Abstract
Aluminum phosphide (AlP) poisoning can be deadly in most cases targeting the heart. To overcome AlP toxicity, exenatide has been studied in the present study due to its pleiotropic effects on cardiac damages. In this study, the rats were exposed to LD50 of AlP (10 mg/kg) by gavage, and exenatide at doses (0.05, 0.1, and 0.2 mg/kg) injected intraperitoneally 30 min after poisoning. The cardiac parameters including heart rate (HR), blood pressure (BP), QRS, corrected QT (QTc), and ST were monitored for 180 min. Blood glucose level was measured in the study groups 30 min after exenatide injection. Evaluation of biochemical parameters including mitochondrial complexes I, II, and IV activities, adenosine diphosphate (ADP)/adenosine triphosphate (ATP) ratio, malondialdehyde (MDA), apoptosis, lactate, troponin I, and brain natriuretic peptide (BNP) was done on heart tissues after 12 and 24 h. Additionally, the tissues were analyzed for any pathological damages including necrosis, hemorrhage, or hyperemia 24 h post-treatment. Our results showed that AlP-induced HR, BP, and electrocardiographic changes were improved by exenatide at all doses. The blood glucose levels of poisoned animals reached control levels after exenatide treatment. Besides, treatment with exenatide at all doses improved complexes I and IV activity, ADP/ATP ratio, and apoptosis. Malondialdehyde, lactate, troponin I, and BNP levels were also diminished after exenatide co-treatment in poisoned animals. On the other hand, administration of exenatide doses improved the histopathology of AlP-induced tissues. Based on our findings, exenatide has a protective effect against phosphine-induced cardiotoxicity in an almost dose-dependent way. However, further investigations are needed on the potential clinical use of exenatide in this poisoning.
Introduction
Aluminum phosphide (AlP), known as a highly lethal solid pesticide, is commonly used to preserve the quality and quantity of agricultural products from pests and insects, especially in developing countries.1,2 High efficiency, cost-effectiveness, ease of use, and not affecting seed viability and stored food are the essential reasons for extensive use of AlP. Although it has numerous advantages, its improper use can lead to severe poisoning of humans and other non-targeted species. More than 70% of AlP-exposed individuals die due to its toxic effects on the body’s various organs.3,4 Aluminum phosphide absorption can be done through ingestion, respiration, or rarely broken skin. Several studies established that the high lethal potential of AlP is due to multi-organ dysfunction, including lungs, kidneys, liver, and especially the heart. Aluminum phosphide toxicity is mediated by fatal phosphine gas 4 released from the AlP tablet and further being in contact with water or hydrochloric acid in the stomach. The toxicity of phosphine has been reported to be due to developing oxidative stress, mitochondrial dysfunction, apoptosis, cellular oxygen utilization in mitochondria, and denaturation of oxyhemoglobin molecules.4–5 According to previous reports, the cardiovascular system is the most vulnerable target of phosphine, resulting in death of poisoned people during the initial 24 h exposure due to cardiovascular collapse. The most important cardiovascular manifestations include heart failure, dysrhythmias, profound and refractory hypotension, subendocardial infarction or pericarditis, and electrocardiographic (ECG) abnormalities. Despite many efforts to find a suitable antidote for AlP poisoning, no effective cure has been discovered.3–7
Exenatide is derived from glucagon-like peptide (GLP-1), which is also used to treat type 2 diabetes mellitus. 8 A wide range of data has demonstrated the ameliorative properties of exenatide on cardiac injuries, including heart failure, 9 acute myocardial infarction,10,11 ischemia-reperfusion injury, 12,13 and arrhythmia. 14 Evidence has shown that exenatide potentially improves cardiovascular disorders through GLP-1 receptors. Additionally, it possesses anti-oxidant, anti-inflammatory, and anti-apoptotic properties.15,16 Exenatide can reduce ventricular arrhythmias during ischemic conditions by activating mitochondrial adenosine triphosphate (ATP)-sensitive potassium (KATP) channels. 14 Based on previous studies, exenatide increases mitochondrial biogenesis. Moreover, it can ameliorate mitochondrial malfunction by maintaining mitochondrial membrane potential (MMP), inhibiting mitochondrial permeability transition pore (mPTP), and improving mitochondrial respiratory capacity.17–21
As mentioned above, although AlP poisoning is a life-threatening condition with irreversible heart tissue damage, there have been no effective medications to eliminate its lethal effects. Therefore, finding a medication suppressing AlP’s cardiovascular toxicity can pave the way for further research in this regard. To our knowledge, there have been no data investigating exenatide effects on cardiac toxicity induced by AlP. It seems that exenatide can be useful in alleviating AlP-induced cardiac toxicity considering its mechanisms of action, that is, cardioprotective properties. Hence, the aim of this study was to examine aforementioned hypothesis in a preclinical rat model.
