Abstract
Administration of the diuretic, spironolactone (SR), can inhibit chronic liver diseases. We determined the effects of SR alone or in combination with the antioxidant α-glycosyl isoquercitrin (AGIQ) on hyperlipidemia- and steatosis-related precancerous lesions in high-fat diet (HFD)-fed rats subjected to a two-stage hepatocarcinogenesis model. Rats were fed with control basal diet or HFD, which was administered with SR alone or in combination with an antioxidant AGIQ in drinking water. An HFD increased body weight, intra-abdominal fat (adipose) tissue weight, and plasma lipids, which were reduced by coadministration of SR and AGIQ. SR and AGIQ coadministration also reduced hepatic steatosis and preneoplastic glutathione S-transferase placental form-positive foci, in association with decrease in NADPH oxidase (NOX) subunit p22phox-positive cells and an increase in active-caspase-3-positive cells in the foci. Hepatic gene expression analysis revealed that the coadministration of SR and AGIQ altered mRNA levels of lipogenic enzymes (Scd1 and Fasn), antioxidant-related enzymes (Catalase), NOX component (P67phox), and anti-inflammatory transcriptional factor (Pparg). Our results indicated that SR in combination with AGIQ had the potential of suppressing hyperlipidemia- and steatosis-related early hepatocarcinogenesis through the reduced expression of NOX subunits.
In contrast to alcoholic liver diseases, nonalcoholic fatty liver disease (NAFLD) is categorized as a hepatic manifestation of metabolic syndrome, which can be diagnosed by the presence of excess hepatocellular lipid droplet accumulation, that is, steatosis, in the absence of other causes of chronic liver diseases with the absence of significant alcohol consumption (Carr and Ahima 2016). NAFLD is a complex disease that includes a wide spectrum of hepatic disorders from benign steatosis to advanced nonalcoholic steatohepatitis (NASH), fibrosis, and cirrhosis, eventually progressing to hepatocellular carcinoma (HCC; Sheedfar et al. 2013). As patients with NAFLD are increasing worldwide, there is an urgent need to develop effective pharmaceutical drugs as well as dietary supplements to mitigate the liver lesions and hyperlipidemia (Chalasani et al. 2012).
NADPH oxidases (NOXs) comprise of membrane-bound subunits that transfer electrons across membrane, where superoxide is generated and rapidly converted into hydrogen peroxide (Brieger et al. 2012). NOX isoforms (NOX1−5, DUOX1 and 2) are each unique in terms of tissue distribution, domain structure, subunit requirements, and mechanisms of activation. Accumulating evidence suggests that NOX-generated reactive oxygen species (ROS) play a critical role in the development of NAFLD and NASH. In mice, an HFD induces liver steatosis, which is closely coordinated with the expression of the NOX subunit p47phox, along with inflammation and oxidative stress (Gabele et al. 2011; Chatterjee et al. 2013). Inflammatory and oxidative stress markers are significantly decreased by the NOX inhibitor apocynin and p47phox deficiency (Chatterjee et al. 2013). In Zucker obese rats, NOX4, p67phox, and p47phox, but not NOX2, are significantly overexpressed in steatotic livers compared with lean rats (Gupte et al. 2009). We recently found that a NOX subunit p22phox is specifically expressed in the preneoplastic hepatic glutathione S-transferase placental form (GST-P)-positive foci in HFD-fed rats in a two-stage (initiation and promotion) hepatocarcinogenesis model (Yoshida et al. 2017).
Spironolactone (SR) is an antagonist of the mineralocorticoid receptor (MR) and is widely used as a diuretic drug for hypertension (Whaley-Connell, Johnson, and Sowers 2010). ROS production through the activation of NOX plays a role in the onset of renal diseases, and the process is stimulated by aldosterone interaction with MR in endothelial cells (Iwashima et al. 2008; Paravicini et al. 2012) and in podocytes (Shibata et al. 2007). Indeed, SR can reverse the increases in mRNA levels of Rac1 (a cytosolic regulator subunit of NOX1 and NOX2) and NOX4 in kidney tissues, consequently preventing hyperoxaluria-related renal injury in vivo (Tsuji et al. 2016). SR can also inhibit liver cholestasis (Ruiz et al. 2007), portal hypertension (Luo et al. 2012), and fibrosis (Fujisawa et al. 2006) by altering the expression of several key targets including multidrug resistance–associated protein-2, endothelial nitric oxide synthetase, and Na/H exchanger isoform 1, respectively. However, the effect of NOX inhibition by SR on liver diseases is not fully elucidated.
Wada et al. (2010) reported that SR was able to suppress hyperlipidemia and hepatic steatosis in mice fed an HFD with fructose-supplementing water by reducing the transcript levels of pro-inflammatory cytokines, and gluconeogenic and lipogenic enzymes in liver tissues. Therefore, we hypothesized that SR could prevent steatosis-related precancerous lesions in rats through the reduced expression of a NOX subunit. In this study, we fed rats with a basal diet or an HFD using a two-stage hepatocarcinogenesis model to determine whether the treatment of SR alone or in combination with α-glycosyl isoquercitrin (AGIQ), also known as enzymatically modified isoquercitrin, can ameliorate the liver lesions. AGIQ is a quercetin–glycoside mixture (Valentova et al. 2014), which can inhibit chemical-induced early hepatocarcinogenesis in rats (Kuwata et al. 2011; Hara et al. 2014), and its metabolites can modulate the expression of NOX subunits (Kim et al. 2013; Jones et al. 2016).
Materials and Method
Chemicals
N-diethylnitrosamine (DEN; CAS No. 55-18-5, purity >99%) and SR (CAS no. 52-01-7, purity >98%) were purchased from Tokyo Kasei Kogyo (Tokyo, Japan) and Sigma-Aldrich Co. (St Louis, MO), respectively. AGIQ (purity >97%) was provided by San-Ei Gen F.F.I., Inc. (Osaka, Japan).
