Abstract
Objective
To explore the mechanism of chromobox 7 (CBX7)-mediated nuclear factor E2-related factor 2 (Nrf2)/hemeoxygenase-1 (HO-1) signaling pathway in the cerebral ischemia/reperfusion (I/R) injury.
Methods
The experimental wild-type (WT) and CBX7-/- mice were used to establish cerebral I/R models using the middle cerebral artery occlusion (MCAO) surgery to determine CBX7 levels at different time points after MCAO injury. For all mice, neurological behavior, infarct size, water content, and oxidative stress–related indicators were determined, and transferase (TdT)-mediated dUTP-biotin nick-end labeling (terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling (TUNEL)) staining method was employed to observe cell apoptosis, while Western blot to measure the expression of CBX7 and Nrf/HO-1 pathway-related proteins.
Results
At 6 h, 12 h, 24 h, 3 days, and 7 days after mice with MCAO, CBX7 expression was gradually up-regulated and the peak level was reached at 24 h. Mice in the WT + MCAO group had increased infarct size, with significant increases in the modified neurological severity scores and water content in the brain, as well as the quantity of TUNEL-positive cells. For the oxidative stress-indicators, an increase was seen in the content of MDA (malondial dehyde), but the activity of SOD (superoxide dismutase) and content of GSH-PX (glutathione peroxidase) and CAT (catalase) were decreased; meanwhile, the protein expression of CBX7, HO-1, and nuclear Nrf2 was up-regulated, while the cytoplasmic Nrf2 was down-regulated. Moreover, CBX7 knockout attenuated I/R injury in mice.
Conclusion
Knockout of CBX7 may protect mice from cerebral I/R injury by reducing cell apoptosis and oxidative stress, possibly via activating the Nrf2/HO-1 pathway.
Keywords
Introduction
Cerebral ischemia/reperfusion (I/R) injury is considered as a threat to the health risks of human beings, bringing about severe burdens to the society and families. 1 Theoretically, cerebral I/R injury refers to the secondary damage of the blood restoration to the ischemic brain tissues, concomitant with more severe neurological dysfunction and morphological change than those before restoration of blood supply.2,3 As reported, cerebral I/R injury involves quite complicated pathophysiological mechanisms, including disrupted energy metabolism, excitatory amino acid toxicity, calcium overload, massive generation of nitric oxide, inflammatory injury, oxidative stress, and neuronal cell apoptosis.4-6 In recent years, even though great progress has been made in the pathophysiological mechanism of cerebral I/R injury, clinical studies have not found any effective neuroprotective drugs after cerebral ischemia. 7 Hence, it is of great significance to explore and discover the novel strategies and targets for the treatment of cerebral I/R injury.
Chromobox (CBX) family, as a group of PcG family, consists of five subgroups, including CBX2, CBX4, CBX6, CBX7, and CBX8, and the expression pattern and biological functions vary in different tissues. 8 Nowadays, the investigation concerning CBX7 was mainly focused on its correlation with the development, progression, invasion, and metastasis of malignant tumors.9,10 The latest evidence suggested that CBX7 was up-regulated in the ischemia-induced endogenous neural progenitor cell (NPC), which could mediate the NPC proliferation. 11 In the work of Jia-Rong Fan et al., 12 the knockout of CBX7 was also found to be able to promote the self-update of adult pluripotent-like olfactory stem cells (APOSCs), and the implantation of APOSCs into the brain could improve the neurological dysfunction of stroke mouse, implicating the potential role of CBX7 in the cerebral ischemia. Worth mentioning, Ye Zhang et al. 13 found that the inhibition of CBX7 may activate the Nrf2/HO-1 signaling pathway to reduce the stress response of endoplasmic reticulum to relieve the renal I/R injury. This pathway, nuclear factor E2-related factor 2 (Nrf2)/hemeoxygenase-1 (HO-1) signal axis, may have a key role in the modulation of anti-oxidative injury. 14 Also, it has been confirmed that the activation of Nrf2/HO-1 pathway may eliminate the reactive oxygen species (ROS) via inducing the transcription of protective genes to antagonize the oxidative stress injury induced by I/R. 15 Hence, we supposed that CBX7 may play a pivotal role via mediating the Nrf2/HO-1 signaling pathway in the cerebral I/R injury.
