Abstract
Keywords
Introduction
The most common type of glomerulonephritisis mesangial proliferative glomerulonephritis (MPGN) in China. 1 MPGN is featured with diffuse mesangial cell overproliferation as well as extracellular mesangial matrix accumulation in mesangial area. 2 Mesangial cell inflammation and overproliferation play a major role in the progression of MPGN. When mesangial cells proliferate abnormally, inflammatory mediators are released, leading to fibrosis, glomerulosclerosis, and, eventually, end-stage kidney disease. 3 An immune-mediated inflammatory disease that causes the body to produce antibodies results in the complex accumulation of immune cells in the glomerular mesangial area by antigens. 4 The etiology of the disease is unclear. MPGN patients are treated clinically with drugs including cyclophosphamide, hormones, mycophenolate, leflunomide, azathioprine, and others. Nonetheless, these drugs frequently have toxicity and side effects that severely limit their clinical application. Finding effective drugs and understanding the pathogenesis of MPGN are thus critical.
The Phosphatidylinositol 3-kinase (PI3K)/Protein kinase B (AKT) signaling pathway contributes to the development and progression of several inflammatory diseases, such as lupus nephritis, 5 rheumatoid arthritis, 6 and osteoarthritis. 7 When AKT is activated, it can directly phosphorylate IKKs, leading to the phosphorylation and the degradation of IκBα and the activation of the NF-κB signaling pathway. The lipopolysaccharides (LPS)-induced inflammatory model has been extensively used in the research of glomerulonephritis at the cellular level. LPS induces an inflammatory response by binding to various membrane receptors, initiating cell signaling, and activating inflammatory pathways, such as the nuclear factor-kappa B (NF-κB) pathway. 8 The responses associated with PI3K/AKT/NF-κB signaling are consistent with MPGN characteristics.
Albiflorin (AF) is the main glycoside component in Paeonia Alba Radix, which is the root of Paeonia lactiflora Pallas or P. veitchii Lynch and is a traditional Chinese prescription with medicinal properties. 9 AF has been linked to cell proliferation in intrinsic dysmenorrhea and coronary artery injury 10 as well as an anti-inflammatory effect in asthma, mood disorders, and osteoblasts.11–13 Studies have proved that AF can modulate the PI3K/AKT signaling,14,15 and since the PI3K/AKT signaling is important in MPGN, we hypothesized that AF could influence MPGN by regulating PI3K/AKT signaling pathway.
Our research was conducted based on the assumption listed above. We investigated the effects of AF on mesangial cell overproliferation, inflammation, and fibrosis in vitro and in vivo for the first time and identified the related signaling pathways. Moreover, we discovered that AF could alleviate kidney injury in MPGN model rats. The renal protective effects of AF may be mediated by inhibiting PI3K/AKT/NF-κB signaling pathway.
Materials and methods
In vitro cell culture and treatment
To obtain the human mesangial cells (HMCs), we purchased them from Central South University’s Advanced Research Center (Wuhan, China). To maintain cell growth, RPMI 1640 medium (Gibco, Grand Island, New York, USA) added with 10% fetal bovine serum (Gibco) was used, and the cells were placed at 37°C in an incubator containing 5% CO2.
HMCs were stimulated for 24 h with 30 μg/mL LPS (Beyotime, Shanghai, China) with various concentrations of AF (Wuhan ChemFaces Biochemical Co., Ltd, Wuhan, China). To investigate the disease mechanism, HMCs induced by LPS were treated for 24 h with 10 nM insulin-like growth factor-1 (IGF-1, PeproTech, Beijing, China) with or without 20 nM AF.
Cell viability measurement
Cell viability was determined using the Cell counting kit-8 (CCK-8, Beyotime) assay, according to the protocol. HMCs were treated with LPS for 24 h, either with or without AF, and then incubated for 2 h at 37°C with CCK-8 solution. The results were visualized using an automated reader (Bio-Rad Model 550, Hercules, California, USA) at 450 nm test wavelength and 650 nm reference wavelength.
