Abstract
Zearalenone (ZEN) is a mycotoxin from Fusarium species commonly found in food commodities and is known to cause reproductive disorders. Several in vivo studies have shown that ZEN is haematotoxic and hepatotoxic and causes several alterations of immunological parameters. Meantime, the available information on the cardiotoxic effects of ZEN is very much limited. In the present study, we investigated the toxic effects of ZEN in heart tissues of Balb/c mice. We demonstrated that ZEN (40 mg kg−1 body weight (b.w.)) increased creatine phosphokinase, lactate dehydrogenase, aspartate transaminase, alanine transaminase, total cholesterol and triglyceride levels and induced oxidative stress as monitored by measuring the malondialdehyde level, the generation of protein carbonyls, the catalase and superoxide dismutase activity and the expression of the heat shock proteins (Hsp 70). We also demonstrated that acute administration of ZEN triggers apoptosis in cardiac tissue. Furthermore, we aimed to evaluate the safety and efficacy of crocin (CRO), a natural carotenoid, to prevent ZEN-induced cardiotoxicity in mice. In fact, combined treatment of ZEN with different doses of CRO (50, 100, and 250 mg kg−1 b.w.) showed a significant reduction of ZEN-induced toxicity for all tested markers in a dose-dependent manner. It could be concluded that CRO was effective in the protection against ZEN-induced toxicity in cardiac tissue.
Introduction
Mycotoxins represent an important category of biologically produced natural toxins with potential effects on animal and human health. The worldwide contamination of food, feed and environment by these natural products, including indoor surfaces and particles, affects thehealth of animals and humans. 1 Special attention was given to zearalenone (ZEN) produced by Fusarium species because of its oestrogenic effects on human and animal health. 2 ZEN occurs naturally all over the world in a variety of food products designed for human and animal consumption, and potentially high concentrations are encountered as contamination in many important crops. 3 In Tunisia, ZEN was found in 64% of the analysed samples of tender wheat, of which 18% exceeded the tolerable levels (100 mg kg−1). 4 The main route of exposure to ZEN in humans is through ingestion of contaminated food products, such as wheat, maize, rye and other cereals. 5 It is known that ZEN is of relatively low toxicity, with a median lethal dose (LD50) value of 500 mg kg−1 body weight (b.w.) determined on experimentation with mice. 6 Nevertheless, its role as a mammalian endocrine disrupter is being recognized, with effects in both males and females of different species. 7 Besides, ZEN was found to be hepatotoxic, and it induces liver lesions. 8 –10 It is also haematotoxic and causes many alterations of immunological parameters. 8,11 –13 In our laboratory, several studies have shown that ZEN is cytotoxic and exhibits a genotoxic potential in both in vitro and in vivo models through induction of chromosome aberrations, micronuclei, DNA fragmentation, cell cycle arrest and so on. 12 –18 It was also shown that ZEN induced the expression of heat shock protein (Hsp 70), which plays a key role in cytoprotection involving mainly the neutralization of oxidative stress induced by this mycotoxin. 19 Nevertheless, there are no data regarding the acute effect of ZEN in heart tissues. Therefore, in this present study, we investigated ZEN-induced toxicity in cardiac tissues. In fact, we aimed to find whether changes in biochemical parameters, oxidative stress and apoptosis could be relevant for ZEN-induced toxicity in heart using Balb/c mice.
