Abstract
Background
Dietary quinic acid given as the nutritional supplement, which may leads to tryptophan and nicotinamide production in the intestinal tract and NAD+ precursor which can prevent from the negative consequences of high fat diet (HFD) consumption.
Objective
The present study was designed to assess in vivo and in vitro effect of D-(−)-Quinic acid in high-fat diet induced hyperlipidemia in mice.
Material and methods
Thirty six albino mice were randomly divided in six groups and each group had six mice. Group I, controlled mice given normal pellet diet, Group-II mice, administered with high fat diet (HFD), Group-III mice given standard drug, Atorvastatin (20 mg/kg, p.o.) along with HFD to mice and Group IV, V and VI mice received D-(−)-Quinic acid at a dose of 75, 150 and 300 mg/kg, respectively in separate group along with HFD to mice. After completion of trial (49 days) the animals were sacrificed and evaluated for body weight, organ fat pad weight, and changes in weight of liver, heart and kidney and also for biochemical parameters, expression of adipogenic and inflammation markers in adipose tissues, and histology examination of liver tissue.
Results
In vitro testing results showed, D-(−)-Quinic acid potentially inhibit α-glucosidase enzyme activity as compared to acarbose. The D-(−)-Quinic acid showed significant hypolipidemic activity by decreasing the increased level of cholesterol, triglyceride level, LDL, VLDL and other hepatic parameters like SGOT and SGPT in serum. D-(−)-Quinic acid reduces the mRNA expression level of PPAR-γ2, TNF-α, IL-1β and IL-6 in adipose tissue in hyperlipidemic mice.
Introduction
Obesity is one of the major causes of many vascular diseases and it is the driving reason for researchers around the world to investigate safe and effective medicines. Every year, approximately 2.8 million people will suffer as a result of high lipid content or changes in vascularity. 1 Obesity is associated with liver damage and leads to preconditioned metabolic abnormalities.2,3 Obesity, hypertension, dyslipidemia, and thyroid problems are all risks for adults who have body issues. 3 Hypertrophic liver or metabolic insufficiency is a kind of hyperlipidemia that happens when there is an increase in the number of fat cells around the liver. Hypertrophic liver refers to the enlargement of fatty cells with virtually no change in the number of fat cells. 4
The prevalence and number of cases of obesity are rapidly increasing. Day by day, around 50 million individuals are clinically diagnosed and more than one billion grown-ups are due to metabolic and diet induced complications. 5 The WHO additionally anticipates that this number may increase to 3.3 billion in the year 2030. Previous research has found that individuals with excessively fatty diets and smoking habits are more prone to hyperlipidemia. 6 Furthermore, the use of work-saving devices such as lifts, automobiles, remote controls, PCs, and stationary recreational exercises has contributed to the population’s obesity.7,8
Obesity is considered as a heterogeneous state in which people with similar BMIs may have different metabolic and CVD risk profiles. Thus, prevalence to obesity-related cardiovascular comorbidities is largely mediated by individual variation in regional body fat accumulation, which adversely affects cardiac structure and function. With the rising prevalence of obesity in longer-lived populations, there is an increased need for future research to investigate the underlying mechanisms for obesity-related cardiac dysfunction and to strengthen the care of patients with obesity and CVD. 9
Since then, strategies have centered on natural sources, where fatty liver-related problems with fewer side effects have been reduced. 10 Various studies have shown that bioactive ingredients such as antioxidants, flavonoids, alkaloids and phenolic content have positive results in hyperlipidemia. Some edible therapeutic plants are used as dietary enhancements for hypolipidemic action in different countries11,12 Such plants are Citrusa urantium, 13 Hibiscus sabdariffa etc. D-(−)-Quinic acid is a phenolic acid that can be found in a wide range of plants and microorganisms.9,14 It cannot be synthesized by mammals, including humans. D-(−)-Quinic acid was reported earlier to have antioxidant and anti-inflammatory properties. 15 It increases tryptophan and nicotinamide production in the intestinal tract, which enhances all lipoprotein concentrations. 9 Earlier research revealed the significant role of α - glucosidase inhibitors in non-insulin - dependent diabetes patients with elevated levels of lipoproteins. 16 The current study also focused on a compound with potent alpha glucosidase inhibition and acts as a modulator of lipoprotein content in mice. However, D-(−)-Quinic acid actions against high fat diet-induced obesity has not been studied yet. The aim of the present study is to suppress obesity and associated cardiac toxicity in mice.