Animals and Treatment
A total of 53 5-week-old male F344/N rats were purchased from Japan SLC, Inc. (Shizuoka, Japan), maintained in an air-conditioned room (room temperature, 22 ± 3°C; relative humidity, 56 ± 11%; 12-hr light/dark cycle), and given free access to a powdered diet (Oriental MF; Oriental Yeast Co., Tokyo, Japan) and tap water. After a 1-week acclimatization period, a medium-term liver carcinogenesis bioassay (Ito, Tamano, and Shirai 2003) was conducted by the following procedure. All animals received an intraperitoneal injection of DEN at a dose of 200 mg/kg body weight and were randomly divided into four groups. The control group (CTL) received normal diet, and all others received HFD (D12451; Research Diets, Lane, NJ) containing 0 (HFD group) or 750/300 ppm SR (SR group) for 7 weeks starting at 2 weeks after DEN initiation. SR at a dose of 750 ppm (correspondent to 50 mg/kg/day) was administered (Fujisawa et al. 2006) from weeks 2 to 4; however, the dose was reduced to 300 ppm (correspondent to 20 mg/kg/day; Luo et al. 2012) from weeks 5 to 9 because the rats showed rough fur at week 4. The fourth group was administered HFD and SR as well as 5,000 ppm AGIQ (SR + AGIQ) in their drinking water. The dose of 5,000 ppm for AGIQ was administered as previously reported (Yoshida et al. 2017). All the animals were subjected to two-thirds partial hepatectomy one week after SR treatment was initiated. Partial hepatectomy enhanced the appearance of preneoplastic liver lesions by increasing the proliferation of DEN-initiated hepatocytes (Ito, Tamano, and Shirai 2003). Animals were observed clinically during the study. At the end of the nine-week experiment, the rats were euthanized by exsanguination under isoflurane anesthesia, and blood was collected from the vena cava without overnight fasting. The livers, kidneys, and abdominal fat (adipose) tissues (surrounding the spermatic cord) were excised and weighed (absolute weight), and organ weight:body weight ratios (relative weight) were calculated. The sliced samples were fixed in 4% paraformaldehyde in 0.1 M phosphate-buffered formalin (pH 7.4; Wako Pure Chemicals Industries, Ltd., Osaka, Japan) for histopathology and immunohistochemistry. The liver pieces were frozen in liquid nitrogen and stored at −80°C until further analysis. All procedures in this study were conducted in compliance with the Guidelines for Proper Conduct of Animal Experiments (Science Council of Japan, June 1, 2006), and the protocol was approved by the Animal Care and Use Committee of the Tokyo University of Agriculture and Technology.
Blood Biochemistry
We obtained plasma from the blood samples described above and measured the following parameters with JCA-BM1250 (JEOL Ltd., Tokyo, Japan): aspartate aminotransaminase (AST), alanine aminotransaminase (ALT), alkaline phosphatase (ALP), triglycerides (TG), and total cholesterol (T.CHOL).
Real-time Reverse-transcription Polymerase Chain Reaction (RT-PCR) Analysis
Analysis of mRNA levels of the genes listed in Online Supplemental Table 1 in liver tissues (n = 6 animals per group) was performed with real-time RT-PCR. Total RNA was extracted with RNeasy® Mini Kits (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. First-strand cDNA was synthesized from 2 μg total RNA in a 20 μl total reaction mixture containing dithiothreitol, deoxynucleoside triphosphates, random primers, RNaseOUT, and SuperScript™III Reverse Transcriptase (Life Technologies, Carlsbad, CA). Real-time RT-PCR was performed using the SYBR® Green PCR Master Mix (Life Technologies) and the StepOnePlus™ Real-Time PCR System (Life Technologies) according to the manufacturer’s protocol. The PCR primers were designed using Primer Express software (version 3.0; Life Technologies). The relative differences in gene expression were calculated using threshold cycle (CT) values that were first normalized to those of the hypoxanthine phosphoribosyl transferase 1 gene, the endogenous control in the same sample, and then relative to a control CT value by the 2−ΔΔC T method (Livak and Schmittgen 2001).
Histopathology and Immunohistochemistry
The liver and kidney slices were dehydrated in graded ethanol, embedded in paraffin, sectioned, and stained with hematoxylin and eosin for histopathological examination. The pathological changes including steatotic cells, ballooning cells, and inflammatory foci were graded by the NAFLD activity score (NAS; Kleiner et al. 2005). Immunohistochemical staining was performed with antibodies against GST-P, Ki-67, proliferating cell nuclear antigen (PCNA), active caspase-3, and p22phox listed in Online Supplemental Table 2. The deparaffinized liver sections were treated with 0.3% H2O2 in methanol for 30 min, and then antigen retrieval was performed if necessary (Fujii et al. 2013; Hara et al. 2014; Yoshida et al. 2017). The liver sections were incubated with primary antibodies overnight at 4°C. An avidin–biotin–peroxidase complex method with VECTASTAIN® Elite ABC Kit (Vector Laboratories Inc., Burlingame, CA) was then employed with 3,3’-diaminobenzidine as a chromogen followed by light hematoxylin staining. The numbers and areas of GST-P-positive foci (>0.2-mm diameter) and the total areas of the liver sections were quantified using Scion Image (Scion Corp., Frederick, MD; Hara et al. 2014). Ki-67, PCNA, active-caspase-3, or p22phox-positive cells were examined in a total of more than 1,000 cells with GST-P-positive foci per animal. In PCNA staining, cells in G1, S, G2, and M phases of the cell cycle were differentially counted, and the total number of cells was calculated (Foley et al. 1993).
Statistical Analysis
All data were expressed as mean + standard deviation. Significance of the differences between the control and the treated group(s) was determined the Tukey’s or Steel–Dwass multiple comparison test. A p value < .05 was considered statistically significant.
Results
SR Alone or in Combination with AGIQ Decreases Body Weight in HFD-fed Rats
There were no treatment-related clinical signs in any group during the study, except for rough fur in the SR group at a higher dose level, as described in Materials and Method section. Final body weight was significantly higher in the HFD group than in the CTL group (Table 1). Treatment with SR alone or in combination with AGIQ significantly decreased body weights as compared to the HFD group. Food and water consumption tended to be lower in the HFD group than in the CTL group. A significant decrease in food consumption was detected in the SR group.