In recent years, special attention was accorded to the study of natural products and healthy foods for prevention of diseases including cancer. 20,21 In this context, several studies were conducted in our laboratory to evaluate the protective potential afforded by natural compounds towards ZEN toxicity. In fact, we have demonstrated that almost all ZEN toxic effects mentioned above are significantly prevented both in vivo and in vitro using vitamin E. 14,16,19,22 Similarly, Abbes et al. 12,13,17 have shown significant prevention by the phyllosilicate clay mainly due to its tight binding to ZEN, resulting in a reduction of toxin bioavailability. Another set of experiments have highlighted the antioxidant potential of cactus cladodes (Opuntia ficus indica) against ZEN-induced oxidative stress in kidney and liver using Balb/c mice. 23 In 2009, the protective effect of isothiocyanate from the Tunisian radish (Raphanus sativus) was shown against ZEN-induced genotoxicity in vivo. 24
In this study, we are interested in evaluating the protective effect afforded by crocin (CRO), a natural carotenoid (Figure 1), towards cardiotoxicity induced by ZEN in vivo. CRO is the major pharmacologically active compound of Crocus sativus L. (saffron), and it represents about 10% of its composition. 25,26 The high antioxidant capacity of CRO has been reported in vivo and in vitro studies. 26 –32 In several pharmacological studies, saffron extracts or its active constituents have shown antitumor effects, 33 radical scavenger properties, 34 antidepressant 35 and memory-improving effects. 36 It was shown that CRO can improve ethanol-induced impairments of learning behaviour in a dose-dependent manner. 37 CRO can decrease lipid peroxidation in kidney 38 and skeletal muscle 39 during ischemia–reperfusion-induced oxidative damage in rats. It is reported that CRO inhibits tumor necrosis factor α-induced apoptosis in PC12 cells by modulating messenger RNA expression of Bcl2 family proteins. 40 Further study indicated that CRO is more potent than α-tocopherol against oxidative stress-induced death of neurons. 26,28 However, it remains unknown whether CRO can antagonize ZEN-induced toxicity, and therefore, we investigated its role towards ZEN-induced damages in heart tissues.

Molecular structure of CRO. CRO: crocin.
Materials and methods
Chemicals
ZEN, CRO and thiobarbituric acid were purchased from Sigma (St Louis, Missouri, USA). Pyrogallol was purchased from Sigma-Aldrich Chemical Company. 2,4-Dinitro-phenylhydrazine (2,4-DNPH) and guanidine were obtained from VWR International (Fontenay-sous-Bois, France). Mouse anti-Hsp 70 (SPA-810) was purchased from Stressgen (Canada), mouse monoclonal anti-p53, anti-Bax and anti-Bcl2 were from Invitrogen (Carlsbad, California, USA) and mouse anti-GAPDH (ab9483) was from Abcam (Cambridge, UK). The secondary antibody (phosphatase-conjugated), anti-mouse immunoglobulin were obtained from Invitrogen. All other chemicals used were of analytical grade.
Animal treatments
Thirty-six Balb/c mice (Sexual, Saint-Doulchard, France) were used (average body weight: 20 ± 0.3 g; age: 6 weeks old). These mice were given standard granulated food and drinking water and were divided into six groups as follows: Group 1: Mice given ethanol/water (1:1, v/v) Group 2: Mice given CRO at 250 mg kg−1 b.w. Group 3: Mice given ZEN in ethanol/water (1:1, v/v) at 40 mg kg−1 b.w., corresponding to 8% of the LD50, and this concentration was chosen according to previous investigations.
12
–14,16,17
Group 4: Mice given CRO at 50 mg kg−1 b.w. and ZEN at 40 mg kg−1 b.w. Group 5: Mice given CRO at 100 mg kg−1 b.w. and ZEN at 40 mg kg−1 b.w. Group 6: Mice given CRO at 250 mg kg−1 b.w. and ZEN at 40 mg kg−1 b.w.
A single administration by intraperitoneal injection of each substance (ZEN and CRO) in 200 µL of solutions was performed. Twenty-four hours after treatments, animals were killed by cervical dislocation.
Preparation of heart extracts
Hearts were homogenized with a glass-teflon Potter-Elvehjem homogenizer in the presence of 10 mM Tris–hydrochloric acid (HCl), pH 7.4 at 4°C and centrifuged at 12,000 r min−1 for 30 min at 4°C. The supernatant was collected for analysis, and the protein concentrations were determined in heart extract using Bradford colorimetric protein determination at 595 nm. 39
Determination of biochemical parameters
Fragments of hearts were quickly dissected, frozen in liquid nitrogen and stored at −20°C for biochemical analysis. The biochemical parameters aspartate transaminase (AST), alanine transaminase (ALT), Total protein, triglycerides (TG), total cholesterol (T-CHOL), lactate deshydrogenase (LDH) and creatine phosphokinase (CPK) were determined with the biochemical parameter counter (C×9 PRO; Bechman Coulter, Brea, CA, USA), and results were expressed as international units per milligram of protein.