Materials and Methods
Chemicals
D-(−)-Quinic acid was procured from Siga Aldrich (B. No. QN27364). Atorvastatin was a gift sample from a Sentiss Pharma Pvt Ltd Chemicals like methanol, chloroform, sesame oil, NaCl, CaCl2, phosphate buffer solution, copper reagent, diethyl dithiocarbamate sodium, oleic acid, bovine serum, albumin serum, Serum Cholesterol, Serum Triglyceride pack, Serum HDL-Cholesterol from High media pvt. Ltd and S D Fine Chemicals Ltd (Mumbai).
In-vitro assay
Determination of hydrogen peroxide scavenging activity
Scavenging of hydrogen peroxide was measured by previously reported method. 17 Different dilutions of D-(−)-Quinic acid were prepared and added to 0.6 mL of 40 mM hydrogen peroxide solution. Absorbance of samples were taken at 230 nm.
H2O2 radical scavenging (%) = (A0 – A1)/A0×100
Where A0 was the absorbance of the control and A1 was the absorbance of sample.
α – Glucosidase inhibition assay
α-Glucosidase inhibitory activity was performed by following the method described by Dong 18 with slight modification 19 . Acarbose is used as a standard in assay. The sample and standard absorbance was measured at 405 nm using a UV–visible spectrophotometer.
The inhibition percentage (%) was determined as follows:
Inhibition (%) = (1-A sample/A control) × 100
Experimental animal
Studies were performed on healthy Swiss Albino mice weighing 20–25 g of either gender (6 weeks old). The animals were placed in a group of six containing six animals each and maintained under controlled temperature (22–24oC) in a 12-h light dark cycle. The mice have been managed to keep in the paddy husk and given equal access to food and water. All tests have been permitted and conducted out in accordance with Administration Committee of Experimental Animals (Approval No. JS-IRB-00I-05).
High fat diet formula
HFD consisting of 58% fat, 25% starch, 17% fiber, 13% lard, 1% cholesterol, and 0.6% vitamin and minerals was provided regularly for 14 weeks. Food consumption was estimated on a regular basis and weight gain was weighed per week (Table 1).
Dose selection
Throughout the current research, three doses of D-(−)-Quinic acid were chosen as 75 mg/kg, 150 mg/kg and 300 mg/kg, also based on the earlier study (LD50 is 5.45 g/kg).9,12 The dose of D-(−)-Quinic acid solution for drug administration was prepared by dissolving measured quantity of D-(−)-Quinic acid in 0.2% of carboxy methyl cellulose (CMC).
Methodology
Mice of either sex with a weight of 20–25 g were selected for study. Mice were allowed free access to normal pellet diet and the water. The study consists of six groups comprising six mice for every group. Group I was normal control group with free access to water and normal pellet diet and given normal saline (0.9%) as vehicle control. Remaining animals except control group were kept on high Fat diet administration for 8 weeks to access the obesity status of animals (animals gain bodyweight more than 35 gm were considered as obese). Later the animals randomly divided in five groups and six animals each. Group II was perceived as negative control category in which high-fat diet treatment (HFD) was given without any treatment to mice. Group III, in which mice were treated with atorvastatin (20 mg/kg, p.o) together with High Fat Diet Group IV, V and VI were test drug treatment groups where mice were treated with three doses (75 mg/kg, 150 mg/kg and 300 mg/kg, p.o) separately along with High fat diet (HFD).
In- Vivo Pharmacological evaluation parameters
Body weight and feed intake
The body weight and feed intake of all treated mice were recorded and changes was noted weekly. The mice has given 5 gm of diet to each mice daily in a group of six animals and this was also constant in vehicle control group with 5 gm of standard chow diet to each mice daily in a group of six animals and weight of animals were recorded after every 2 weeks till the end of experiment.
Weight of organs
Mice organs including Kidney, Liver, fat pads have been carefully dissected and measured using an electronic weighing balance.
Body mass index
Body mass index (BMI) = ¼ body weight(g)/length2 (cm)2.
Lee index
The lee index was determined using formula described by Bernardis. 8
Histopathological studies
Excised quickly the organ and fixed in 10% buffered formalin. The tissue was cut in fine section (5 μm) and on glass slide mounted it. The section was stained with hematoxylin (0.6% w/v) for 15 min and eosin (1% w/v) for 2 min and then examined under the light microscopy.
