Abstract
To investigate the protective effects of curcumin (Cur) on gastric mucosal injury induced by cisplatin (DDP), and explore possible molecular mechanisms. A mouse of gastric mucosal injury was established by intraperitoneal injection of DDP (27 mg/kg). Thirty mice were randomly divided into control group, DDP group and DDP + Cur group. Serum and gastric mucosal samples were collected on the 7th day after Cur treatment. The index of gastric mucosa injury was calculated, and the expression levels of inflammation, apoptosis and signaling pathway proteins were evaluated using hematoxylin and eosin staining, ELISA and western blotting analysis. These data showed that Cur treatment significantly attenuated DDP-induced decrease in body weight, food intake, fat and muscle ratios, and improved the gross gastric injury, scores of ulcer index, and histopathology changes triggered by DDP (p < .05). Meanwhile, Cur significantly decreased serum IL-23 and IL-17 proteins, reduced the expression levels of gastric mucosal IL-1β, TNF- α and MPO, and restored the level of IL-10 protein (p < .05). Moreover, Cur treatment significantly inhibited the expression levels of Caspase-3, PARP and Bax, and increased the expression of Bcl-2 protein. Furthermore, Cur treatment significantly decreased the expression levels of IL-1R, MyD88 and TAK1, and also repressed the activation of NF-κB and nuclear translocation of NF-κB p65. And more importantly, Cur treatment significantly inhibited DDP-induced gastric mucosal JNK1/2, ASK1, P38 and JUN phosphorylation, and promoted the phosphorylation of ERK1/2 and C-Myc proteins. Our data suggest that Cur treatment alleviates DDP-induced gastric mucosal inflammation and apoptosis, which may be mediated through the NF- κ B and MAPKs signaling pathway.
Introduction
Cisplatin (DDP) is a widely used chemotherapeutic agent in the treatment of lung, head and neck, ovary, bladder and testicular cancer.1–3 DDP not only killed tumor cells, but also damaged normal cells, showing serious side effects. Digestive tract reaction is one of the most common side effect, lead to physical weakness of patients, seriously hinder patient’s treatment will, then affect the efficacy of chemotherapy. 4 The commonly used drugs in the treatment of digestive tract reactions are serotonin antagonist, antihistamines, phenothiazines. 5 However, the mechanism of gastric mucosal injury caused by DDP has not been fully elucidated. Therefore, how to treat gastric mucosal injury triggered by DDP reasonably and effectively is one of the difficult problems to be solved urgently, which has important clinical significance.
It has been found that the mechanisms of gastric mucosal injury triggered by many factors including mucosal oxidative stress,6,7 inflammation,8,9 and epithelial cell apoptosis. 10 When gastric mucosal injury occurs, neutrophils dissociated from circulating blood will pour into gastric mucosal tissue, which triggers the release of a variety of pro-inflammatory cytokines, and further aggravates the inflammatory process of gastric mucosa. 11 Nuclear factor kappa B (NF- κ B) pathway has always been considered to play a central role in triggering gastric mucosal inflammation.12,13 Driven by pro-inflammatory cytokines, the activated NF- κ B can further amplify the inflammatory response by increasing the transcription of tumor necrosis factor-α (TNF-a) and other pro-inflammatory signals. Moreover, the activation of NF-κB was closely related to neutrophil recruitment and the level of gastric mucosal inflammatory cytokines. 14 Some stress signals, such as reactive oxygen species (ROS) and inflammatory cytokines, were reported to cause the degradation of I κ B subunit, and then released NF-κB as transcription factors of pro-inflammatory adhesion molecules and cytokines. 15 Mitogen-activated protein kinase (MAPKs) pathway, including p38MAPK, extracellular signal-regulated kinase (ERK) and c-jun N-terminal kinase (JNK), is one of the important pathways for the expression of pro-inflammatory mediators and apoptosis signals. 16 A large number of studies reported that MAPKs signaling pathway was widely involved in the process of gastric mucosal inflammation and apoptosis caused by many factors.17,18 However, the expression of NF-κB and MAPKs signaling pathways in DDP-induced gastric mucosal injury remain unclear.
