Abstract
Eleutheroside B, also known as syringin, has been shown to have various pharmacological activities such as anti-inflammatory, anti-irradiation and antidepressant, but there are few studies on its anti-cancer activity. Its anti-tumor effect on SMMC-7721 cells has not been revealed. Moreover, whether it induces autophagy is still unclear. Thus, the present study investigated whether Eleutheroside B induces apoptosis, autophagy and cellular motility in SMMC-7721 cells and HeLa cells, and explored the underlying molecular mechanisms. SMMC-7721 cells and HeLa cells treated with Eleutheroside B and cell viability measured by MTT assay and trypan blue dye exclusion assay. Apoptosis checked by flow cytometry combined, fluorescent staining. Apoptotic signal proteins and autophagy proteins were checked by Western blot. This study showed that Eleutheroside B inhibited the cell proliferation and blocked cell cycle, migration and invasion as well. Moreover, Eleutheroside B induced apoptosis in SMMC-7721 cells and HeLa cells. It upregulated Bax expression, while simultaneously decreasing Bcl-2 expression. Further elucidation of the mechanism revealed that Eleutheroside B induced mitochondrial dysfunction, with mitochondrial membrane potential collapse and cytochrome c release, suggesting that Eleutheroside B induced apoptosis by triggering mitochondrial pathway. Most importantly, Eleutheroside B could induce autophagy in SMMC-7721 cells and HeLa cells. Taken together, these results suggested Eleutheroside B is a potential therapeutic candidate for HCC and Human cervical cancer.
Introduction
Cancer has been regarded as one of the most important public health problems, and the incidence and mortality of cancer are increasing rapidly in the world. The most important factor affecting the choice of treatment is the degree of tumor differentiation (preoperative stage). As cancer is diagnosed, various treatment options that may cure cancer are considered. There are three basic strategies for the treatment of cancer: chemotherapy, radiotherapy and surgery, each of which could be used alone or in combination with other methods. 1 Surgery and radiotherapy are part of local treatment. They are only effective for local tumors, but they are not effective for potential metastatic lesions and tumors with clinical metastasis. Chemotherapy belongs to systemic therapy. With any administration method, Chemotherapeutic drugs are spread through the bloodstream and affect most organs and tissues throughout the whole body. Therefore, chemotherapy, which is known to us, is the main treatment for some middle and advanced tumors with systemic dissemination tendency and metastasis. The two major categories of chemotherapeutic drugs are synthetic drugs and natural drugs. Traditional natural products are the source of drugs for the treatment of many diseases including cancer, and plants are an important source of new natural products. Therefore, Chinese herbal medicine for the treatment of cancer has great potential.
Eleutheroside B (C17H24O9, Figure 1(a), is also calledSyringin found in Acanthopanaxsenticosus Harms (Araliaceae), a woody medicinal plant originally from Northeastern Asia. In traditional medicine, Eleutheroside B (Syringin) is widely used to treat a variety of diseases, is a naturally occurring phenylpropanoid glucoside that showed extensive attention because of its rich biological activities. Eleutheroside B (Syringin) has been proven possessing various pharmacological activities such as anti-inflammatory, anti-irradiation, antioxidant and anti-allergic properties.2,3 It also can be used to enhance memory, relieve fatigue, improve human cognition and protect cardiovascular and cerebrovascular against ischemia damage, etc. Acanthopanaxsenticosus Harms (Araliaceae), also known as Siberian Ginseng, Eleutherococcus senticosus, and Ciwujia in Chinese, is a traditional Chinese medicine with the effects of invigorate, strengthen the spleen, and nourish kidney.