5-Ethynyl-2′-deoxyuridine assay
The HMCs were seeded into 96-well plates and fixed with 4% paraformaldehyde for 15 min, followed by incubating with 2 mg/mL glycine for 5 min. Cells were permeabilized with 0.5% Triton-X 100 at indoor temperature for 20 min, after being washed with 3% BSA/phosphate buffer solution (PBS) (v/v). For staining, cells were administrated with 5 g/mL DAPI (Beyotime) for five minutes and then incubated in a 10 mM EdU solution (KeyGEN, Nanjing, China) for 30 min. The images was captured using a fluorescence microscope (DMI3000B, Leica, Wetzla, Hessen, Germany). The percentage of EdU positive cells (%) was calculated using Image-Pro Plus 6.0.
Quantitative real-time polymerase chain reaction
The total RNAs extracted from HMCs were precipitated in 75% ethanol using TRIzol (Invitrogen, Carlsbad, California, USA). For reverse transcription, a SYBR primeScriptTM RT-PCR was used (Takara, Dalian, China). The ExScriptTM RT-PCR kit (Takara) was then used to perform qRT-PCR in accordance with the manufacturer’s instructions. This was carried out at 95°C for 5 min, followed by 40 cycles under the condition of 95°C for 10 s and 60°C for 30 s. The primer sequences were listed below: IL-1α: (F) 5′-CGAAGACTACAGTTCTGCCATT-3′, (R) 5′-GACGTTTCAGAGGTTCTCAGAG-3’; IL-2: (F) 5′-TGAGCAGGATGGAGAATTACAGG-3′, (R) 5′-GTCCAAGTTCATCTTCTAGGCAC-3’; IL-10: (F) 5′-GCTCTTACTGACTGGCATGAG-3′, (R) 5′-CGCAGCTCTAGGAGCATGTG-3’; IFN-γ: (F) 5′-CTCGGAAACGATGAAATATAC-3′, (R) 5′-TTACTGGGATGCTCTTCG-3’; GAPDH (internal control): (F) 5′-CCTCGTCTCATAGACAAGATGGT-3′, (R) 5′-GGGTAGAGTCATACTGGAACATG-3’. The 2−∆∆Ct method was used to calculate the relative gene expression levels.
Animal model establishment
Twenty four male Sprague Dawley (SD) rats (6–8 weeks; 180–220 g) were provided by Shanghai Alac Laboratory Animal Co., Ltd. (Shanghai, China). During the experiment, rats were kept in pathogen-free conditions with 65% humidity at 25°C and 12 h of lighting and darkness. The 24 rats were randomly divided into 4 groups: Sham group (n = 6), model group (n = 6), model + 20 mg/kg AF group (n = 6), and model +5 mg/kg Predisone group (n = 6). The MPGN animal model was established using an improved Hogendoorn’s method, as described in previous research. 16 Except for the Sham rats, the other rats were given 35 mg/kg sodium pentobarbital intraperitoneally to be anesthetized before undergoing left nephrectomy. After one week, the rats were subcutaneously injected with 3 mg of bovine serum albumin (BSA, Beyotime) emulsified in 0.1 mL of Freund’s complete adjuvant (Beyotime), twice in 2 weeks. At the end of the 3rd week, the rats were given intraperitoneal injections of 0.5, 1.0, 1.5, and 3 mg BSA every 1 h. Each rat received 0.5–3.0 mg BSA into the tail vein every other day, with a 0.5 mg increase each time, for a total of 11 days. The dose was then increased by 0.5 mg per week until the 9th week. During the interval between tail vein injections, each rat received twice the amount of BSA injected intraperitoneally. At the end of the 5th week, each rat was administrated with 100 μg of LPS in the tail vein. Prednisone is considered as a positive drug that is commonly used in clinical trials to treat various chronic kidney diseases. From the 6th week to 10th weeks, rats (Sham + 20 mg/kg AF group, model + 20 mg/kg AF group, and model + 5 mg/kg Prednisone group) were given AF or Prednisone drugs (Guangdong Huanan Pharmaceutical, Dongguan, Guangdong, China) orally. The AF and Prednisone dosages in this section were selected based on previous work. 17 All animal experiments were approved by the Ethics Committee of Affiliated Hospital of Guizhou Medical University(2021-LUNSHEN-055).
