Abstract
We previously demonstrated that high-fat diet (HFD)–induced hepatic lipid accumulation is more severe in BALB/c mice than in C57BL/6J (B6) mice. To understand the changes in liver metabolism, we studied blood chemistry, gene expression, and histopathological changes of the liver in nine-week HFD-fed BALB/c and B6 mice and one- or four-week HFD-fed BALB/c mice. Serum total cholesterol and triglyceride levels were significantly increased in all HFD-fed groups, and one- and four-week HFD-fed BALB/c groups, respectively. Histopathology revealed that vacuolation of hepatocytes was severe in nine-week HFD-fed BALB/c mice, although it was less severe in the other groups. Microarray analysis of mRNA expression of nine-week HFD-fed BALB/c mice showed up-regulation of genes involved in fatty acid uptake and biosynthesis, such as Cd36, Acaca, Acly, and Fasn. Some changes were observed in the one- and four-week HFD-fed BALB/c groups and the nine-week HFD-fed B6 group, however these changes in mRNA expression were not so marked. In conclusion, the fatty accumulation observed in BALB/c mice may be caused, at least in part, by up-regulation of fatty acid uptake and biosynthesis. Cd36, Acaca, Acly and Fasn may be involved in these metabolic processes.
Introduction
Hepatic lipid metabolism is influenced by the balance between the degradation and synthesis and/or import and export of triacylglycerol (TAG) and fatty acids. Disruption of this balance causes hepatic steatosis.
In laboratory animals, many models of hepatic steatosis have been created. Among genetically modified animal models, acyl-coenzyme A oxidase null mice, methionine adenosyltransferase-1A null mice, nuclear respiratory factor 1 knockout mice, phosphatase and tensin homolog deleted on chromosome 10 null mice, ob/ob mice, and db/db mice have been reported (Fan and Qiao 2009). Among dietary mouse models, there are methionine- and choline-deficient diet–induced and high-fat diet–induced models (Fan and Qiao 2009). Moreover, combined mouse models induced by the combination of genetic modification and nutritional/dietary challenges have also been reported (Fan and Qiao 2009). In zebrafish, exposure of larvae to alcohol leads to hepatic steatosis in a similar manner to that observed in humans (Passeri et al. 2009). In neonatal piglets, total parenteral nutrition induces liver steatosis and apoptosis (Wang et al. 2006). There are also ethanol- and protein-deficient diet–induced steatotic liver models in pigs (Spannbaure et al. 2005). In particular, in view of their usability and the similarity to human diet–induced steatosis, high-fat diet (HFD)–induced rodent models are useful tools for characterizing the pathophysiological and molecular mechanisms involved in the progression of hepatic steatosis.
West et al. reported that C57BL/6J (B6) mice are a particularly obesity-prone strain among nine different inbred mouse strains examined (West et al. 1992). B6 mice have been used in many studies as obesity and steatosis models, whereas A/J mice have been used as an obesity-resistant mouse strain in some reports.
In fatty liver disease, various potential candidate genes have been reported, including sterol regulatory element binding factor (Srebf) 1c, stearoyl CoA desaturase-1 (Scd1), microsomal triglyceride transfer protein (Mttp), phosphatidylethanolamine methyltransferase, and peroxisome proliferator-activated receptor (PPAR)-α (Wilfred de Alwis and Day 2007). Several studies analyzing gene expression in animal models of HFD-induced hepatic steatosis have been reported. However, many of these reports examined only one time-point or did not include any histopathology.
We previously observed that HFD-induced hepatic lipid accumulation is more severe in BALB/c mice than in B6 mice (Nishikawa et al. 2007; Nishikawa et al. 2008). In this study we analyzed changes in gene expression and histopathological patterns as well as important metabolic changes underlying the development of hepatic steatosis.
Materials and Methods
One- and Four-Week-Fed BALB/c Groups
Animals
Nine-week-old and twelve-week-old male BALB/cAJcl mice were used for these experiments. The animals, purchased from CLEA Japan, were housed individually in plastic mouse cages and maintained under controlled conditions (twelve-hour light:dark cycle, temperature of 23°C ± 3°C, relative humidity of 50% ± 20%). For acclimatization, mice were fed a standard rodent chow (CE-2, CLEA Japan, Tokyo, Japan), with ad libitum access to water for one week. After this period, the ten-week-old mice were fed either High Fat Diet 32 (HFD32, CLEA Japan), or CE-2 for four weeks. The thirteen-week-old mice were fed in the same way for one week. The composition of each of the two diets is shown in Table 1 . At the end of the experiment, all the mice were fourteen weeks old. They were fasted for twenty hours and were then exsanguinated from the caudal vena cava under ether anesthesia, and the liver and peri-epididymal fat tissues were collected and weighed. The left lateral lobe of the liver was fixed in 10% neutral buffered formalin for histopathology, whereas the right and caudate lobes were stored at –80°C for RNA extraction. All these procedures were performed according to the “Rules for Feeding and Storage of Experimental Animals and Animal Experiments” and were approved, from the point of view of animal welfare, by the Institutional Animal Care and Use Committee of the testing facility.
