Abstract
Acute methanol poisoning is a global health concern. This study was designed to compare the prognostic roles of neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and their combination in the prediction of clinical outcomes in methanol-intoxicated patients as well as to evaluate their associations with all initial patients’ characteristics. We conducted a cross-sectional study among methanol-intoxicated patients. A total of 109 patients were enrolled in the study. Thirty-four (31%) patients died during hospital admission while 30 (27.5%) patients developed visual loss. Most of the unfavorable findings were evident in patients with high NLR and PLR. Neutrophil-to-lymphocyte ratio and PLR can excellently differentiate between survivors and non-survivors with an area under the curve (AUC) of 0.991 vs 0.923, respectively. Platelet-to-lymphocyte ratio showed an accepted discrimination ability to differentiate between patients who developed and patients who did not develop visual loss, AUC of 0.734, however, NLR showed no discrimination, AUC of 0.558. We concluded that NLR and PLR can serve as valuable tools in risk-stratifying patients and prognosticating outcomes in acute methanol poisoning. Platelet-to-lymphocyte ratio is superior to NLR as a predictive factor in patients with permanent visual impairment. However, a combination of NLR with PLR can develop a more powerful prediction for overall clinical outcomes.
Keywords
Introduction
Methanol is considered as one of the top-five most greatly used chemicals in industrial fields, agriculture, and by consumers all over the world.1,2 Acute methanol poisoning is still a global health concern in both developing and developed countries. Intake of illicit and homemade alcoholic beverages along with inhalation and transdermal exposures can lead to acute methanol poisoning.3–7 Methanol itself initiates the toxicity. While formic acid, the main metabolite of methanol, is responsible for metabolic derangement and permanent neurological sequelae. Methanol is metabolized by the alcohol dehydrogenase (ADH) enzyme into formaldehyde. Subsequently, formaldehyde is metabolized into formic acid by the aldehyde dehydrogenase (ALDH) enzyme. 8 Other mechanisms of methanol toxicity include, but are not limited to, oxidative stress, lipid peroxidation, production of pro-inflammatory cytokines, and expression of key proteins responsible for maintaining cellular homeostasis.9–11 Clinical manifestations of methanol toxicity include gastrointestinal abnormalities, neurological impairment, and metabolic acidosis, in addition to, a visual impairment that ranges from blurred vision to early or delayed blindness.12,13 In Iran, during the COVID-19 pandemic, an outbreak of methanol poisoning was pronounced with a report of more than 300 deaths and nearly 1000 critically ill patients following household methanol-containing disinfectants and sanitizers ingestion as false beliefs regarding the methanol preventive ability against COVID-19 infection.14–16 Similarly, other sporadic cases of methanol toxicity were described during the COVID-19 pandemic worldwide.17–19 Methanol poisoning represents 92% of the incidents stated by the global event-based surveillance with at least one or more deaths. 20 In addition, methanol poisoning puts major burdens on the healthcare system because of its associated high mortality and morbidity rates despite the effective patients management. 21
Neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) denote the proportions of absolute neutrophil count and platelet count, respectively, to the lymphocyte count. They are relatively novel,simple, and inexpensive biomarkers and can be easily estimated from a routine complete blood count (CBC) test. Elevated NLR and PLR values reflect an impaired cell-mediated immunity in association with systemic inflammation.22–25
Neutrophil-to-lymphocyte ratio reflects the equilibrium between systemic inflammation and immunity in peripheral blood. 26 Also an elevation in NLR is related to the elevation in neutrophils and/or reduction in lymphocytes.27,28 Prior studies have extensively demonstrated the prognostic role of NLR as a significant predictor of morbidity and mortality in different diseases.26,29–33 Although absolute neutrophil and lymphocyte counts could be affected by different physiological, pathological, and physical changes, NLR has been demonstrated to be more stable compared to other leukocyte subpopulations. 24 Stimulated neutrophils produce a multitude of proteolytic enzymes such as myeloperoxidase (MPO) which mediates tissue injury. Myeloperoxidase is an enzyme abundantly expressed by polymorphonuclear neutrophils and macrophages upon release during their activation. It may contribute directly to tissue damage owing to its powerful proinflammatory action.34,35 Platelet-to-lymphocyte ratio infers an interplay concurrent between platelet count and lymphocyte count, reflecting platelet aggregation as well as initiation of inflammatory cascades. Elevated PLR is reported in several acute and chronic inflammatory conditions and is associated with an unfavorable prognosis.36–42
Neutrophil-to-lymphocyte ratio and PLR are currently emerging as prognostic biomarkers in many disease processes, however, their predictive values for poor outcomes in acute methanol poisoning have not been explored yet. We hypothesized that NLR and PLR could be utilized as inflammation-based prognostic biomarkers. Consequently, this study was designed to compare the prognostic roles of NLR, PLR, and their combination in the prediction of clinical outcomes of methanol-intoxicated patients, in terms of in-hospital mortality and visual loss. Furthermore, NLR and PLR of the enrolled patients were stratified into tertials (low, intermediate, and high) to evaluate their associations with all initial patients’ characteristics (baseline, clinical, laboratory) and the received medical care. To the best of our knowledge, there is no study indicating a link between NLR, PLR, and methanol poisoning.