Curcumin (Cur) is a traditional Chinese medicine, which is a monomeric compound extracted from Curcuma Longa. 19 It has been found that Cur has the effects of anti-inflammation, anti-oxidation, anti-rheumatism and anti-tumor, and has a protective effect on gastrointestinal mucosal injury.20–22 Meanwhile, it has been reported that Cur increased the production of anti-inflammatory cytokine IL-10 and stimulated the expression of SOCS-1 through JAK/STAT/SOCS signaling pathway, thus improving the development of cerebral ischemia. 23 Moreover, Cur was also reported to ameliorate stress-induced intestinal barrier injury, improve the function of intestinal barrier and mitochondrial through the induction of Parkin-dependent mitophagy via AMPK activation, and subsequent TFEB nuclear translocation. 24 However, it is unclear whether Cur protected gastric mucosal against DDP-induced gastric mucosal injury. Therefore, the purpose of this study was to investigate the protective effect of Cur on DDP-induced gastric mucosal injury, and to clarify its possible molecular mechanism.
Materials and methods
Animals and experimental groups
A total of 30 male C57BL/6 mice (aged 6–8 weeks) were used in this study, which provided by Liaoning Experimental Animal Resource Center (Liaoning Changsheng Biotechnology Co., Ltd). Cur (No. TC04341 G, CAS: 458–37-7) and DDP (No. SP439401, CAS: 15,663–27-1) were provided by Sinopharm Group Co., Ltd, China. One week before the experiment, all mice were kept in an environment of 20–24°C with a humidity of 55% + 5% and a light/dark cycle of 12/12 h. During the period, ad libitum access to water and food were provided to allow all mice to adopt the environment. These mice were then randomly and equally divided into the control group, DDP group, and DDP + Cur group, 10 animals in each group. All animal protocols were approved by the Institutional Animal Care and Use Committee of General Hospital of Northern Theater Command, and all care and handling of animals was in accordance with the criteria outlined by National Institutes of Health guidelines.
Drug administration and samples collection
On the first day 1 after the experiment, the mice in the DDP and DDP + Cur group were injected intraperitoneally with DDP aqueous solution (27 mg/kg). 2 h after DDP administration, the mice in the DDP + Cur group were intraperitoneally injected with Cur solution (25 mg/kg) for 6 consecutive days. The mice in the control group were injected with the same amount of normal saline as control. Body weight and food intake of the mice were weighed daily. On the 7th day of the experiment, blood and gastric mucosal samples were collected. All mice were anesthetized by the abdominal injection of 2% pentobarbital sodium (1.5 mL/kg; Cat No: 57-33-0; Shanghai Rongbai Biological Technology Company, China) to reduce the fear of mice in the process of model preparation. Moreover, to alleviate post-operative pain, analgesia in the form of buprenorphine (0.2 mg/kg; Cat No: #117299; year 2015; Alstoe Animal Health, York, UK) was administered intramuscularly prior to recovery and killing of animals.
Physique analysis
The mice in each group were weighed, and then put into the physique analyzer. After entering the data of body weight, the system will automatically calculates the fat and muscle weight of the mice. Body/fat rate (BF%) = (fat weight/body weight) × 100%, muscle percentage (Muscle ratio, M%) = (muscle weight/body weight) × 100%.
Ulcer index and the area of gastric ulcers
The digital photos were analyzed to calculate the UI using a damage scoring as described. 25 Briefly, each lesion was scored from 0 to 5 as follows (a) undetectable lesions (score of zero); (b) tiny circular hemorrhagic lesions (score of 1); (c) lesions < 2 mm long (score of 2); (d) lesions with 2–3 mm length (score of 3); (e) lesions of 3–4 mm length (score of 4); (f) lesions > 4 mm (score of 5). When the width of lesion exceeded 1 mm, the score was doubled. Summation of all of the lesion scores was calculated as the UI. The digital photos of gastric injury were quantified using Image J software and the area of gastric lesions was calculated in mm2.
Histological analysis
The gastric mucosal samples for histological analysis was fixed in 10% neutral-buffered formalin, and then embedded in paraffin by Leica Microsystem tissue processor (ASP 300S, Germany). The paraffin-embedded tissues were subsequently cut into 3–4 μm thick slices using the Leica Microsystem microtome (Model RM 2265, Germany), and stained with hematoxylin and eosin (H&E).