Statistical analysis
High Fat diet Formula.

Results
Determination of hydrogen peroxide scavenging activity
Hydrogen peroxide scavenging and α– Glucosidase inhibition activity of D-(−)-Quinic acid.

Concentrations of D-(−)-Quinic acid and standard were in the range of 0.001 μg–1000 μg/mL.
α– Glucosidase inhibition assay
The maximum α– glucosidase inhibition was observed at higher concentration of D-(−)-Quinic acid at1000 μg/mL and order of inhibition was concentration dependent, 10<50<100<250<500<1000. The IC50 value of D-(−)-Quinic acid was calculated 93.75 μg/ml (Table 2).
Effect of quinic acid on serum lipid profile
Effect of quinic acid on serum lipid parameter.

The data were expressed as mean ± SEM (n = 6) and the analysis of variance was performed using one way ANOVA with post hoc test.
aWhen compared with control group.
bwhen compared with inducer group.
cp < 0.005.
dp < 0.01.
ep < 0.001, ns p > 0.05.
Results showed that mice recieved HFD (Group II)alone for 49 days showed significant increase (p < 0.001) in serum SGOT and SGPT level as compared with control mice group I. The treatment of mice with Quinic acid showed a significant decrease (p < 0.001) in SGOT and SGPT levels, which indicates a hepatoprotective effect (Table 3).
Effect of quinic acid on body weight
Effect of D-(−)-Quinic acid on body weight.

The data were expressed as mean ± SEM (n = 6) and the analysis of variance was performed using one way ANOVA with post hoc test. When compared with control group in fourth week of study.
ap < 0.005.
bp < 0.01.
cp < 0.001, ns p > 0.05.
Effect of quinic acid on BMI
Effect of D-(−)-Quinic acid on BMI.

The data were expressed as mean ± SEM (n = 8) and the analysis of variance was performed using one way ANOVA with post hoc test. When compared with control group in fourth week of study.
ap < 0.005.
bp < 0.01.
cp < 0.001, ns p > 0.05.
Effect of quinic acid on rectal body temperature
Rectal body temperature of mice fed with HFD (Group II) was decreased comparative to mice fed with normal pellet diet (Group I) while treatment with Quinic acid showed an increase in body temperature after administration of quinic acid.
Effect of D-(−)-Quinic acid on organ body weight
Effect of D-(−)-Quinic acid on Organ weight.
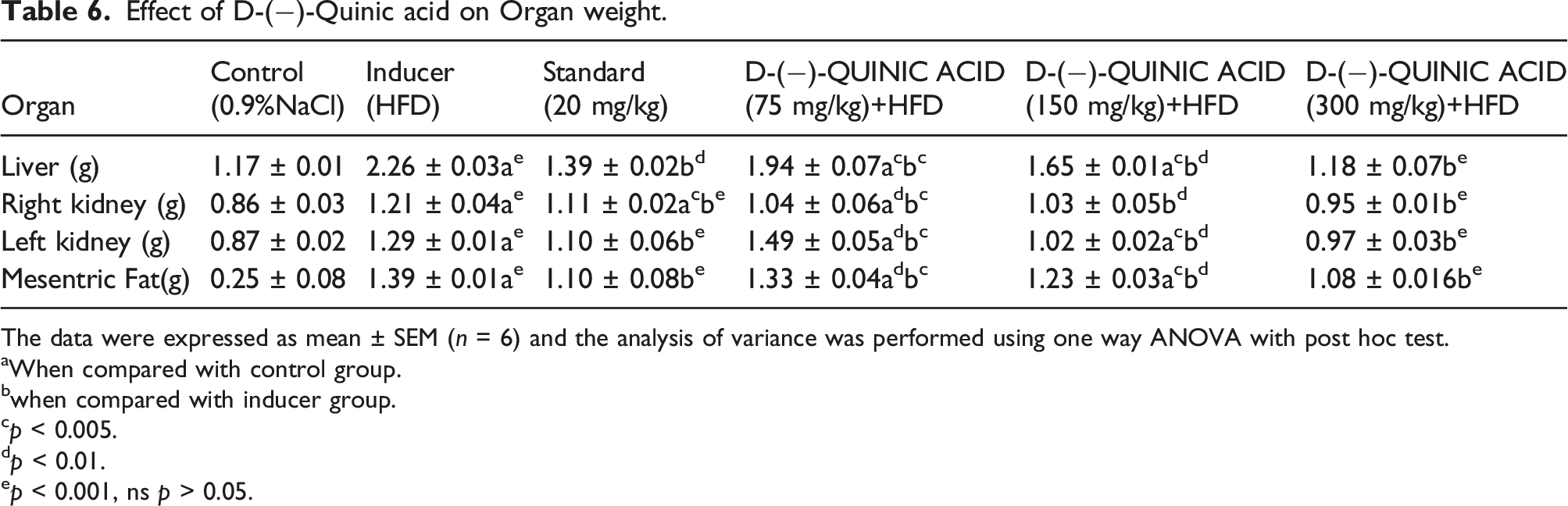
The data were expressed as mean ± SEM (n = 6) and the analysis of variance was performed using one way ANOVA with post hoc test.