Methods
Study design and settings
A cross-sectional analysis of 109 patients with acute methanol intoxication admitted to Poison Control Center of Ain Shams University Hospitals (PCC-ASUH), Cairo, Egypt, from January 2020 to December 2021. The Institutional Review Board of Faculty of Medicine, Ain Shams University approved the study proposal. Administrative approval was attained from PCC-ASUH. The informed consent was obtained by the patients or their caregivers/family members (in comatose patients). The eligibility criteria included patients with acute methanol poisoning who were aged 18 years and older. While patients with the following conditions were excluded from the study: receiving chemotherapy or radiation, known blood disorders, malignancy, history of blood transfusion in the last 90 days, and recent infections in the last 14 days. Diagnosis of methanol poisoning was confirmed by history, physical examination, and toxicology laboratory evaluation following the PCC-ASUH protocols.
Study procedures
All eligible patients underwent detailed history taking including (age, gender, residence, history of substance use, mode of poisoning, and comorbidities). A thorough initial clinical assessment was performed including Glasgow coma scale scoring, vital signs, hemodynamics, and electrocardiogram (ECG) findings along with ophthalmic examination. Arterial blood samples were used for arterial blood gas analysis along with venous blood samples (whole blood, serum, and plasma) for routine laboratory investigations using standards supplied in the kits, including [(CBC, blood methanol level, random blood sugar, serum urea, and serum creatinine)]. Pre-and in-hospital management was also assessed, including received antidotes, mechanical ventilator support, inotropes or vasopressors use, and extracorporeal elimination. Eligible patients were stratified into tertials (low, intermediate, and high) according to their NLR and PLR.
Sample size
The current study had more than 95% power. A post hoc power calculation was computed by G*power 3.1 based on the mean difference between two independent means (NLR values in survivors versus non-survivors, 3.8 ± 1.8 vs 10.6 ± 1.7, respectively) at a sample size of 109 and a type I error threshold (α) <.05.. 43
Statistical analysis
Continuous variables were presented as the mean ± SD or median (25th to 75th percentiles) according to the normality of data. Categorical variables were presented by frequency and percentage. The normality of continuous variables was checked by the Kolmogorov-Smirnov test. Homogeneity of variances was checked by Leven’s test. The chi-squared test was used to discover if there was a relationship between two categorical variables. Fisher’s exact test for (R × C) table was used as an alternative to chi-squared test when the expected cell count is less than five. The ordinary one-way analysis of variance (ANOVA) was used to determine whether there were any significant differences between the means of two or more independent groups on a continuous dependent variable while Welch’s ANOVA test was used if equal variances were not assumed. Post-hoc tests following ANOVA test (Tukey test if equal variances were assumed; Games-Howell test if equal variances were not assumed) were used for multiple comparisons between groups. The Kruskal-Wallis H test is a non-parametric alternative to the one-way ANOVA. Dunn’s is a post-hoc test following Kruskal-Wallis H test. Mann–Whitney
Results
All patients’ baseline characteristics along with the results of the clinical and ophthalmic assessment, laboratory findings, and pre-and-hospital management of methanol-intoxicated patients were stratified by NLR and PLR tertiles into low, intermediate, and high.
Baseline characteristics of the studied patients
Baseline characteristics of methanol-intoxicated patients stratified by NLR and PLR on admission.

Bold values indicate a statistically significant difference at the
aOrdinary one-way analysis of variance (ANOVA) test.
bFisher’s exact test (R
cChi-squared test (
dAlcohol, cannabis, and opioids.
eBenzodiazepines, cannabis, and opioids.
fChronic obstructive pulmonary disease, depression, diabetes, hypertension, and ischemic heart disease.
Abbreviations: NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SD, standard deviation.
Clinical assessment on admission of the studied patients
Clinical assessment of methanol-intoxicated patients stratified by NLR and PLR on admission.

Data are median (25th to 75th percentiles), unless otherwise mentioned.