Enzyme-linked immunosorbent assay detection
Enzyme-linked immunosorbent assay was used to determine serum concentrations of the IL-23 (ab119545, Abcam, UK) and IL-17 by ELISA kits (ab100702, Abcam, UK) according to the manufacturers’ instructions. The optimal density at 450 nm was used to measure the concentration of IL-23 and IL-17 in a microplate reader. The curve equation and r value were calculated to determine the concentrations of samples.
Western blot analysis
Western blotting for this study was performed as described previously. 26 Briefly, the gastric mucosal samples were immersed in RIPA lysis buffer containing 1× protease inhibitor cocktail and lysed homogeneously by Sonic Dismembrator 100. Bio-Rad Protein Assay was used to determine the protein concentration. An equal amount of soluble proteins was separated on 10% sodium dodecyl sulfate poly-acrylamide gel electrophoresis (SDS-PAGE), and then transferred onto a Poly vinylidene fluoride (PVDF) membrane. After blocking with 5% nonfat milk in TBS, the membrane was probed with primary antibody as follows. Antibody against TNF-α (ab215188), IL-1β (ab254360), TNF-α(ab215188), myeloperoxidase (MPO) (EPR20257) and IL-10 (ab189392), Caspase-3 (ab184787), PARP (ab32064), and Bax (ab32503) and Bcl-2 (ab182858) were purchased from Abcam (UK) at 1:2000 dilution. Antibody against NF-κB, NF-κB p65, p-NF-κB p65, JNK (sc-7345), p-JNK (sc-293,136), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (sc-365,062) were purchased from Santa Cruz Biotechnology (USA) and used at a 1:1000 dilution. The blotted membranes were then washed twice with TBS containing 0.1% of Tween 20 to remove unbounded primary antibody and then blotted with corresponding horseradish peroxidase-conjugated secondary antibody. Goat anti-mouse, goat anti-rabbit, and goat anti-rat secondary antibody (Abcam, UK) were used at 1:4000 dilution. The chemiluminescent signal was visualized using a Clarity Western ECL Substrate, and the signal was imaged by Tanon 5200 imaging system.
Statistical analysis
Data were expressed as the means ± standard error (SE) and analyzed using T-Student Test and one-way ANOVA with Tukey post hoc test. All statistical analyses were carried out using IBM SPSS Statistics (Version 20.0, Armonk, New York, USA). A two-sided p < .05 was considered statistically significant.
Results
Curcumin treatment ameliorated DDP-induced body weight, food intake, fat and muscle ratios
The body weight and food intake which a marker of the gastric mucosal injury in the DDP group were significantly decreased compared with the control group, whereas Cur treatment significantly increased the body weight and food intake caused by DDP (Figures 1(a) and (b), p < .05). Moreover, physique analysis showed that DDP treatment decreased fat and muscle ratios compared with the control group, and Cur treatment significantly ameliorated fat and muscle ratios induced by DDP (Figures 1(c) and (d), p < .05), indicating that Cur can effectively ameliorate DDP-induced gastric mucosal injury in mice. Curcumin treatment ameliorated DDP-induced body weight, food intake, fat and muscle ratios. Cur treatment significantly increased DDP-induced (a) body weight and (b) average food intake as compared to the mice in the DDP group. Moreover, Cur treatment also promoted (c) body/fat rate (BF%) and (d) muscle percentage (Muscle ratio, M%) Data were represented as the mean ± SEM *p < .05, versus control group. #p < .05, versus DDP group.
Curcumin treatment attenuated gastric mucosa injury triggered by DDP
The results showed that the mucosal surface of gastric tissue in control group was light red, smooth, and no macroscopic damage was detected. Obvious edema, massive bleeding or patchy bleeding were observed on the mucosal surface of gastric tissue in the DDP group, and only slight hyperemia, edema and a small amount of bleeding were observed on the mucosal surface of gastric tissue in Cur group (Figure 2(a)). Moreover, administration of DDP caused severe hemorrhagic gastritis marked with increased UI damage scores compared with the control group, whereas Cur treatment significant improved the UI damage scores triggered by DDP (Figure 2(b), p < .05). Curcumin treatment attenuated gastric mucosa injury triggered by DDP. Gastric mucosa tissue was dissected along the long side of stomach and washed in saline solution for observation. (a) Representative gross stomach images in each group. (b) The digital photos were analyzed to calculate the UI using a damage scoring. Data were represented as the mean ± SEM *p < .05, versus Control group. #p < .05, versus DDP group.
