Abstract
Background:
The risk of symptomatic infarct swelling has been reported to be higher in patients treated with recombinant tissue plasminogen activator (rt-PA). The aim of this study was to evaluate the timing of symptomatic infarct swelling after rt-PA treatment.
Methods:
We retrospectively analyzed 14 868 patients with acute ischemic stroke from a stroke registry databank. We recruited patients with massive middle cerebral artery (MCA) infarction and symptomatic infarct swelling and excluded those with parenchymal or symptomatic hemorrhage. Multiple linear regression and multivariate logistic regression analyses were used to estimate the impact of rt-PA on the timing of symptomatic infarct swelling.
Results:
A total of 23 patients with rt-PA treatment and 117 patients without rt-PA treatment were included. The rt-PA treatment group had a lower rate of coronary artery disease (8.7% vs 32.5%; P = .023), lower severity of baseline National Institutes of Health Stroke Scale score (19 vs 23; P = .014), shorter duration of infarct swelling (27.6 vs 45.4 hours; P < .001), and higher rate of hemicraniectomy surgery (65.2% vs 28.2%; P =.001) than those without rt-PA treatment. After adjusting for variables in multiple linear regression analysis, rt-PA treatment and an elevated C-reactive protein level were associated with early symptomatic infarct swelling (P = .014 and P = .041, respectively). The rt-PA treatment was an independent factor related to early symptomatic infarct swelling within 36 hours (P = .005; odds ratio [OR]: 5.3; confidence interval [CI]: 1.65-17.0) or 48 hours (P = .009; OR: 16.4; CI: 2.00-134).
Conclusion:
Intravenous rt-PA treatment may hasten the onset of cerebral edema and subsequent cerebral herniation in large MCA territory infarction.
Background
Intravenous (IV) recombinant tissue plasminogen activator (rt-PA) is one the most effective treatments for ischemic cerebral infarction within 4.5 hours of symptom onset. 1,2 Although thrombolysis is of net benefit for patients with ischemic stroke, it has also been shown to increase the risk of early symptomatic intracranial hemorrhage and mortality within 7 days of stroke onset. 3 The third International Stroke Trial (IST-3) reported that in addition to symptomatic intracranial hemorrhage, symptomatic infarct swelling, defined as the worsening of neurological status accompanied by major infarct swelling on imaging, may contribute to a higher mortality rate within 7 days of stroke onset in patients with thrombolysis. 4
Space-occupying cerebral edema occurs in 5% of all ischemic strokes and is termed malignant middle cerebral artery (MCA) infarction. It has been reported to be associated with a high mortality rate ranging from 41% to 79% and to most frequently occur within 48 hours of symptom onset. 5,6 In the European Cooperative Acute Stroke Study (ECASS) I trial, herniation was the most frequent cause of death during the first 7 days of stroke in both placebo and thrombolysis groups. 7 In an analysis of the ECASS I trial, neurological deterioration within 24 hours of stroke was independently associated with brain swelling, with a 44% increased risk of early mortality. 8 Taken together, rt-PA may result in the early onset of brain edema and subsequently poor outcomes. To the best of our knowledge, no study has investigated the timing of symptomatic brain edema in patients with thrombolysis. Therefore, the aim of this study was to evaluate the timing of symptomatic infarct swelling after rt-PA treatment in patients with large territorial infarcts.
Patients and Methods
Study Design and Patient Selection
We retrospectively analyzed 14 868 patients with acute ischemic stroke who arrived at a Chang Gung Medical System-affiliated hospital within 24 hours after stroke onset from January 2007 to April 2014. The clinical data of these patients with acute stroke were prospectively registered in the Chang Gung Stroke Registry database from the Keelung, Linkou, Chiayi, and Kaohsiung branches in Taiwan. At these branches, 527 patients with consecutive ischemic stroke received IV rt-PA treatment. The study was approved by the institutional review board of Chang Gung Memorial Hospital (102-1684B).