Final Body Weight, Organ Weight, Food and Water Intake in Rats Treated with SR with or without AGIQ.a
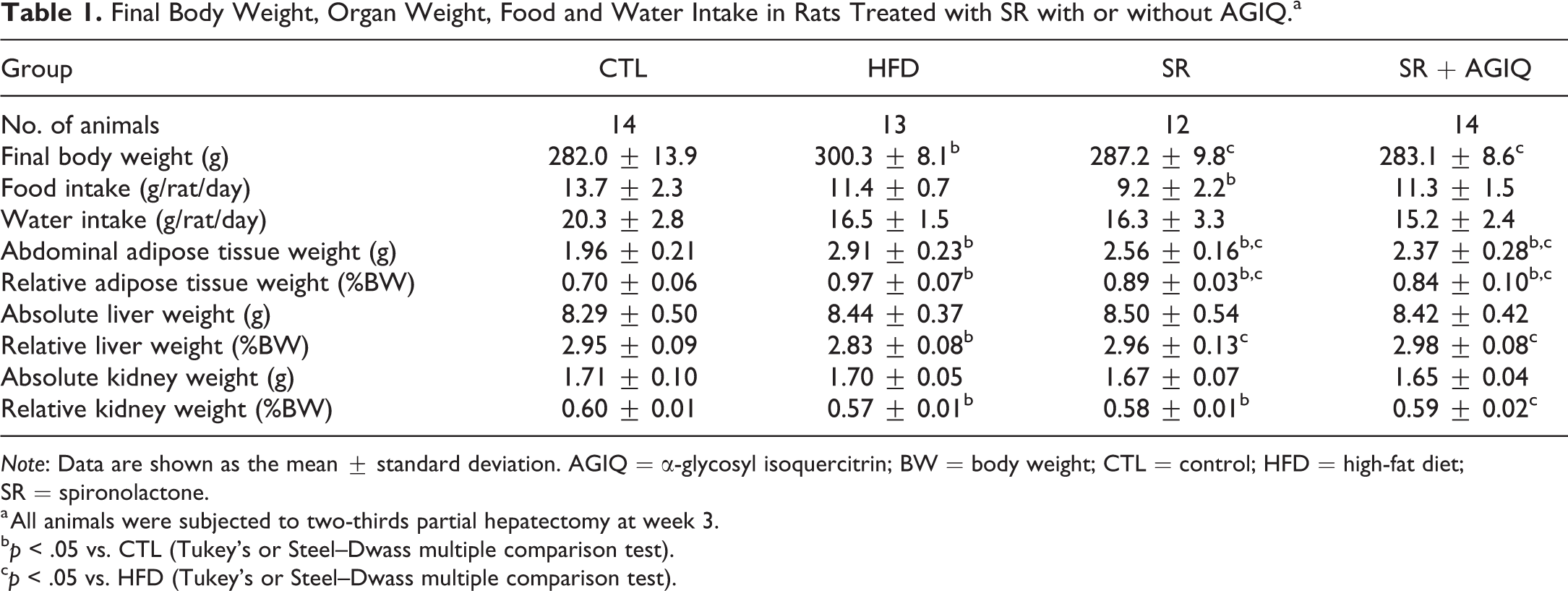
Note: Data are shown as the mean ± standard deviation. AGIQ = α-glycosyl isoquercitrin; BW = body weight; CTL = control; HFD = high-fat diet; SR = spironolactone.
a All animals were subjected to two-thirds partial hepatectomy at week 3.
b p < .05 vs. CTL (Tukey’s or Steel–Dwass multiple comparison test).
c p < .05 vs. HFD (Tukey’s or Steel–Dwass multiple comparison test).
Treatment of SR Alone or in Combination with AGIQ Decreases the Abdominal Adipose Tissue Weight in HFD-fed Rats
Absolute and relative abdominal adipose tissue weights were significantly higher in the HFD group than in the CTL group with or without the treatment of SR alone or in combination with AGIQ (Table 1). Treatment with SR alone or in combination with AGIQ significantly decreased the abdominal adipose tissue weights as compared to the HFD group. The relative liver weights were significantly lower in the HFD group than in the CTL group but were significantly higher in rats treated with SR alone or in combination with AGIQ than the HFD group. The relative kidney weights were significantly lower in the HFD group than in the CTL group with or without SR treatment. Cotreatment with SR and AGIQ significantly increased the relative kidney weight as compared to the HFD group. No significant changes were detected in the absolute weights of the liver and kidney among the groups.
Treatment of SR Alone or in Combination with AGIQ Attenuates Hyperlipidemia in HFD-fed Rats
Among the liver-related parameters, AST was significantly lower in the HFD group than the CTL group, with or without treatment with SR alone or in combination with AGIQ (Figure 1A). A similar effect was detected in ALT; however, there were no significant differences between the groups (data not shown). ALP was significantly increased in the HFD group as compared to the CTL group and was significantly decreased by the treatment with SR in combination with AGIQ (Figure 1B). TG was significantly higher in the HFD group with or without the treatment with SR alone or in combination with AGIQ than in the CTL group but was significantly attenuated by the cotreatment of SR and AGIQ (Figure 1C). T.CHOL tended to increase in the HFD group as compared to the CTL group; this was not a significant change but was significantly decreased by the treatment with SR alone or in combination with AGIQ as compared to the HFD group (Figure 1D).