Diet composition.
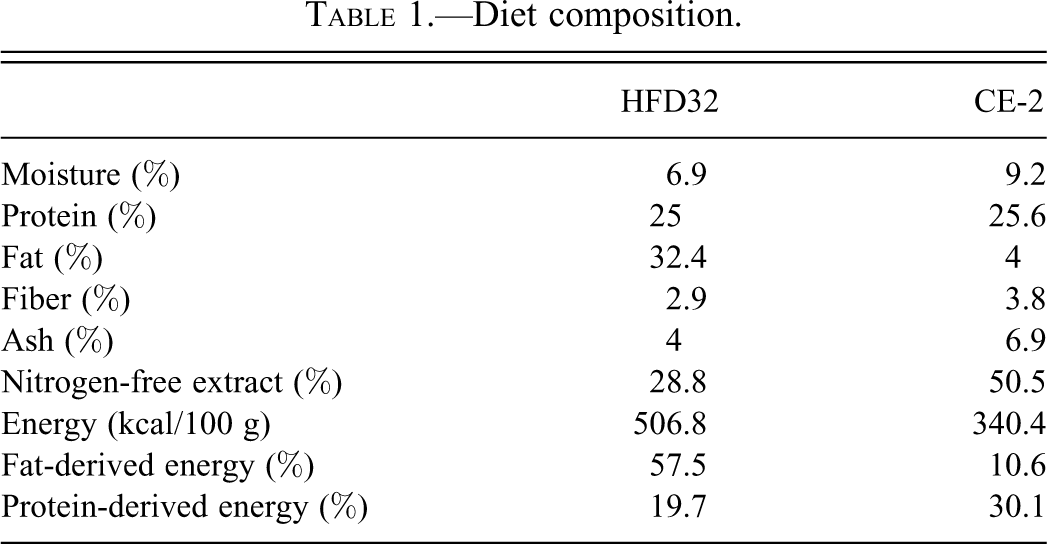
Blood Biochemistry
Serum samples were prepared from the blood collected through the caudal vena cava by centrifugation at 2,000–3,000 rpm for ten minutes. Serum TAG, total cholesterol (Cho), glucose, and phospholipid levels were determined using a Hitachi 7180 autoanalyzer (Hitachi High-Technologies Corp., Tokyo, Japan).
Nine-Week–Fed BALB/c and B6 Groups
The method used has been previously described in detail (Nishikawa et al. 2008). Briefly, five-week-old male BALB/cAJcl and C57BL/6JJcl mice were fed a CE-2 or HFD32 diet for nine weeks. At the end of the experiment, mice were fasted for twenty hours, then blood, liver, and peri-epididymal fat tissues were collected. The left lateral lobe of the liver was fixed in 10% neutral buffered formalin for histopathology, whereas the right and caudate lobes were stored at –80°C for RNA extraction. Serum Cho, TAG, and plasma glucose were measured using commercially available kits (Cholesterol E-test Wako, Triglyceride E-test Wako, and Glucose CII-test Wako; all from Wako, Osaka, Japan). All procedures involving mice were conducted according to the guidelines for the care and use of laboratory animals approved by the Graduate School of Agricultural and Life Sciences of Tokyo University. The following methods were used in the one- or four-week–fed BALB/c groups and the nine-week–fed BALB/c and B6 groups.
Histopathological Examination
The liver samples fixed in 10% neutral buffered formalin were dehydrated and embedded in paraffin. Four-µm-thick paraffin sections were stained with hematoxylin and eosin.
Calculation of Energy Intake
Dietary intake was obtained by subtracting the weight of the remaining feed from the weight of the feed initially supplied. Energy intake was calculated on the basis of energy values of 3.404 kcal/g and 5.068 kcal/g for the CE-2 and the HFD feed, respectively.
Statistical Analysis
Results are presented as mean ± standard deviation (SD). The Student t test was used to compare HFD-fed groups with control groups.
Isolation of Total RNA
Each liver sample of approximately 30 mg was lysed in 600 µL of RLT buffer (QIAGEN Co. Ltd., Tokyo, Japan) using a Mixer Mill (QIAGEN). Total RNA was extracted from the liver homogenate using RNeasy Plus (QIAGEN) according to the manufacturer’s protocol. The ratio of absorbance at 260 nm and 280 nm (A260/A280) was calculated for each RNA sample from measurements taken using a NanoDrop ND-1000 spectrophotometer (Isogen, Maarssen, The Netherlands). The integrity of total RNA was checked by the RNA integrity number generated by a 2100 Bioanalyzer (Agilent Technologies, Amsterdam, The Netherlands) with 6000 Nano Chips according to the manufacturer’s instructions. Since the A260/A280 and RNA integrity numbers were all above 1.8 and 7, respectively, this finding confirmed that all the total RNA samples were appropriate for use in the subsequent GeneChip experiment. Two RNA samples from each group were used for hybridization to microarrays.