Curcumin treatment mitigated DDP-induced gastric histopathological changes
In the present study, 10 sections in each group were used for evaluation of histo-pathological changes. Histological examination showed that in the control group, the structure of each layer of gastric wall was normal, the cells were arranged neatly, the boundary between each layer was clear, and no inflammatory cell infiltration was found. DDP treatment caused severe gastric structural damage marked with severe mucosal erosion and ulcer, accompanied by epithelial loss and bleeding. However, Cur treatment significantly improved the pathological alterations and microscopic score of gastric mucosa, and reduced gastric ulcer, edema and inflammatory cell infiltration (Figure 3, P < .05). Curcumin treatment mitigated DDP-induced gastric histopathological changes. (a) Representative images showing histopathological gastric injury (HE staining, bar: 50 μm). (b) Ulcer index scores. 10 sections in each group were used for evaluation of histopathological changes. Data were represented as the mean ± SEM *p < .05, versus Control group. #p < .05, versus DDP group.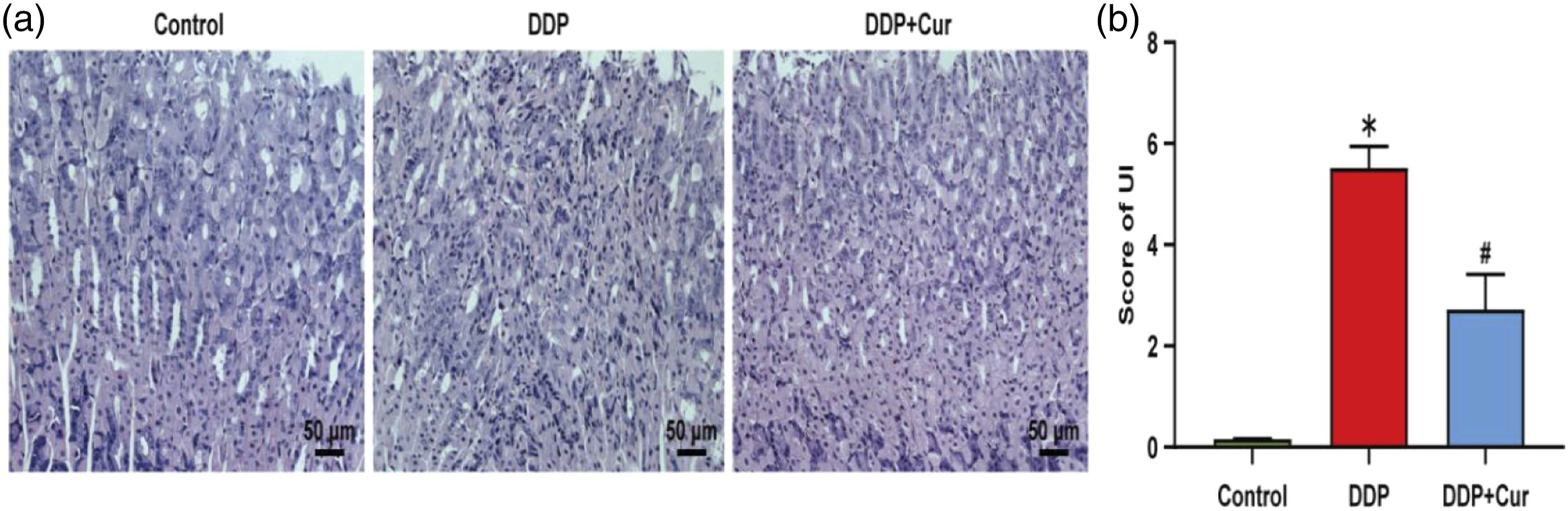
Curcumin treatment inhibited DDP-induced serum and gastric mucosal inflammation
The inflammatory events were reflected by the expression of IL-23 and IL-17 in serum, and the expression of TNF-α, IL-1β, MPO and IL-10 protein in gastric mucosa. Compared with the control group, DDP significantly increased the expression levels of IL-23, IL-17, IL-1β, TNF- α and MPO, and decreased the expression level of IL-10. However, Cur significantly decreased serum IL-23 and IL-17 proteins, reduced the expression levels of gastric mucosal TNF- α, IL-1β and MPO, and restored the expression level of IL-10 (Figure 4, P < .05). These data suggest that Cur is involved in the anti-inflammatory effect of relieving DDP-induced gastric mucosal injury. Curcumin treatment inhibited DDP-induced serum and gastric mucosal inflammation. (a) The expression level of serum IL-23. (b) The expression level of serum IL-17. (c) Representative western blot images of the expression of TNF-α, IL-1β, MPO, IL-10, and GAPDH. (d–g) Quantitative analysis of the density of TNF-α (d), IL-1β (e), MPO (f), IL-10 (g), and with normalized to internal control GAPDH. All experiments were repeated at least three times. Data were represented as the mean ± SEM *p < .05, versus Control group. #p < 0.05, versus DDP group.