aWhen compared with control group.
bwhen compared with inducer group.
cp < 0.005.
dp < 0.01.
ep < 0.001, ns p > 0.05.
Effect of D-(−)-Quinic acid on histology of liver tissue
Histopathological studies revealed that steatosis caused by high fat diet and the metabolism of unsaturated lipids is associated with inflammation, congestion, and nonalcoholic fatty liver disease resulting in hepatic failure. HFD induced hyperlipidemia that increases the enzymes of liver and observation of hepatocellular damage which correlates with increased liver weight. Histopathological analyses in Quinic acid treated mice demonstrated reduced hepatic disruption and increased cell viability (Figure 1(a)–(f)). Histopathology of liver: the figure showing different changes were seen in histology of hepatic cells where H- hepatocytes, N-Necrosis, PT-periphery of lobule, (a) control group, (b) Inducer HFD group, (c) Standard drug treated group, (d) quinic acid treated (75 mg/kg), (e) quinic acid treated (150 mg/kg) and (f) Quinic acid quinic acid treated (300 mg/kg) treated groups.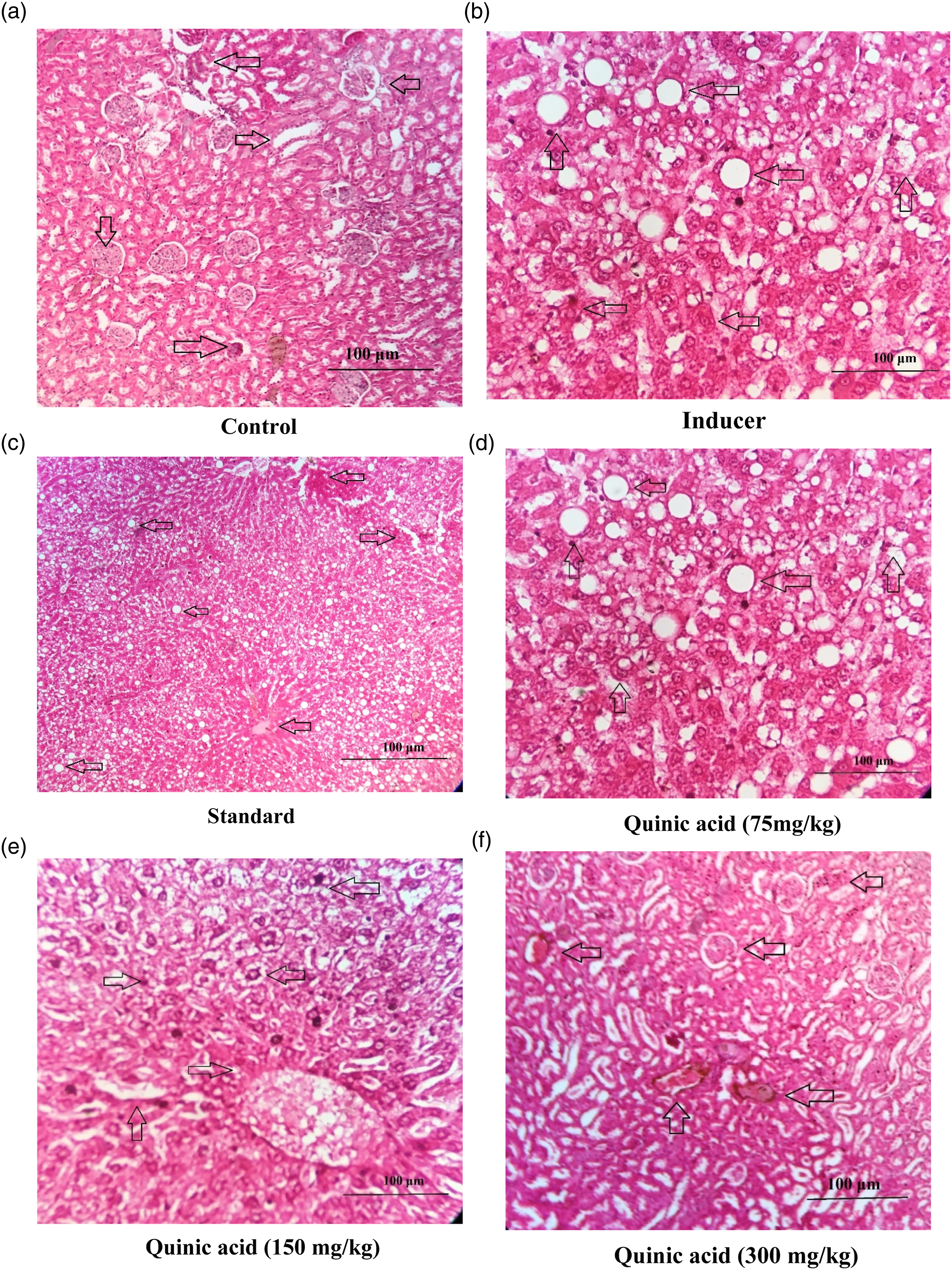
Expression of adipogenic and inflammation markers in Adipose tissues of HFD treated mice. Relative mRNA expression level of (a) PPAR-γ2, (b) TNF-α, (c) IL-1β and (d) IL-6 in adipose tissue of experimental mice were assessed by quantitative real time PCR. All the values were expressed as mean ± SD of six rats in each group. The statistically significant difference among groups was determined by oneway ANOVA using SPSS version 20. *p < 0.05, ***p < 0.001, ***p < 0.01, ns p >0.05, when compared with the HFD induced obesity, #p < 0.05, ##p < 0.001, ###p < 0.01 when compared with control group.
Discussion
Hyperlipidemia is considered a disease that leads to severe complications like liver cirrhosis and cardiac insufficiency as it progresses. 21 Hyperlipidemia is distinguished by the extra fat layer deposited in the liver cells. Hyperlipidemia is seen as very common in patients with obesity. 22 The prevalence of weight gain is rapidly increasing in all over the world, and there is a great deal of interest in inventing more safe and effective medications for hyperlipidemia treatment. 23 This disease has progressed highly in the 21st century, becoming one of the most serious public health issues.
