Abstract
Ferroptosis is potential to relieve drug resistance in hepatocellular carcinoma (HCC). Glutathione peroxidase 4 (GPX4) is a critical modulator of ferroptosis. This study discussed the mechanism of GPX4-inhibited ferroptosis in sorafenib resistance in HCC. HCG18 in HCC cells was detected. Sorafenib resistant (SR) cell line Huh7-SR cells were treated with sorafenib (0, 2.5, 5, 7.5, 10 μM). After silencing HCG18 in Huh7-SR cells, cell activity, proliferation and apoptosis were detected. The levels of iron, the concentration of MDA, GSH and lipid reactive oxygen species (ROS) were measured to evaluate the ferroptosis. The downstream mechanism of HCG18 was predicted and verified. Huh7-SR cells were infected with lentivirus sh-HCG18 to establish xenograft tumor model. HCG18 was elevated in HCC cells and associated with sorafenib resistance. Silencing HCG18 inhibited cell proliferation, promoted apoptosis, and impaired sorafenib resistance. Ferroptosis was inhibited in Huh7-SR cells, while silencing HCG18 inhibited sorafenib resistance by promoting ferroptosis. GPX4 overexpression averted the promotion of sh-HCG18 on ferroptosis, thereby reducing sorafenib resistance. HCG18 sponged miR-450b-5p to regulate GPX4. Collectively, Silencing HCG18 inhibits GPX4 by binding to miR-450b-5p, promotes GPX4-inhibited ferroptosis, and averts sorafenib resistance in HCC.
Introduction
Hepatocellular carcinoma (HCC) is a prevailing malignancy of the liver and the histological subtype of liver cancer.1,2 HCC is the seventh most predominant cancer and the fourth critical cause of cancer mortality worldwide. 3 Although therapeutic strategies have recently advanced, tumor metastasis and drug resistance continue to pose challenges in the treatment of HCC. 4 The multikinase inhibitor sorafenib is the only systemic treatment identified to prolong the overall survival of HCC patients. 5 Sorafenib is acknowledged as the standard drug option for advanced HCC, 6 which enjoys strong inhibition both in tumor proliferation and tumor angiogenesis. 7 However, tumor cells can acquire primary or secondary resistance to sorafenib 8 and the mechanism of sorafenib resistance (SR) remains poorly characterized. Therefore, it is urgent to discover new alternative strategies to overcome SR.
Sorafenib exerts anti-tumor effects by regulating ferroptosis. 9 Ferroptosis is a newly unveiled mode of programed cell death induced by lipid peroxidation and is promising for cancer management. 10 Furthermore, ferroptosis plays a predominant role in the pathology of the liver, including HCC.11,12 Preventing ferroptosis is likely to stimulate SR to cancer cells.13,14 But the mechanism of ferroptosis affecting the SR in HCC is largely unknown.
As crucial molecules in biological functions, long noncoding RNAs (lncRNAs) have obtained widespread attention for their actions in drug resistance in cancers, including HCC.15–17 LncRNA HCG18 is markedly increased in HCC and their upregulation indicates poor prognosis. 18 HCG18 silencing prevents proliferation and migration while induces the apoptosis of HCC cells. 19 Evidence has supported the significant roles of competing endogenous RNA (ceRNA) network in HCC. 18 Aberrant expression of microRNAs (miRNAs) has frequently been observed in human cancers including HCC, 3 and is concerned with liver metabolism, liver injury, liver fibrosis and tumor development. 20 miRNAs are also major regulators of many intracellular processes related to SR in HCC. 21 miR-450b-5p could inhibit cell viability, proliferation rate, and invasion of HCC cells. 22 miR-450b-5p is also implicated in chemoresistance to 5-fluorouracil in colorectal cancer and radioresistance of nasopharyngeal carcinoma.23,24 HCG18 is included in the ceRNA network in disease progression.25,26 However, the mRNA-miRNA-lncRNA analysis involving HCG18 and miR-450b-5p in HCC is unclear. This study attempted to identify the mechanism of HCG18 in regulating ferroptosis and SR in HCC.
Materials and methods
Ethical approval
The animal experiment was approved by the ethics committee of The Second Hospital of Shandong University (KYLL-2021(KJ)A-0468). The use, care, and operation procedures of all animals complied with the National Institutes of Health Guide for the Care and Use of Experimental Animals. 27
Cell culture and construction of drug-resistant strain
Four human hepatoma cell lines including SK-hep1, HepG2, HCCLM3, and Huh7 and normal control cell line LO2 were from ATCC (Manassas, VA, USA). All the cells were cultured in Dulbecco’s modified eagle’s medium (DMEM; GIBCO, Gaithersburg, MD, USA) and 10% fetal bone serum (FBS; HyClone, Logan, UT, USA) and 1% penicillin/streptomycin (100 IU/mL penicillin, 100 μg/mL streptomycin) at 37°C/5% CO2.28,29
To establish sorafenib resistant (SR) cells, HCC cell line Huh7 was first exposed to low dose (2.5 μM) of sorafenib. When the cells showed stable growth, we began to enhance the dose of sorafenib (5, 7.5, and 10 μM). The medium containing sorafenib was changed every two days for 6 months. In further experiments, SR cells grew slowly in the medium containing 10 μM sorafenib (clinically relevant dose). SR cells (Huh7-SR) are usually maintained in a constant culture condition containing sorafenib. 30 Sorafenib was purchased from Bayer Corporation (Nexavar, Beijing, China) and dissolved in 100% dimethyl sulfate (DMSO). 31
Cell transfection
Ferrostatin-1 (1 μM 13 ) was from MedChemExpress (HY-100,579, Shanghai, China). miR-450b-5p inhibitor, shRNA sequence targeting HCG18 (sh-HCG18), overexpressed GPX4 plasmid (oe-GPX4) and corresponding controls were provided by GenePharma (Shanghai, China). The cells were delivered according to Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA).