4
Acanthopanaxsenticosus Harms (Araliaceae) is widely distributed in Japan, China, Korea and the Far East of Russia. At present, there are few studies on the anticancer activity of Eleutheroside B. Studies have shown that Eleutheroside B has been proved to be cytotoxic to human breast cancer cells, prostate (Du145), glioblastoma (A1235).5-7 However, the systematic screening remains unknown and whether it causes autophagy in tumor cells remains unclear. In this study, a variety of cells were screened, and sensitive strains were selected for analysis. We studied whether Eleutheroside B can regulate cellular motility and apoptosis, autophagy of SMMC-7721 cells and HeLa cells, and examined its potential molecular mechanisms. Eleutheroside B inhibits the survival of six tumor cells. (A) Structure formula of Eleutheroside B. (B) Different concentrations of Eleutheroside B on six kinds of tumor cells for 24 h, detected by MTT assay. (C) Eleutheroside B on SMMC-7721 cells, HeLa cells for 6 h, 12 h, 24 h, detected by trypan blue dye exclusion assay. * p < 0.05, ** p < 0.01, *** p <0.001 compared to untreated cells.
Materials and Methods
Materials
Eleutheroside B (≥98% purity by HPLC) was purchased from MREDA (China, Beijing). The stock solution was stored at −20°C with a concentration of 54 mm in DMSO. The final concentration of DMSO did not exceed 0.1% in the culture medium.
Cell culture and cell viability assay
Human hepatoma HepG2, Bel-7402, Huh7 and SMMC-7721 cell lines, human breast adenocarcinoma MDA-MB-231 cells line, human cervical cancer HeLa cell line were obtained from the cryopreservation in our laboratory and supplemented with 10% FBS in DMEM high-glucose medium (Invitrogen-Gibco, Paisley, UK) at a humidified 5% CO2 atmosphere and 37°C. In short, cells were inoculated on 96-well culture plates and treated with different concentrations of Eleutheroside B for 24 h. One day after treatment, a solution containing 10% MTT (sigma-aldrich, St Louis, MO, USA) solution was added and incubated at a humidified 5% CO2 atmosphere and 37°C for 4 h. After the end of the incubation period, viability was measured using a microplate reader (TECAN, Switzerland) and the absorbance of 570 nm was recorded.
Trypan blue dye exclusion assay
After treatment with different concentrations of Eleutheroside B (0–15 μM) for 24 h, 0.4% trypan blue (sigma, St Louis, MO, USA) were mixed into SMMC-7721 cells and HeLa cells. Finally, stained (nonviable) cells and unstained (viable) cells were counted separately under inverted microscope (Olympus, Tokyo, Japan).
Clonogenic survival assay
A clonogenic survival assay measure the ability of a single cell to grow into a colony. Cells were seeded on a 6-well culture plate at a humidified 5% CO2 atmosphere and 37°C with different concentrations of Eleutheroside B (0–15 μM) for 24 h. Then, the old medium was replaced with fresh medium and cells fixed with 4% Paraformaldehyde. Finally, Colony formation was observed using an inverted microscope (Olympus, Tokyo, Japan).
Cell morphological assessment and Hoechst 33,342 staining
SMMC-7721 cells and HeLa cells were inoculated on a culture plate and treated with different concentrations of Eleutheroside B (0–15 μM) for 24 h. After the cells were fixed, the nuclei stained with Hoechst 33,342 solution (Sigma, St Louis, MO, USA). A fluorescence microscopy (Olympus, Tokyo, Japan) was used to examine the morphological changes of apoptosis and nuclear condensation.
AO/EB double-staining of apoptotic cells
SMMC-7721 cells and HeLa cells were treated with Eleutheroside B at specified concentrations (0–15 μM) for 24 h. After the cells were fixed, stained with AO/EB double-staining (Solarbio, Beijing, China). The nuclear changes were observed using fluorescence microscopy (Olympus, Tokyo, Japan). The chromatin of living cells was green and normal in structure. The nuclear chromatin of early apoptotic cells nuclei was green and shrunken in a round shape. The chromatin of late apoptotic cells was orange and shrunken in a round shape. The chromatin of dead cells was orange and normal in structure.