All of the patients underwent a standard neurological examination, electrocardiography, blood chemistry analysis, and noncontrast computed tomography (CT) in the emergency department. Baseline clinical status was assessed using the National Institutes of Health Stroke Scale (NIHSS) score by a trained neurologist. Data on demographics, vascular risk factors, and medication use were also recorded. The inclusion criteria were (1) age between 18 and 79 years, (2) symptoms and signs of clinically definite acute stroke with an involved MCA territory, (3) definite stroke onset time, and (4) cerebral edema occurring within the first 7 days of stroke onset. The inclusion and exclusion criteria for IV rt-PA treatment were mainly adopted from the National Institute of Neurological Disorders and Stroke protocol 1 and the protocol of the Taiwan Guidelines for the Management of Stroke. 9 However, a few patients with a NIHSS score over 25 points also received IV rt-PA treatment. Because the onset time of brain swelling is presumably delayed in elderly patients with brain atrophy, patients in the control group aged older than 80 years were therefore excluded to avoid selected bias.
The patients who received treatment were given a 0.6- to 0.9-mg/kg IV dose of rt-PA and then admitted to the neurology intensive care unit (ICU) for observation. The majority of the patients were given 0.9 mg/kg as the standard dose. A few patients who were older or had higher NIHSS scores received low-dose treatment (0.6 mg/kg), and this was based on the judgment of the clinical physician in line with the conclusion of Japan Alteplase Clinical Trial. 10 In the nonthrombolysed patients, an antiplatelet agent, mainly aspirin at 100 or 200 mg/d, was used during the acute stage. All patients were admitted to the stroke unit or neurology ICU and were treated according to the protocols of the Taiwan Guidelines for the Management of Stroke. 9
Definition and Outcome Measures
We classified the patients into 2 groups: those who received IV rt-PA and those who did not. During hospitalization, all of the patients were evaluated daily for NIHSS score and Glasgow Coma Scale (GCS) score by neurologists. In cases of clinical neurological deterioration, defined as an increase in NIHSS score ≥4, increase in GCS score ≥2, or asymmetric pupillary size, we performed CT and completed a laboratory survey to exclude medical causes of clinical deterioration other than brain swelling. The CT scans were retrospectively assessed by an experienced neurologist (T. C. Lin) who was blinded to the clinical information.
The time from neurological deterioration to follow-up CT was less than 60 minutes. To verify the onset time of cerebral edema, we recorded the time of the follow-up CT scan in which ventricular compression, midline shift, or herniation was noted. The patients with a parenchymal hemorrhage with a space-occupying effect and symptomatic hemorrhage detected on CT scans, defined as ECASS II, 11 were excluded to prevent confounding with an intracerebral hemorrhage. The patients with a hemorrhagic infarction, defined as petechiae within the infarct area but no space-occupying effect, were identified for further analysis.
Statistics
All values are expressed as mean ± standard deviation or median and interquartile range. All statistical analyses were performed with χ2 test or Fisher exact test for categorical variables as appropriate. For continuous variables, we used the independent samples Student t test or Mann-Whitney U test after testing for normality. To evaluate an independent effect of rt-PA on the timing of symptomatic infarct swelling, we performed multiple linear regression analysis to estimate other baseline variables. To assess the influence of rt-PA treatment on early symptomatic infarct swelling within 24, 36, or 48 hours, a multivariate logistic regression model was constructed adjusting for age, initial NIHSS score, hemorrhagic infarct, and any factor with a P value <.1 in the univariate logistic regression analysis, which was applied to analyze age, sex, rt-PA treatment, hypertension, diabetes mellitus, coronary artery disease, congestive heart failure, valvular heart disease, atrial fibrillation, hyperlipidemia, smoking, old stroke, systolic and diastolic blood pressure, initial NIHSS score, serum white blood cell count, serum total cholesterol, serum triglycerol, serum creatinine, serum alanine aminotransferase, glycohemoglobin, and serum C-reactive protein (CRP), given that some of these variables have previously been associated with the development of infarct swelling. 12 Significance was defined as a P value <.05. All statistical analyses were performed using SPSS software version 18.0 for Windows (SPSS, Chicago, Illinois).