Reverse transcription quantitative polymerase chain reaction
Primers for RT-qPCR.

3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay
Cells (2.5 × 103) were cultured in 96-well plates and exposed to different concentrations of sorafenib (0, 2.5, 5, 7.5, 10 μM) 30 for 48 h. The medium was replaced by fresh DMEM containing 10 μL MTT (5 mg/mL, Sigma-Aldrich, St. Louis, MO, USA). 33 After 4 h, the medium was discarded and cells were treated with 100 μL DMSO at 37°C for 10 min. Finally, the optical density (OD) was detected at 490 nm.
Colony formation assay
The transfected Huh7-SR cells were incubated in the medium containing 5 μM sorafenib for 48 h, and the cell proliferation was detected by colony formation assay. Briefly, 0.7% agarose was added to the fresh medium, 2 mL agar was put into the culture dish (100 mm in diameter) until the agar was evenly spread on the bottom, and the culture dish was preserved for subsequent experiments after cooling and solidification. Next, the diluted 1 mL cell suspension was mixed with an equal volume of 0.7% agarose solution, and the cells were seeded at 11 × 104 cells/100 cm2. After coagulation, the upper agar was supplemented with 2–3 mL medium at 37°C and incubated in 5% CO2 for 2 weeks (the medium was changed every 2–3 days). The cells were counted and the images were obtained by DMM-300D microscope (Caikon, Shanghai, China). The experiment was carried out three times to obtain the average colonies. 34
Flow cytometry
The transfected Huh7-SR cells were incubated in the medium containing 5 μM sorafenib for 48 h. Then, cells (1 × 105) were placed in 10 μL binding buffer containing 5 μL Annexin V and 5 μL propidium iodide for 15 min at room temperature, and the apoptosis percentage (%) was determined using flow cytometry. 17
Iron assay
According to the instruction of an iron assay kit (MAK025, Sigma-Aldrich), the cells were supplemented into the buffer to measure the iron level. 35 Next, the cells were centrifuged for 10 min at 13,000 g at 4°C to get the supernatant. In 96-well microplates, the supernatant (50 μL) was mixed with 50 μL buffer at 25°C for 30 min, and 200 μL reagent mixture at 25°C for 30 min devoid of light. The OD value was evaluated at 593 nm.
Detection of malondialdehyde and glutathione
According to the instructions, MDA assay kit (MAK085, Sigma-Aldrich) was utilized to assess the level of MDA and GSH assay kit (S0053, Beyotime, Shanghai, China) was utilized to measure the level of GSH. 35
Detection of lipid reactive oxygen species
The cells were plated on glass cover slides and then treated with C11-BODIPY™ 581/591 probe was incubated for 30 min. After washing with 1 × phosphate buffer saline (PBS) for 3 times, the cells were fixed with 4% paraformaldehyde. Zeiss fluorescence axial microscope (SensiCam-PCO computer Optics GmbH, Germany) with CCD black and white TV camera was used to collect images. Image J software was adopted to quantify lipid ROS (green fluorescence).
Western blot
The cells or tissues were lysed with enhanced RIPA lysate containing protease inhibitor (Boster, Wuhan, Hubei, China), and the concentration was assessed using the bicinchoninic acid (Beyotime). The protein was isolated by SDS-PAGE (10% (w/v)) and moved to polyvinylidene fluoride membranes, which were blocked for 2 h with 5% bovine serum albumin to block nonspecific binding. Next, the membranes were added with diluted primary antibodies rabbit anti-GPX4 (ab125066, 1:2000, Abcam, Cambridge, MA, USA) and GAPDH (ab181602, 1: 10,000, Abcam) at 4°C overnight. Then, the membranes were added with HRP-labeled goat anti-rabbit IgG secondary antibody (1:2000, ab205718, Abcam) for 1 h. The enhanced chemiluminesence was used for development and Image Pro Plus 6.0 (Media Cybernetics, San Diego, CA, USA) was used to quantify gray scale. 35 GAPDH was used as an internal reference.
Bioinformatics analysis
GEPIA website (https://gepia.cancer-pku.cn/detail.php) 36 was used to predict the expression of HCG18 in HCC, UALCAN website (https://ualcan.path.uab.edu/analysis.html) 37 was used to predict the relationship between the expression of HCG18 and the prognosis of HCC. GEPIA website was used to predict the expression of GPX4 in HCC, and UALCAN website was used to predict the relationship between the expression of GPX4 and the prognosis of HCC. Starbase website (https://starbase.sysu.edu.cn/index.php) 38 was used to predict miRNAs binding to HCG18 and GPX4, respectively. ONCOMIR website (https://www.oncomir.org/oncomir/search_cancer_form_miR.html) 39 was used to predict miRNAs associated with HCC tumorigenesis, and the intersection was taken via the online website (https://bioinformatics.psb.ugent.be/webtools/Venn/). The Starbase website was used to predict the possible binding sites of HCG18 and miR-450b-5p and the possible binding sites of miR-450b-5p in GPX4 3′UTR.
Dual-Luciferase assay
Through the Starbase website, the binding sites of miR-450b-5p with HCG18 and GPX4 were predicted. The synthesized HCG18 gene fragments HCG18-wt and GPX4-wt containing miR-450b-5p binding sites and corresponding HCG18-mut and GPX4-mut fragments according to the binding sites were constructed and inserted into pmiR reporter plasmids (Huayueyang Biotechnology, Beijing, China). The constructed plasmids were cotransfected into HEK293 T cells (Beinuo Biotechnology, Shanghai, China) with mimic NC or miR-450b-5p mimic. After 48 h, the cells were lysed and luciferase activity was assessed using a luciferase detection kit (K801-200; Biovision, Mountain View, CA, USA). 40
RNA immunoprecipitation
The binding of miR-450b-5p to GPX4 and HCG18 was detected by RIP kit (Millipore, MA, USA). 41 Huh7-SR cells were rinsed with precooled PBS and the supernatant was removed. The cells were lysed using equal volume RIPA lysate (P0013b, Beyotime) for 5 min, and centrifuged for 10 min at 4°C to get the supernatant. One part of the cell extract was used as input, and the other part was incubated with the antibody for coprecipitation. The RNA was extracted from the sample after digestion with proteinase K and used for RT-qPCR detection of HCG18, miR-450b-5p and GPX4. The antibody Ago2 (1:100, ab32381, Abcam) was mixed for 30 min, and rabbit anti IgG (1:100, ab109489, Abcam) was used as a negative control.