High-fat diet induced obesity is a well reported model and used previously for screening of drugs to treat hyperlipidemia. 24 Weight loss occurs when the fat deposition in cells decreases as a result of a decrease in the arrangement of new adipocytes from preexisting cells or a decrease in adipocyte size as a result of fat storage. 25 Here, this study propose that D-(−)-Quinic acid may protect or prevent high fat diet-induced obesity, fat accumulation, and fat deposition in the liver. In this study, the decrease in body fat percentage that could be due to decreased adipocyte production and fatty livers. The accumulation of triglycerides in hepatocytes is responsible for increased unsaturated fat deposition and decreased unsaturated fat oxidation. The results showed in present study indicates increased level of triglycerides’ content in blood. One of the critical nuclear receptors is PPARα that controls lipid metabolism 26 and the PPARα agonist acts to decrease the formation of TG and stimulation of fatty acid oxidation. 27 This study reported to decrease in expression of PPAR-γ2, TNF-α, L-1β and IL-6 in adipose tissue which were found similar in drugs which used to evaluate anti-hyperlipidemia in mice. 28 The quinic acid treated mice showed decreased in expression of PPAR-γ2, TNF-α, L-1β and IL-6 in adipose tissue. Oral administration of D-(−)-Quinic acid has decreased food intake in animals and altogether stifled higher weight increases, fat rate, and total fat and HFD-feed in mice in a dose dependent manner. Adipogenesis, or fat cell arrangement, is a type of differentiation in which undifferentiated preadipocytes are transformed into fully differentiated adipocytes, which store energy as fat and make the subject’s stout.28,29 Matsuzawa Nagata30,31 reported that ROS generation and oxidative stress were also correlated with greater risk of obesity. The present study also estimated the Invitro hydrogen peroxide scavenging activity which indicates it antioxidant activity. Obesity is also related to oxidative stress and induces other degenerative conditions, such as diabetes, hypertension and hyperlipidemia. As a result, in the search for plants that have the dual capacity of preventing cellular stress and combating obesity, later it also affects liver functions. Mice with high total cholesterol, glyceride levels, or LDL concentrations have been linked to coronary vascular disease, insulin resistance, and non-alcoholic fatty liver. 32 HFD raises free unsaturated fat to cause metabolic diseases and chronic inflammation activation. 33 In the present study, it was observed that D-(−)-Quinic acid is one such therapeutic compound which provides a significant reduction high cholesterol and decrease in LDL levels in mice. The change in body weight was accounted for high fat diet administration in mice and, which is a reasonable model for obesity, also showed a similar change as in humans.