Considering the above, we, first, detected the level of CBX7 in the mouse after cerebral I/R injury and explored the neuroprotective effect of CBX7 knockout-mediated Nrf2/HO-1 signal pathway on the mouse, aiming to provide novel insights for treatment of cerebral I/R injury.
Materials and methods
Ethical statement
This study conformed to Guide for the Care and Use of Laboratory Animals published by National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals, 16 and the design of this study had been approved by the Ethical Board of Medical Laboratory Animal of our University.
Construction of mouse cerebral ischemia-reperfusion model
The modified Longa method was adopted to prepare mouse cerebral I/R models by using middle cerebral artery occlusion (MCAO) surgery. 17 In brief, C57BL/6 mice in anesthesia were positioned on an operative table, where an incision was made in the middle of the neck to isolate the right carotid artery and external carotid artery that was later ligated and dissected. Then, a fish line with a ball-shaped end was inserted from the end of external carotid artery, and when the operator sensed the resistance, the line was maintained for 120 min. Later, the fish line was removed to restore the blood flow in the common carotid artery and internal carotid artery. The same procedures were also carried out for the mice in the Sham group, but with no entrance of fish line. After operation, mice were placed back into the cages and fed separately with free access to the food and water. Mice were divided into the Sham group and MCAO group, while those in the MCAO group were furthered classified into 6-h MCAO group (n = 6), 12-h MCAO group (n = 6), 24-h MCAO group (n = 6), 3 days MCAO group (n = 6), and 7 days MCAO group (n = 6) according to the time after operation. Then, Western blot was conducted to detect the level of CBX7 in the brain of mice.
Treatment and grouping of animals
The wild-type (WT), male C57BL/6 mice (20–25 g), and CBX7-/- mouse were purchased from the Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). WT and CBX7-/- mice were divided into the WT + Sham group, CBX7-/- + Sham group, WT + MCAO group, and CBX7-/- + MCAO group, with six mice in each group.
Measurement of neurological behavior scores
For measurement of neurological behavior scores, the modified neurological severity scores (mNSS) was adopted, 18 including the motor, sense, balance, and reflex, with a total of 18 points. As for the result, 0 point represents normal and the higher scores represent more severe damages. Measurement was conducted at 24 h after I/R injury.
Measurement of water content in the brain of mouse
At 24 h after I/R, mice were subjected to harboring the brain tissue by decapitation, and the brain was placed on a filter paper to remove the water. Then, with the rhinencephalon, lower brain stem, and cerebellum, the wet weight of the brain was measured by using the following formulae: Water content of brain (%) = (wet weight - dry weight)/wet weight × 100%. 19
TTC (2,3,5-triphenyltetrazolium chloride) staining
At 24 h after cerebral I/R, mice were decapitated and the head was frozen rapidly at −20°C for 20 min and then the olfactory bulb, cerebellum, and low brain stem were all removed. The remaining brain tissues were placed on an operation table to prepare the coronal sections in thickness of 2 mm which were later placed in 1% TTC solution for 30 min at 37°C in the dark. Stained sections were serially scanned to measure the infarct size in brain by using the Image J software and calculate the ratio of cerebral infarction volume to the total volume of brain.
TUNEL (terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling) staining
The brain tissues of mouse were fixed in 4% paraformaldehyde and then sliced into sections, followed by rinsing in PBS. Sections were preserved in 1% paraformaldehyde for later use. Paraffin-covered sections were roasted to remove the paraffin and then hydrated. Protease K working solution was prepared in concentration of 20 mg/mL and added dropwise onto the sections, followed by 15 to 20 min of incubation at 30°C and washes in PBS for 3 times (5 min/wash). Tissues were then incubated with the TUNEL working solution at 37°C for 1 h in the dark, and then, the reaction solution was discarded, followed by three washes in PBS (5 min/wash). Sections were incubated with DAPI (4’, 6-diamidino-2-phenylindole) at 37°C in the dark for 10 min and mounted. Then, sections were observed by using the confocal fluorescent microscope.