Tumor xenograft in nude mice
BALB/c nude mice (4–6 weeks old) from Beijing Vital River Laboratory Animal Technology (Beijing, China) were reared in a standard laboratory with free access to food and water. Lentivirus LV-sh-NC and LV-sh-HCG18 were from GenePharma (Shanghai, China). In order to establish subcutaneous xenograft tumor models, Huh7-SR cells were infected with lentivirus LV-sh-NC or LV-sh-HCG18 and then resuspended in PBS at 5 × 105/mL. Totally 100 μL cells were subcutaneously injected into the right dorsal area of each nude mouse. When the tumor volume reached 150 mm3, sorafenib (10 mg/kg) was orally administered to nude mice once a day to the end. Tumor volume (V) was calculated: V = 0.5 × L × W2, where L and W were defined as tumor length (L) and width (W). After 28 days of cell injection, the nude mice were euthanized by intraperitoneal injection of excessive pentobarbital sodium (100 mg/kg). 42 Then, the subcutaneous transplanted tumor was stripped and the tumor weight was recorded, followed by RT-qPCR and Western blot.
Statistical analysis
SPSS21.0 software (IBM Corp. Armonk, NY, USA) and GraphPad Prism 8.0 (GraphPad Software Inc., San Diego, CA, USA) were used for data processing and mapping. The data are described as mean ± standard deviation (SD). The normal distribution and homogeneity of variance tests were performed and the values were in normal distribution and the variance was homogeneous. Independent t test was adopted for comparison between two groups and one-way or two-way analysis of variance (ANOVA) was adopted for comparison among multi-groups, followed by Tukey’s multiple comparisons test. p < 0.05 indicated statistical significance.
Results
High expression of HCG18 in HCC is associated with SR
LncRNA HCG18 is upregulated in gastric cancer,
43
lung adenocarcinoma,
44
colorectal cancer
45
and other cancers, and participates in tumor development. However, the action mechanism of HCG18 on HCC still needs to be studied, and the regulation of HCG18 on SR of HCC has not been reported. Through the GEPIA website prediction, we discovered that HCG18 was elevated in HCC (Figure 1(a)), and HCG18 was linked with the prognosis of HCC (Figure 1(b)). HCG18 is highly expressed in HCC and is associated with sorafenib resistance. a: GEPIA website predicted that HCG18 is highly expressed in HCC; b: Ualcan website predicted that HCG18 is related to HCC prognosis; c: HCG18 expression in four human hepatoma cell lines and normal control cell line LO2 was detected by RT-qPCR; d: Huh7 and Huh7-SR cells were treated with sorafenib of different concentrations, and the cell activity was detected by MTT; e: HCG18 expression in cells detected by RT-qPCR. Cell experiment was repeated three times, and the data were described as mean ± SD. One-way ANOVA was used in Figure c, two-way ANOVA was used in Figure d, followed by Tukey’s multiple comparisons test; t test was used in Figure E; *p < 0.05.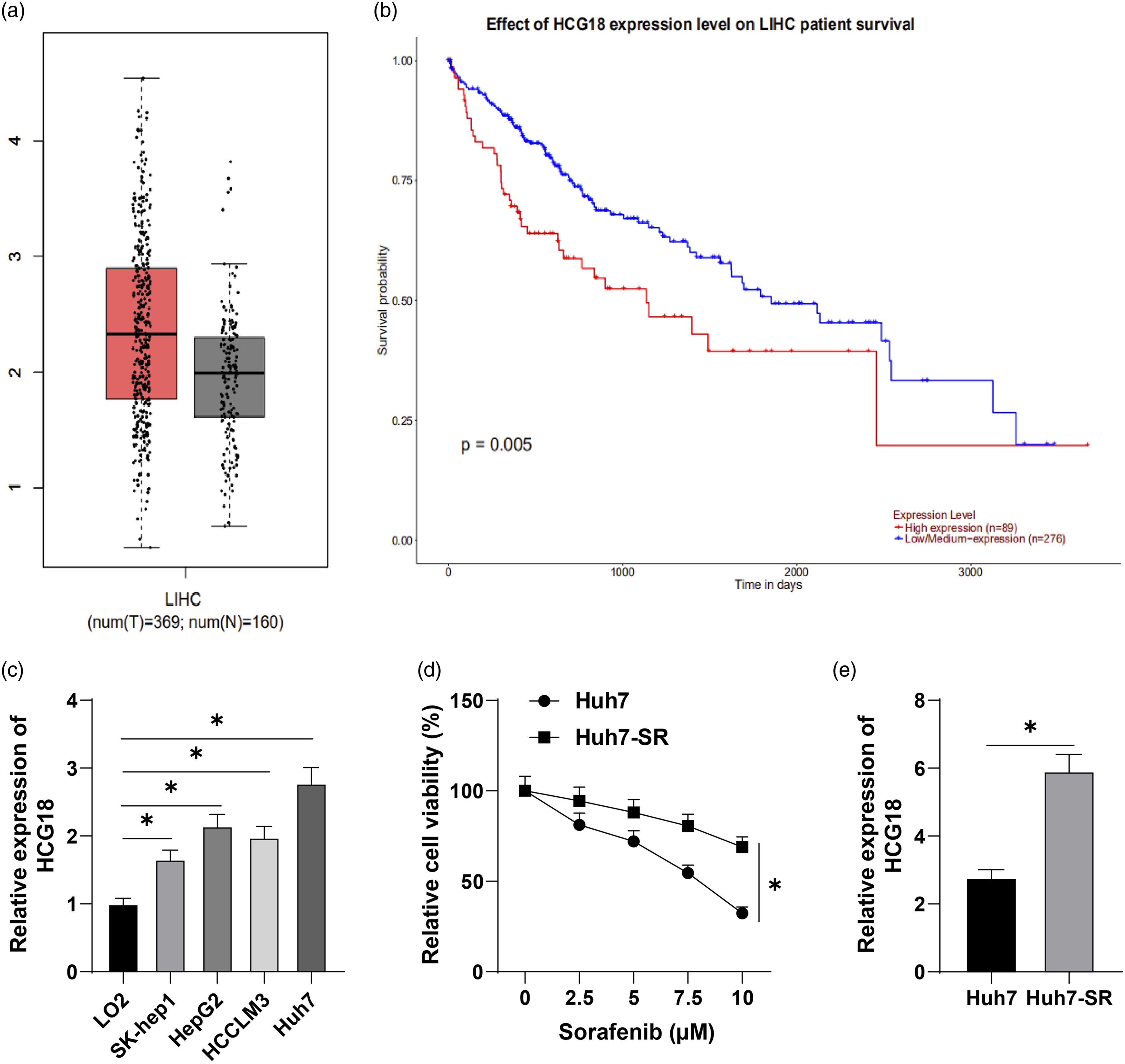