Bold values indicate a statistically significant difference at the
aSignificant vs (NLR≤ 3.2 or PLR ≤ 290).
bsignificant vs (NLR = 3.3–7.9 or PLR = 291–363).
cKruskal-Wallis H test.
dOrdinary one-way analysis of variance (ANOVA) test.
eWelch’s one-way analysis of variance (ANOVA).
fBundle branch block, inverted T wave, prolonged QTc, sinus bradycardia, sinus tachycardia, and ST segment elevation.
gChi-squared test (
Abbreviations: NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; ECG, electrocardiogram.
Ophthalmic findings of the studied patients
Ophthalmic findings of methanol-intoxicated patients stratified by NLR and PLR on admission.

Bold values indicate a statistically significant difference at the
aChi-squared test (
bFisher’s exact test (R
Abbreviations: NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Laboratory findings of the studied patients
Laboratory findings of methanol-intoxicated patients stratified by NLR and PLR on admission.

Data are mean ± standard deviation or median (25th to 75th percentiles).
Bold values indicate a statistically significant difference at the
aSignificant vs (NLR ≤ 3.2 or PLR ≤ 290).
bsignificant vs (NLR = 3.3–7.9 or PLR = 291–363).
cWelch’s one-way analysis of variance (ANOVA) test.
dKruskal-Wallis H test.
eMean rank for PaCO2 stratified by PLR (48.1, 50.8, and 66.3, respectively).
fOrdinary one-way analysis of variance (ANOVA) test.
Abbreviations: ABG, arterial blood gas; HB, hemoglobin; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Medical care of the studied patients
Pre-and in hospital management of methanol-intoxicated patients stratified by NLR and PLR on admission.
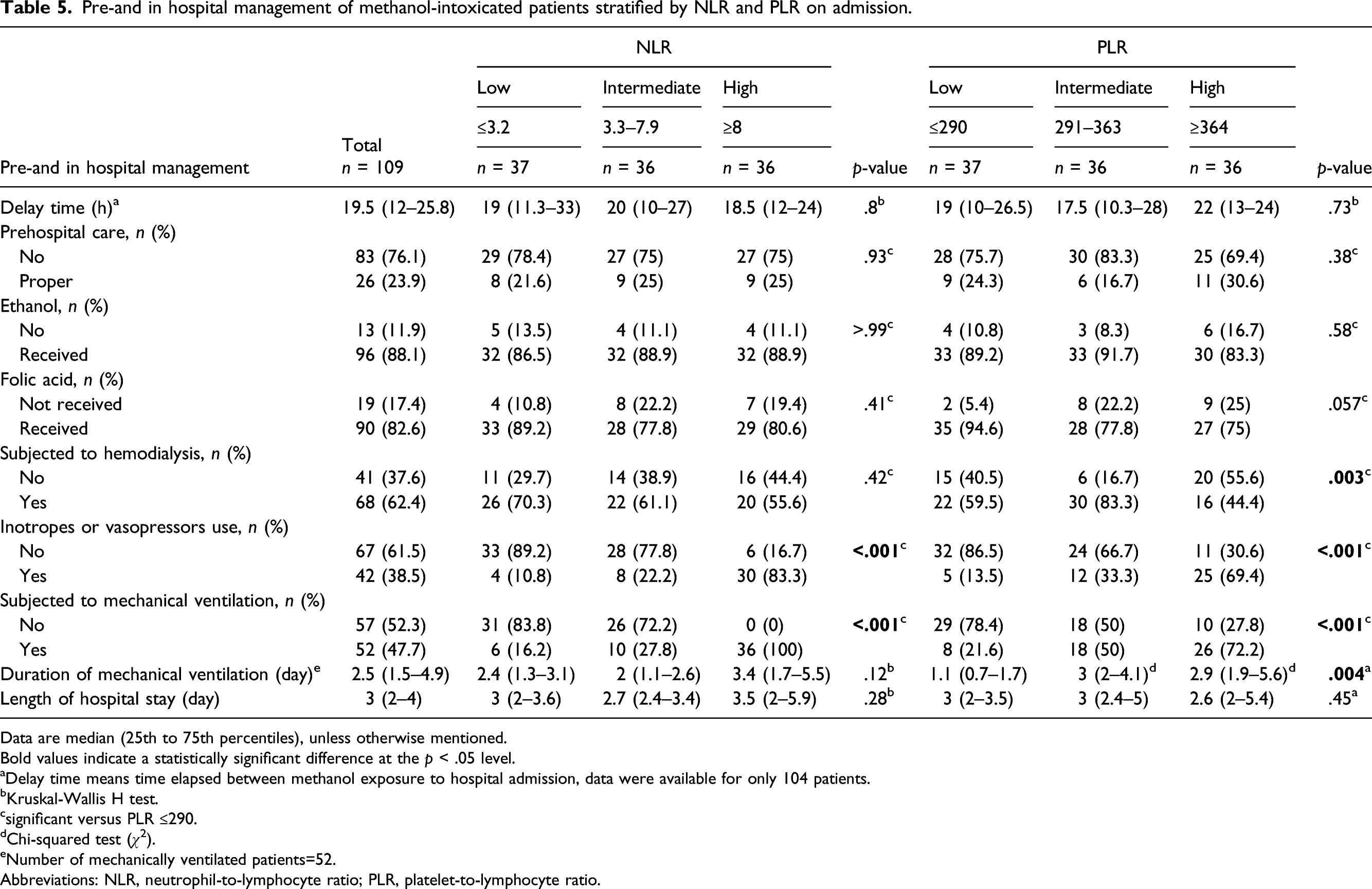
Data are median (25th to 75th percentiles), unless otherwise mentioned.