Apoptotic cell assay
SMMC-7721 cells and HeLa cells were inoculated on a 6-well culture plate and treated with 0–15 μM Eleutheroside B for 24 h. Then, according to the instructions provided by the manufacturer, use the Annexin V and Dead Cell Assay Kit (BD Pharmingen, NJ, USA) to detect apoptotic cells. Cells were incubated with Annexin V and Dead Cell Detection Reagent in the dark at room temperature for 20 min. Finally, apoptotic cells were detected by flow cytometry (Beckman Coulter, Pasadena, California, USA) and the results were analyzed using summit 5.2 and FlowJo®v. 10.
Cell cycle analysis
The cells were inoculated on a 6-well culture plate and treated with Eleutheroside B for 24 h. Subsequently, the cells were washed with PBS and add 200 μL Cell Cycle reagent (BD, Franklin Lake, New Jersey, USA) in the dark at room temperature for 30 min. Finally, the cell cycle was observed and analyzed by flow cytometry (Beckman Coulter, Pasadena, California, USA).
Wound healing assay
Cells were inoculated on 6-well culture plate for 24 h. Next, make scratches with the gun head and treated with different concentrations of Eleutheroside B (0–15 μM) for 24 h. The cell migration at 0 and 24 h was observed under an inverted microscope (Olympus, Tokyo, Japan).
Transwell migration and invasion assay
SMMC-7721 cells and HeLa cells were inoculated on a 6-well culture plate and treated with various concentrations of Eleutheroside B (0–15 μM) for 24 h. Then, the upper chamber of the Transwell plate was coated with Matrigel TM (BD Pharmingen, NJ, USA) for 1 h to form an 8 micron upper cavity. DMEM medium containing 15% FBS was added to the lower chamber and cells in DMEM medium containing 0.5% FBS were added to the upper chamber and cultured 24 h. Next, the upper chamber was treated with methanol, and the cells were stained with 0.1% Giemsa (Biotopped, Beijing, China). The number of invading and migrating cells was observed by inverted microscope (Olympus, Tokyo, Japan).
Western blot analysis
SMMC-7721 cells and HeLa cells treated with Eleutheroside B (0–15 μM) for 24 h were lysed with RIPA buffer containing protease and phosphatase inhibitors (Sigma, St Louis, MO, USA). The extracted proteins were separated by 10–15% SDS polyacrylamide gel electrophoresis, and then proteins were transferred from the gel to PVDF membrane. Next, the PVDF membrane was incubated with specific primary antibodies (1:1000 for p53, Cyclin D1, Bcl-2, Bax, Cytochrome C, Survivin, E-cadherin, p62, LC3) overnight and secondary antibodies (1:1000) at room temperature for 1 h. Proteins demonstrated and measured by the Azure Biosystems cSeries system (Azure Biosystems, Dublin, California, USA). Primary antibodies included p53, Bcl-2, Bax, Cyclin D1, Cytochrome C, Survivin, E-cadherin (Cell Signaling Technology, Danvers, MA, USA), LC3, p62 (Proteintech, Chicago, USA).
Statistical analysis
All data, obtained from at least three independent experiments, were presented as the mean ± SD. Data were analyzed using one-way analysis of variance (ANOVA) followed by the SPSS 19.0 software (SPSS, Inc, Chicago, IL). p < 0.05 was considered significant.
Results
Eleutheroside B inhibits the viability of tumor cells
The IC50 of Eleutheroside B against different cells.