Curcumin treatment improved DDP-induced gastric mucosal apoptosis
Compared with the control group, DDP significantly increased the expression levels of Caspase-3, PARP and Bax, and decreased the expression level of anti-apoptotic protein Bcl-2. It is worth noting that Cur treatment significantly inhibited the expression levels of Caspase-3, PARP and Bax, and increased the expression level of Bcl-2 protein (Figure 5, P < .05). These data suggest that Cur treatment can significantly improve gastric mucosal apoptosis induced by DDP. Curcumin treatment improved DDP-induced gastric mucosal apoptosis. (a) Representative Western blot images of the expression of Caspase-3, PARP, Bax, Bcl-2, and GAPDH. (b–e) Quantitative analysis of the density of Caspase-3(B), PARP(C), Bax(D), Bcl-2(E), and with normalized to internal control GAPDH. All experiments were repeated at least three times. Data were represented as the mean ± SEM *p < .05, versus Control group. #p < .05, versus DDP group.
Curcumin treatment targeted DDP-induced gastric mucosal injury through NF- κ B signaling pathway
To investigate whether Cur treatment attenuated DDP-induced gastric mucosal inflammation through the NF- κ B signaling pathway, the expression levels of IL-1R, MyD88 TAK1, NF- κ B and nuclear translocation of NF-κ B p65 proteins were assessed by western blotting analysis. Compared with the control group, DDP significantly increased the expression levels of gastric mucosa IL-1R, MyD88 and TAK1 proteins, and also activated the expression of NF- κ B and nuclear translocation of NF- κ B p65. However, Cur treatment significantly decreased the expression levels of IL-1R, MyD88 and TAK1 proteins, and also inhibited the activation of NF- κ B and nuclear translocation of NF-κB p65 (Figure 6, p < .05). These results suggest that Cur may alleviate DDP-induced gastric mucosal inflammation through NF- κ B signaling pathway. Curcumin treatment targeted DDP-induced gastric mucosal injury through NF-κB signaling pathway. (a) Representative Western blot images of the expression of IL-1R, MyD88, TAK1, NF-κB, NF-κB p65, p-NF-κB p65, and GAPDH. (b–f) Quantitative analysis of the expression levels of IL-1R (B), MyD88 (C), TAK1 (D), NF-κB (E), and p-NF-κB p65 (F). All experiments were repeated at least three times. Data were represented as the mean ± SEM *p < .05, versus Control group. #p < .05, versus DDP group.