Preparation of Biotin-Labeled cRNA for Hybridization
Two µg of total RNA was used for the preparation of biotin-labeled cRNA for hybridization. The cRNA was prepared according to the manufacturer’s instructions using the GeneChip Array Station (Affymetrix).
Hybridization and Signal Detection
A GeneChip Mouse Genome 430 2.0 Array (Affymetrix) cartridge was filled with the hybridization cocktail, and then incubated at 45°C overnight. Arrays were then washed and stained in a GeneChip Fluidics Station 450 (Affymetrix) according to Fluidics Protocol FS450_0001, and the hybridization signal was detected by a GeneChip Scanner 3000 (Affymetrix). The scanned images were processed by GeneChip operating software (Affymetrix).
Data Normalization
GeneChip data were normalized using GeneSpring Gx 7.3.1 (Agilent Technologies). After data transformation (set measurements < 0.01–0.1), per-chip normalization was performed to equalize the 50th percentile of each GeneChip (normalize to 50th percentile). After this step, the intensities were normalized for each gene by setting the mean intensity to mean signal of control chips.
Data Analysis
Extraction of Affected Genes
Genes that exhibited changes greater than 1.5-fold were categorized as up-regulated (in the HFD-fed animals, genes that exhibited the “present call” (labeled as present by Affymetrix detection call) were subjected to fold-change analysis. Genes that exhibited changes less than 0.67-fold were categorized as down-regulated (in the CE-2-fed animals, genes that exhibited the “present call” were subjected to fold-change analysis).
Identification of Lipid Metabolism-Related Genes
Based on the KEGG PATHWAY (Kyoto Encyclopedia of Genes and Genomes), the genes related to fatty acid biosynthesis, fatty acid elongation in mitochondria, biosynthesis of unsaturated fatty acids, steroid biosynthesis, terpenoid backbone biosynthesis, glycerolipid metabolism, synthesis and degradation of ketone bodies, primary bile acid biosynthesis, and the PPAR-α signaling pathway were selected from those genes that were differentially expressed. From the genes involved in PPAR-α signaling, those related to adipocyte differentiation, adaptive thermogenesis, cell survival, ubiquitination, and gluconeogenesis were excluded. In addition, important genes involved in fatty acid biosynthesis, ATP-citrate lyase (Acly); lipid influx, low-density lipoprotein receptor (Ldlr), very low-density lipoprotein receptor (Vldlr), and scavenger receptor class B member 1 (Scarb1); lipid efflux, apolipoprotein B (ApoB), Mttp, ATP-binding cassette subfamily A member 1 (Abca1), as well as the transcription factors Srebf1 and Srebf2, were also analyzed.
Results
Results of the nine-week–fed BALB/c and B6 groups and the one- and four-week–fed BALB/c groups are presented together.
Energy Intake, Body Weight, and Organ Weight
The energy intake, body weight, and organ weight of the animals in the study are shown in Table 2 . The total energy intake and body weight of HFD-fed mice were significantly greater than those of control mice in the one-week–fed BALB/c group and in the nine-week–fed BALB/c and B6 groups. In all HFD-fed groups, the energy intake was higher than that of the respective control groups during the first few days of the feeding period followed by a decrease to control levels (data not shown); therefore, the increased energy intake and body weight in the one-week–fed BALB/c group may be transient. The liver weight was increased only in the nine-week–fed BALB/c group, but it was not significant with respect to relative liver weight. The reason for the decrease in relative liver weight in the four-week–fed BALB/c group was not clear. In the nine-week–fed B6 group, the relative liver weight decreased because the liver weight was unchanged and body weight had markedly increased. The peri-epididymal fat weight had increased in all HFD-fed groups. Between the nine-week–fed BALB/c and B6 groups, the fat weight was almost equal.
Energy intake, body weight, and organ weight.