24 h urinary protein assay and measurement of biochemical parameters
The total protein content of rat urea after 24 h of exposure to AF and Prednisone was determined using the biuret method and expressed as mg/24 h. Rats were fasted and given free access to water during urine collection. After the animals were euthanized, aortic blood samples were collected and centrifuged at 3000 rpm for 10 min at 4°C, and urine samples were centrifuged at 600 g for 5 min. Serum creatinine (SCr), serum total cholesterol (TC), serum total triglycerides (TG), and urine blood urea nitrogen (BUN) were measured using an ELISA kit (Nanjing Jiancheng Institute of Biological Engineering, Nanjing, Jiangsu, China).
Hematoxylin-eosin and Masson staining
An ethanol fixation period of 48 h was followed by dehydration in ethanol, embedding in paraffin, and sectioning with a thickness of 5 μm was performed on the kidney tissues. Hematoxylin-eosin (H&E) staining was performed after dewaxing with gradient series ethanol. After washing with 95% ethanol and staining with acid ponceau solution for 1 min, sections were stained with 1% hematoxylin for 5 min, washed again by 95% ethanol, and stained with masson stain for 5 min. The results were observed using an Olympus BX-42 microscope (Tokyo, Japan).
Western blot
Total proteins were extracted and quantified using RIPA lysis buffer (Beyotime). The proteins were electrophoresed on 12% SDS-PAGE and transferred to PVDF membranes (Millipore, Boston, Massachusetts, USA) and blocked with 5% skim milk in the following steps. The membranes were then incubated with different primary antibodies against ki67 (1:2000, ab16667, Abcam, Cambridge, UK), Collagen IV (Col-IV, 1:1000, ab214417, Abcam), alpha-smooth muscle actin (α-SMA, 1:1000, ab7817), PI3K (1:2000, ab278545, Abcam), p-AKT (1:1000, #4060, Cell signaling, Beverly, Massachusetts, USA), AKT (1:2000, #4691, Cell signaling), p-NF-κB (1:1000, #3033, Cell signaling), NF-κB (1:2000, #8242, Cell signaling), and GAPDH (1:2000, ab8245, Abcam) overnight at 4°C. After washing with Tris Buffered Saline Tween (TBST), the HRP-labeled secondary antibody (1:5000, P48010, Yeasen, Shanghai, China) was used for incubation at indoor temperature for 1 h. Finally, the ptotein bands were observed under enhanced chemiluminescence (GE Healthcare, Chicago, Illinois, USA).
Statistics
