing oxidative damage.63,64 As cell-culture studies focus on isolated cells apart from tissues or organisms, the influence of crocin or any substance that can interfere with the pharmacological receptors cannot be studied. Importantly, cell cultures cannot study some pharmacokinetic parameters, and animal experiments remain required to achieve this purpose. Nanocrocin shows several unique physicochemical properties that are exploited in various high-performance products or novel applications. So, these properties may represent major obstacles such as cytotoxicity in dose-dependent studies and have to be carefully characterized in advance. Nanocrocin as a nanoparticle can be generated confounding or even conflicting data, and evidence is accumulating that nano crocin may interfere.
This study provided insight into ways to modulate the toxicity of the commonly encountered mycotoxins as ZEA. We showed that ZEA induces oxidative injuries in renal cells and natural antioxidants such as crocin, which has antioxidant properties, protect organisms from ZEA-induced oxidative damage. Our result also suggests that nano antioxidant treatment might be helpful more than primary materials to prevent ZEA-related toxicity. Also, we recommend that crocin, when administered as a niosomal structure has more beneficial effects in reducing ZEA-induced nephrotoxicity than conventional form of crocin. However, more studies should be done to confirme this application in animal models and consecutively in humans.
Footnotes
Author contributions
Fatemeh Ghafari: Methodology, visualization, software, writing-original draft preparation.
Zohre Sadeghian: Data curation, methodology, software, writing-original draft preparation.
Akram Oftadeh Harsin: Data curation, visualization, software, editing, methodology.
Sodabe khodabandelo: Validation, writing- reviewing and editing.
akram ranjbar: conceptualization, methodology, software, supervision.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hamadan University of Medical Sciences; 140004083175.