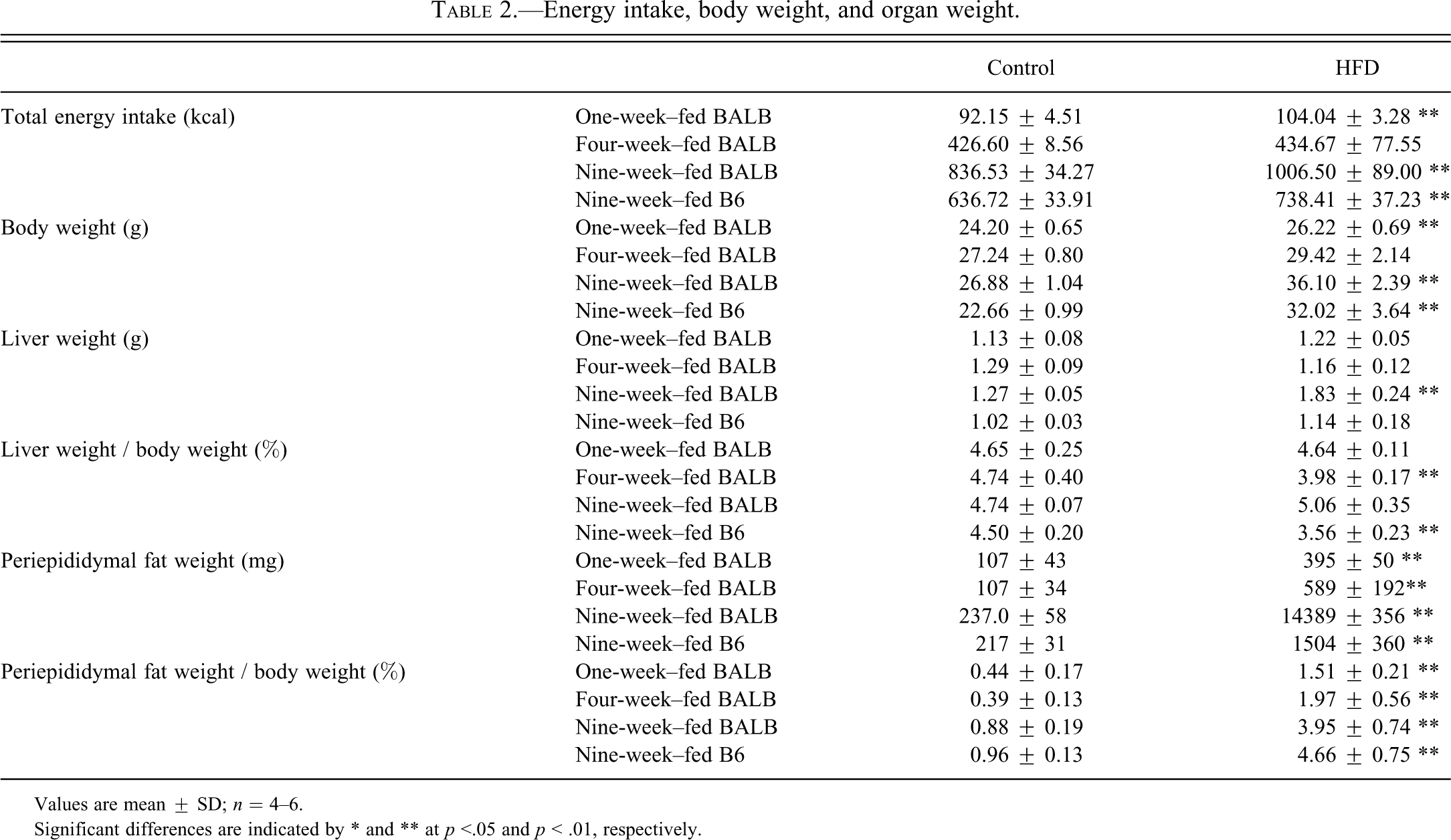
Values are mean ± SD; n = 4–6.
Significant differences are indicated by * and ** at p <.05 and p < .01, respectively.
Blood Biochemistry
As shown in Table 3 , total Cho levels were significantly increased in all HFD-fed groups. The TAG and phospholipid levels in the one- and four-week–fed BALB/c groups and the glucose level in the nine-week–fed BALB/c group were also all significantly higher. For phospholipids, only the one- and four-week–fed BALB/c groups were examined.
Blood chemistry.

-: not examined. Values are means ± SD; n = 4–6.
Significant differences are indicated by * and ** at p < .05 and p < .01, respectively.
Histopathology
In all control (CE-2–fed) groups, no vacuolation of hepatocytes was observed. In the HFD groups, the severity of vacuolation of hepatocytes was moderate in the nine-week–fed BALB/c group, and mild in the one- and four-week–fed BALB/c groups and the nine-week–fed B6 group. In the four-week–fed BALB/c mice and the nine-week–fed BALB/c and B6 mice, macrovacuoles were observed (inset of Figure 1 ).