Each experiment was repeated at least for three times. The data were expressed as mean ± standard deviation (SD). SPSS 18.0 (Chicago, Illinois, USA) was used to analyze the data. The comparisons among groups were performed by One-Way ANOVA, followed by Duncan’s test. p <0.05 was recognized as significant.
Results
AF reduces LPS-induced proliferation of HMCs
The molecular structure of AF was exhibited in Figure 1(a). HMCs were stimulated with LPS and different concentrations of AF for 24 h to determine the role of AF in HMCs proliferation. The results of the CCK-8 assay depicted that LPS dramatically enhanced cell vitality of HMCs, while AF treatment (from 5 μM to 40 μM) dramatically suppressed the growth of HMCs (Figure 1(b)). Since AF concentrations from 5 μM to 20 μM had obvious effects on cell viability, the dose of AF in this range was used for subsequent experiments. The EdU incorporation assay revealed that LPS stimulation significantly increased the number of EdU-positive cells, whereas AF markedly decreased the EdU incorporation into HMCs (Figure 1(c) and (d)). According to Western blot, the LPS-induced elevation of ki67 protein expression was gradually reduced (Figure 1(e)). The findings indicated that AF could restrain the LPS-induced proliferation of HMCs. AF reduces LPS-induced proliferation of HMCs. (a) The molecular structure of AF was depicted. (b) CCK-8 assay was used to detect the viability of HMCs treated for 24 h with LPS (30 μg/mL) or AF at various concentrations (2.5, 5, 10, 20, and 40 μM). (c) The quantification (%) of EdU-positive cells. (d) Representative images of EdU results under a light microscope. (e) Ki67 protein expression was assessed using Western blot. The statistical data of protein bands were analyzed by Image J 1.48. Data were shown as mean ± SD **p < 0.01 versus control (con) group, #p < 0.05 and ##p < 0.01 versus LPS group.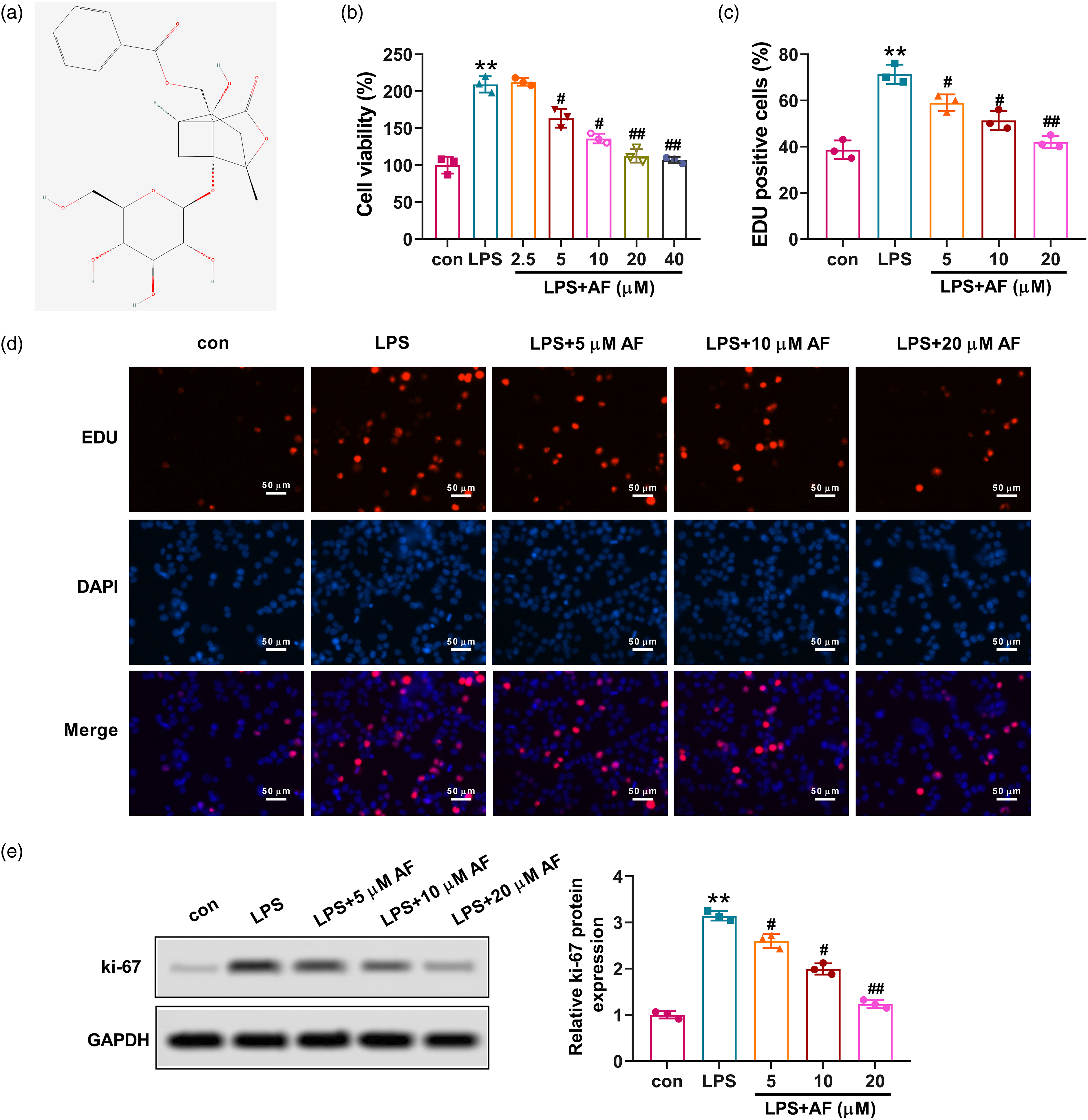
AF reduces inflammatory factors and fibrosis factors in LPS-induced HMCs
To determine the anti-inflammatory effect of AF on HMCs, the mRNA levels of IL-1α, IL-10, IL-2, and IFN-γ were measured by qRT-PCR. AF at concentrations ranging from 5 μM to 20 μM notably reduced the elevated cytokines induced by LPS (Figure 2(a)–(d)), indicating that AF reduced LPS-induced inflammation of HMCs. The fibrosis factors were measured to better understand the impact of AF on the HMCs. The Western blot results manifested that the fibrotic protein levels of Col-IV and α-SMA were significantly up-regulated by LPS induction, while