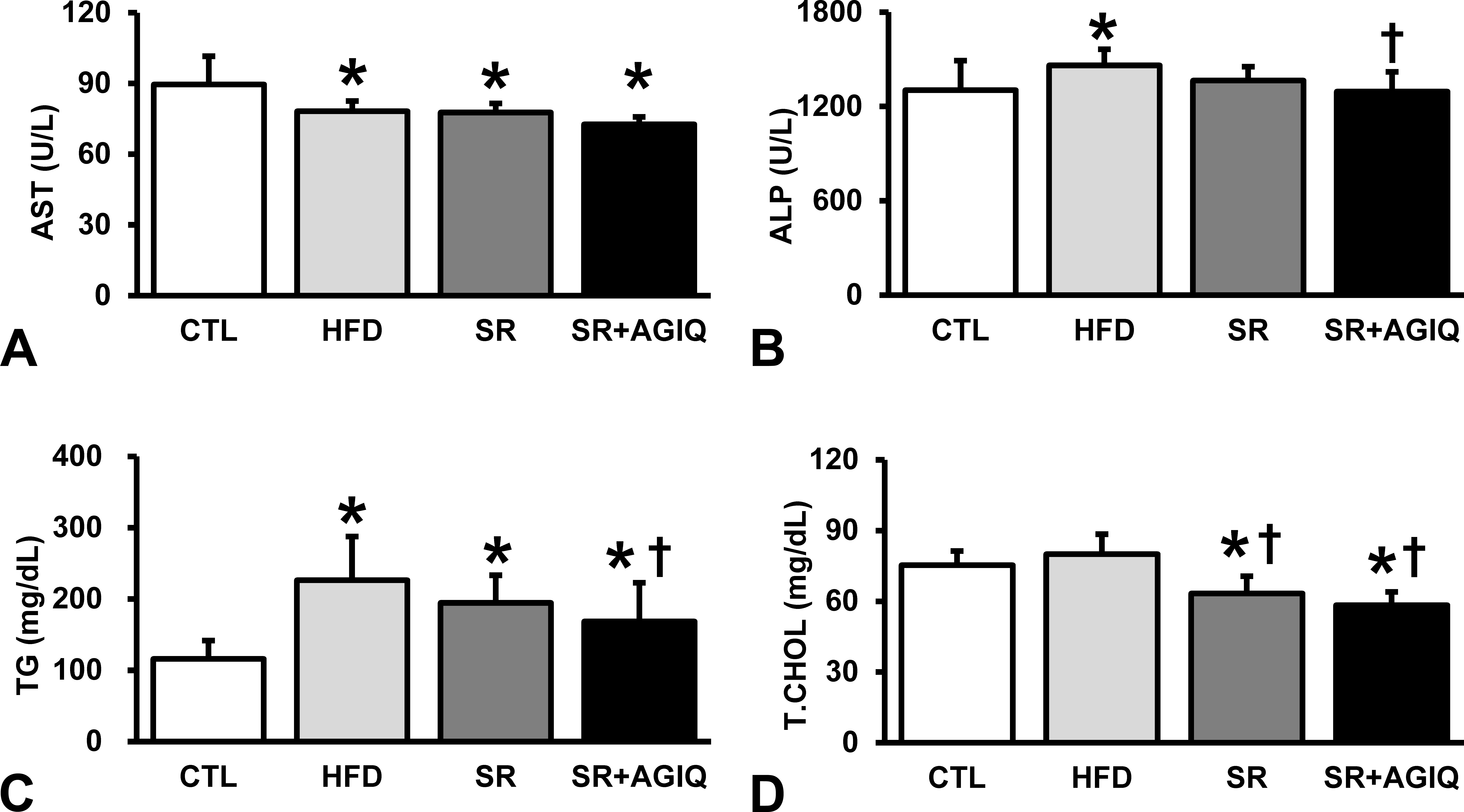
Blood biochemistry in plasma samples of rats after N-diethylnitrosamine initiation followed by basal diet (CTL) or high-fat diet (HFD) with or without cotreatment with spironolactone alone (SR) or SR in combination with α-glycosyl isoquercitrin (AGIQ). (A) Aspartate aminotransaminase (AST). (B) Alkaline phosphatase (ALP). (C) Triglycerides (TG). (D) Total cholesterol (T.CHOL). Columns represent mean + standard deviations. *Significantly different between the CTL group and HFD, SR or SR+AGIQ group, p < .05; †Significantly different between the HFD group and SR or SR + AGIQ group, p < .05 (Tukey’s or Steel–Dwass multiple comparison test). CTL = control.
Treatment of SR Alone or in Combination with AGIQ Alters the mRNA Levels of Oxidative Stress and Antioxidant Enzymes, and Lipogenic Enzymes and Transcriptional Factors
The p67phox mRNA levels in the HFD group were similar to the levels in the CTL group but were significantly decreased upon treatment with SR with or without AGIQ (Table 2). Catalase expression was significantly increased by SR treatment in combination with AGIQ as compared to the CTL and HFD groups. Pparg mRNA levels were significantly increased due to treatment with SR alone or in combination with AGIQ as compared to the CTL group; a significant increase was also detected in the SR and AGIQ cotreatment group as compared to the HFD and SR only groups. Fasn and Scd1 mRNA levels were significantly decreased by the treatment with SR alone or in combination with AGIQ. There were no significant changes in the other mRNA levels.
Gene Expression Analysis in Rats Treated with SR with or without AGIQ.a
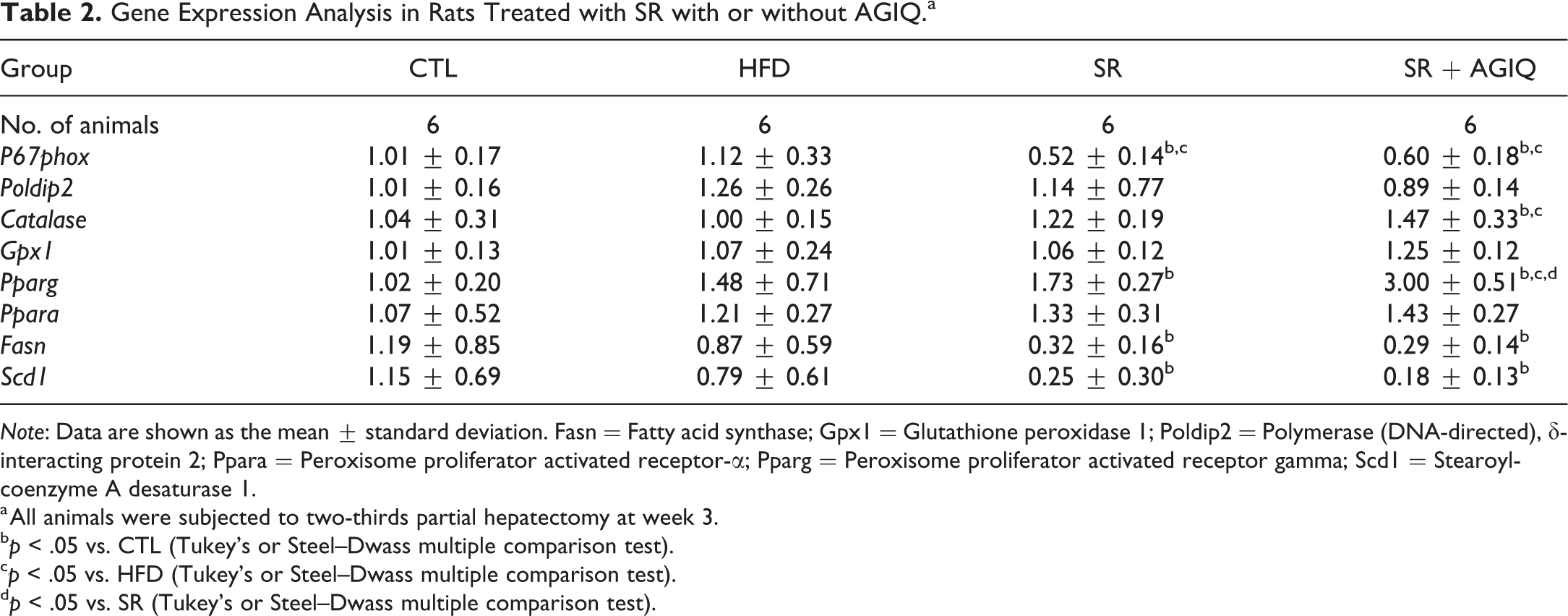
Note: Data are shown as the mean ± standard deviation. Fasn = Fatty acid synthase; Gpx1 = Glutathione peroxidase 1; Poldip2 = Polymerase (DNA-directed), δ-interacting protein 2; Ppara = Peroxisome proliferator activated receptor-α; Pparg = Peroxisome proliferator activated receptor gamma; Scd1 = Stearoyl-coenzyme A desaturase 1.