Eleutheroside B inhibits the proliferation of SMMC-7721 cells and HeLa cells
To evaluate the Influence of Eleutheroside B on the proliferation of SMMC-7721 and HeLa cells, SMMC-7721 cells and HeLa cells were treated with Eleutheroside B (0–15 μM) for 24 h, and the effect on cell clone formation was detected. It was found that Eleutheroside B significantly reduced colonies in a concentration-dependent manner (Figures 2(a) and (b)). In addition, Eleutheroside B significantly increased the proportion of G0/G1 phase cells in a dose-dependent (Figures 2(c) and (d)). Therefore, we detected the expression of cyclin D1, which regulates the transition from G0/G1 phase to G1/S phase. The results of Western blot showed that Eleutheroside B significantly inhibited the expression of cyclin D1 in SMMC-7721 cells and HeLa cells (Figure 2(e)). Effects of Eleutheroside B on clonogenic survival and cell cycle analysis in SMMC-7721 and Hela cells. (A) SMMC-7721 cells were seeded on a cuture plate and treated with Eleutheroside B for 24 h and then stained with Giemsa to detect the effect on cell clone formation using an inverted microscope. (B) HeLa cells were seeded on a cuture plate and treated with Eleutheroside B for 24 h and then stained with Giemsa to detect the effect on cell clone formation using an inverted microscope. The data are presented as mean ± SD of three independents. * p < 0.05, ** p < 0.01, *** p < 0.001 compared to untreated cells. (C) SMMC-7721 cells were seeded on a cuture plate and treated with Eleutheroside B for 24 h and the cell cycle status were detected by flow cytometry. (D) HeLa cells were seeded on a cuture plate and treated with Eleutheroside B for 24 h and the cell cycle status were detected by flow cytometry. The data are presented as mean ± SD of three independents. * p < 0.05, ** p < 0.01, *** p < 0.001 compared to untreated cells. (E) SMMC-7721 cells and HeLa cells were treated with Eleutheroside B for 24 h and the expression of Cyclin D1 was detected by Western blot.
Eleutheroside B induced apoptosis in SMMC-7721 and HeLa cell
To test the ability of Eleutheroside B on inducing apoptosis of SMMC-7721 and HeLa cells, SMMC-7721 and HeLa cells were incubated with different concentrations of Eleutheroside B (0–15 μM) for 24 h, and the effect on the formation of apoptotic bodies was detected. The results of Hoechst 33,342 staining showed that Eleutheroside B increased the formation of apoptotic bodies in SMMC-7721 cells (Figure 3(a)) and HeLa cells (Figure 3(b)). The results of AO/EB double-staining showed that the apoptotic and dead cells of SMMC-7721 cells (Figure 3(c)) and HeLa cells (Figure 3(d)) increased significantly with the increase of Eleutheroside B concentration. In addition, the results of flow cytometry with Annexin V/PI staining further confirmed the apoptosis inducing ability of Eleutheroside B in SMMC-7721 and HeLa cells (Figures 3(e) and (f)). Apoptosis of SMMC-7721 and HeLa cells induced by different concentrations of Eleutheroside B for 24 h. The morphological changes of SMMC-7721 cells (A) and HeLa cells (B) were observed under microscope and stained with Hoechst33342 fluorescent nucleus. The arrow represents apoptotic cell (× 200). SMMC-7721 cells (C) and HeLa cells (D) were stained with AO/EB. The arrow represents apoptotic cell (× 200). The number of apoptotic cells in SMMC-7721 cells (E) and HeLa cells (F) was determined by flow cytometry with Annexin V/PI double-staining. * p < 0.05, ** p < 0.01, *** p < 0.001 compared to untreated cells.
Mitochondrial pathway is involved in the apoptosis induced by Eleutheroside B
To identify the underlying mechanism, apoptosis related proteins were detected. Eleutheroside B treatment group decreased the level of Survivin in SMMC-7721 cells, and increased the level of p53 and cytochrome C in SMMC-7721 cells (Figure 4(a)). Mitochondria are also known to play a key role in activating of apoptosis, among which the ratios and the expression level of Pro-apoptotic proteins Bax and anti-apoptotic proteins Bcl-2 and are the key factors.