Results
After excluding the patients with a parenchymal hemorrhage, a total of 140 patients with symptomatic brain edema were included, including 23 with IV rt-PA treatment. Petechial hemorrhagic infarction was observed in 6 (26.1%) patients with thrombolysis and 46 (39.3%) nonthrombolysed patients. The patients’ characteristics and vascular risk factors were comparable between the 2 groups with or without thrombolysis, both in those with or without hemorrhagic infarct (Table 1). In the patients with hemorrhagic infarct, those with thrombolysis were younger (60.1 ± 12.4 years vs 67.6 ± 10.6 years; P = .003), had a lower rate of coronary artery disease (8.7% vs 32.5%; P = .023), and lower baseline NIHSS score (median NIHSS score: 19 vs 23; P = .014) but significantly shorter time interval between symptom onset and initiation of infarct swelling (median 27.6 vs 45.4 hours; P < .001). Patients with thrombolysis also had higher rates of early infarct swelling (<24, <36, or <48 hours) and hemicraniectomy surgery. Even after excluding the patients with a hemorrhagic infarct, patients with thrombolysis still had the same results of a lower rate of coronary artery disease, lower baseline NIHSS score, shorter time interval to infarct swelling, higher rate of early infarct swelling (<36 or <48 hours), and higher rate of hemicraniectomy surgery.
Demographic Data of the Patients With and Without rt-PA Treatment.a
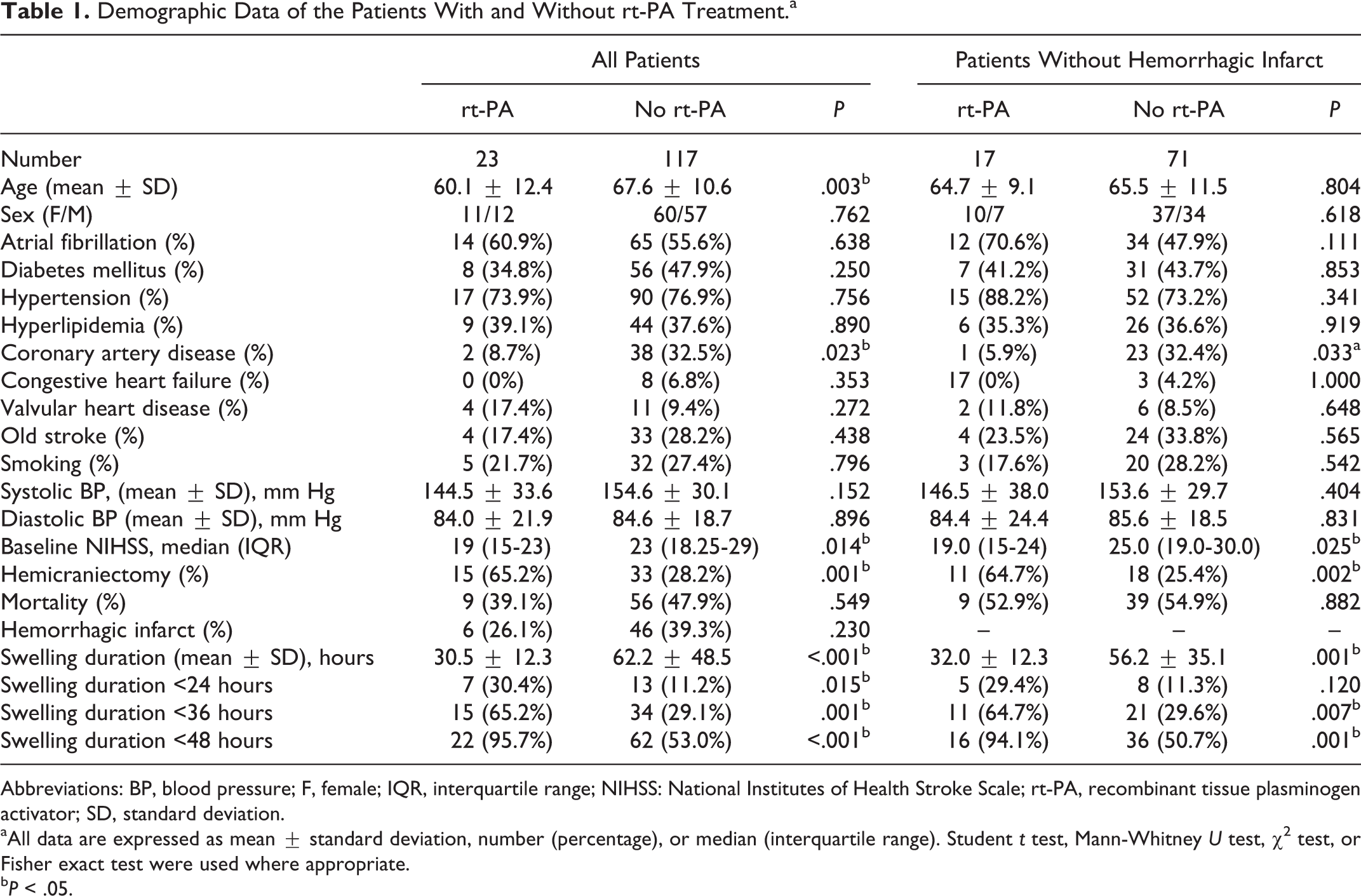
Abbreviations: BP, blood pressure; F, female; IQR, interquartile range; NIHSS: National Institutes of Health Stroke Scale; rt-PA, recombinant tissue plasminogen activator; SD, standard deviation.