a All animals were subjected to two-thirds partial hepatectomy at week 3.
b p < .05 vs. CTL (Tukey’s or Steel–Dwass multiple comparison test).
c p < .05 vs. HFD (Tukey’s or Steel–Dwass multiple comparison test).
d p < .05 vs. SR (Tukey’s or Steel–Dwass multiple comparison test).
Treatment with SR Alone or in Combination with AGIQ Reduces Steatosis and Preneoplastic Liver Lesions in HFD-fed Rats
Slight to moderate hepatocellular steatosis and ballooning were observed in the liver of rats fed HFD (Figures 2A and 2B). Inflammatory foci were observed in the liver in all groups. Scoring of NAS revealed a significant increase in the HFD group as compared to the CTL group, and the effect was significantly decreased by treatment with SR in combination with AGIQ as compared to the HFD group (Figures 2C-E [E on following page]).
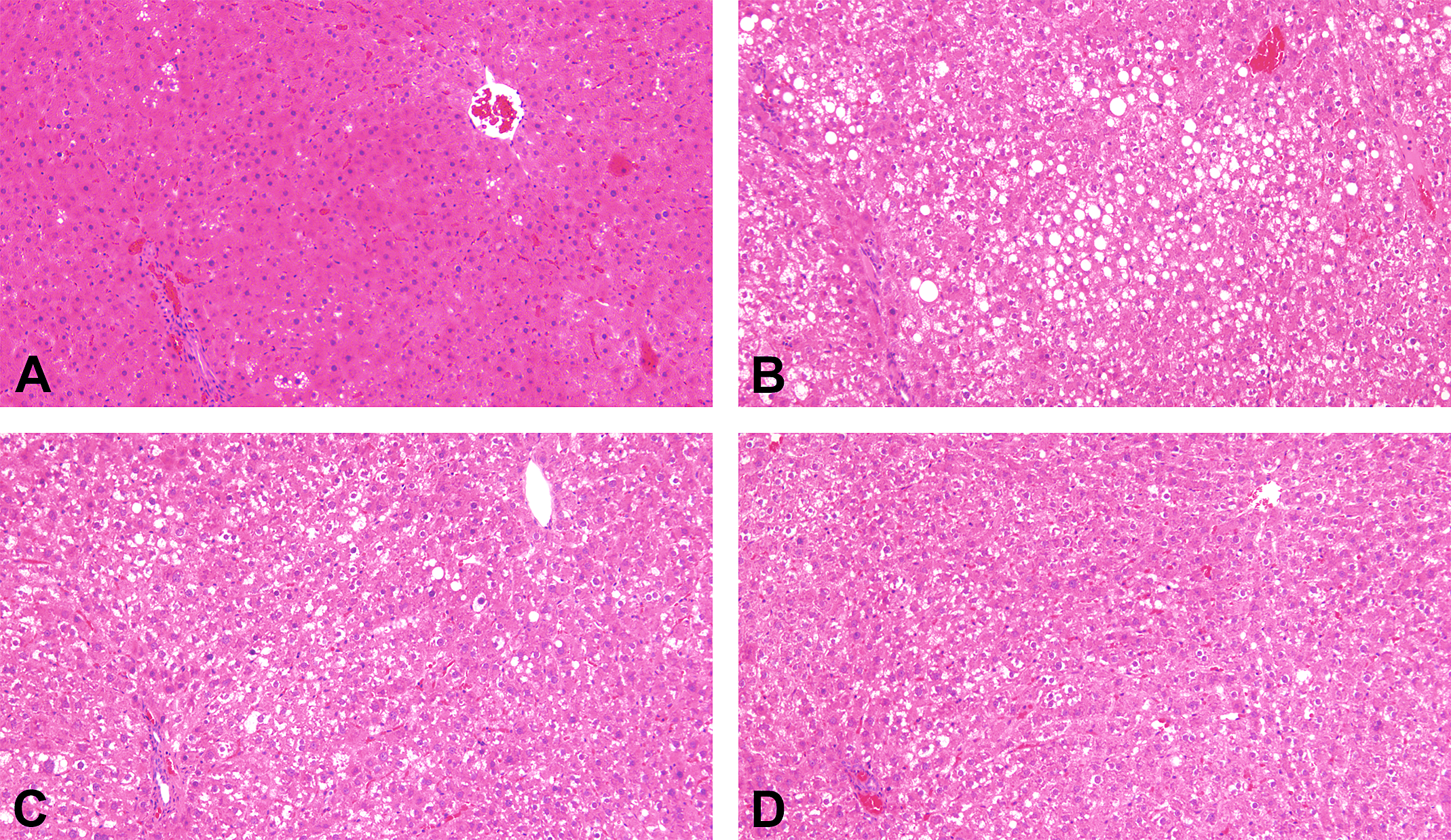
Nonalcoholic fatty liver disease (NAFLD) activity score and representative images of steatosis of hepatocytes in the liver. Rats were treated as described in Figure 1. Representative images of liver lesions in the CTL (A), HFD (B), SR (C), and SR + AGIQ groups (D): (A) normal liver, (B) steatosis of hepatocytes, (C, D) reduction of steatosis compared with the HFD group, (E) the NAFLD activity score (NAS) graded for steatotic cells, ballooning cells, and inflammatory foci. Columns represent mean + standard deviation. *Significantly different between the CTL group and HFD group, p < .05; †Significantly different between the HFD group and SR + AGIQ group, p < .05 (Tukey’s or Steel–Dwass multiple comparison test). AGIQ = α-glycosyl isoquercitrin, CTL = control, HFD = high-fat diet, SR = spironolactone.