8
Inthe present study, Eleutheroside B up-regulated Bax and simultaneously decreasing Bcl-2, indicating that Eleutheroside B elevated the ratio of Bax/Bcl-2 (Figure 4(a)). A similar effectof Eleutheroside B was also showed in HeLa cells (Figure 4(b)). These data suggest that Eleutheroside B induces apoptosis through the mitochondrial pathway. Next, we detected mitochondrial membrane potential with fluorescent probe JC-1, the collapse of mitochondrial membrane potential is an important parameter of mitochondrial dysfunction related to apoptosis. Compared with the control group, the number of SMMC-7721 cells and HeLa cells with reduced mitochondrial membrane potential treated with Eleutheroside B increased (Figures 4(c) and (d)). These results suggest that Eleutheroside B induces apoptosis through mitochondrial apoptosis pathway, accompanied by mitochondrial dysfunction. Different concentrations of Eleutheroside B induced apoptosis through mitochondrial pathway after 24 h. The levels of p53, survivin, cytochrome c, Bax and Bcl-2 in SMMC-7721 cells (A) and HeLa cells (B) were detected by Western blot. By flow cytometry, Eleutheroside B increased the number of SMMC-7721 cells (C) and HeLa cells (D) with decreased membrane potential. The data are presented as mean ± SD of three independent experiments. * p < 0.05, ** p < 0.01, *** p < 0.001 compared to untreated cells.
Eleutheroside B inhibited cell migration and invasion
In order to evaluate the Influence of Eleutheroside B to inhibit the migration and invasion of SMMC-7721 and HeLa cells, SMMC-7721 cells and HeLa cells were treated with Eleutheroside B (0–15 μM) for 24 h, and the effect on cellular motility was detected. In the wound healing assay, Eleutheroside B significantly inhibited the migration ofSMMC-7721 cells (Figure 5(a)) and HeLa cells (Figure 5(b)) in a concentration dependent manner. Eleutheroside B also reduced invasion and migration of SMMC-7721 cells (Figures 5(c) and (e)) and HeLa cells (Figures 5(d) and (f)) compared to the untreated group in the transwell assay. So far, some studies have confirmed that E-cadherin and vimentin prevent cancer cell metastasis by modulating