aAll data are expressed as mean ± standard deviation, number (percentage), or median (interquartile range). Student t test, Mann-Whitney U test, χ2 test, or Fisher exact test were used where appropriate.
b P < .05.
Overall, 7 (30.4%) of the patients with thrombolysis had infarct swelling within 24 hours of symptom onset, compared to 13 (11.2%) of the nonthrombolysed patients. By 48 hours, most of the patients with thrombolysis had infarct swelling (95.7%) compared to the nonthrombolysed patients (53.0%). Although more patients with thrombolysis underwent hemicraniectomy surgery, the overall mortality rate was not significantly different between the 2 groups (P = .549).
In multiple linear regression analysis (Table 2), rt-PA treatment and an elevated CRP level were significantly associated with a shorter duration of symptomatic infarct swelling (P = .014 and P = .041, respectively) after adjusting for other variables.
Simple and Multiple Linear Regression Analyses of Factors Associated With the Timing of Symptomatic Infarct Swelling.
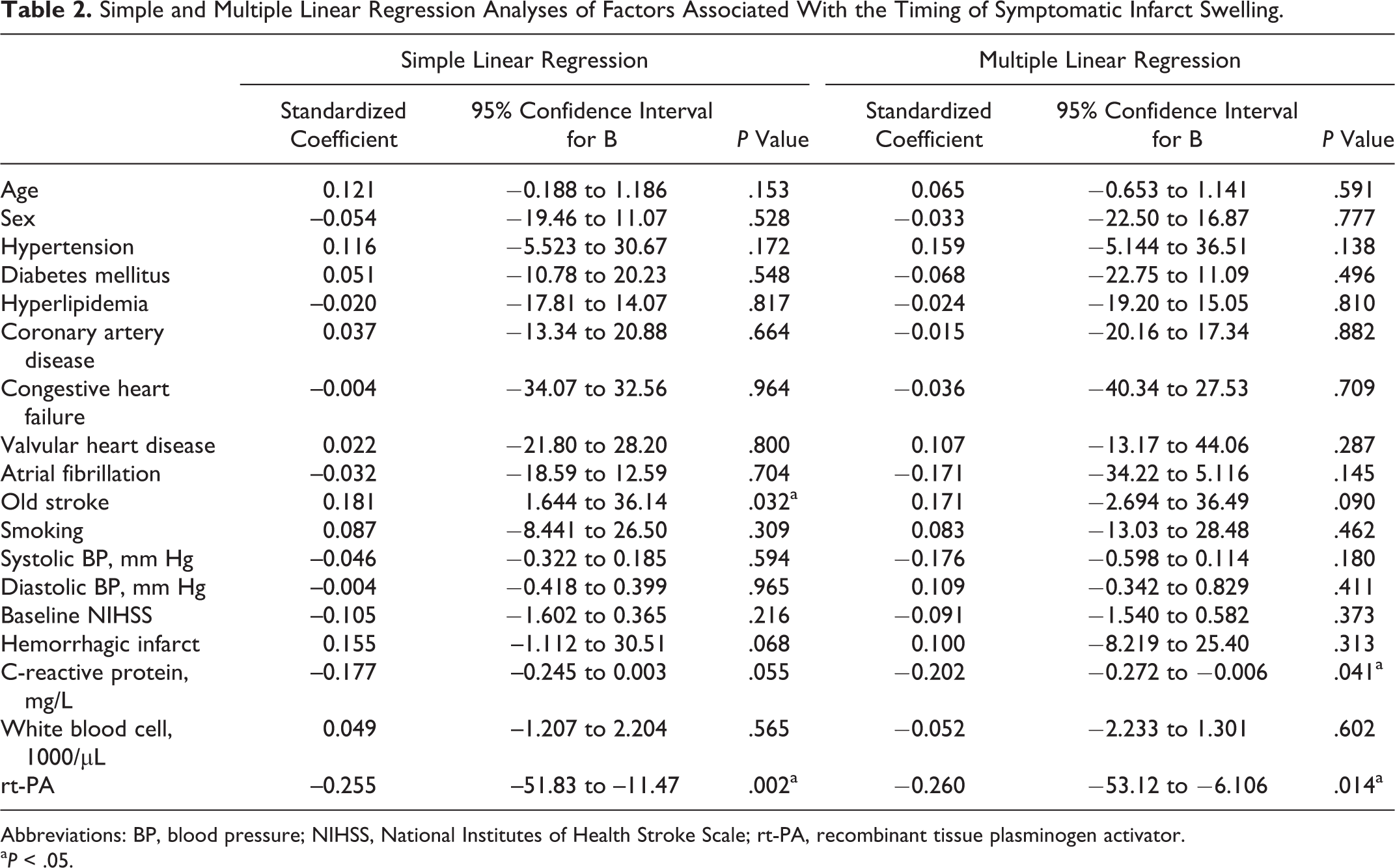
Abbreviations: BP, blood pressure; NIHSS, National Institutes of Health Stroke Scale; rt-PA, recombinant tissue plasminogen activator.