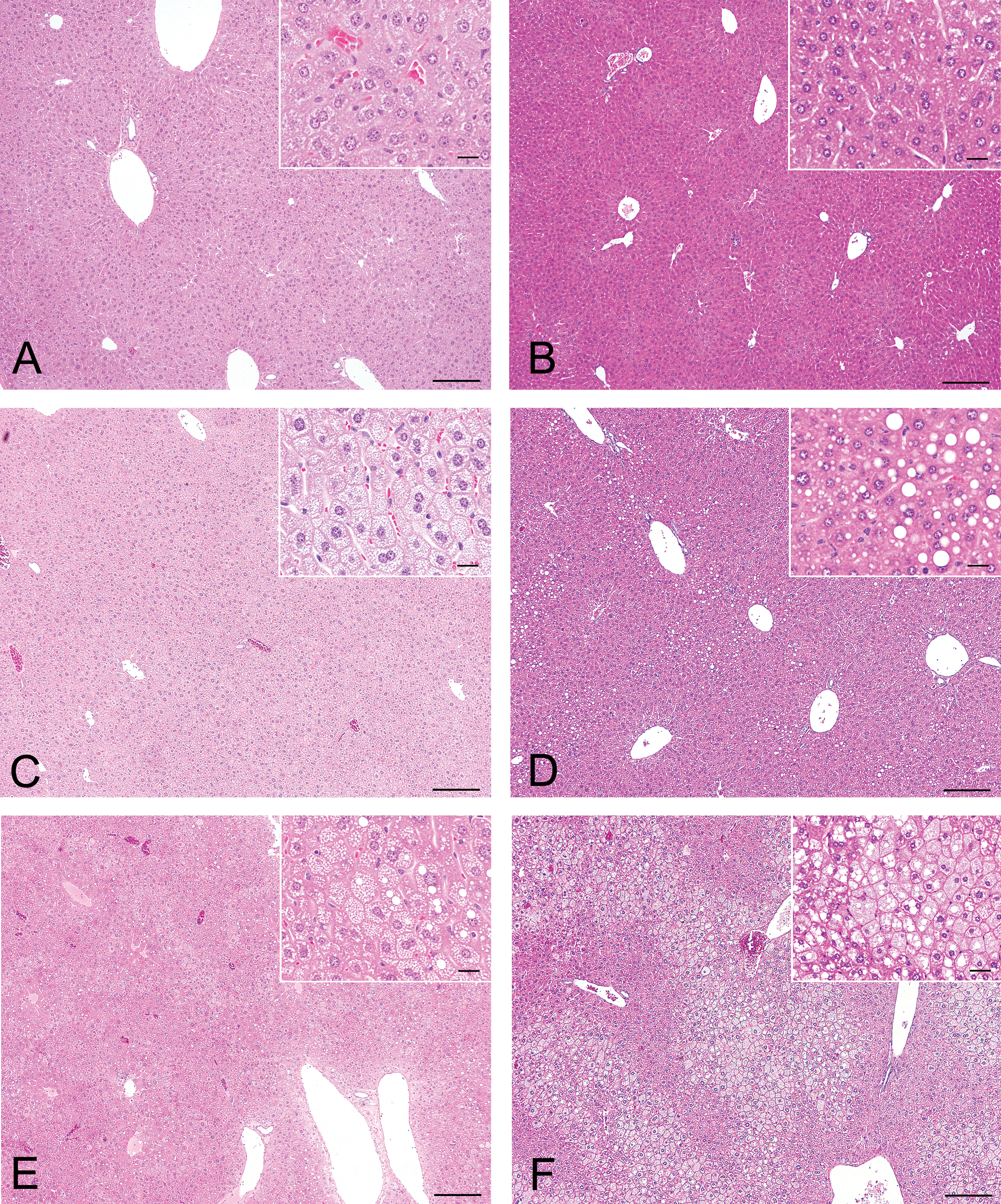
Photomicrographs of hematoxylin and eosin–stained liver sections. Scale bar = 200 μm (20 μm for insert). (A) One-week CE-2–fed BALB/c mice. (B) Nine-week CE-2–fed B6 mice. (C) One-week HFD-fed BALB/c mice. (D) Nine-week HFD-fed B6 mice. (E) Four-week HFD-fed BALB/c mice. (F) Nine-week HFD-fed BALB/c mice. Insets show a higher magnification of the respective figure. Vacuolation of hepatocytes was severe in the nine-week HFD-fed BALB/c mice. In four-week HFD-fed BALB/c mice and nine-week HFD-fed B6 mice, a few macrovacuoles were observed (inset).
Microarray Analysis (Table 4 )
List of genes differentially expressed under the influence of a high-fat diet in one-, four-, and nine-week–fed B6 mice.
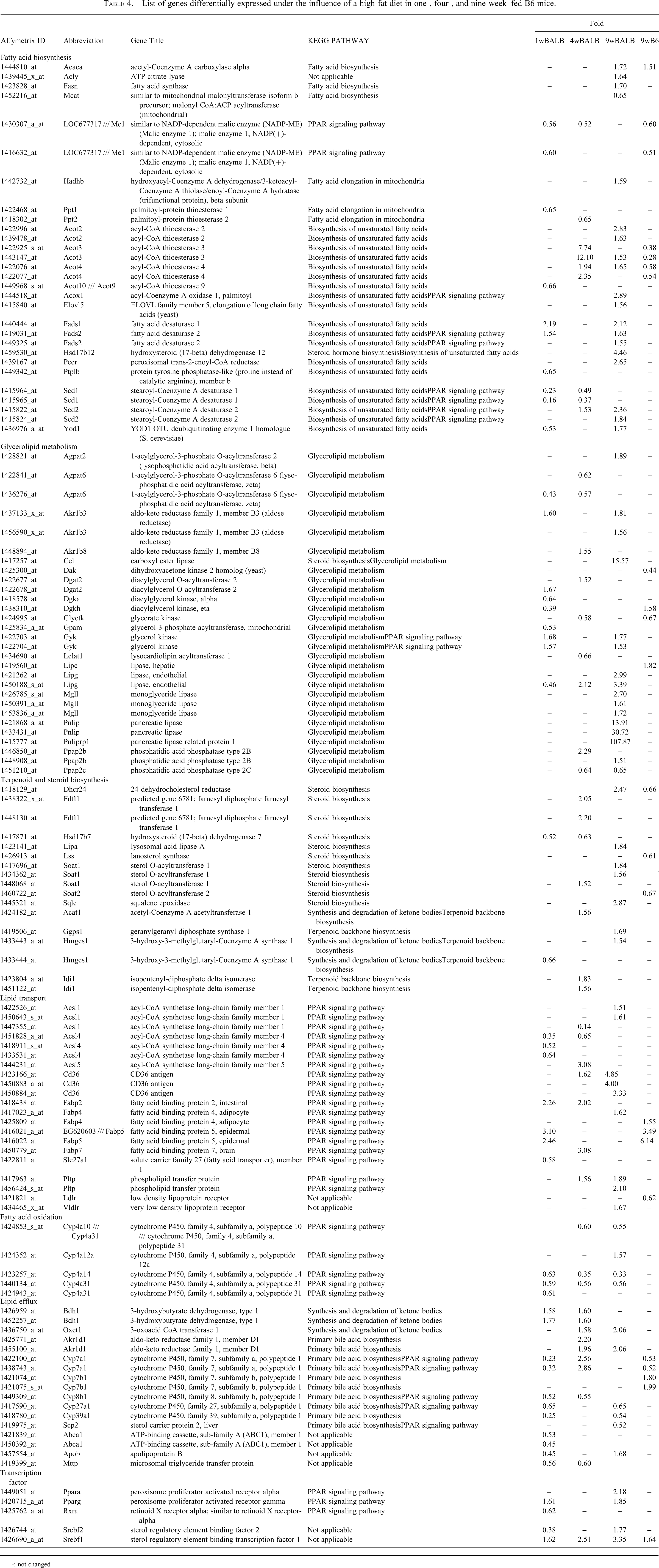
-: not changed
A summary of differentially expressed genes related to lipid metabolism is shown in Table 4. Among the genes related to fatty acid biosynthesis, acetyl-coenzyme A carboxylase α (Acaca), Acly, and fatty acid synthase (Fasn), among others, showed increased expression in the nine-week HFD-fed BALB/c group, suggesting that fatty acid biosynthesis may be activated. Other genes related to fatty acid biosynthesis, including Scd1 and malic enzyme (Me), showed decreased expression in both the one- and four-week HFD-fed BALB/c groups, suggesting that fatty acid biosynthesis may be down-regulated in these groups. In the nine-week HFD-fed B6 group, the expression of some genes was decreased; however, Acaca expression was increased.
In TAG biosynthesis, glycerophosphate acyltransferase, lysophosphatidate acyltransferase, and diacylglycerol acyltransferase may act as regulatory enzymes (Nguyen et al. 2007). As shown in Table 4, 1-acylglycerol-3-phosphate O-acyltransferase (Agpat) 2 and Agpat6, diacylglycerol O-acyltransferase 2 (Dgat2), mitochondrial glycerol-3-phosphate acyltransferase (Gpam), and lysocardiolipin acyltransferase 1 (Lclat1) are the genes encoding the abovementioned enzymes. In the nine-week HFD-fed BALB/c group, although the expression of many genes related to glycerolipid metabolism increased, of the genes related to TAG metabolism, only Agpat2 showed increased expression. In both the one- and four-week HFD-fed BALB/c groups, there were both increases and decreases in the expression of genes involved in TAG biosynthesis.