Then, we selected four human hepatoma cell lines SK-hep1, HepG2, HCCLM3 and Huh7, and normal control cell line LO2, and detected HCG18 expression by RT-qPCR, which demonstrated that HCG18 was significantly overexpressed in hepatoma cell lines (p < 0.05, Figure 1(c)). We further successfully constructed sorafenib resistant cell line Huh7-SR by selecting the cell line Huh7 with the highest expression of HCG18. After Huh7 and Huh7-SR cells were treated with sorafenib (0, 2.5, 5, 7.5, 10 μM), MTT assay was utilized to test cell activity. It was found that sorafenib had a weaker inhibitory effect on Huh7-SR cells than Huh7 cells (p < 0.05, Figure 1(d)). Meanwhile, HCG18 expression in Huh7-SR cells was higher than that in Huh7 cells (p < 0.05, Figure 1(e)). These results suggest that HCG18 is elevated in HCC and is associated with SR.
Silencing HCG18 inhibits SR in HCC
The above results were further verified in vitro. After Huh7-SR cells were transfected with shRNA targeting HCG18, HCG18 in Huh7-SR cells was significantly decreased (p < 0.05, Figure 2(a)), and sh-HCG18-1 with the best silencing efficiency was used for subsequent experiments. Then, the transfected Huh7-SR cells were treated with sorafenib (0, 2.5, 5, 7.5, 10 μM). MTT assay elicited that the cell activity was significantly inhibited after silencing HCG18 (p < 0.05, Figure 2(b)). The results of colony formation assay and flow cytometry revealed that silencing HCG18 significantly suppressed proliferation (p < 0.05, Figure 2(c)) and enhanced apoptosis (p < 0.05, Figure 2(d)). Briefly, silencing HCG18 can inhibit cell proliferation, promote cell apoptosis, and inhibit SR in HCC. Silencing HCG18 inhibits sorafenib resistance in HCC. Huh7-SR cells were transfected with sh-NC or sh-HCG18. a: The expression of HCG18 in Huh7-SR cells was detected by RT-qPCR; b: Huh7-SR cells were treated with sorafenib of different concentrations, and the cell activity was detected by MTT; c: After 5 μM sorafenib treatment, the proliferation of Huh7-SR cells tested by colony formation assay; d: Apoptosis detected by flow cytometry. The cell experiment was repeated three times, and data were described as mean ± SD. One-way ANOVA was used in Figure a, two-way ANOVA was used in Figure b, Tukey’s multiple comparisons test was used for post hoc test; t test was used in Figure c-d; *p < 0.05.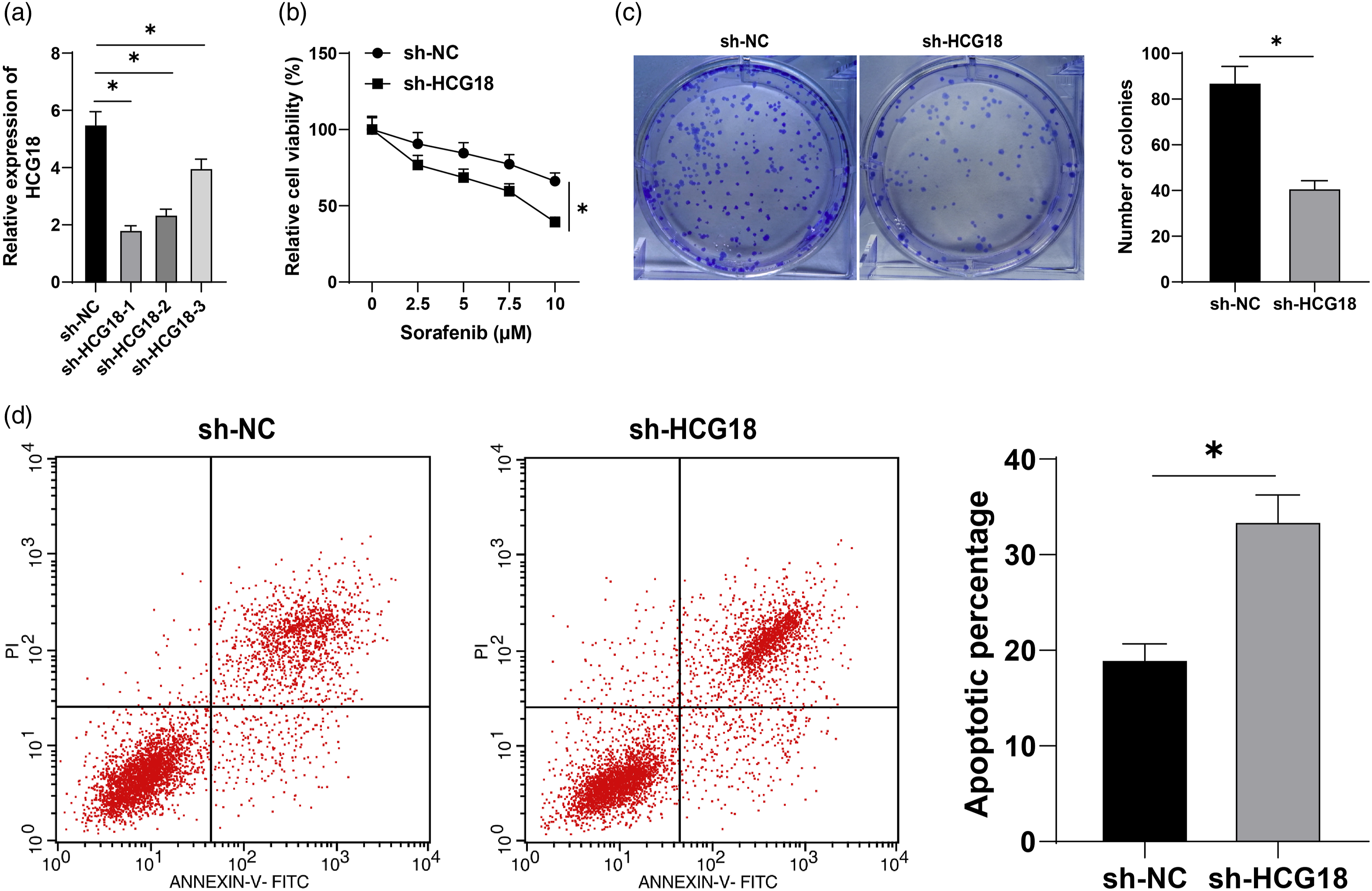