Determination of oxidative stress–related indicators
Brain tissues were harbored from the mice of each group to prepare the homogenate in PBS. Homogenate was then centrifuged at 3500 r/min for 15 min at 4°C to collect the supernatant, while the SOD (superoxide dismutase) activity and content of GSH-PX (glutathione peroxidase), MDA (malondial dehyde), and CAT (catalase) in the supernatant were determined using the corresponding kits provided by Nanjing Jiancheng Bioengineering Institute.
Western blot
Brain tissues were prepared to collect the total proteins from the whole cell, nucleus, and cytoplasm according to the instruction of kits (NanJing KeyGen Biotech Co., Ltd.), and the collected proteins were subjected to the measurement of protein concentration by using the BCA kit. Protein samples were then subjected to the SDS-PAGE (sodium dodecyl sulphate polyacrylamide gel electrophoresis) and then transferred onto the PVDF (polyvinylidene fluoride) membrane, where the unoccupied sites were blocked by the non-fat milk, followed by being washed in PBST. Proteins on the membrane were detected by incubation with the rabbit anti-mouse monoclonal anti-CBX7 (ab21873, Abcam, UK), Nrf2 (ab137550, Abcam, UK), HO-1 (ab13243, Abcam, UK), Lamin B1 (ab16048, Abcam, UK), and β-actin antibodies (ab8226, Abcam, UK) at 4°C overnight, followed by five washes in PBST, 3 min/wash. The resulting immunoblots were further incubated with the horseradish peroxidase–labeled secondary antibodies (ab205718, Abcam, UK) for 1 h at room temperature. The final immunoblots were developed in ECL (chemiluminescence), and the band of protein was analyzed using the Image J software to determine the average of band intensity, with GAPDH and Lamin B1 as the loading controls. The relative expression of targeted protein was expressed by the ratio of the band intensity of targeted protein to that of the GAPDH/Lamin B1.
Statistical analysis
All data were subjected to the statistical analysis using the SPSS 21.0 software. Measurement data were expressed in mean ± standard deviation (SD). Difference among groups was analyzed by using the one-way ANOVA, followed by the Tukey’s post hoc test for pairwise comparison. p < 0.05 suggested that the difference had statistical significance.
Results
Expression of CBX7 after cerebral I/R
As shown in Figure 1, the results of Western blot for determining the level of CBX7 in the brain after MCAO injury at different time points, and the results demonstrated that at 6 h, 12 h, 24 h, 3 days, and 7 days after MCAO, mice had the significant up-regulation of CBX7 as compared to the Sham group, and the peak level was reached at 24 h (p < 0.05). Expression of CBX7 in the brain tissues of mouse after cerebral I/R injury. A: Western blot was used to determine the level of CBX7 in the brain tissue of mouse after MCAO; B: Comparison of the CBX7 expression at different time points after MCAO; The data were presented as the mean ± SD (n = 6); * p < 0.05 versus the Sham group. CBX7: chromobox 7, MCAO: middle cerebral artery occlusion.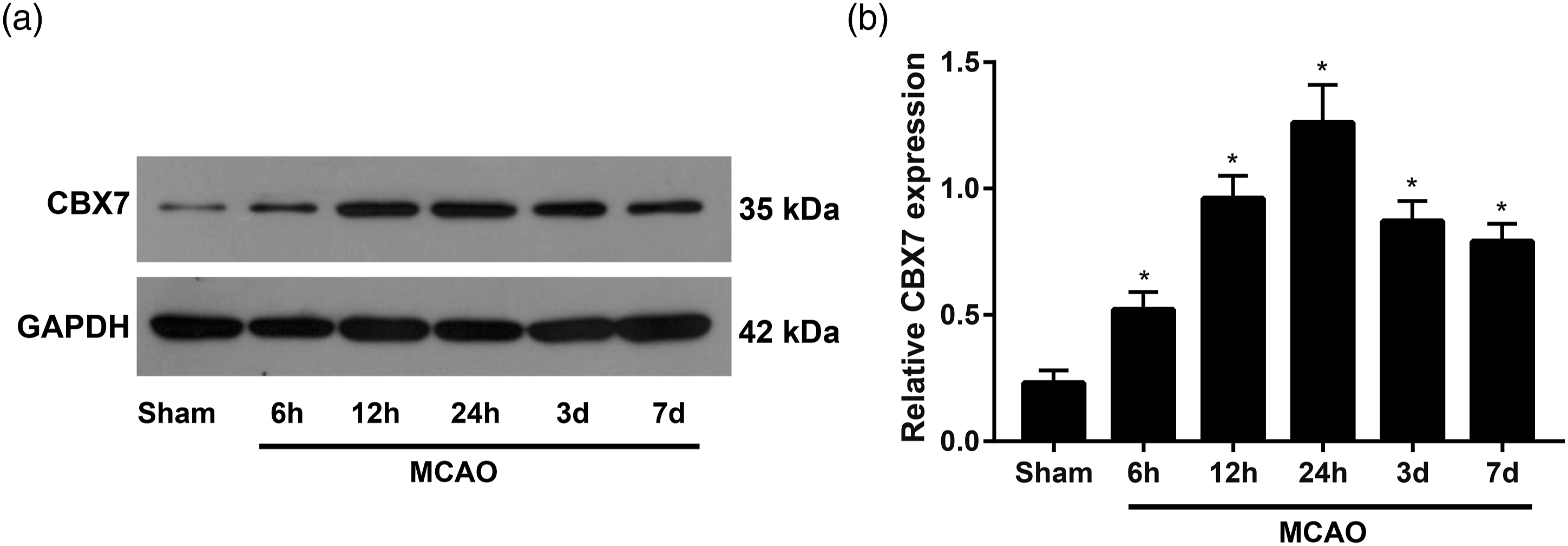