The number and area of GST-P-positive liver foci were significantly higher in the HFD group than the CTL group (Figures 3A, C; 4A, B). The area of GST-P-positive foci was significantly decreased by treatment with SR in combination with AGIQ. Ki-67 labeling index in the liver foci was significantly higher in the HFD group, and SR and AGIQ cotreatment group than in the CTL group (Figure 4C). A similar effect on cell proliferation was shown in PCNA labeling index. The total number of PCNA-positive cells and cells in G1, G2, and M phases were significantly higher in the HFD group with or without SR alone or in combination with AGIQ (data not shown). Cells in S phase were higher in the HFD group and were significantly lower in the treatment group of SR (Figure 4D). Active caspase-3 labeling index in the liver foci was significantly higher in the SR and AGIQ cotreatment group than the other groups (Figure 4E). p22phox, a component of NOX1-4, was expressed in the cell membrane and cytoplasm of hepatocytes inside and outside foci (Figure 3B, D). The labeling index of p22phox was significantly higher in the HFD group than in the CTL group but was significantly decreased by treatment with SR alone or in combination with AGIQ (Figure 4F).
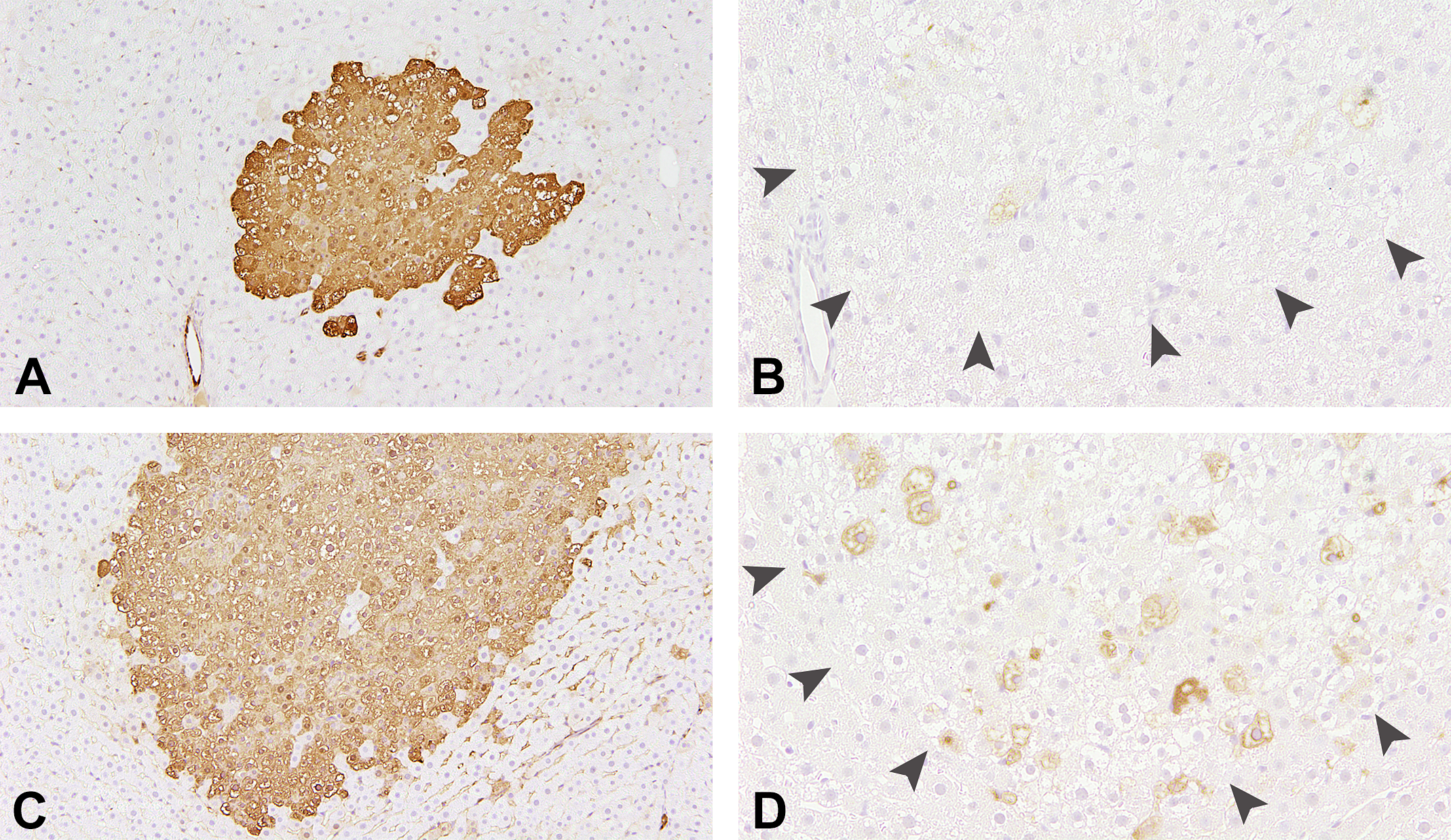
Representative images of glutathione S-transferase placental (GST-P)-positive foci with p22phox-positive cells. GST-P-positive foci are larger in the HFD group (B) than in the CTL group (A). A higher number of 22phox-positive cells is observed in the foci in the HFD group (D) compared with the CTL group (C). CTL = control, HFD = high-fat diet.