AF treatment (from 5 μM–20 μM) significantly decreased the expression. Taken together, AF could reduce inflammatory and fibrosis factors induced by LPS in HMCs. AF reduces inflammatory factors and fibrotic factors induced by LPS in HMCs. (a–d). qRT-PCR assay was used to assess the mRNA levels of IL-1α, IL-2, IL-10, and IFN-γ in HMCs. (e) Col-IV and α-SMA protein levels as determined by Western blot. Quantitative analysis of Col-IV and α-SMA protein levels. Data were shown as mean ± SD **p < 0.01 versus con group, #p < 0.05 and ##p < 0.01 versus LPS group.
AF affects LPS-induced injury of HMCs by regulating the PI3K/AKT/NF-κB signaling pathway
The protein expression associated with PI3K/AKT/NF-κB pathway was detected in LPS-induced HMCs. As depicted in Figure 3(a), the protein levels of PI3K, p-AKT, and p-NF-κB were dramatically enhanced in the LPS group, while AF significantly inhibited the expression of these proteins, showing that AF possibly prevented injury of HMCs by regulating PI3K/AKT/NF-κB signaling pathway. The effects of AF under PI3K agonist (IGF-1) intervention were detected in HMCs, to further confirm that the protective effects of AF were mediated by this signaling. Remarkably, IGF-1 reversed the inhibition effect of AF on PI3K/AKT/NF-κB signaling (Figure 3(b)), which indicated the definite suppression of AF on PI3K/AKT/NF-κB pathway. The CCK-8 assay demonstrated that treatment with IGF-1 significantly mitigated the anti-proliferative effect of AF on LPS-induced HMCs (Figure 3(c)). As expected, IGF-1 counteracted the anti-inflammatory effect of AF on HMCs, as evidenced by increased mRNA expressions of IL-1α, IL-10, IL-2, and IFN-γ (Figure 3(d)–(g)). These results proved that AF could ameliorate the LPS-induced injury of HMCs via modulating the PI3K/AKT/NF-κB pathway. AF affects LPS-induced injury of HMCs by regulating the PI3K/AKT/NF-κB pathway. HMCs exposed to LPS (30 μg/mL) were treated with IGF-1 (10 nM) with or without AF (20 μM) for 24 h. (a-b) The protein expression of PI3K, p-AKT, AKT, p-NF-κB, and NF-κB was analyzed by Western blot. The statistical data of the proteins were analyzed using Image J 1.48 software. (c) The cell viability of HMCs was assessed by CCK-8 assay. (d-g) The mRNA levels of IL-1α, IL-2, IL-10, and IFN-γ were determined by qRT-PCR. Data were shown as mean ± SD **p < 0.01 versus con group, #p < 0.05 and ##p < 0.01 versus LPS group, &p < 0.05 versus LPS + 20 μM AF group.