Measurement of SOD activity
Superoxide dismutase (SOD) is one of the most important antioxidant enzymes. It catalyzes the dismutation of the superoxide anion into hydrogen peroxide (H2O2) and molecular oxygen. SOD activity was determined according to the method described by Marklund and Marklund 41 by assaying the autoxidation and illumination of pyrogallol at 440 nm for 3 min. One unit of SOD activity was calculated as the amount of protein that caused 50% pyrogallol autoxidation inhibition. The SOD activity is expressed as units per milligram protein.
Measurement of CAT
Catalase (CAT) is an antioxidant enzyme that catalyses the decomposition of H2O2 to water and oxygen. CAT activity was measured according to the method described by Aebi 42 by assaying the hydrolysis of H2O2 and the resulting decrease in absorbance at 240 nm over a 3 min period at 25°C. The activity of CAT was calculated using the molar extinction coefficient of 0.04 mM−1 cm−1. The results were expressed as micromoles per minute per milligram protein.
Protein carbonyl assay
Protein carbonyls content was determined as described by Mercier et al. 43 in heart homogenate by measuring the reactivity of carbonyl groups with 2,4-DNPH, as DNP hydrazones formed from the reaction are easily quantifiable at 370 nm. Thus, 200 µL of supernatant of heart homogenate were placed in two glass tubes. Then, 800 µL of 10 mM 2,4-DNPH in 2.5 M HCl were added. Tubes were left for 1 h of incubation at room temperature in the dark. Samples were vortexed every 15 min. Then, 1 mL of 20% TCA was added to the samples, and the tubes were left in ice bucket for 10 min and centrifuged for 5 min at 4000 r min−1 to collect the protein precipitates, and the supernatants were discarded. Next, another wash was performed using 1 mL of 10% Trichloroacetic acid (TCA), and protein pellets were broken mechanically with the aid of a glass rod. Finally, the pellets were washed with 1 mL of ethanol–ethyl acetate (1:1, v/v) to remove the free 2,4-DNPH. The final precipitates were dissolved in 500 µL of guanidine hydrochloride (6 M) and were left for 10 min at 37°C with general vortex mixing. Any insoluble materials were removed by additional centrifugation. Protein carbonyls concentration was determined from the absorbance at 370 nm, applying the molar extinction coefficient of 22 mM−1 cm−1. A range of nanomoles of carbonyls per millilitre is usually obtained for most proteins and is related to the protein content in the pellets.
Evaluation of lipid peroxidation status
Lipid peroxidation is the degradation of lipids that occurs as a result of oxidative damage. Polyunsaturated lipids are susceptible to an oxidative attack, typically by reactive oxygen species (ROS), resulting in a well-defined chain reaction with the production of end products such as malondialdehyde (MDA). The measurement of MDA was assayed according to the method of Ohkawa et al. 44 Samples containing 0.2 mL of heart homogenate were combined with 0.2 mL of 8.1% sodium dodecyl sulphate (SDS), 1.5 mL of 20% acetic acid adjusted to pH 3.5 and 1.5 mL of 0.8% thiobarbituric acid. The mixture was brought to a final volume of 4 mL with distilled water and heated to 95°C for 120 min. After cooling to room temperature, 5 mL of mixture of n-butanol and pyridine (15:1, v/v) was added to each sample, and the mixture was shaken vigorously. After centrifugation at 4000 r min−1 for 10 min, the supernatant fraction was isolated and the absorbance measured at 546 nm. The concentration of MDA was determined according to a standard curve.
Western blot analysis
Equal amounts of proteins (25 μg) were separated by 12% SDS-polyacrylamide gel electrophoresis. Separated proteins were electroblotted in the transfer buffer (10 mL Tris base, pH 8.3, 96 mM glycine and 10% methanol). The membrane was then blocked in Tris-buffered saline (TBS) (20 mM Tris-HCl, pH 7.5; 500 mM sodium chloride) containing 5% of Bovine serum albumin (BSA). The membrane was washed in TBS containing 0.3% Tween 20 and probed with an antibody for Hsp 70, Bax, Bcl2 and p53 at a 1:1000 dilution for 2 h at room temperature, and the membrane was then washed and incubated with goat anti-mouse alkaline phosphate conjugated at a 1:3000 dilution for 1 h. Next, the membrane was washed and the chromogenic substrate 5-bromo-4-chloro-3-indolyl phosphate/nitro blue tetrazolium was added to localize antibody binding. Hsp 70, Bax, Bcl2 and p53 levels were then determined by computer-assisted densitometric analysis (densitometer, GS-800, BioRad Quantity One, Hercules, California, USA).