cancer cell adhesion. In the present study Eleutheroside B increased the expression of E-cadherin compared to the untreated group (Figure 5(g)). Different concentrations of Eleutheroside B inhibited cell migration and invasion after 24 h. Cell migration in wound healing assay of SMMC-7721 (A) and HeLa (B) (×40). Transwell migration assay of SMMC-7721 (C) and HeLa (D), Giemsa established cells (×100). Transwell invasion assay of SMMC-7721 (E) and Hela (F), Giemsa established cells (×100). The levels of E-cadherin in SMMC-7721 cells and HeLa cells were detected by Western blot (G). * p < 0.05, ** p < 0.01, *** p < 0.001 compared to untreated cells.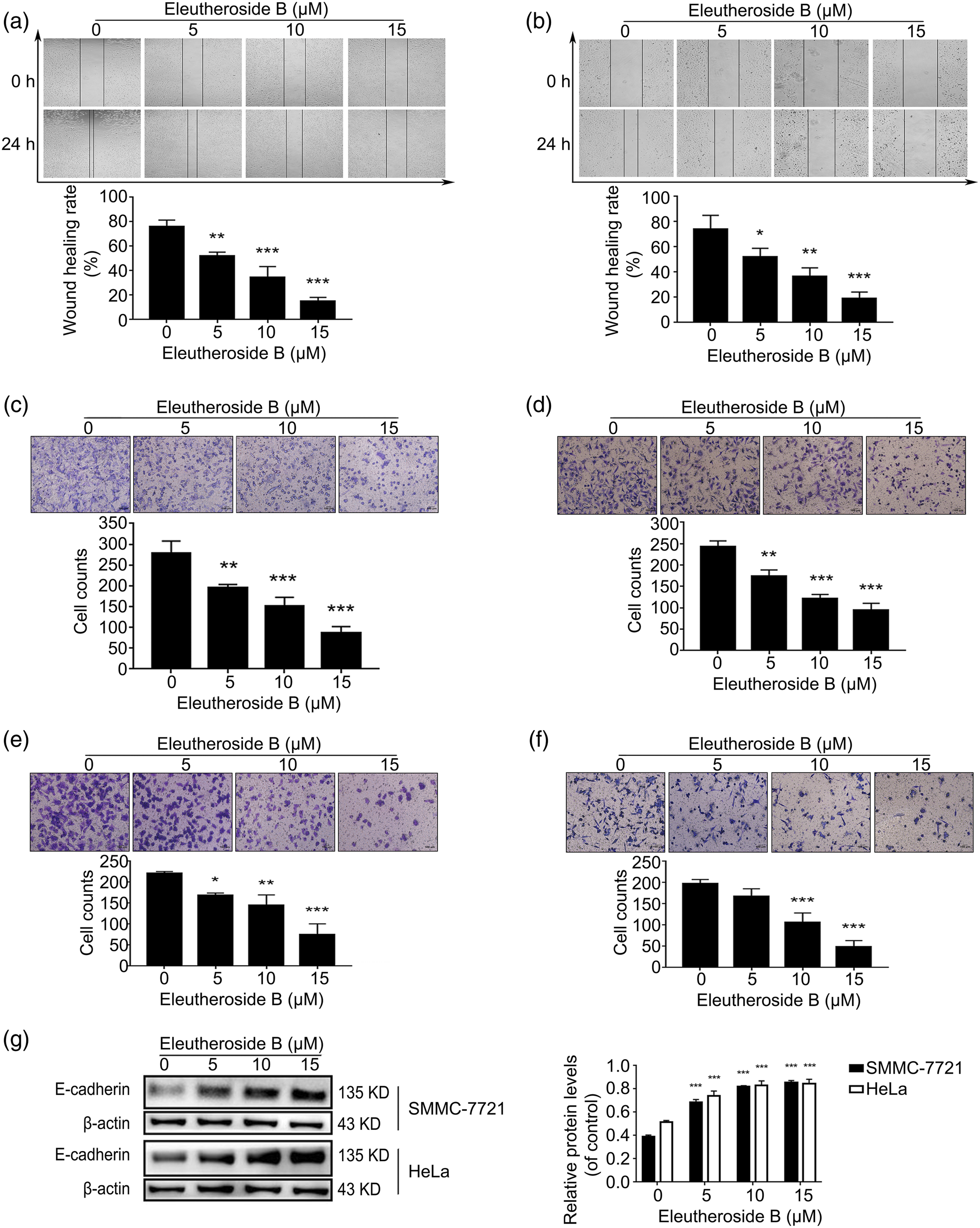
Eleutheroside B induced autophagy in SMMC-7721 and HeLa cells
Due to the role of autophagy in the treatment of cancer, and Eleutheroside B showed obvious anti-tumor effect in SMMC-7721 and HeLa cells, we speculated that Eleutheroside B would induce autophagy in the two cells. Autophagy-related cell death is also manifested by the accumulation of lysosomal vesicles and the conversion of LC3-I to autophagosome-associated LC3-II.
9
The conversion of LC3 generally related to the number of intracellular autophagosomes and can be increased due to either induction or late stage suppression of autophagy. P62 is an autophagic substrate which is used in measuring autophagic activity. In this study, Eleutheroside B significantly decreased the expression of LC3-I and increased the expression of LC3-II compared to untreated Eleutheroside B in the SMMC-7721 cells (Figure 6(a)) and HeLa cells (Figure 6(b)). In addition, SMMC-7721 cells (Figure 6(a)) and HeLa cells (Figure 6(b)) treated with Eleutheroside B decreased p62 protein levels compared to untreated SMMC-7721 cells and HeLa cells in a concentration-dependent manner. Therefore, these results suggest that Eleutheroside B could induce autophagy in SMMC-7721 cells and HeLa cells. Different concentrations of Eleutheroside B induced autophagy after 24 h. Western blot assay of the expression of LC3- I, LC3- II and p62 in SMMC-7721 (A) and HeLa (B) cells. * p < 0.05, ** p < 0.01, *** p < 0.001 compared to untreated cells.