a P < .05.
In univariate logistic regression, rt-PA treatment (P = .02; odds ratio [OR]: 21.2; confidence interval [CI]: 1.21-10.1) and higher initial NIHSS score (P = .02; OR: 0.92; CI: 0.85-0.99) were associated with early symptomatic infarct swelling within 24 hours, and rt-PA treatment (P = .002; OR: 4.58; CI: 1.78-11.8) and younger age (P = .011; OR: 0.96; CI: 0.93-0.99) were associated with early symptomatic infarct swelling within 36 hours. However, only rt-PA treatment (P = .004; OR: 19.5; CI: 2.55-149) was associated with early symptomatic infarct swelling within 48 hours. In multivariate logistic regression analysis (Table 3), rt-PA treatment was an independent factor associated with early symptomatic infarct swelling within 36 hours (P = .005; OR: 5.3; CI: 1.65-17.0) or within 48 hours (P = .009; OR: 16.4; CI: 2.00-134), after adjusting for baseline confounders.
Multivariate Logistic Regression Model for Predicting Early Symptomatic Infarct Swelling.a
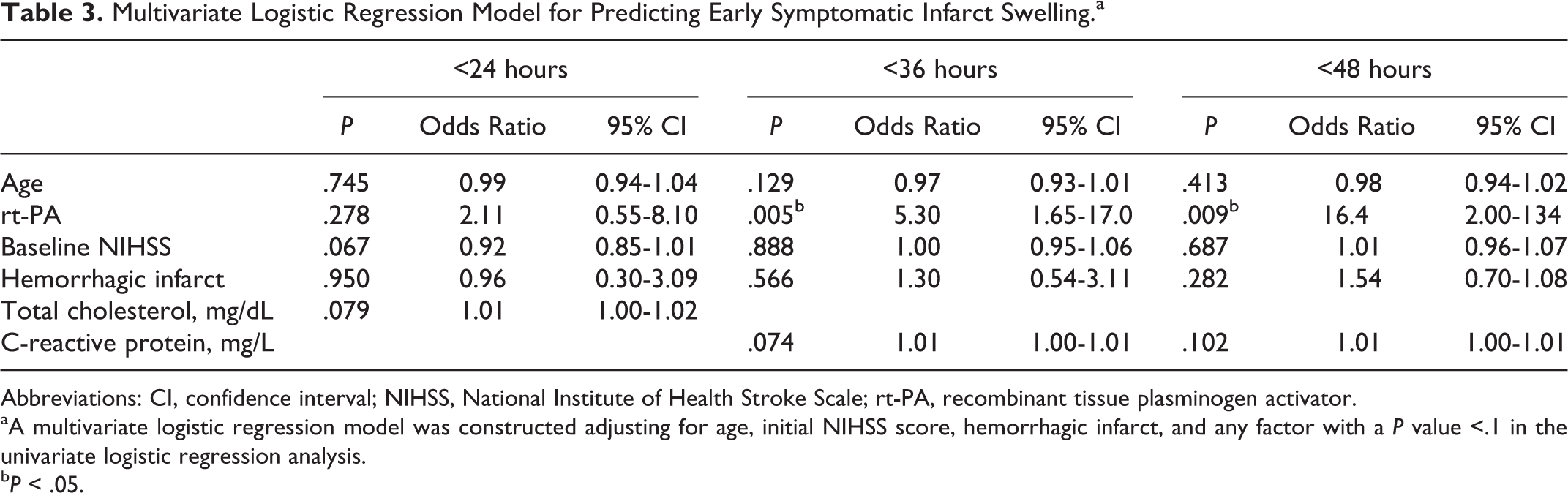
Abbreviations: CI, confidence interval; NIHSS, National Institute of Health Stroke Scale; rt-PA, recombinant tissue plasminogen activator.