Many genes related to steroid biosynthesis showed increased expression in both the four- and nine-week HFD-fed BALB/c groups. On the other hand, a few genes showed decreased expression in the one-week HFD-fed BALB/c and the nine-week HFD-fed B6 groups.
Among the lipid transport genes, CD36 antigen (Cd36) and solute carrier family 27 member 1 (Sld27a1) are responsible for the transport of fatty acids. Cd36 expression increased in four-week and nine-week HFD-fed BALB/c groups. In addition, the number of probes increased more in the nine-week than in the four-week HFD-fed BALB/c group. Therefore, fatty acid import in the liver was increased in these groups, and may have been more active in the nine-week HFD-fed BALB/c group. Pospholipid transfer protein (Pltp), which is related to reverse cholesterol transport to the liver, was up-regulated in the four-week and nine-week HFD-fed BALB/c groups.
As to the reduction in lipid accumulation in the liver, lipid oxidation or lipid efflux could be related.
The expression of genes related to lipid oxidation was down-regulated in the one-, four-, and nine-week HFD-fed BALB/c groups. However, the gene expression of carnitine palmitoyltransferase I (Cpt1), which transports fatty acids through the inner mitochondrial membrane for beta-oxidation, was not changed.
Expression of genes involved in synthesis and degradation of ketone bodies was increased in the one-, four-, and nine-week HFD-fed BALB/c groups. Genes related to primary bile acid biosynthesis, including Cyp7a1 (the first and rate-limiting enzyme in the bile acid biosynthetic pathway) were down-regulated in the one-week–fed BALB/c group and up-regulated in the four-week–fed BALB/c group. Srebp1, a key transcription factor of lipid biosynthesis, was up-regulated in all groups. In the nine-week fed BALB/c group, PPAR-α, PPAR-γ, and Srebp2 were also up-regulated.