Discussion
Cervical cancer is often diagnosed among women. The main cause is Human papillomavirus infection, followed by human immunodeficiency virus infection, smoking and oral contraceptives. 10 Hepatocellular carcinoma (HCC) is the second leading cause of cancer-related death worldwide. About 80% of liver cancer cases are caused by chronic infection of hepatitis B virus (HBV) and hepatitis C virus (HCV). 11 Although HCC patients can be treated by surgery, radiofrequency ablation and liver transplantation, the 5-years survival rate of HCC patients is still very low. The widely use of Chinese Herbal Medicine (CHM) in cancer patients attracts extensive attentions in recent years.12,13 Eleutheroside B, as a Chinese herbal medicine, has significant anti-tumor effect by blocking cell proliferation and regulating apoptosis signal pathway. It also regulates E-cadherin and inhibits invasion and migration to block the metastasis of SMMC-7721 cells and HeLa cells. Moreover, Eleutheroside B also induced autophagy.
Dysregulated cellular proliferation, one of the hallmarks of cancer, is mediated by aberrant activation of the cell cycle machinery through the biological effects of cyclin-dependent kinases (CDKs). Uncontrolled cell proliferation is a key feature and a target of many anticancer strategies primarily aimed at stopping cellular divisions. These strategies can be triggered by programmed cell death (PCD) (apoptosis, autophagy, or programmed necrosis. P53, the most common anti-tumor gene, is involved in cell cycle regulation, DNA damage repair and apoptosis induction. It has been reported that p53 can cause cell cycle arrest by regulating cyclin D1. CyclinD1, a key regulator of the cell cycle, forms complexes with cyclin-dependent kinase 4 or 6 in the cytoplasm. These complexes then enter the nucleus and cause cell cycle arrest. Notably, in a recent article, show a novel mechanism that CyclinD1 can be degraded by an autophagic process in hepatocellular carcinoma in addition to proteasome pathway. 14 In this study, Eleutheroside B can reduce the proliferation and viability of SMMC-7721 cells and HeLa cells, significantly increase the level of p53, down regulate CyclinD1, and cause cell cycle arrest.
Apoptosis is a type of programmed cell death in which target cells are eliminated without damaging adjacent cells. Apoptosis is an important event to maintain the stability of the internal environment of all human organs. 15 Previous studies have shown that p53 promotes apoptotic cell death by activating many positive regulators such as Bax, a pro-apoptotic member of Bcl-2. There are two major apoptotic pathways, namely the mitochondria-mediated intrinsic pathway and the death receptor-mediated extrinsic pathway. Among the Bcl-2 family proteins, the Bcl-2 belongs to the anti-apoptotic proteins and Bax belongs to the pro-apoptotic proteins. 16 Therefore, an elevated Bax/Bcl-2 ratio contributes to mitochondrial dysfunction and further apoptosis. Interestingly, in respect to mitochondrial metabolism and cellular survival, the Ca2+ concentration within the mitochondrial matrix plays an important role in mediating oxidative phosphorylation and ATP production via regulation of the TCA cycle, while also regulating the opening of the permeability transition pore (PTP) which plays a role in apoptosis. The intrinsic pathway initiates within the cell bythe permeabilization of the external mitochondrial membrane due to the formation of transmembrane channels in the mitochondrial membrane. These channels permit the release of cytochrome c, as well as other apoptosis effectors, that can associate with apoptotic protease-activating factor-1 and caspase-9 to form a complex called the apoptosome, leading to cell death, activates caspase-3, which leads to cell death. It was found that the release of cytochrome C could cause the decrease of membrane potential. Survivin is an anti-apoptotic protein discovered in recent years. Survivin participates in the progression and development of malignant tumors. 17 Caspases are controlled by specific cellular inhibitors called Inhibitor of Apoptotic Proteases (IAP), which can bind to them, thus blocking their function. As a member of IAP gene family, survivin can restrain apoptosis by inhibiting the activation of Caspase-3. In this study, Eleutheroside B significantly increased the expression of Bax, while simultaneously decreasing the expression of Bcl-2 in SMMC-7721 and HeLa cells, showing that Eleutheroside B elevated the Bax/Bcl-2 ratio, increased the cytochrome C in the cytoplasm, and down regulated survivin, which indicated that apoptosis triggered by mitochondria-mediated signaling pathway. In the process of apoptosis, there are a series of morphological changes such as cell contraction, plasma membrane blistering, chromatin condensation and chromosomal DNA fragmentation. 18 Here, the SMMC-7721 and HeLa cells treated with Eleutheroside B showed the above typical apoptotic characteristics. In addition, Annexin V/PI staining further confirmed the effect of apoptosis induced by Eleutheroside B. These results indicate that Eleutheroside B can induce apoptosis in SMMC-7721 and HeLa cells.