AF alleviates renal injury in MPGN model rats
The 24 h urinary protein was tested to validate the renoprotective effects of AF on MPGN rats. When compared to the model group, AF significantly lowered 24 h urinary protein levels (Figure 4(a)). AF also notably lowered the contents of SCr and BUN (Figure 4(a)), which exhibited an improvement in renal function. Additionally, AF markedly lessened the levels of TC and TG (Figure 4(b)), demonstrating that AF suppressed dyslipidemia. H&E staining showed that AF improved the glomerular structures (arrows) and ameliorated inflammatory infiltration and tubule dilation (Figure 4(c)). Prednisone, the positive drug that showed protective effects against MPGN, produced similar results to AF. Our results revealed that AF alleviated renal injury by improving renal function and mitigating dyslipidemia and pathological changes. AF alleviates renal injury in MPGN model rats. (a) The content of 24 h urinary protein of rats from Sham group, model group, model + 20 mg/kg AF group, and model + 5 mg/kg Prednisone (positive control) was tested by biuret method. The levels of SCr and BUN in rats were tested by the ELISA kits. (b) The levels of TC and TG in serum of rats were detected by ELISA kits. (c) H&E-stained kidney tissue sections from each group. Arrows indicate glomerular structures. Data were presented as mean + SD **p < 0.05 versus Sham group, ##p < 0.05 versus model group.
AF may mitigate the inflammatory response and fibrosis by inhibiting PI3K/AKT/NF-κB signaling pathway in MPGN model rats
We examined the inflammatory cytokines that have been linked to renal inflammation. Rats treated with AF or Prednisone depicted a reduction in inflammatory response, including the contents of factors, IL-1α, IL-10, IL-2, and IFN-γ in serum (Figure 5(a)). Additionally, increased fibrosis was observed in the kidneys of MPGN model rats with Masson staining, but the AF or Prednisone groups displayed mitigating effects compared with the model group (Figure 5(b)). Furthermore, the evaluation of the PI3K/AKT/NF-κB signaling