The α-Glucosidase enzyme is essential and it is engaged in the assimilation of starches. 34 In this examination, a study has been done to access the inhibition capacity of the alpha-glycosidase. The alpha-glucosidase inhibitors concede the degradation of sugar, which backs off the accumulation of glucose. One of the α-glucosidase inhibitors is Acarbose, which inhibits the metabolism of starch and disaccharides. Digestion of lipids assumes a vital role in the management of energy homeostasis. In this way, inhibition of enzymes might be considered as a primary focus for the treatment of obesity.35–37 The present investigation additionally demonstrated that administration of D-(−)-Quinic acid restrains the alpha glucosidase enzyme. In this way, the most adequate procedure to treat liver dysfunction is by utilizing herbal extracts with inhibitory potential. 38 Flavonoids are one of the potential free radical scavengers with a structural similarity to phenolic acids. 39 The inhibitory action of D-(−)-Quinic acid on weight gain is most likely credited to the phenolic and flavonoid content. In the current study, a D-(−)-Quinic acid had a significant effect on the hepatic disease. Restriction of pancreatic lipase movement and growth of lipolysis is being viewed as a powerful approach to weight control. 39 The present investigation uncovered that inhibition of the metabolism of lipids in the digestive system is the potential mechanism by which D-(−)-Quinic acid averted weight gain in the HFD-fed mice. Previous literature has additionally supported this report. 40 In the present investigation, it was uncovered that D-(−)-Quinic acid essentially protects from the damage induced by HFD.
Histopathological studies have revealed that steatosis caused by a high fat diet and the metabolism of unsaturated lipids is associated with inflammation, congestion, and non-alcoholic fatty liver disease, resulting in hepatic failure. 41 HFD induces hyperlipidemia that increases the enzymes of the liver and the observation of hepatocellular damage, which correlates with an increased liver weight. Histopathological analyses of D-(−)-Quinic acid treated mice demonstrated reduced hepatic disruption and increased cell viability.
Conclusion
The present findings propose that the Quinic acid exhibit hepatoprotective and hypolipidemic action against fat diposition induced by HFD. The histopathological investigations of the kidney, liver, heart, mesenteric fat likewise upheld that Quinic acid lowered the hepatotoxicity of HFD. Our outcomes recommended that Quinic acid may be valuable to keep the improvement of weight.
Summary
This study will help researchers identify the potential reason of the pharmacological effects of quinic acid and help other researchers design an improved clinical treatment plan for metabolic disorders.
Supplemental Material
Supplemental Material - Protective effect of D-(−)-quinic acid as food supplement in modulating AMP-activated protein kinase signalling pathway activation in HFD induced obesity
Supplemental Material for Protective effect of D-(−)-quinic acid as food supplement in modulating AMP-activated protein kinase signalling pathway activation in HFD induced obesity by Jie Dong, Hai Long Zheng, Qiuyao Zeng, Xian Zhang, Liang Du and Souravh Bais in Human & Experimental Toxicology
Footnotes
Acknowledgements
All the authors want to thank all the institutes affiliated to this work and providing necessary research facility and institutional funding for it.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Institutional funding.
Statement of human and animal rights
All experimental procedures involving animals were conducted in accordance with the institutional animal care review board guidelines of Dongfeng Hospital, Hubei University of Medicine, Shiyan, Hubei, 442008, P.R. China, and approved by the Administration Committee of Experimental Animals, Shiyan, Hubei, China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