Effect of CBX7 on the cerebral infarct size and neurological behavior of mouse after cerebral I/R
By comparison with the WT + Sham group, cerebral infarct size of mouse in the CBX7-/- + Sham group showed no obvious variance (p > 0.05), while mice in the WT + MCAO group had significant enlargement in the cerebral infarct size (p < 0.05, Figure 2). As compared to the WT + MCAO group, mice in the CBX7-/- + MCAO group had a sharp shrinkage in the cerebral infarct size (p < 0.05). Moreover, no obvious difference was shown between the WT + Sham group and CBX7-/- + Sham group concerning the mNSS and water content in brain (p > 0.05), but the increased mNSS and water content in brain were found in the WT + MCAO group (p < 0.05). In addition, when compared to the WT + MCAO group, mice in the CBX7-/- + MCAO group manifested the sharp decreases in the mNSS and water content in the brain (p < 0.05). Comparison of the cerebral infarct size, mNSS, and water content in the brain among groups. A: 2,3,5-triphenyltetrazolium chloride staining was performed to reflect the cerebral infarct size of mouse; B: Comparison of the cerebral infarct size among groups; and C-D: Comparison of the mNSS C: and water content in the brain D: of mouse; The data were presented as the mean ± SD (n = 6); * p < 0.05 vs. WT + Sham group or chromobox 7-/- + Sham group; # p < 0.05 vs. the wild-type + MCAO group. mNSS: modified neurological severity scores. WT: wild-type.
Comparison of cell apoptosis in the brain of mouse
As seen from Figure 3, mice of CBX7-/- + Sham group showed no significant difference in the TUNEL-positive cells in the brain from those in the WT + Sham group (p > 0.05), but those in the WT + MCAO group presented with obvious increased TUNEL-positive cells (p < 0.05). Furthermore, the quantity of TUNEL-positive cells in the mice of CBX7-/- + MCAO group was found to be decreased evidently when compared to the WT + MCAO group (p < 0.05). Comparison of cell apoptosis in the brain of mouse. A: TUNEL-DAPI staining was conducted to reflect cell apoptosis in the brain of mouse; B: Comparison of the TUNEL-positive cells in the brain of mouse among groups; The data were presented as the mean ± SD (n = 6); * p < 0.05 versus WT + Sham group or chromobox 7-/- + Sham group; # p < 0.05 versus. the WT + middle cerebral artery occlusion group. TUNEL: terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick-end labeling, WT: wild-type.