Silencing HCG18 inhibits SR in HCC by promoting cell ferroptosis
The research on ferroptosis in tumors has become one of the hot spots in recent years. Sorafenib can promote ferroptosis in HCC cells.
46
In order to further study the role of ferroptosis in SR of HCC, iron levels in Huh7 and Huh7-SR cells were detected by iron assay kit. The results showed that iron levels in sorafenib-resistant cells were clearly decreased (p < 0.05, Figure 3(a)). Additionally, MDA concentration was decreased (p < 0.05, Figure 3(b)), and GSH concentration was significantly enhanced (p < 0.05, Figure 3(c)). Then, C11-bodipy fluorescent probe was used to detect lipid ROS in cells, it was found that lipid ROS was decreased significantly (p < 0.05, Figure 3(d)). In brief, ferroptosis was inhibited in sorafenib resistant cells. Silencing HCG18 suppresses sorafenib resistance of HCC by promoting cell ferroptosis. a: Iron levels detected by iron assay kit; b-c: MDA and GSH concentrations detected by kits; d: C11-bodipy fluorescent probe detected the level of lipid ROS; e: After transfection of sh-NC or sh-HCG18 into Huh7-SR cells, iron levels in the cells were detected by iron assay kit; f–g: MDA and GSH concentrations in the cells were detected by kits; h: C11-bodipy fluorescent probe detected the level of lipid ROS; i: Huh7-SR cell activity evaluated by MTT. The cell experiment was repeated three times, and the data were expressed as mean ± SD. The t test was used in Figure a–h; Two-way ANOVA was used in Figure i, Tukey’s multiple comparisons test was used for post hoc test; *p < 0.05.
Then, we detected ferroptosis in sh-HCG18-transfected Huh7-SR cells. Compared with the sh-NC group, iron level (p < 0.05, Figure 3(e)) and MDA concentration (p < 0.05, Figure 3(f)) in HCG18 silenced Huh7-SR cells were increased, GSH concentration was decreased (p < 0.05, Figure 3(g)), and lipid ROS was significantly increased (p < 0.05, Figure 3(h)). These results suggest that silencing HCG18 can promote ferroptosis. Furthermore, we treated the sh-HCG18-transfected Huh7-SR cells with ferrostatin-1 (ferroptosis inhibitor) and treated the cells with sorafenib (0, 2.5, 5, 7.5, 10 μM). MTT assay showed that inhibition of ferroptosis significantly improved the cell viability (p < 0.05, Figure 3(i)). Taken together, silencing HCG18 suppresses SR of HCC by promoting cell ferroptosis.
Silencing HCG18 promotes GPX4-inhibited ferroptosis in sorafenib-resistant hepatoma cells
Ferroptosis is an iron-dependent cell death closely concerned with GPX4.