Curcumin targeted DDP-induced gastric mucosal apoptosis through MAPKs signaling pathway
To further clarify whether Cur ameliorated DDP-induced gastric mucosal apoptosis through MAPKs signaling pathway, the expression levels of p-ERK1/2, JNK1/2 and p-p38MAPK protein were detected by western blotting analysis. The results showed that DDP significantly increased the phosphorylation of JNK1/2, P38, ASK1 and JUN proteins, and decreased the phosphorylation of ERK1/2 and C-Myc proteins. However, Cur treatment significantly decreased DDP-induced gastric mucosal JNK1/2, ASK1, P38 and JUN phosphorylation, and increased the phosphorylation of ERK1/2 and C-Myc proteins (Figure 7, p < .05). These results suggest that MAPKs signaling pathway are involved in the regulation of DDP-induced gastric mucosal apoptosis. Curcumin targeted DDP-induced gastric mucosal apoptosis through MAPKs signaling pathway. (a) Representative Western blot images of the expression of JNK1/2, p-JNK1/2, p38MAPK, p-p38MAPK, ASK1, p-ASK1, JUN, p-JUN, ERK1/2, p-ERK1/2, C-Myc, p-C-Myc and GAPDH. (b–f) Quantitative analysis of the expression levels of p-JNK1/2, p-p38MAPK, p-ASK1, p-JUN, p-ERK1/2, and p-C-Myc. All experiments were repeated at least three times. Data were represented as the mean ± SEM *p < .05, versus Control group. #p < .05, versus DDP group.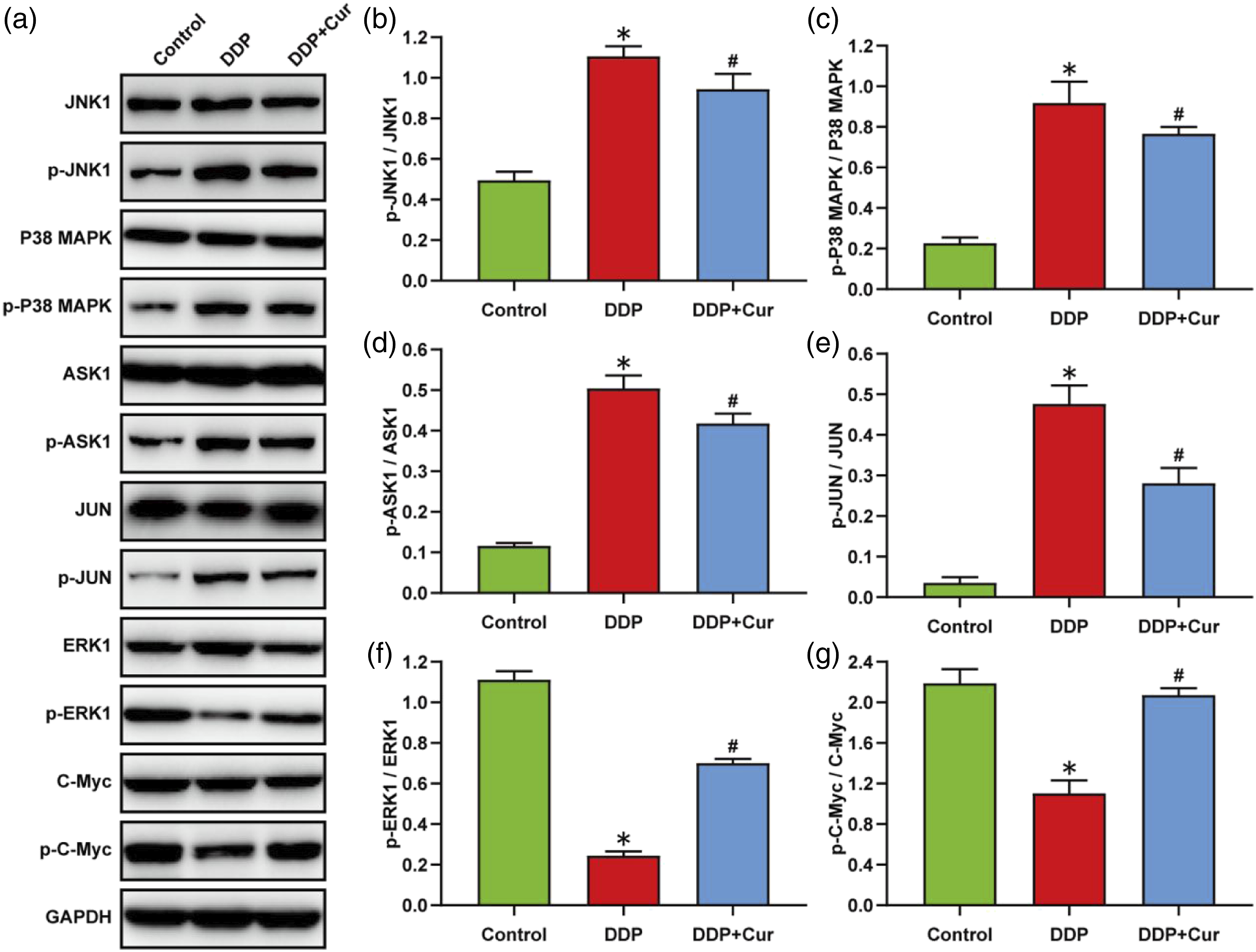
Discussion
Cur has been reported to have obvious anti-inflammatory, anti-oxidative stress and apoptosis effects, and has an important protective effect on gastric mucosal injury caused by many factors.27–29 However, the effects of Cur on DDP-induced gastric mucosal injury remain unclear. In the present study, our data showed that Cur ameliorated DDP-induced body weight, food intake, fat and muscle ratios, attenuated gastric mucosa injury and gastric histopathological changes. Meanwhile, Cur treatment significantly inhibited DDP-induced serum and gastric mucosal inflammation, and improved gastric mucosal apoptosis. Moreover, Cur treatment also significantly decreased the expression levels of IL-1R, MyD88 and TAK1, and decreased the expression of NF-κ B and nuclear translocation of NF-κB