aA multivariate logistic regression model was constructed adjusting for age, initial NIHSS score, hemorrhagic infarct, and any factor with a P value <.1 in the univariate logistic regression analysis.
b P < .05.
Discussion
Symptomatic infarct swelling accounts for most cases of mortality in patients with massive MCA infarction in the first week during hospitalization, with most occurring within 48 hours of symptom onset. A previous study suggested that early hemicraniectomy within 48 hours was beneficial for patients with massive MCA infarction. 13 However, there are limited data on the timing of the onset of symptomatic infarct swelling in patients with thrombolysis. The present study revealed that thrombolysis may hasten the onset of symptomatic infarct swelling in patients with acute massive MCA infarction, even after adjusting for baseline confounding factors. Therefore, to improve the outcomes of patients with acute massive MCA infarction after IV thrombolysis treatment but without clinical improvement, early follow-up brain imaging and aggressive treatment such as early hemicraniectomy may be required.
Several early clinical and radiological predictors of the development of symptomatic infarct swelling and poor outcome have been reported. 12,14 The infarct size seems to play a critical role in the development of infarct swelling in massive MCA infarcts. Reliable predictors include an infarct size larger than 50% of the MCA territory on CT and an infarct volume in diffusion-weighted imaging greater than 145 mL within 14 hours 15 or greater than 82 mL within 6 hours of stroke onset. 16 Although early recanalization may be associated with a smaller infarct and better outcomes in MCA territory infarcts, 17 spontaneous and induced recanalization may induce vasogenic edema due to blood–brain barrier (BBB) disruption, thereby worsening the edema of the original cerebral infarction. 18 –20 The IST-3 study reported that in addition to symptomatic intracranial hemorrhage, neurological deterioration due to swelling of the infarct occurred more frequently in patients with rt-PA treatment. 4 The Diffusion and Perfusion Imaging Evaluation for Understanding Stroke Evolution (DEFUSE) study reported that recanalization was beneficial in patients with a small infarct and existing penumbra, although not in those with an infarct size over 100 mL regardless of whether or not a penumbra existed, in which case recanalization was harmful and usually resulted in cerebral edema and hemorrhage after rt-PA treatment. 21 Taken together, the infarct size seems to be the most important factor to predict symptomatic infarct swelling in massive MCA infarction after thrombolysis and that reperfusion injuries may worsen the edema. In the current study, we found a significantly faster development of symptomatic infarct swelling in the patients with a large MCA territory infarction after thrombolysis. However, it is unclear whether the earlier timing of symptomatic infarct swelling was related to the reperfusion injury or rt-PA itself.
Thrombolysis treatment was an independent predictor of an earlier onset of infarct swelling in this study. In addition to reperfusion injury, rt-PA itself may play a harmful role in the development of infarct swelling following ischemic stroke. The adverse effects of rt-PA may be mediated through matrix metalloproteinases (MMPs) that increase BBB permeability by degrading components of the extracellular matrix in endothelial cells. 22 Kidwell et al investigated the effect of thrombolysis and mechanical embolectomy on BBB disruption and confirmed an independent association between thrombolysis therapy and BBB disruption independent of reperfusion. 23 Based on these observations, we hypothesize that rt-PA may have induced BBB disruption in ischemic areas immediately after stroke, resulting in the earlier onset time of infarct swelling in our patients.