47
GEPIA website predicted that GPX4 was highly expressed in HCC (Figure 4(a)) and correlated with HCC prognosis (Figure 4(b)). In Huh7-SR cells transfected with sh-NC or sh-HCG18, GPX4 was significantly decreased after silencing HCG18 (all p < 0.05, Figures 4(c) and (d)), suggesting that silencing HCG18 inhibited GPX4. Therefore, we speculate that the regulation of HCG18 on ferroptosis of sorafenib-resistant hepatoma cells is inhibited by GPX4. Silencing HCG18 promoted GPX4-inhibited ferroptosis of sorafenib-resistant hepatoma cells. a: GEPIA website predicted that GPX4 is highly expressed in HCC; b: Ualcan website predicted that the expression of GPX4 is related to the prognosis of HCC;c–d: After sh-NC or sh-HCG18 was transfected into Huh7-SR cells, GPX4 expression was tested by RT-qPCR and Western blot; e–f: After sh-HCG18 and oe-GPX4 were transfected into Huh7-SR cells, GPX4 levels were tested by RT-qPCR and Western blot; g: iron levels detected by iron assay kit; h–i: MDA and GSH concentrations in the cells were detected by kits; j: C11-bodipy fluorescent probe was used to detect the level of lipid ROS; k: cell activity was detected by MTT. The cell experiment was repeated three times, and the data were expressed as mean ± SD. The t test was used in Figure c-d; one-way ANOVA was used in Figure e-j, two-way ANOVA was used in Figure k, followed by Tukey’s multiple comparisons test; *p < 0.05.
To verify the above hypothesis, we delivered sh-HCG18 and oe-GPX4 into Huh7-SR cells at the same time. RT-qPCR and Western blot elicited that oe-GPX4 significantly promoted GPX4 expression compared with the sh-HCG18 + oe-NC group (all p < 0.05, Figures 4(e) and (f)). Compared with the sh-HCG18 + oe-NC group, the iron level (p < 0.05, Figure 4(g)) and MDA concentration (p < 0.05, Figure 4(h)) of the sh-HCG18 + oe-GPX4 group were decreased, and GSH (p < 0.05, Figure 4(i)) was increased; meanwhile, C11-bodipy fluorescent probe showed that the level of lipid ROS was significantly decreased (p < 0.05, Figure 4(j)). Finally, cells were treated with different concentrations of sorafenib (0, 2.5, 5, 7.5, and 10 μM) and the cell activity was detected by MTT. It was found that oe-GPX4 significantly increased cell activity compared with the sh-HCG18 + oe-NC group (p < 0.05, Figure 4(k)). In summary, the effect of silencing HCG18 on ferroptosis of sorafenib-resistant hepatoma cells is mediated by GPX4.
HCG18 as a ceRNA regulates GPX4 through miR-450b-5p in Huh7-SR cells
To further clarify the regulatory mechanism of HCG18 on GPX4, HCG18 was mainly located in the cytoplasm in Huh7-SR cells through nuclear/cytosol fractionation experiment, suggesting that HCG18 may regulate GPX4 through ceRNA mechanism (p < 0.05, Figure 5(a)). Then, the Starbase website was used to respectively predict the miRNAs binding to HCG18 (supplementary Table 1) and GPX4 (supplementary Table 2); 389 miRNAs related to HCC occurrence were obtained (supplementary Table 3), and the intersection of the above three results was obtained. Three miRNAs (hsa-miR-196b-5p, hsa-miR-450b-5p, hsa-miR-214-3p) were obtained (Figure 5(b)). Among them, miR-450b-5p and miR-214-3p were poorly expressed in HCC, and the regulatory mechanism between HCG18 and miR-450b-5p has not been reported in literature, so we chose miR-450b-5p to study. HCG18 as a ceRNA regulates GPX4 expression through miR-450b-5p in Huh7-SR cells. a: The localization of HCG18 in Huh7-SR cells was detected by nuclear/cytosol fractionation experiment; b: miRNAs predicted from Starbase website and ONCOMIR website; c: Starbase website predicted the possible binding sites of HCG18 and miR-450b-5p; d–e: Dual-luciferase assay and RIP assay confirmed the binding of HCG18 and miR-450b-5p; f: Starbase website predicted the possible binding sites of miR-450b-5p and GPX4;g–h: Dual-luciferase assay and RIP assay confirmed the binding of miR-450b-5p and GPX4; i: the expression of miR-450b-5p was detected by RT-qPCR; j–k: After silencing HCG18 and miR-450b-5p in Huh7-SR cells, miR-450b-5p and GPX4 expressions were examined by RT-qPCR and Western blot. The cell experiment was repeated three times, and the data were expressed as mean ± SD. The t test was used in Figure i; one-way ANOVA was used in Figure k, two-way ANOVA was used in Figure a-k and j, Tukey’s multiple comparisons test was used for post hoc test; *p < 0.05.
Starbase website predicted the possible binding sites of HCG18 and miR-450b-5p (Figure 5(c)) and the possible binding sites of miR-450b-5p and GPX4 3′UTR (Figure 5(f)). Dual-luciferase assay and RIP assay showed that HCG18 could bind to miR-450b-5p (all p < 0.05, Figures 5(d) and (e)) and confirmed that miR-450b-5p could bind to GPX4 (all p < 0.05, Figures 5(g) and (h)). RT-qPCR showed that miR-450b-5p in Huh7-SR cells was obviously increased after silencing HCG18 (p < 0.05, Figure 5(i)); compared with sh-HCG18 + inhibitor NC group, miR-450b-5p in sh-HCG18 + miR-450b-5p inhibitor group was decreased (p < 0.05, Figure 5(j)) and GPX4 was significantly elevated (all p < 0.05, Figure 5(j) and (k)). These results suggest that HCG18 as a ceRNA regulates GPX4 expression through miR-450b-5p in Huh7-SR cells.