was conducted using Western blot. The MPGN model rats had higher levels of PI3K/AKT/NF-κB signaling-related proteins, whereas AF or Prednisone effectively reduced PI3K, p-AKT, and p-NF-κB protein expression (Figure 5(c)). Collectively, AF might ameliorate the inflammation and fibrosis via inhibiting the PI3K/AKT/NF-κB pathway in MPGN model rats. AF may mitigate the proliferation of mesangial cells, inflammatory response, and fibrosis by inhibiting PI3K/AKT/NF-κB pathway in MPGN model rats. (a) The contents of inflammatory factors, IL-1α, IL-2, IL-10, and IFN-γ in the serum of rats were measured by ELISA. (b) The renal fibrosis of rats was examined by Masson staining, and collagen was statistically displayed in the histogram. (c) The protein expression of PI3K, p-AKT, AKT, p-NF-κB, and NF-κB was analyzed by Western blot. The statistical data of proteins were analyzed with Image J 1.48. Data were presented as mean ± SD **p < 0.05 versus Sham group, ##p < 0.05 versus model group.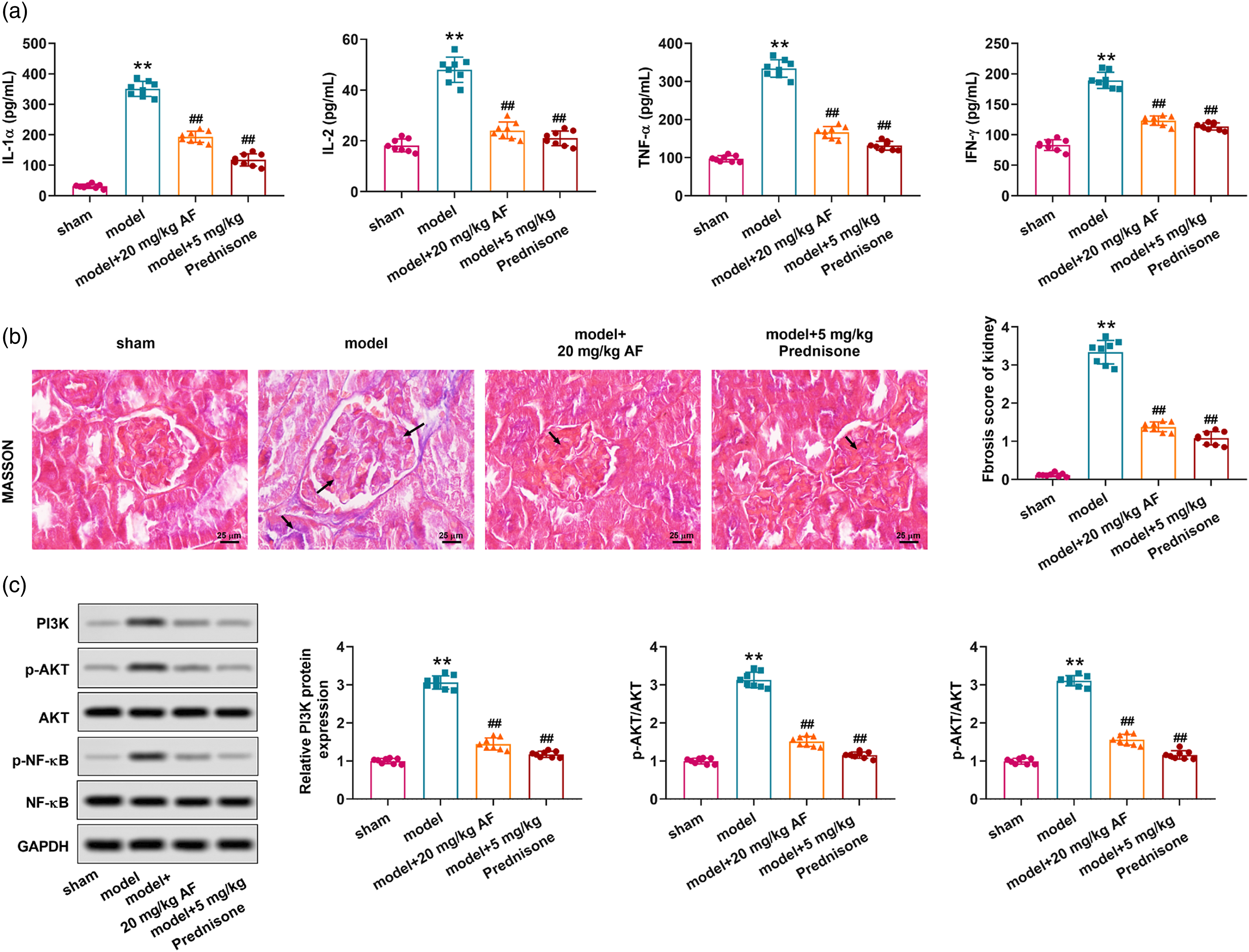
Discussion
The MPGN is featured with mesangial cell overproliferation and mesangial matrix expansion. Mesangial cell overproliferation and inflammation are important in MPGN development, but the potential molecular mechanisms are unclear, and there is currently no special therapeutic drug for MPGN. 18 In the glomerulus, mesangial cells are active intrinsic cells, and the most common response to a variety of stimuli is proliferative response. For this reason, it will be imperative to inhibit the overproliferation of mesangial cells in order to treat MPGN. As an active compound in Paeonia Alba Radix, AF has been demonstrated to exert pharmacological effects, such as anti-oxidation and anti-inflammation.11,12 From the perspective of traditional Chinese medicine, Chinese herbal medicines that are low-toxic, have few side effects, and have significant curative effects will be considered a significant advancement in renal failure treatment and prognosis. However, the potential therapeutic application of AF for MPGN remains undetermined.