Bold values indicate a statistically significant difference at the
aDelay time means time elapsed between methanol exposure to hospital admission, data were available for only 104 patients.
bKruskal-Wallis H test.
csignificant versus PLR ≤290.
dChi-squared test (
eNumber of mechanically ventilated patients=52.
Abbreviations: NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Clinical outcomes of the studied patients
Figure 1 shows the probability of methanol-intoxicated patients’ survival during the hospital length of stay. Figure 2 demonstrates that approximately 31%, 34 patients died during hospital admission while 27.5%, 30 patients developed visual loss. Non-survivors had statistically significantly higher both NLR and PLR than survivors (median 10.5 and 411.5 [25th to 75th percentiles: 9.5–11.95, 362.5–430], respectively) vs 3.3 and 292 (25th to 75th percentiles: 2.4–4.8, 254–331], respectively, Kaplan-Meier survival plot showing the probability of methanol-intoxicated patients’ survival during the hospital length of stay. Clinical outcomes of methanol-intoxicated patients. Clinical outcomes of methanol-intoxicated patients stratified by NLR and PLR. Data are expressed as median (25th to 75th percentiles).


Prognostic utility of NLR, PLR, and their combination
NLR, PLR, and their combination in the prediction of hospital mortality of methanol-intoxicated patients.
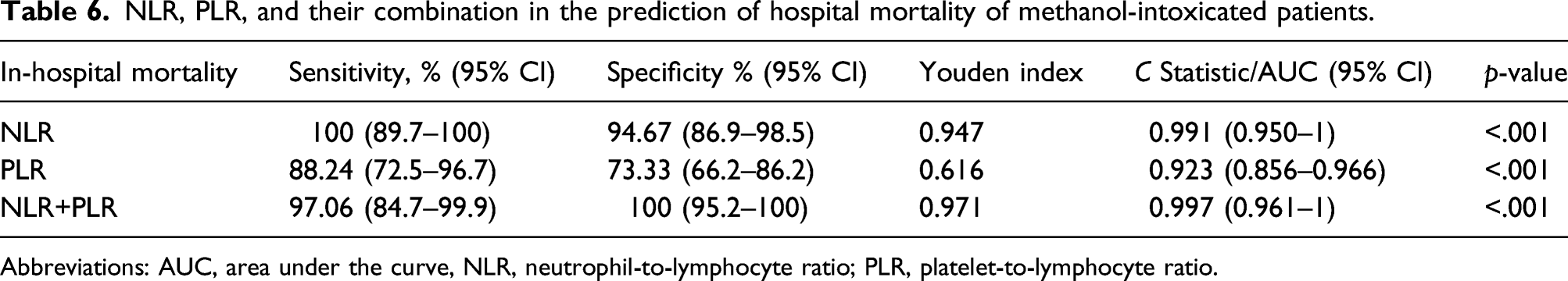
Abbreviations: AUC, area under the curve, NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.

The receiver operating characteristic (ROC) curves for NLR and PLR, and their combination in the prediction of clinical outcomes of methanol-intoxicated patients. Panel A: Comparing ROC curves for NLR and PLR in the prediction of hospital mortality of methanol-intoxicated patients. Panel B: Comparing ROC curves for NLR and PLR in the prediction of visual loss of methanol-intoxicated patients. Panel C: Combined ROC curve for NLR and PLR in the prediction of hospital mortality of methanol-intoxicated patients. Panel D: Combined ROC curve for NLR and PLR in the prediction of visual loss of methanol-intoxicated patients.