The metastasis process of tumor cells is complex, including the acquisition of invasiveness, the change of tumor matrix interaction, the survival in circulation and the extravasation to the distant. Epithelial mesenchymal transition (EMT) is the first and most important step in the process of metastasis. EMT promotes tumor progression, by endowing cells with migratory and invasive properties, inducing stem cell properties, suppressing apoptosis and senescence, and contributing to immunosuppression. Cell adhesion is closely associated with tumor invasiveness and metastasis, and the cadherin superfamily is a group of transmembrane glycoproteins that mediate calcium-dependent homophilic intercellular adhesion. Perturbations in cadherins have been shown to be associated with the development of cancer, particularly invasion and metastasis. The majority of evidence indicates that E-cadherin is associated with EMT, and that the loss of E-cadherin may promote invasive and metastatic behavior in many types of epithelial tumors. 19 Therefore, whether to inhibit cell invasion and migration by increasing E-cadherin is one of the goals of this study. Our results showed that Eleutheroside B enhanced the expression of E-cadherin in SMMC-7721 and HeLa cells. Our data show that Eleutheroside B can significantly inhibit the movement of SMMC-7721 cells and HeLa cells. Therefore, it has great potential to block the metastasis of SMMC-7721 cells and HeLa cells.
Autophagy is another type of programmed cell death. The term ‘autophagy’ was chosen to distinguish the lysosomal degradation, or ‘eating’ (phagy), of part of the cell’s self (auto) from the breakdown of extracellular material (heterophagy). Western blot assessment of the increase in either the LC3-II form or of the relationship between LC3-II and LC3-I content is currently considered as a simple, quick procedure to verify the presence of cell autophagy and its use is advised in recently published guidelines for the interpretation of assays monitoring autophagy. 20 The p62 protein through its LIR domain interacts with LC3 for attachment to the autophagosomes, thereby delivering the ubiquitinated target cytotoxic materials attached to it through its UBA domain for transportation to the lysosome and degradation. 21 The development of selective autophagy modulating drugs could be a useful therapeutic objective to improve therapeutic responses in cancer. 22 In this experiment, we found that Eleutheroside B can trigger the autophagy through regulating the expression of LC3 and p62 in SMMC-7721cells and HeLa cells.
Conclusions
Overall, this study showed that Eleutheroside B inhibited the proliferation and induced apoptosis through Mitochondrial pathways in SMMC-7721 and HeLa cells. Eleutheroside B also induced cell cycle arrest by suppressing the expression of cyclin D1. Further more, this research demonstrated that Eleutheroside B could prevent cell migration and invasion by increasing E-cadherin expression. Eleutheroside B regulated autophagy as well and blocked the growth of SMMC-7721 cells and HeLa cells. Therefore, Eleutheroside B has the potential to be used in the development of drugs for the treatment of liver cancer and cervical cancer in the future.
Footnotes
Authors' Contributions
Designed the experiments: S.F.S and J.C.Performed the experiments: S.F.S., Y.Z and W.L.X.Analysis and interpretation of data: S.F.S and B.W.Drafting the manuscript: S.F.S and J.C.
Declaration of transparency and scientific rigor
This Declaration acknowledges that this paper adheres to the principles for transparent reporting and scientific rigor of preclinical research recommended by funding agencies, publishers and other organizations engaged with supporting research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Natural Science Foundation of Hebei Province (H2021209004).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