Statistical analysis
Data are expressed as the mean ± standard deviation of the means. The analysis parameters were tested for homogeneity of variance and normality, and they were found to be normally distributed. The data were therefore analyzed using a one-way analysis of variance with a post hoc Tukey–Kramer test to identify significance between groups and their respective controls. In all cases, p < 0.05 was considered statistically significant.
Results
Biochemical parameters analysis
Table 1 summarizes the results of measurement of biochemical parameters in heart tissues after 24 h exposure to ZEN (40 mg kg−1 b.w.) alone or in combination with different doses of CRO (50, 100 and 250 mg kg−1 b.w.). Relative to control, ZEN promoted a significant increase in ALT, AST, TG, LDH, CPK and T-CHOL levels (p < 0.05). Regarding pretreatment with CRO, these levels were significantly reduced in a dose-dependent manner. We observed a significant decrease in ALT, AST, TG, LDH, CPK and T-CHOL levels (p < 0.05) across mice pretreatment with 250 mg kg−1 of CRO. Therefore, ZEN disturbed significantly the homeostasis of cardiac tissues in mice by changing the biochemical parameters, whereas CRO provided great cardioprotection against ZEN-induced toxicity.
Effect of CRO at different doses and its protective role on biochemical markers in heart tissues of Balb/c mice treated with ZEN by intraperitoneal route.a

AST: aspartate transaminase; ALT: alanine transaminase; CRO: crocin; CTL: control; CPK: creatinin phosphokinase; LDH: lactate deshydrogenase; SD: standard deviation; TG: triglycerides; T-CHOL: total; ZEN: zearalenone.
aValues are expressed as mean ± SD (six animals were treated per group).
b p < 0.05: Values are significantly different from CTL.
c p < 0.05: Values are significantly different from treated groups with ZEN alone.
Induction of lipid peroxidation
Results of the effect of ZEN alone and in combination with CRO on the induction of lipid peroxidation in heart extracts as determined by MDA level are shown in Figure 2. ZEN, at 40 mg kg−1 b.w., induces a significant increase in MDA formation as compared to the control groups in heart extract. Thus, the MDA level increases from a basal level of 4.57 ± 0.57 nmol mg−1 of protein to reach 32.63 ± 5.28 nmol mg−1 of protein. The increase in MDA levels was about sevenfold as compared to the control group (p < 0.05).
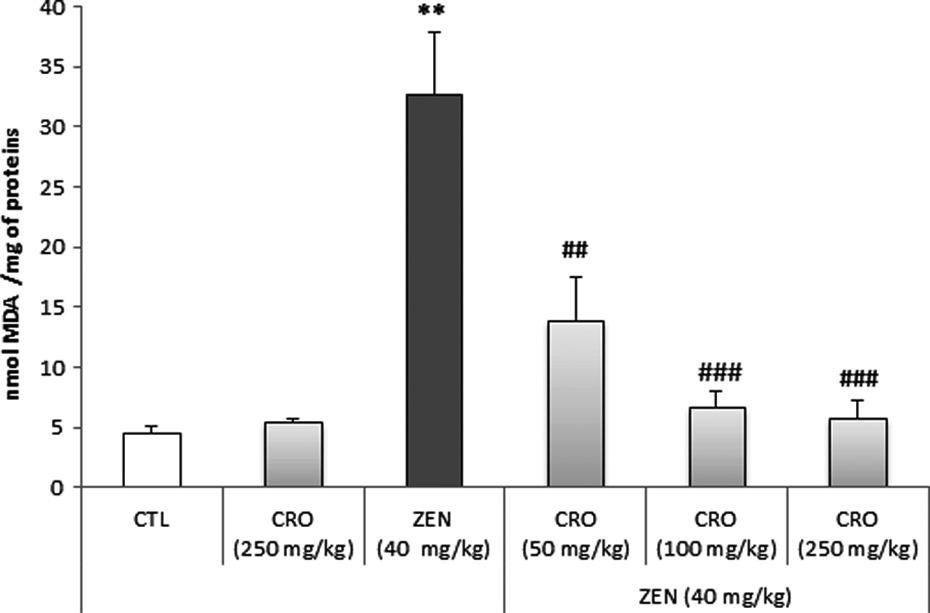
Lipid peroxidation as determined by MDA level in heart tissues of Balb/c mice exposed to (i) a single dose of ZEN (40 mg kg−1 b.w.) for 24 h and (ii) CRO (50, 100 and 250 mg kg−1 b.w.). Results are expressed as means ± SD from three independent experiments. **p < 0.01: from control; ## p < 0.01 and ### p < 0.001: from groups treated with ZEN. CTL: control; MDA: malondialdehyde; ZEN: zearalenone; b.w.: body weight; CRO: crocin.