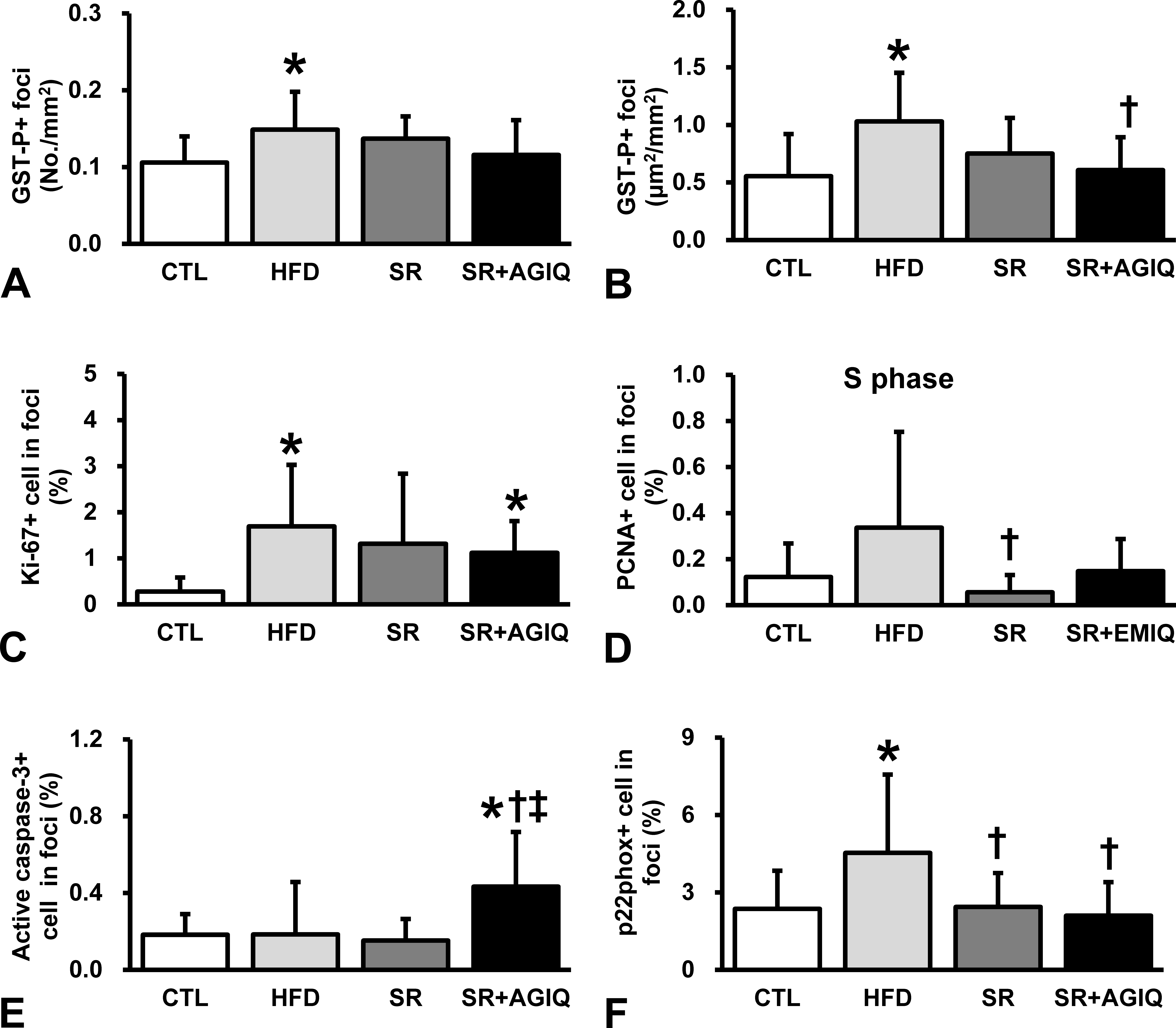
Quantitative analysis of glutathione S-transferase placental (GST-P)-positive foci with Ki-67-positive cells, PCNA-positive cells, active caspase-3-positive cells, and p22phox-positive cells in the liver. Rats were treated as described in Figure 1. The number (A) and area (B) of GST-P-positive foci, and quantitative data (%) of Ki-67-positive cells (C), PCNA-positive S phase cells (D), active caspase-3-positive cells (E), and p22phox-positive cells (F) in GST-P-positive foci. Columns represent mean + standard deviation. *Significantly different between the CTL group and HFD or SR+AGIQ group, p < 05; †significantly different between the HFD group and SR or SR + AGIQ group, p < .05; ‡significantly different between the SR group and SR + AGIQ group, p < .05 (Tukey’s or Steel–Dwass multiple comparison test). AGIQ = α-glycosyl isoquercitrin, CTL = control, HFD = high-fat diet, PCNA = proliferating cell nuclear antigen, SR = spironolactone.
Discussion
Delineating the mechanism(s) of steatosis-related HCC and identifying therapeutic targets are important steps toward improving the health of patients with NAFLD or NASH. To this end, we first established a model for evaluating steatosis-related early hepatocarcinogenesis using a medium-term liver assay in rats fed HFD. The medium-term (8 week) liver assay developed by Ito, Tamano, and Shirai (2003) has been used for evaluating a variety of genotoxic or nongenotoxic hepatocarcinogens and nonhepatocarcinogens using a critical marker of precancerous lesions, GST-P-positive foci in liver sections. This assay would also be valuable for identifying the candidate chemopreventive drugs against liver disorders. As steatosis is closely related to the development of liver cancer in humans (Sheedfar et al. 2013), we hypothesized that an HFD was capable of enhancing liver cancer promotion in this well-established 2-stage (initiation and promotion) liver model. We observed that an HFD increased GST-P-positive liver foci, along with hyperlipidemia (i.e., increased TG) and steatosis. Hepatocellular damage (i.e., increased ALT and AST) with increased liver weight was not evident in the present protocol, probably dependent on the duration of study period. Decreased plasma levels of ALT and AST in the HFD group might be caused by altered liver metabolism including a reduction of cofactor (pyridoxal 5 phosphate) of the liver enzymes (Hall et al. 2012). Previous HFD studies demonstrated that ALP was increased in accordance with higher levels of ALT and AST, and the changes might be related to damage, steatosis, and inflammation in the liver (Vedanarayanan and Krishnan 2011). However, only ALP was increased in the HFD group in the present study, and the finding might be related to jejunal ALP activity induced by the HFD (Lallès et al. 2012). The data on the precancerous lesions were consistent with previous findings that HFD increased GST-P-positive liver foci in Sprague-Dawley rats initiated by DEN administered via an intraperitoneal injection (Wang et al. 2009) or through drinking water (Kumamoto et al. 2013). The liver-promoting effects by an HFD are considered to be mediated by the activation of tumor necrosis factor (TNF)/nuclear factor-κB (Nf-κB) and extracellular signal-regulated kinase signaling (Wang et al. 2009) or oxidative damage, that is, an increase in 8-hydroxydeoxyguanine (Kumamoto et al. 2013) in liver tissues. In the present study, we confirmed the higher expression of the NOX subunit, p22phox as well as cell proliferation markers, Ki-67 and PCNA, in GST-P-positive liver foci in HFD-fed rats. These findings suggest that NOX-derived ROS might enhance cell proliferation in precancerous lesions, subsequently leading to liver cancer development. As NOX plays a critical role in liver abnormalities in NAFLD and NASH in humans (Dattaroy et al. 2015) and experimental animals (Janevski et al. 2011; Chatterjee et al. 2013), our data indicate that the model we proposed here is useful to evaluate NOX-related liver cancer promotion as well.