p65. And more importantly, Cur treatment significantly decreased DDP-induced gastric mucosal JNK1/2, ASK1, P38 and JUN phosphorylation, and increased the phosphorylation of ERK1/2 and C-Myc proteins. Our data suggest that Cur treatment alleviates DDP-induced gastric mucosal inflammation and apoptosis, which may be mediated through the NF-κ B and MAPKs signaling pathway.
Cur is a polyphenol extracted from plant Curcuma longa, and used to treat diseases related to oxidative stress and inflammation. It is reported that oral pretreatment of Cur dose-dependently reduced the formation of gastric antral ulcerations and histopathological changes induced by naproxen. 30 Moreover, Cur analogue FM0807 was found to significantly inhibit the expression levels of TNF- α, IL-6 and Il-1 β, and increase the level of IL-10 in synovial fluid of rheumatoid arthritis rats. 31 In the present study, our data revealed that Cur ameliorated DDP-induced gastric mucosa injury and gastric histopathological changes. Meanwhile, Cur significantly decreased serum IL-23 and IL-17 proteins, reduced the expression of gastric mucosal IL-1β, TNF- α and MPO proteins, and restored the level of IL-10 protein. IL-23 and IL-17 are a class of proinflammatory cytokine that are reported to be involved in chronic inflammatory disease in many tissues throughout the body, including the lungs, brain, gastrointestinal tract and skin.32,33 The presence of IL-23 plays an important role in the proliferation and survival of TH17 lymphocyte and promotes the ability of producing IL-17. 34 Similarly, Cur inhibited the expression of IL-17 by inducing the production of indoleamine2,3-dioxygenase in human gastric mucosa infected with Helicobacter pylori, suggesting that Cur alleviates gastric mucosal injury induced by H. pylori infection through anti-inflammatory effect. 35 Interestingly, oral administration of Cur-loaded Soluplus® micelles (Cur-Ms) significantly reduced oxidative stress in mice with alcohol induced-tissue injury, and protected the liver and gastric mucosa from alcohol damage. 36 These data suggest that Cur treatment can alleviate DDP-induced gastric mucosal inflammation.