Interestingly, treatment of animals with ascending doses of CRO (50, 100 and 250 mg kg−1 b.w.) showed a sharp decrease of MDA level in heart extract. MDA level has decreased significantly to reach the control level already with the highest tested dose of 250 mg kg−1 b.w. of CRO (Figure 2).
Protein carbonyl assay
The formation of protein carbonyls, indicative of severe protein oxidation, was assayed in heart homogenate, and the results are illustrated in Figure 3. ZEN generates the formation of protein carbonyls as compared to control groups in heart extract. In fact, the protein carbonyls level increases from basal value of 0.41 ± 0.11 nmol mg−1 of protein in the control group to reach 1.30 ± 0.23 nmol mg−1 of protein in ZEN-treated group. However, ascending doses of CRO remarkably decreased the formation of protein carbonyls induced by ZEN (40 mg kg−1 b.w.) in a dose-dependent manner in heart extracts. Indeed, the concentration of protein carbonyls reached the control level with CRO at 250 mg kg−1 b.w. (Figure 3).

Concentrations of protein carbonyls in heart tissues of Balb/c mice exposed to (i) a single dose of ZEN (40 mg kg−1 b.w.) for 24 h and (ii) CRO (50, 100 and 250 mg kg−1 b.w.). Results are expressed as means ± SD from three independent experiments. **p < 0.01: from control; # p < 0.05 and ## p < 0.01: from groups treated with ZEN. CTL: control; ZEN: zearalenone; b.w.: body weight; CRO: crocin.
CAT activity
As shown in Figure 4, ZEN induced a marked increase in CAT activity in heart extracts. This increased activity was about fourfold as compared to the control group (p < 0.05). Yet, when mice were given CRO, a significant decrease of this activity in dose-dependent manner was noticed. At the highest dose of CRO (250 mg kg−1 b.w.), the CAT activity reached the control level (Figure 4).

CAT activity measured in heart tissues of Balb/c mice exposed to (i) a single dose of ZEN (40 mg kg−1 b.w.) for 24 h and (ii) CRO (50, 100 and 250 mg kg−1 b.w.). Results are expressed as means ± SD from three independent experiments. ***p < 0.001: from control; ## p < 0.01 and ### p < 0.001: from groups treated with ZEN. CTL: control; CAT: catalase; ZEN: zearalenone; CRO: crocin; b.w.: body weight.
SOD activity
Figure 5 illustrates the effect of ZEN and CRO on SOD activity. Compared with the control group, there was a significant increase in the SOD activity in the ZEN-treated group. In fact, the SOD activity measured in heart homogenate increased from basal value of 0.70 ± 0.19 U mg−1 of protein in control group to reach 6.82 ± 0.72 U mg−1 of protein in ZEN-treated group. Thus, the SOD activity was about ninefold. Nevertheless, pretreatment with CRO at different doses showed a remarkable decrease in the SOD activity (Figure 5).