Comparison of the oxidative stress–related indexes in the brain of mice
As shown in Figure 4, in comparison with the WT + Sham group, mice in the CBX7-/- + Sham group presented with no significant difference in the oxidative stress–related indicators (all p > 0.05), while in the WT + MCAO group, MDA increased and SOD activity content of GSH-PX and CAT decreased significantly (all p < 0.05). Additionally, in comparison with the mice of WT + MCAO group, mice in the CBX7-/- + MCAO group presented with a sharp decrease in the content of MDA but increases in the SOD activity and content of GSH-Px and CAT (all p < 0.05). Comparison of the oxidative stress–related indexes in the brain among groups. A-D: Comparison of malondial dehyde content A: glutathione peroxidase content B: superoxide dismutase activity C: and catalase content D: in the brain of mouse among groups; The data were presented as the mean ± SD (n = 6); * p < 0.05 versus WT + Sham group or CBX7-/- + Sham group; # p < 0.05 versus the WT + middle cerebral artery occlusion group. WT: wild-type.
Comparison of CBX7 expression and Nrf2/HO-1 pathway-related proteins in the brain of mice
Western blot (Figure 5) demonstrated that CBX7 protein was not expressed in the CBX7-/- mice. As compared to the WT + Sham group, CBX7, HO-1, and nuclear Nrf2 were up-regulated significantly in the mice of WT + MCAO group, while the cytoplasmic Nrf2 expression was down-regulated (all p < 0.05). Besides, when compared to the WT + MCAO group, the expression of HO-1 and nuclear Nrf2 were further up-regulated in the mice of CBX7-/- + MCAO group, with a significant decrease in the cytoplasmic Nrf2 expression (all p < 0.05). Comparison of CBX7 expression and Nrf2/HO-1 pathway-related proteins in brain of mice among groups. A: Western blot was performed to detect the level of CBX7 and Nrf2/HO-1 pathway-related proteins in the brain of mouse; B-E: Comparison of CBX7, HO-1, cytoplasmic Nrf2, and nuclear Nrf2 in the brain of mouse; The data were presented as the mean ± SD (n = 6); * p < 0.05 vs. WT + Sham group or CBX7-/- + Sham group; # p < 0.05 vs. the WT + middle cerebral artery occlusion group. CBX7: chromobox 7, Nrf2/HO-1: nuclear factor E2-related factor 2/hemeoxygenase-1, WT: wild-type.
Discussion
In this work, we, first, determined the level of CBX7 in mice after cerebral I/R injury and found MCAO injury triggered the significant up-regulation of CBX7 at different time points, and the peak level was reached at 24 h. Results from H-Y Chiu et al. 11 noted that CBX7 was up-regulated in the ischemic/hypoxic brain, and under the hypoxic condition, HIF-1α can activate the expression of CBX7 via binding to the promoter of CBX7. Importantly, HIF-1α is credited as a key transcription regulator under the hypoxic condition, 20 which played key roles in development of various ischemic/hypoxic disease or injury. 21 It has been confirmed that the attack of ischemia or hypoxia indicated the up-regulation of HIF-1α expression in the ischemic penumbra. 22 Therefore, the activation and induction of HIF-1α might provide a possibility for the up-regulation of CBX7 in mice after cerebral I/R injury, further demonstrating a crucial role of CBX7 in the cerebral I/R injury.