Discussion
In the present study, in order to increase our understanding of the key changes in hepatic steatosis in HFD-fed BALB/c mice identified in our previous study (Nishikawa et al. 2007; Nishikawa et al. 2008), we compared gene expression in the liver between HFD-fed BALB/c mice and B6 mice. Moreover, to understand the metabolic changes in the liver leading up to steatosis, we examined gene expression in BALB/c mice at two different feeding periods (one week and four weeks).
In the HFD-fed groups, peri-epididymal fat weight and serum Cho were increased at all time points examined. However, liver weight increased only in the nine-week HFD-fed BALB/c group. Histopathology showed that lipid accumulation in the liver was severe only in the nine-week HFD-fed BALB/c group. Therefore, lipid accumulation in the liver may occur subsequent to abdominal fat accumulation and the increase in serum Cho.
Metabolic changes predicted from the results of our microarray analysis are shown in Figure 2 . In the nine-week HFD-fed BALB/c group, marked differences from other groups are increased expression of fatty acid biosynthesis–related genes (Acaca, Acly, and Fasn) and increases in fatty acid uptake–related genes (Cd36).
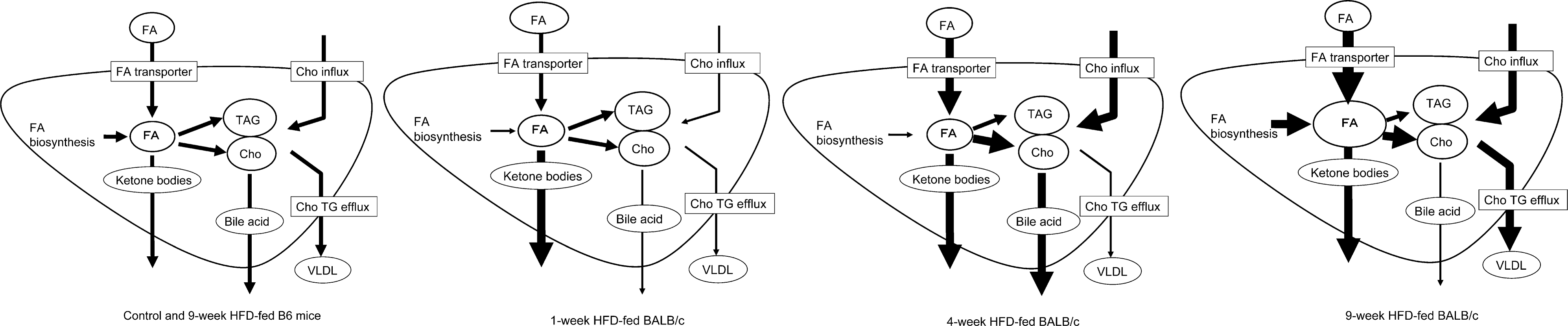
Predicted lipid metabolic change for each HFD-fed group. In the nine-week HFD-fed BALB/c group, fatty acid uptake and fatty acid biosynthesis may be markedly up-regulated. In the four-week HFD-fed BALB/c group, fatty acid uptake may be up-regulated, but biosynthesis may be down-regulated. In the one-week HFD-fed BALB/c group, fatty acid biosynthesis may be down-regulated. In both the control and the nine-week HFD-fed B6 groups, lipid metabolism may be balanced. Cho, cholesterol; FA, fatty acids; TAG, triacylglycerol; VLDL, very low-density lipoprotein.
In fatty acid biosynthesis, ACACA, FASN, and ACLY are all important. ACACA, which is involved in carboxylation of acetyl-CoA to malonyl-CoA, is the most important enzyme in the regulation of lipogenesis (Botham and Mayes 2006). ACLY catalyzes the cleavage of citrate to oxaloacetate and acetyl-CoA, releasing acetyl-CoA, which can then be used in fatty acid biosynthesis in the extramitochondrial cytosol (Botham and Mayes 2006). In leptin receptor–deficient db/db mice where hepatic ACLY is abnormally elevated, targeted suppression of ACLY in the liver can correct obesity-associated fatty liver (Wang et al. 2009). FASN catalyzes the last step in fatty acid biosynthesis and thus is believed to be a major determinant of the maximal hepatic capacity to generate fatty acids by de novo lipogenesis (Dorn et al. 2010). In twelve-week HFD-fed BALB/c mice with steatosis and in humans with nonalcoholic fatty liver disease, FASN expression in the liver was higher compared to the respective normal control (Dorn et al. 2010). Therefore, induction of ACLY, ACACA, and FASN may be one of the causes of excessive fat accumulation in the livers of the nine-week HFD-fed BALB/c group.