SOD activity measured in heart tissues of Balb/c mice exposed to (i) a single dose of ZEN (40 mg kg−1 b.w.) for 24 h and (ii) CRO (50, 100 and 250 mg kg−1 b.w.). Results are expressed as means ± SD from three independent experiments. ***p < 0.001: from control; # p < 0.05 and ## p < 0.01: from groups treated with ZEN. CTL: control; SOD: superoxide dismutase; ZEN: zearalenone; b.w.: body weight; CRO: crocin.
Hsp 70 expression
ZEN induces the expression of Hsp 70 genes in heart as evidenced by the example of immunoblotting illustrated in Figure 6(a), which was further confirmed by results of scanning densitometry (Figure 6(b)). However, the administration of ascending doses of CRO reduces progressively the amounts of Hsp 70 (Figure 6(a) and (b)).

(a) Immunochemical detection of Hsp 70 in heart tissues of Balb/c mice exposed to (i) a single dose of ZEN (40 mg kg−1 b.w.) for 24 h and (ii) CRO (50, 100 and 250 mg kg−1 b.w.). (b) Scanning densitometry of Hsp 70 levels are expressed as percentage of control. Results are expressed as means ± SD from three independent experiments. **p < 0.01: from control; ## p < 0.01: from groups treated with ZEN. CTL: control; Hsp 70: 70 kilodalton heat shock protein; b.w.: body weight; ZEN: zearalenone; CRO: crocin.
P53 and Bax/Bcl2 expression
To investigate the involvement of apoptosis in ZEN-induced cardiotoxicity in Balb/c mice, we evaluated the expression of p53, Bax and Bcl2 after 24 h of ZEN exposure. Our results indicate that the expression of apoptosis biomarker, p53, increased after ZEN treatment. This induction was correlated with the overexpression of pro-apoptotic protein, Bax, and the decrease of the anti-apoptotic protein, Bcl2 (Figure 7(a)). These data were confirmed by scanning densitometry (Figure 7(b)). The expression of p53 increased about fivefold compared with the control (p < 0.05), and the ratio of Bax to Bcl2 changed about 6.6-fold versus control (p < 0.05). However, pretreatment with CRO regulated ZEN-induced apoptosis. The expressions of p53 and Bax were decreased accompanied by an increase in Bcl2 (Figure 7; p < 0.05). Consequently, CRO inhibit ZEN-induced apoptosis in mice heart tissues.
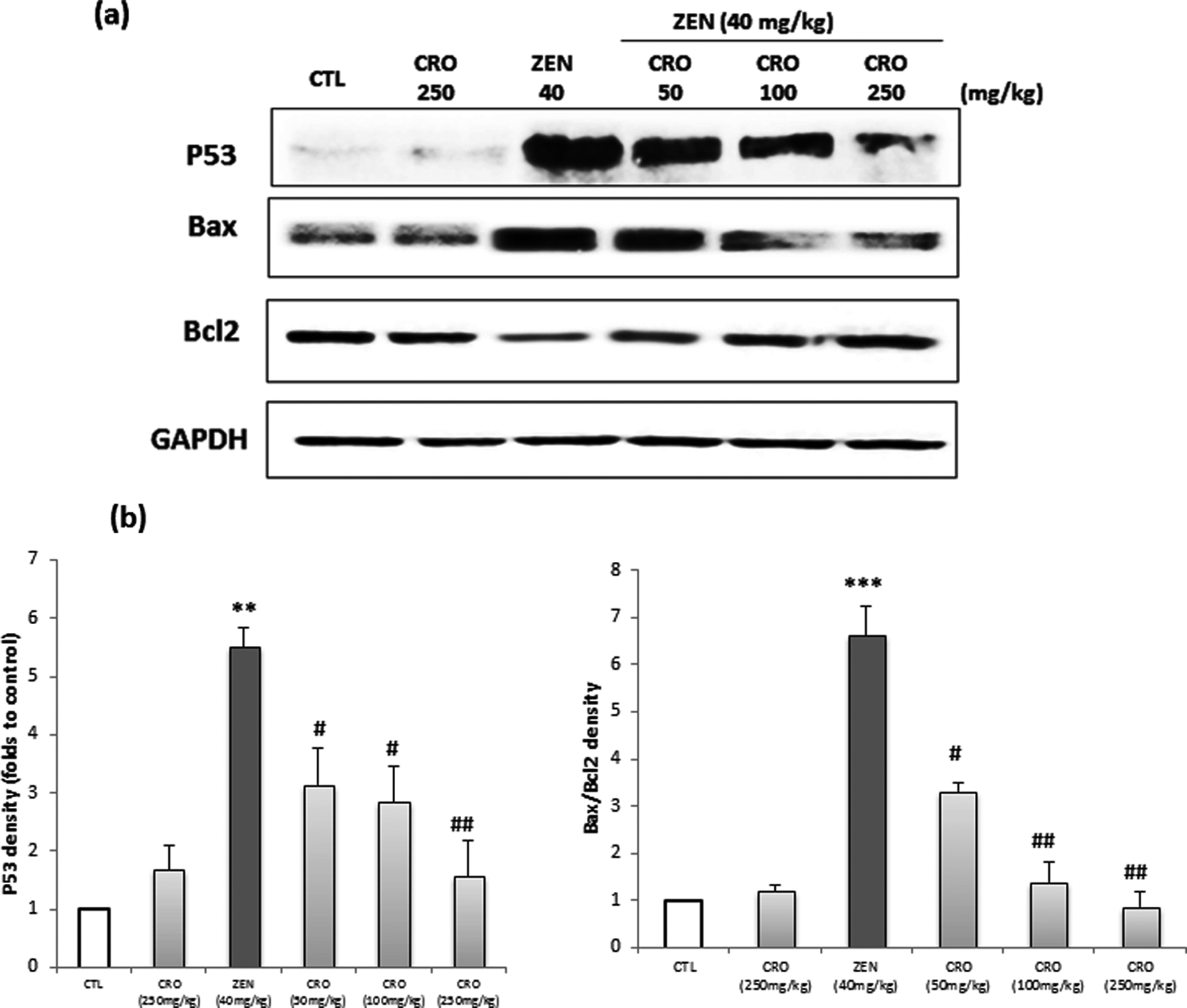
(a) Immunochemical detection of p53, Bax and Bcl2 in heart tissues of Balb/c mice exposed to (i) a single dose of ZEN (40 mg kg−1 b.w.) for 24 h and (ii) CRO (50, 100 and 250 mg kg−1 b.w.). (b) Scanning densitometry of p53, Bax and Bcl2 levels are expressed as percentage of control. Results are expressed as means ± SD from three independent experiments. **p < 0.01 and ***p < 0.001: from control; # p < 0.05 and ## p < 0.01: from groups treated with ZEN. CTL: control; ZEN: zearalenone; b.w.: body weight; CRO: crocin.
Discussion