Cerebral ischemia is accompanied by a marked inflammatory reaction that is initiated by the ischemia-induced expressions of cytokines, adhesion molecules, and other inflammatory mediators, leading to the accumulation of inflammatory cells. 24 Inflammatory cells such as microglia and mast cells may regulate BBB permeability and brain edema formation. 25,26 In addition, elevated blood levels of systemic inflammatory markers such as interleukin 6 and CRP have been associated with an unfavorable functional outcome and increased mortality after stroke. 27 Patients with an increased CRP level have been reported to have a higher risk of mortality after IV rt-PA treatment. 28 Our results revealed that an elevated CRP level was an independent factor associated with early symptomatic infarction swelling, suggesting that inflammatory reactions may play a critical role in the development of cerebral edema, particularly in patient with IV rt-PA treatment, as rt-PA is known to promote neutrophil degranulation and MMP release. 29
Even though more of our patients with thrombolysis received surgical decompression, the mortality rate was not different between the 2 groups. Although treatment of space-occupying infarction with surgical decompression has been proven to reduce mortality, 13 the optimal timing of decompressive surgery has yet to be elucidated. 12 If the surgical procedure is performed after the development of herniation, it is certainly too late. Although our findings are unable to fully support the benefit of surgical intervention in infarct swelling following thrombolysis, they may suggest that intensive monitoring is mandatory for patients with large MCA infarction who fail to improve after thrombolysis.
There are several limitations to this study due to its retrospective design and small sample size. This methodology may diminish the generalizability of our findings. However, we recruited patients from 4 hospitals located throughout Taiwan to strengthen the study’s external validity. In addition, there was sufficient power of our data to increase the internal validity, despite the selection bias with the case–control design. We believe that the results may be applied to most Asian patients, however, further studies are needed to validate whether our results are applicable to other populations. Given that data on the baseline infarct size were unavailable, it is hard to say whether the infarct size was smaller in the rt-PA group despite a lower baseline NIHSS score. Furthermore, follow-up angiography findings were also unavailable to determine whether the brain edema was due to reperfusion injuries or rt-PA alone. In addition, the effect of reperfusion injury on BBB disruption has to date mostly been studied through experimental models, and it has not been proven in humans. We cannot confirm the mechanism by which rt-PA damaging the BBB in this study, however, the early onset of symptomatic edema may be due, in part, to thrombolysis. Finally, it is possible that patients with thrombolysis may receive more attention during hospitalization, thus introducing some degree of bias in the detection of cerebral edema, which may have effected the study outcome. Despite intensively monitoring patients with massive MCA infarction, there could be an inherited uncertainty in determining the exact timing of symptomatic infarct swelling.
In conclusion, this study suggests that thrombolysis therapy is an independent predictor of the early onset of symptomatic brain edema in patients with acute massive MCA infarction. Intensive monitoring and aggressive interventions should be considered for these patients.
Footnotes
Acknowledgments
The authors would like to thank all of the Chang Gung Stroke Registry databank collaborators for their support and help in the realization of the original database. The authors also thank Yi-Chen Kuo for assisting us with this study.
Author Contributions
Yen-Chu Huang contributed for designing the study, data analysis, and revision of the manuscript. Ting-Chun Lin contributed for drafting the manuscript and analysis of data. Ya-Hui Lin and Chen Jin-Hua (Biostatistics Center Taipei Medical University) and Hsu-Huei Weng contributed for statistical analysis. Jiann-Der Lee and Rey-Yue Yuan contributed for critical revision of the manuscript. Ying-Chih Huang, Meng Lee, Chih-Ying Wu, Huan-Lin Hsu, Chia-Yu Hsu, Tsong-Hai Lee, Shan-Jin Liu, Yeu-Jhy Chang, Chien-Hung Chang, Tsung-I Peng, Chia-Wei Liou, Ku-Chou Chang, and Yi-Ting Pan contributed for acquisition of data. All authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Chang Gung Memorial Hospital grants CMRPG6D0041 and CORPG6D0281.