CD36 antigen acts on hepatocytes as a facilitator of long-chain fatty acid transport, but the mechanism by which it facilitates fatty acid uptake remains vague (Silverstein and Febbraio 2009). Fatty acids in hepatocytes may be converted to TAG or oxidized as fuel. When intrahepatic fatty acids are present in excess, the majority of the excess fatty acid is likely to be converted to TAG, which may be stored (Bradbury 2006). In models in which CD36 was specifically induced in the liver by pharmacological means or by cDNA transduction, CD36 contributed to steatosis (Silverstein and Febbraio 2009). In subjects with extreme steatosis, CD36 gene expression is up-regulated (Greco et al. 2008). Considering these reports, the increase of lipid uptake induced by CD36 may contribute to the severe hepatic fatty accumulation seen in the nine-week HFD-fed BALB/c group.
In the nine-week HFD-fed B6 group, a few genes related to lipid metabolism were differentially expressed, suggesting that lipid metabolism may be balanced by the gene expression level at this point.
In both the one- and four-week HFD-fed BALB/c groups, fatty acid biosynthesis was considered to be down-regulated. SCD1 catalyzes the synthesis of monounsaturated long-chain fatty acids from saturated fatty acyl-CoAs. SCD1-deficient mice had decreased liver fat content, suggesting that the lack of SCD1 provided protection from fatty accumulation in the liver (Peter et al. 2011).
An increase in Cd36 expression was also observed in the four-week HFD-fed BALB/c group, although not to the extent observed in the nine-week–fed BALB/c group, suggesting that the increased lipid uptake may occur at least by four weeks and then increase with time. In the one-week HFD-fed BALB/c group, no change was observed.
Several studies examining gene expression in HFD-fed mice have been reported. However, the relationship between histopathology and gene expression is not clear. In a study of HFD-fed B6 mice, several genes involved in lipogenesis (e.g., Acly, Me, and Fasn) were induced and they later returned to baseline levels, whereas genes involved in Cho synthesis (e.g., HMG-CoA reductase, farnesyl pyrophosphate synthetase, squalene synthase) were repressed after eleven days of feeding HFD; no steatosis of the liver was observed at this point (Gregoire et al. 2002). In twelve-week HFD-fed C57BL/6J mice, the expression of genes involved in fatty acid beta-oxidation and ketone body synthesis (e.g., Aox1, acetyl-CoA acyltransferase 1, HMG-CoA lyase) and fatty acid uptake into the liver (Cd36/Fat) was increased, and that of genes involved in lipogenesis (e.g., Fasn, acetyl-CoA synthetase 2, Gpam, Me) and Cho synthesis (squalene epoxidase (Sqle), farnesyl diphosphate farnesyl transferase 1) was decreased (Kim et al. 2004). In twenty-six-week HFD-fed mice, genes involved in fatty acid oxidation (e.g., Cyp4a10, Cyp4a14) were up-regulated, whereas those involved in lipogenesis (e.g., Me, Scd1) were suppressed (Patsouris et al. 2006). In a nine-month study of HFD-fed obese C57BL mice, genes related to beta-oxidation (e.g., carnitine O-acetyltransferase, Cpt1) were up-regulated and those related to Cho biosynthesis (e.g., Cyp7a1, Sqle) were down-regulated. On the other hand, in nine-month HFD-fed lean C57BL mice, genes involved in fatty acid biosynthesis (e.g., Scd1, Me, Fasn) were down-regulated (de Fourmestraux et al. 2004). From these reports, it appears that short-term HFD feeding may lead to induction of lipogenesis, but long-term HFD-feeding may lead to a return to baseline or suppression of fatty acid biosynthesis and Cho synthesis, and induction of beta-oxidation and fatty acid intake. Kim et al. hypothesized that in the twelve-week HFD-fed mice, the uptake of fatty acids into the liver was augmented, resulting in subsequent accumulation of TAG in the liver, and that hepatic TAG accumulation might have driven the up-regulation of genes involved in lipid catabolism and the down-regulation of lipogenic genes by a feedback mechanism. Therefore, down-regulation or no change of lipogenesis in long-term HFD-fed animals is considered to be an adaptive change to excessive lipid intake in the liver, and it can be applied to the one-and four-week HFD-fed BALB/c groups in this study. Moreover, at these time points, the histopathological changes were not so severe. In the nine-week HFD-fed BALB/c group, although the up-regulation of fatty acid uptake corresponded with the above reports, the up-regulation of fatty acid biosynthesis may not occur in the same way as in the short-term HFD induction of lipogenesis and probably indicates the collapse of the lipid metabolic system, inducing marked fat accumulation, as observed morphologically.
In conclusion, the severe fatty accumulation in BALB/c mice may be caused, at least in part, by up-regulated fatty acid uptake and fatty acid biosynthesis in BALB/c mice. Cd36, Acaca, Acly, and Fasn may be involved in these metabolic processes.
Footnotes
Acknowledgments
The authors would like to thank Mr. Naoya Masutomi, Mr. Toshinobu Shimizu, Ms. Natsuko Terasaki, Ms. Manami Miyake, and Mr. Yoshimi Inoue for skillful technical assistance and helpful advice concerning the microarray analysis.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article. The author(s) received no financial support for the research and/or authorship of this article.