The present study showed that ZEN (40 mg kg−1 b.w.) exhibits cardiac toxicity with acute exposure (24 h) by disturbing the biochemical parameters and inducing elevated cardiac marker CPK. These changes were associated with increased oxidative stress and apoptosis in cardiac tissues. Our results indicate that ZEN increased the level of intracellular enzymes, AST and ALT. It’s reported that an increase in the levels of these transaminases is related to the level of damaged cells. 45 We also observed that CPK level was increased by ZEN. CPK is one of the sensitive markers existing for the evaluation of damage to the heart muscle. 46 We have also shown that the cardiac level of MDA was significantly increased following ZEN administration. MDA, the end product of lipid peroxidation, is currently used to evaluate the presence of free radicals and lipid peroxidation-induced cardiovascular toxicity. 47,48 To further assess ZEN-induced oxidative damages in Balb/c mice, the generation of protein carbonyls was monitored. Protein carbonylation is a sign of irreversible oxidative damage, mostly leading to a loss of protein function, which may have lasting detrimental effects on cells and tissues. 49,50 Our results clearly showed that ZEN induced a marked increase in the generation of protein carbonyls in heart extract. Based only on the analysis of MDA and protein carbonyls (presumed late biomarkers of oxidative damage), it is difficult to define severe oxidative stress as a cause or a consequence of cellular toxicity. Thus, the levels of early markers of oxidative stress, including antioxidant enzymes and Hsp, may be altered in the presence of lower levels of oxidative stress and before the biomarkers of severe oxidative stress attributed to cytotoxicity appear. The induction of oxidative stress by ZEN in cardiac tissues was also investigated by assessing SOD and CAT activities. Our results clearly showed that ZEN enhances both SOD and CAT activities. We have also examined the induction of a range of cellular cytoprotective proteins known as Hsp, another early marker of oxidative injury. In fact, after oxidative stress enhancement, Hsps are induced and play a key role in cell protection and repair. 51,52 This induction is triggered by structural damage caused to cell proteins, mainly thiol oxidation and on general perturbations of the cellular redox status level. 53 –55
Our results clearly demonstrated that ZEN induced a sharp elevation in the expression level of Hsp 70 in heart homogenate. These findings are in accordance with other in vivo studies showing that ZEN disrupted the oxidative status in mice liver and kidney. 9,10,23
Evidence suggests that oxidative stress is a major apoptotic stimulus in various diseases such as cardiovascular problems. 56 According to the results, lipid peroxidation could be considered as a mechanism of ZEN cardiotoxicity, therefore to understand whether acute exposure to ZEN can induce apoptosis in heart tissue, some key factors involved in apoptosis pathway were evaluated in this study. The p53 expression has been shown to orchestrate the process of apoptosis, 57,58 and its upregulation is associated with the activation of apoptosis process. The proteins of Bcl2 family are involved in the regulation of the intrinsic apoptotic pathway. 