Silencing HCG18 annuls SR in HCC by regulating GPX4-inhibited ferroptosis through miR-450b-5p
Finally, to verify the regulation of HCG18 to SR in vivo, Huh7-SR cells were infected with lentivirus LV-sh-NC or LV-sh-HCG18 to establish subcutaneous xenograft tumor models. Compared with the LV-sh-NC group, the volume and mass of mouse tumors in the LV-sh-HCG18 group were significantly decreased (Figures 6(a)–(c), all p < 0.05). Meanwhile, HCG18 and GPX4 expressions were decreased (Figures 6(d) and (e), p < 0.05), and miR-450b-5p was significantly increased (Figure 6(d), p < 0.05). Ferroptosis test results showed that compared with the LV-sh-NC group, the iron level and MDA concentration in the LV-sh-HCG18 group were increased (Figures 6(f) and (g), p < 0.05), while the GSH concentration was significantly decreased (Figure 6(h), p < 0.05). These results suggest that silencing HCG18 can reverse SR in HCC by regulating GPX4-inhibited ferroptosis through miR-450b-5p (Figure 6(i)). HCG18 can reverse sorafenib resistance in HCC by regulating GPX4-inhibited ferroptosis through miR-450b-5p. Huh7-SR cells were infected with lentivirus LV-sh-NC or LV-sh-HCG18 to establish subcutaneous xenograft tumor models. a: Typical tumor photos; b: Tumor volume; c: Tumor mass; d: HCG18, miR-450b-5p, and GPX4 expressions detected by RT-qPCR; e: GPX4 expression tested by Western blot; f: Iron level detected by iron assay kit; g–h: MDA and GSH concentrations in tumors detected by kits; i: Flow Chart of HCG18 promoting sorafenib resistance in HCC. N = 6 in each group. The data were described as mean ± SD. The t test was used in Figures b–c and e–h; two-way ANOVA was used in Figure d, followed by Tukey’s multiple comparisons test; *p < 0.05.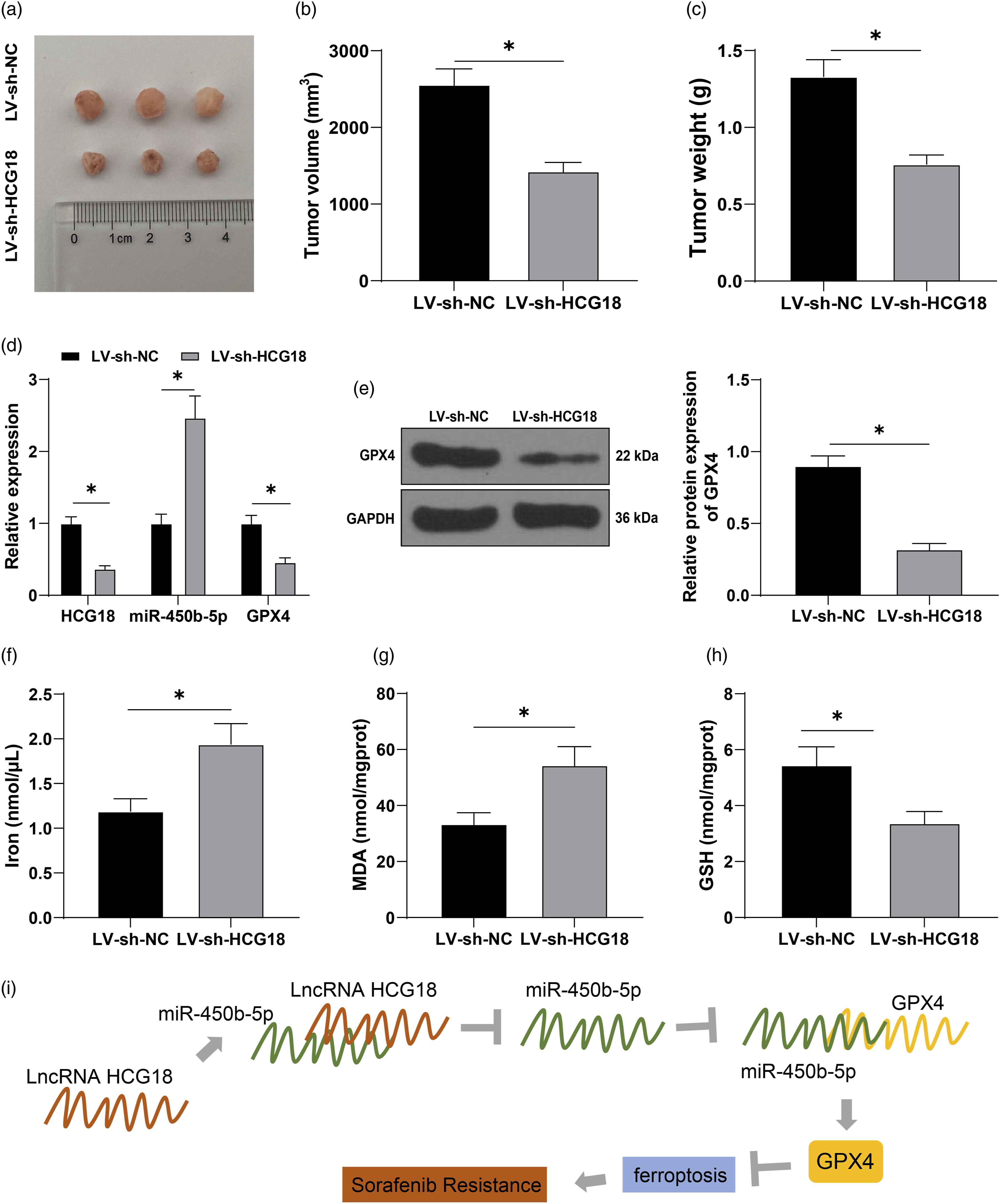
Discussion
Sorafenib is an approved systemic therapy for advanced HCC that extends the overall survival and improve the prognosis.1,48 However, the long-term survival of HCC patients is still suboptimal mainly due to the acquired drug resistance. Preventing ferroptosis has been evidenced to potentially facilitate SR in cancer cells.13,14 This study supported that silencing HCG18 could promote GPX4-inhibited ferroptosis and reverse SR in HCC via the ceRNA network of HCG18/miR-450b-5p/GPX4.