NLR, PLR, and their combination in the prediction of visual loss of methanol-intoxicated patients.

Abbreviations: AUC, area under the curve, NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Discussion
We found that initial NLR significantly increased in non-survivors but not significantly increased in patients with permanent visual impairment while initial PLR significantly increased in non-survivors and patients with permanent visual impairment. In addition, NLR and PLR on admission can serve as promising prognostic biomarkers in the early detection of patients with poor outcomes in case of acute methanol poisoning, however, their combination demonstrated more predictive power.
In this work, significantly more patients with high NLR had a history of medical comorbidities. In corroboration, previous studies showed that NLR values significantly increased in many systemic diseases.44–47 We found that degree of consciousness and most hemodynamic parameters were significantly worse in patients with high NLR and PLR. Similarly, a study by Efros et al. reported that patients with elevated NLR (>5.12) had significantly lower blood pressure measurements than those with lower NLR (≤5.12) among 2182 patients diagnosed with acute pulmonary embolism. 47
Our findings demonstrated a significant association between abnormal ECG findings and NLR and PLR. In support, NLR was significantly linked with carbon monoxide-induced cardiotoxicity as indicated by abnormal ECG findings (transmyocardial repolarization parameters) in the observational study by Temrel and coworkers. 48
The present work revealed that most patients with high NLR and PLR developed abnormal ophthalmic findings. Elevated NLR and PLR were reported in patients with many ocular diseases, e.g., dry eye disease, idiopathic acute anterior uveitis, neovascular age-related macular degeneration, and primary angle-closure glaucoma, compared to healthy individuals. It seems that inflammation, either local or systemic, is the shared mechanism involved in these ocular disorders.25,49,50
Neutrophil-to-lymphocyte ratio alone poorly differentiated between patients with permanent visual loss and those without permanent visual loss. But adding PLR to NLR demonstrated more predictive power. In contrast to our results, a systematic review by Kurtul and Ozer 51 demonstrated the clinical utility of NLR as a diagnostic and/or a prognostic marker in different eye diseases.
The patients in the current study had high anion and osmolal gaps metabolic acidosis on admission. Moreover, results of arterial blood gas analysis were significantly impaired mainly in patients with high NLR and PLR. A high anion plus osmolal gap metabolic acidosis is a common finding of toxic alcohol ingestion.52,53 Accumulation of formic acid in the body is the fundamental mechanism of methanol-inducing metabolic acidosis during the clinical course of toxicity. 54
Initial renal functions were significantly elevated in patients with high NLR and PLR. In corroboration, several studies correlated increased levels of the NLR, and other inflammatory ratios to the higher risk of development of acute kidney injury among critically ill patients.55–57 Methanol may induce nephrotoxicity due to direct contributing factors, e.g., high blood methanol and formic acid levels, or indirect contributing factors, e.g., hemolysis and myoglobinuria. 58 In the same context, our study demonstrated that patients with the high NLR tertile had significantly higher blood methanol levels compared to low and intermediate NLR tertiles.
In the present work, all patients with high NLR were subjected to mechanical ventilation. Similarly, several studies described NLR as a promising biomarker for the prediction of poor outcomes in patients with respiratory illness such as the need for mechanical ventilation, disease severity, and mortality.59–62
Recent studies throw light on NLR and PLR, being novel markers of morbidity and mortality in various diseases. 63 Our results demonstrated that non-survivors had significantly higher both NLR and PLR than survivors. In support, a study by Yoldas and colleagues showed that both NLR and PLR of non-survivors treated in the intensive care unit significantly increased than the survivors. 63
Conclusions
Identifying novel, simple, and widely available indices such as NLR and PLR in risk-stratifying patients and prognosticating outcomes in the setting of acute methanol poisoning may help to guide effective management to minimize most methanol-related morbidity and mortality. Platelet-to-lymphocyte ratio is superior to NLR as a predictive factor in patients with permanent visual impairment but NLR is more specific. A combination of NLR with PLR can develop a more predominant prediction for overall clinical outcomes in those patients.
Limitations of the study
This study is limited by the lack of previous studies on the association between NLP, PLR, and methanol poisoning. In addition, no cause-and-effect relationships can be proven due to the cross-sectional design of the study.
Footnotes
Acknowledgements
The authors acknowledge the support of Taif University Researchers Supporting Project number (TURSP-2020/132), Taif University, Taif, Saudi Arabia.
The authors would like to thank Miss. Gihad Mahmoud Diab (English teacher, British Council, Heliopolis, Cairo) for her help with the manuscript English language editing.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study is a revision of the doctoral dissertation of the first author.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Taif University TURSP-2020/132.