59 Bax is a pro-apoptotic protein, and Bcl2 possesses antiapoptotic properties by stabilizing mitochondrial membrane and suppressing the release of cytochrome c. The balance between these proteins is important in the progression of apoptosis. 60 In this study, ZEN increased the p53 expression and also the ratio of Bax/Bcl2 (p < 0.05). This increase in p53 and in Bax/Bcl2 ratio by ZEN could be considered as a predictor of ZEN-induced apoptosis in cardiac tissue. The induction of the apoptotic process by ZEN was also reported in vitro using different cell lines. 18,61,62
In the present study, we evaluated the protective role of CRO, a natural carotenoid, against ZEN-induced toxicity in heart. Our results showed that mice pretreatment by CRO in different doses downregulated the levels of CPK, ALT, AST, TG, LDH and T-CHOL near to normal levels when compared with the ZEN-treated group. CRO is a potent antioxidant agent. 31 Cardioprotective effects of CRO via antioxidant effect has been well reported. 46,63 Thus, we have demonstrated that CRO significantly reduced the levels of MDA and protein carbonyls, modulated the activities of SOD and CAT and decreased the expression of Hsp70. Antioxidants are known to regulate gene expression and signal transduction pathways so that the occurrence of apoptosis can be prevented. Antiapoptotic effect of CRO has been shown through inhibitory effect against TNF-α–induced cell death, 39 modulatory properties on expression of Bcl2 family proteins and inhibition of release of cytochrome c to the cytosol. 64 Our results showed that CRO (25, 50 and 250 mg kg−1 b.w.) plus ZEN clearly reduced the increase of the protein level of p53 and the Bax/Bcl2 ratio, compared to the ZEN-treated group. It could be concluded that CRO could maintain a redox balance in cardiac tissue and modulate the susceptibility of biological membranes to interact with ROS, as indicated by the marked decrease in MDA level and subsequently prevent tissue against apoptosis. Also, it can maintain the mitochondria membrane stability and reduce the increase of Bax/Bcl2 ratio induced by ZEN. Regarding the hydrophilicity of CRO, it may be suggested that like hydrophilic antioxidant vitamin C, CRO can restore the antioxidant property of oxidized α-tocopherol by recycling the tocopheroxyl radical. 65
In conclusion, we believe that this is the first report regarding the induction of oxidative stress and apoptosis by ZEN in cardiac tissue. Our data also indicate that CRO has protective effects against ZEN-induced cardiotoxicity by regulating the biochemical parameters, attenuating lipid peroxidation and protein carbonyls levels and modulating SOD and CAT activities and Hsp 70 expression. CRO also alleviated apoptosis by decreasing the p53 expression and Bax/Bcl2 ratio.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Ministère Tunisien de l’Enseignement Superieur et de la Recherche Scientifique et de la Technologie (Laboratoire de Recherche sur les Substances Biologiquement Compatibles, LRSBC).