Then, WT and CBX7-/- mice were selected for this study, and the results demonstrated mice in the CBX7-/- + MCAO group had significant reductions in infarct size of brain, mNSS, and water content in the brain, as compared to the WT + MCAO group, indicating that CBX7 knockout could rescue the cerebral I/R injury. A similar study using animals with MCAO also demonstrated knockout of the Wip1 gene decreased the mNSS and cerebral infarct size, so as to exert the neuroprotective effect. 23 Consistently, the knockout of AQP4 could reduce the water content and the apoptosis of nerve cells in mice after cerebral I/R injury with increasing the survival rate of neurons and mitigating the neuroinflammation injury. 24
Massive generation of free radicals and oxidative stress constitutes the core pathogenesis of I/R injury that can induce the lipid peroxidation, mitochondrial injury, and cell apoptosis. 25 At the attack of cerebral ischemia, the excessive consumption of ROS consumes the endogenous anti-oxidative enzymes, resulting in alterations in the expression and activity of various enzymes, including SOD, GSH-Px, and CAT. 26 In turn, these decreased activities of enzymes would fail to eliminate ROS in excess, while the excessive ROS accumulation triggers oxidative stress and causes cell and tissue damages, or even cell death.27,28 However, MDA, as one of the final products in the peroxidation, could reflect the changes in the free radicals and the degree of damage to the tissue through its variation. 29 More importantly, recent evidence has shown a close correlation between oxidative stress and cell apoptosis. 30 During the cerebral ischemia, ROS could induce the neuronal apoptosis via many pathways.31,32 In this work, we found that after cerebral I/R injury, CBX7-/- mice had a decrease in the content of MDA but increases in the SOD activity and content of GSH-PX and CAT, presenting with a decrease in the cell apoptosis. Likewise, Rong Jiang et al. 33 found that lidocaine could reduce the level of oxidative stress in rats after cerebral I/R injury by reducing the level of MDA and increasing the levels of GSH-PX and SOD, thereby exerting the neuroprotective effect . Ya-Xuan Sun et al. 34 hold that liquiritin, possibly via increasing the levels of SOD, CAT, and GSH-Px and decreasing the content of MDA, can reduce the cell apoptosis in the brain to antagonize the oxidative stress and apoptosis. Taken together, CBX7 knockout may protect cells from the cerebral I/R injury via mitigating cell apoptosis and oxidative stress.
Last, we also found that in the MCAO mouse, HO-1, and nuclear Nrf2 were up-regulated, while Nrf2 in the cytoplasm was down-regulated. As reported, under the physiological condition, Nrf2 exists in the cytoplasm without activity by binding to Keap1 to sustain the low transcription activity. 35 However, damage to cells could dissociate the Nrf2 from Keap1, and Nrf2 could thus be translocated into the nucleus to bind to the anti-oxidant response elements (ARE) to initiate the transcription of gene harboring the anti-oxidant genes within the promotor,36,37 including HO-1, SODs, GPx, CATs, and NAD(P)H. 38 HO-1, as the major anti-oxidative stress pathway, can catalyze the oxidation of heme into dehydrobilirubin, free iron, and NO to protect cells from oxidative stress and inflammation. 39 Thus, after cerebral ischemia, Nrf2/HO-1 signaling pathway is activated, and as a result, Nrf2 is translocated into the nucleus to initiate the expression of HO-1, a downstream anti-oxidative gene, to mitigate I/R-induced brain injury, which is taken as a protective response.40,41 Our findings also supported that in the mice of CBX7-/- + MCAO group, Nrf2/HO-1 signaling pathway was further activated. Interestingly, Xiaofeng Lei et al. 42 reported that after I/R, an acute increase in ROS was seen in the brain, with up-regulations of Nrf2 in nuclei and HO-1, but the intervention of lycopene could further increase the expression of Nrf2 and HO-1 in the nucleus, thereby protecting tissues from I/R injury. Tremendous evidence has shown that the activation of Nrf2/HO-1 pathway could also improve the oxidative stress injury and cell apoptosis after I/R.43-45 Ye Zhang et al. 13 also reported that inhibiting CBX7 could activate Nrf2/HO-1 to mitigate the renal I/R injury. Thus, knockout of CBX7, possibly by activating the Nrf2/HO-1 pathway, can antagonize oxidative stress and apoptosis to protect the brain from I/R injury.
In summary, CBX7 is up-regulated in mice after cerebral I/R injury, while the knockout of CBX7 could exert neuro-protections from cerebral I/R, possibly via activating the Nrf2/HO-1 pathway, to reduce cell apoptosis and oxidative stress. Results of this study may provide novel theoretical evidence for the treatment of cerebral I/R injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