The high expression of HCG18 in HCC has been identified previously.18,19 Drug resistance has always been an essential issue affecting the outcome of HCC. 49 However, the mechanism of HCG18 on SR in HCC has not been reported. GEPIA website predicted that HCG18 was elevated in HCC and associated with HCC prognosis. RT-qPCR noted that HCG18 was overexpressed in hepatoma cell lines. Next, we constructed sorafenib-resistant cell line Huh7-SR and treated Huh7 and Huh7-SR cells with sorafenib (0, 2.5, 5, 7.5, 10 μM). MTT assay verified that sorafenib had a weaker inhibitory effect on Huh7-SR cells than on Huh7 cells. HCG18 in Huh7-SR cells was elevated relative to that in Huh7 cells. In short, HCG18 was highly expressed in HCC and associated with SR. Then, the in vitro experiences were performed. After Huh7-SR cells were delivered with sh-HCG18 and different concentrations of sorafenib, MTT assay, colony formation assay, and flow cytometry elicited that silencing HCG18 inhibited cell activity and proliferation and increased apoptosis. The subcutaneous xenograft tumor model induced by injecting Huh7-SR cells and lentivirus LV-sh-HCG18 and administering sorafenib also showed consistent results. Briefly, silencing HCG18 inhibited SR in HCC. Many lncRNAs have been identified to correlate with SR.50–52 We were the first to discover the correlation between HCG18 and SR in HCC.
Sorafenib has been documented to promote ferroptosis in HCC cells, 46 and SR can be strengthened by inhibiting ferroptosis. 13 Ferroptosis is triggered and augmented by lipid peroxide accumulation, 53 ROS production, 54 and lipid ROS formation.55,56 To confirm the role of ferroptosis in SR of HCC, ferroptosis levels in Huh7 and Huh7-SR cells were determined. Accordingly, iron levels and MDA concentration in Huh7-SR cells were clearly decreased, GSH concentration was enhanced, and lipid ROS was lowered significantly. GSH depletion leads to ferroptosis54,57 and ROS production. 56 In brief, ferroptosis was inhibited in sorafenib-resistant cells. Then, we detected ferroptosis in sh-HCG18-transfected Huh7-SR cells, and found that iron level and MDA concentration in HCG18 silenced Huh7-SR cells were increased, GSH concentration was decreased, and lipid ROS was significantly increased. These results suggest that silencing HCG18 promoted ferroptosis. Furthermore, we treated the sh-HCG18-transfected Huh7-SR cells with ferrostatin-1 and sorafenib. MTT assay showed that ferrostatin-1 significantly improved cell viability. Cell proliferation is suppressed with GPX4 knockdown, which can be averted by ferrostatin-1.58,59 Taken together, silencing HCG18 suppresses SR of HCC by promoting cell ferroptosis. The interplay between ferroptosis and lncRNAs in cancer cells has been repeatedly validated.60–62 But the regulation of HCG18 in ferroptosis in HCC cells has not been studied at home and abroad, which potently indicated the novelty of our study.
Ferroptosis is specifically activated by losing GPX4 activities. 55 GPX4 is potential to kill drug-resistant cancer cells by regulating ferroptosis. 63 GEPIA website predicted that GPX4 was elevated in HCC and correlated with HCC prognosis. Consistently, GPX4 was overexpressed in HCC tissues and regarded as a probable biomarker for HCC diagnosis/prognosis. 64 In Huh7-SR cells, GPX4 was significantly decreased after silencing HCG18. To verify the action of GPX4, we delivered sh-HCG18 and oe-GPX4 into Huh7-SR cells together. The iron level and MDA concentration were decreased, and GSH was increased, lipid ROS was decreased. GPX4 upregulation alleviated ferroptosis aggravation in HCC cells. 65 GPX4 depletion strengthened the antitumor action of sorafenib in HCC cells by promoting ferroptosis. 66 In summary, the effect of silencing HCG18 on ferroptosis of sorafenib-resistant HCC cells was achieved via GPX4.
To further clarify the mechanism of HCG18 on GPX4, we found that HCG18 was mainly located in the cytoplasm in Huh7-SR cells, suggesting that HCG18 may regulate GPX4 through ceRNA mechanism. Then, Starbase website and Ualcan website predicted the miRNAs binding to HCG18 and GPX4 and miRNAs related to HCC occurrence, and the intersections (hsa-miR-196b-5p, hsa-miR-450b-5p, and hsa-miR-214-3p) were obtained. Among them, miR-450b-5p and miR-214-3p were poorly expressed in HCC, and the mechanism between HCG18 and miR-450b-5p has not been reported, so we chose miR-450b-5p to study. miR-450b-3p is downregulated in HCC. 67 Dual-luciferase assay and RIP assay verified the binding relations of HCG18 and miR-450b-5p and of miR-450b-5p and GPX4. miR-450b-5p in Huh7-SR cells was increased after silencing HCG18; miR-450b-5p was decreased and GPX4 was significantly elevated in the sh-HCG18 + miR-450b-5p inhibitor group. To sum up, HCG18 as a ceRNA regulates GPX4 through miR-450b-5p in Huh7-SR cells.
The innovation of this study lies in the discovery of the molecular mechanism that lncRNA HCG18 regulates GPX4-inhibited ferroptosis by adsorbing miR-450b-5p to regulate SR in HCC. However, this study has not yet explored the regulatory mechanism of abnormal expression of HCG18 in sorafenib-resistant hepatoma cells, which will be the focus of our future study.
Supplemental Material
Supplemental Material - Silencing lncRNA HCG18 regulates GPX4-inhibited ferroptosis by adsorbing miR-450b-5p to avert sorafenib resistance in hepatocellular carcinoma
Supplemental Material for Silencing lncRNA HCG18 regulates GPX4-inhibited ferroptosis by adsorbing miR-450b-5p to avert sorafenib resistance in hepatocellular carcinoma by Xiaoming Li, Yunhui Li, Peilong Lian, Qigang lv, and Fangfeng Liu in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Major R&D plan of Shandong Province (Grant 2017GSF18170) and Jinan science and technology development plan (Grant 201907071).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