The MPGN rat model was characterized by immune complex deposition in the glomeruli, matrix expansion, and cell hypercellularity. Our findings showed that AF inhibited cell viability, decreased EdU-stained proliferating cells, and reduced ki67 expression in LPS-induced mesangial cells, demonstrating that AF has potent anti-proliferative effects on mesangial cells. Mesangial cells that have been activated and proliferated in glomerulus can produce multiple cytokines and vasoactive substances, such as IFN and IL-1, which promote the continuous proliferation of mesangial cells. 19 The anti-inflammatory effects of AF on kinds of diseases have been recognized. 20 For instance, AF mitigates the inflammation caused by ovalbumin in asthmatic mice, 11 reduces the inflammation caused by chronic constriction injury in the hippocampal NLRP3 inflammasome in rats, 12 ameliorates streptozotocin-induced cognitive impairment via modulating inflammation and oxidation in the brain of rats, 14 and alleviates oxidative stress and inflammation in Methotrexate-induced enteritis. 21 Our data demonstrated that AF reversed the inflammation by lessening the factor levels of IL-1α, IL-10, IL-2, and IFN-γ in vitro and in vivo. Inflammatory mediators are released when mesangial cells proliferate abnormally, leading to renal fibrosis and progressive glomerulosclerosis. 22 Glomerulosclerosis and tubulointerstitial fibrosis are caused by an excessive accumulation of extracellular matrix. A persistent inflammatory response may eventually lead to fibrosis. Proinflammatory cytokines increase extracellular matrix in the kidney, resulting in renal fibrosis. 23 Pathological conditions stimulate myofibroblasts by promoting the synthesis of α-SMA in the interstitial space. 24 Col-IV is an extracellular matrix component that is thought to be important in myofibroblast differentiation. Furthermore, the hepatoprotective effect against chronic liver fibrosis of AF has been recognized. 25 In this work, we proved that AF weakened the expression of Col-IV and α-SMA in vitro and alleviated kidney fibrosis in vivo, with similar effects on MPGN.
The activation of PI3K/AKT signal transduction is critical for maintaining cell cycle progression and promoting cell growth and proliferation. 26 IGF-1 is thought to combine to IGF-1 receptor and the PI3K agonist. 27 According to our results, IGF-1 antagonized the inhibitory effects of AF on PI3K/AKT/NF-κB signaling activation and renal injury. The effects of AF on the PI3K/AKT signal and the NF-κB signal have also been reported,14,15 which is in line with our data. Inflammatory factors that activate the PI3K/AKT signaling pathway can cause glomerular mesangial cell overgrowth. 28 In an anti-Thy1 MPGN rat model, AKT phosphorylation is linked to mesangial cell proliferation. 29 Small molecule blockers of PI3K/AKT pathway were also found to significantly attenuate the pro-proliferative effect of PDGF-BB on mesangial cell abnormal proliferation, as well as the expression and release of inflammatory mediators in mesangial cells. 30 When PI3K/AKT signaling is blocked, HMCs can be prevented from proliferating abnormally. 31 Our data is in line with the prior work. NF-κB is a downstream component of the PI3K/AKT signaling pathway and is activated by the PI3K/AKT signal via phosphorylation of IκB kinase, resulting in the degradation of IκB. 32 A study showed that NF-κB inhibitors mitigated kidney injury through suppressing inflammatory response and oxidation. 33 In our results, AF depicted similar effects in terms of reducing inflammatory cytokines and possibly inhibiting inflammation in combination with NF-κB. PI3K/AKT signaling has been implicated in numerous studies in regulating inflammatory factors on glomerular mesangial cell proliferation 28,34 and the synthesis and expression of extracellular matrix, such as type I collagen. 35 Another review demonstrated that NF-κB activation aggravates renal fibrosis by increasing Col-IV expression. 36 We also found that AF might alleviate renal fibrosis by inhibiting PI3K/AKT/NF-κB pathway.
Conclusion
In a word, AF treats MPGN by inhibiting mesangial cell overproliferation, inflammation, and fibrosis. AF can also significantly reduce the renal pathological injury in MPGN model rats. We also showed that AF effectively prevents MPGN, at least in part, by restraining PI3K/AKT/NF-κB pathway. Our data are encouraging and manifested that AF could be a new therapeutic agent for MPGN. The mechanism of traditional Chinese medicine has its particularity, and how it works is not fully determined. In the future, some bioinformatics methods will be used to explore the targets of AF in MPGN disease and delineate the complex network, so as to provide new strategies for the treatment of MPGN.
Footnotes
Author contributions
Ye Chen designed the experiments; Haiyan Yu, Yu Wang, Zida He, and Ruixue Chen carried out the experiments; Yingni Dai and Yingqian Tang analyzed the data and prepared figures; Haiyan Yu drafted the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the this work was supported by Guizhou Provincial Department of Science and Technology (Intellectual Property Office) joint fund project [Qian Ke He LH Zi (2017) No. 7183].
Data availability statement
All data and models generated or used during the study appear in the submitted article.