To further study the signaling pathway of Cur in regulating DDP-induced gastric mucosal inflammation, our data demonstrated that Cur significantly decreased the expression levels of IL-1R, MyD88 and TAK1, and decreased the expression of NF-κ B and nuclear translocation of NF-κB p65. Similar to the results of our study, Cur was reported to inhibit the inflammatory response, and regulate the levels of vasoactive intestinal peptide and gastrin by targeting I κ B-α/NF-κ B in reserpine treated rats. 37 Cur@SFNPs not only removes iron from tissues and reduces oxidative stress, but also enhances the biological formation of iron-sulfur clusters, thus compensating for FXN deficiency and improving the morphology and function of mitochondria. 38 Meanwhile, Cur has also been shown to improve neuroinflammation and neuronal cell death in vitro and in vivo through regulating the NF-κB/Akt signaling pathway. 39 Moreover, Cur has been revealed to relieve oxaliplatin-induced peripheral neuropathic pain in rats via inhibiting the activation of NF- κ B, and reduced the release of inflammatory factors such as TNF-α, Il-1 β and IL-6. 40 Cur was also found to reduce the expression of M1 markers (Il-1 β and IL-6), increase the expression of M2 markers (IL-4 and IL-10), inhibit the activation of downstream NF- κ B, and attenuate LPS-induced the inflammatory response. 41 Furthermore, compared with the control group, tannic acid/curcumin-nanoparticles oral administration obviously improved DSS-induced colitis symptoms in mice via suppressing the TLR4/NF-κB signaling pathway. 42 These data suggest that Cur alleviates DDP-induced gastric mucosal inflammation possible through inhibition of NF- κ B signaling pathway.
In the present study, our data also showed that Cur significantly inhibited the expression of PARP, Caspase-3 and Bax protein, and increased the expression of Bcl-2 protein, and then improved DDP-induced gastric mucosal apoptosis. It is reported that Cur pretreatment significantly decreased the expression of Wnt5a, IL6, TNF-a, JNK1 phosphorylation, and also significantly reduced the nuclear translocation level of NF-κB induced by ischemia-reperfusion. 43 Meanwhile, Cur was revealed to significantly decrease the expression levels of Bax and caspase-3, and increase the expression of Bcl-2 in diabetic nephropathy mice. 44 Moreover, our data also demonstrated that Cur treatment significantly decreased DDP-induced gastric mucosal JNK1/2, ASK1, P38 and JUN phosphorylation, and increased the phosphorylation of ERK1/2 and C-Myc proteins. Similar to our results, oral administration of Cur induced activation of ERK1/2 and JNK in gastric mucosa of H. pylori infected mice, and promoted c-JUN phosphorylation and nuclear translocation. 45 Meanwhile, Cur has been found to inhibit influenza A virus-induced activation of p38/JNKMAPK and NF-κB pathways. 46 Furthermore, Cur derivative CNB-001 has been revealed to significantly inhibit the phosphorylation of ERK, JNK and p38 MAPK induced by thrombin therapy, which suggest that CNB-001 improves the activation of thrombin-stimulated microglia by inhibiting the ERK and p38MAPK signaling pathways. 47 Cur can prevent cardiac tissue injury, reduce serum MDA, NF- κ B p65, TNF- α and IL-6, and enhance cellular antioxidants. Cur down-regulated TLR4 and AP-1, and decreased the phosphorylation level of p38-MAPK, JNK and ERK1/2. 48 Therefore, these data suggest that Cur treatment alleviates DDP-induced gastric mucosal apoptosis, which may be mediated through the MAPKs signaling pathway. The limitation of this study is that no gene knockout mice or corresponding neutralizing antibodies were used to further verify that Cur reduced DDP-induced gastric mucosal injury. Meanwhile, several different Cur dose groups were not set to study the dose-effect relationship between Cur and gastric mucosal injury.
In summary, our data suggest that DDP triggers significant gastric mucosal damage, whereas Cur treatment alleviates DDP-induced gastric mucosal inflammation and apoptosis, which may be mediated through the NF- κ B and MAPKs signaling pathway.
Footnotes
Author contributions
Jinping Gao and Ying Piao designed experiments and wrote the manuscript. Yunen Liu revised and polished the manuscript. Juan Chen completed animal experiment, pathological experiment. Changci Tong and Qian Wang completed molecular biology experiment and statistical analysis of data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Key Projects of Liaoning Provincial Department of Education (No.LJKZ1153).
Ethical approval
The animal use protocol was approved by the Institutional Animal Care and Use Committee of General Hospital of Northern Theater Command (Shenyang, China), and the permit number is 2020W0024.