In this study, we found that the MR antagonist SR inhibited the expression of p67phox mRNA in liver tissues of HFD-fed rats. Further, SR significantly reduced HFD-induced p22phox-expressing hepatocytes in preneoplastic GST-P-positive foci. These results suggested that SR has a potential of inhibiting expression of NOX subunits in liver tissues with steatosis. The effect of SR on hyperlipidemia, but not steatosis and preneoplastic foci, was evident in the present study. An MR antagonist eplerenone can inhibit experimental NASH and hyperlipidemia in mice fed an HFD and a high-fructose diet (Wada et al. 2013) or choline-deficient and amino acid–defined diet (Pizarro et al. 2015). This compound effectively attenuated hepatic TNF production, thereby ameliorating steatosis, fibrosis, and insulin resistance (Wada et al. 2013), although expression levels of lipogenic and NOX genes were not examined. Hepatic gene expression analysis revealed that SR treatment reduced mRNA levels of lipogenic enzymes (Scd1 and Fasn) that regulate elongation or desaturation of fatty acids (Softic, Cohen, and Kahn 2016) as compared to the CTL group. A previous study suggested that SR could downregulate a crucial transcriptional factor involved in lipogenesis, carbohydrate response element binding protein, followed by decreased expression of downstream Scd1 and Fasn (Wada et al. 2010). Interestingly, by analyzing PCNA-positive cells in different phases of cell cycle, we confirmed that SR significantly inhibited HFD-increased S phase cells. This finding was supported by evidence that SR can directly reduce cell number of cancer cell through K-RAS suppression, which may be regulated by MR (King et al. 2014). Taken together, SR alone might ameliorate HFD-induced hyperlipidemia and proliferative activity in the foci through the modulation of intrahepatic lipogenic and NOX cascades.
The present experiment demonstrated that SR in combination with AGIQ significantly reduced HFD-increased NAS in liver sections. The effect of AGIQ on steatosis has not been fully understood to our knowledge. AGIQ is readily soluble in water, is mostly deglycosylated before absorption, and it likely exists as quercetin and/or quercetin glucuronide in humans (Makino et al. 2009) and rats (Nyska et al. 2016). Our findings are supported by previous reports in which administration of quercetin reduced hepatic lipid accumulation together with inflammation and portal fibrosis in diet-mediated obese animals through reduced expression of inflammation and lipid metabolism-related genes (Kobori et al. 2011; Marcolin et al. 2012; Pisonero-Vaquero et al. 2015). Hepatic gene expression analysis revealed that SR in combination with AGIQ treatment significantly increased mRNA levels of Pparg, which is the transcriptional factor regulating lipid transporter genes. Ppar-γ might attenuate hepatic inflammation and tumor induction because Pparg knockout mice induce hepatic inflammation through the inhibition of NF-κB and COX1 (Wang et al. 2017) and increase HCC through the inhibition of apoptosis (Yu et al. 2010). We also demonstrated that AGIQ in the presence of SR increased apoptosis in the preneoplastic GST-P-positive foci. The effect on apoptosis was probably mediated by AGIQ because SR alone did not increase apoptosis in the foci. A similar enhanced effect of AGIQ on apoptosis was reported in a medium-term liver bioassay of rats (Hara et al. 2014). Quercetin or quercetin glucuronide induced apoptosis by the activation of caspase-3 in a human hepatoma cell line (Granado-Serrano et al. 2006; Sudan and Rupasinghe 2014) and could sensitize TNF-related apoptosis-inducing ligand (TRAIL)-resistant HCC cells to TRAIL-induced apoptosis (Kim et al. 2008). As we did not observe the effects of AGIQ alone on the pathological changes in the present study, this limitation will be overcome by our additional study using this animal model.
The mechanism of combined effects of SR and AGIQ on chemoprevention toward early hepatocarcinogenesis might involve NOX-related pathways, since the combined treatment with SR and AGIQ reduced expression of p22phox in the liver foci. SR inhibits expression of NOX components RAC1 and NOX4 by modulating MR in a normal rat kidney cell line NRK-52E (Tsuji et al. 2016), while isoquercitrin is also known to inhibit overexpression and phosphorylation of p47phox in RAW249.7 cells and endothelial cells (Kim et al. 2013; Jones et al. 2016), respectively. Indeed, the effects of the combined treatment with SR and AGIQ might be related to oxidative stress, since both the treatment increased expression antioxidant gene, Catalase. Therefore, the combined treatment might provide synergistic inhibition of NOX-related oxidative stress in liver tissues and thereby mitigate the development of NAFLD-related early hepatocarcinogenesis.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623318778508 - Spironolactone in Combination with α-glycosyl Isoquercitrin Prevents Steatosis-related Early Hepatocarcinogenesis in Rats through the Observed NADPH Oxidase Modulation
Supplemental Material, DS1_TPX_10.1177_0192623318778508 for Spironolactone in Combination with α-glycosyl Isoquercitrin Prevents Steatosis-related Early Hepatocarcinogenesis in Rats through the Observed NADPH Oxidase Modulation by Hirotada Murayama, Ayumi Eguchi, Misato Nakamura, Masahi Kawashima, Rei Nagahara, Sayaka Mizukami, Masayuki Kimura, Emi Makino, Naofumi Takahashi, Ryoichi Ohtsuka, Mihoko Koyanagi, Shim-mo Hayashi, Robert R. Maronpot, Makoto Shibutani, and Toshinori Yoshida in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to thank Mrs. Shigeko Suzuki for her technical assistance in preparing the histological specimens.
Author Contributions
Authors contributed to conception or design (HM, TY, AE, MN, MKaw, RN, SM, MKim, EM, NT, RO, MKay, MH, RM, MS); data acquisition, analysis, or interpretation (HM, TY, AE, MN, MKaw, RN, SM, MKim, EM, NT, RO, MKay, MH, RM, MS); drafting the manuscript (HM, TY, AE, MN, MKaw, RN, SM, MKim, EM, NT, RO, MKay, MH, RM, MS); and critically revising the manuscript (TY, RM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Food Chemical Research Foundation.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