Materials and methods
Chemicals
The AlP tablets were purchased from Samiran Pesticide Formulating Co. (Iran). The kit of mitochondria isolation was purchased from BioChain Inc (USA). ApoFlowEx® fluorescein isothiocyanate (FITC) kit was gotten from Exbio (Vestec, Czech Republic). Also, the lactate and troponin I kits were obtained from ZellBio (Germany). Other chemicals were provided from Sigma-Aldrich (Germany).
Animals
The male Wistar rats (200–250 g weight range) were used in the current study. The rats were housed in standard cages of polycarbonate in a room with 20–25°C temperature, 50–55% humidity, and light–dark cycle of 12:12 with free access to the standard diet and water. All animal procedures were conducted according to ethical guidelines and standard protocols in the Ethics Committee of the Tehran University of Medical Science (code number: IR.TUMS.VCR.REC.1397.782).
Determination of AlP’s LD50 and effective exenatide dosage
Previous studies reported that LD50 of AlP ranged from 8.7 to 12.5 mg/kg. 6 Since the constant decomposition of AlP during the storage process may alter its LD50, it is needed to determine the AlP’s LD50 separately for each study. To this end, the AlP tablets were dissolved in almond oil after powdering, and four doses of AlP, including 9, 10, 11, and 12 mg/kg, were gavaged to rats in each cage (six rats per cage). Rats’ mortality was recorded every 6 h during 24 h following the gavage. Finally, the LD50 was calculated using Probit statistical software.
In order to assay the cardiac parameters, doses of exenatide (0.05, 0.1, and 0.2 mg/kg) and administration route were chosen according to the literature 22 supported by our pilot PowerLab study.
Experimental design
In this study, animals were randomly divided into eight groups, each contained six rats as follows: (1) control (CON) group: animals were treated with normal saline; (2) EXE 0.05 group: animals were treated with 0.05 mg/kg exenatide; (3) EXE 0.1 group: animals were treated with 0.1 mg/kg exenatide; (4) EXE 0.2 group: animals were treated with 0.2 mg/kg exenatide; (5) AlP group: animals were treated with LD50 dose of AlP (10 mg/kg); (6) AE 0.05 group: animals were treated with AlP (10 mg/kg) + exenatide (0.05 mg/kg); (7) AE 0.1 group: animals were treated with AlP (10 mg/kg) +exenatide (0.1 mg/kg); and (8) AE 0.2 group: animals were treated with AlP (10 mg/kg) +exenatide (0.2 mg/kg). It is worth noting that almond oil and normal saline were AlP and exenatide solvents, respectively. As well, exenatide was injected into rats via intraperitoneal (i.p.) injection, 30 min after AlP gavage.
Following treatments, animal studies were assigned into the following three steps: (1) in step one, cardiac hemodynamic parameters were assessed for 180 min in anesthetized rats after 30 min of AlP poisoning; (2) in step two, biochemical parameters were assessed during 12 h and 24 h after AlP poisoning; and (3) in step three, the heart tissues were evaluated for pathological changes 24 h after AlP poisoning.
Survival time investigation
To evaluate the survival time, four groups containing 10 rats were used as follows: (1) AlP group: animals were treated with LD50 dose of AlP; (2) AE 0.05 group: animals were treated with AlP (10 mg/kg) +exenatide (0.05 mg/kg); (3) AE 0.1 group: animals were treated with AlP (10 mg/kg) +exenatide (0.1 mg/kg); and (4) AE 0.2 group: animals were treated with AlP (10 mg/kg) +exenatide (0.2 mg/kg). Exenatide was injected 30 min following AlP gavage. The rats in each group were monitored for 48 h after treatments, and the rate of deaths was recorded.
ECG and hemodynamic parameters assessment
The cardiovascular parameters, including heart rate (HR), blood pressure (BP), QRS complexes, corrected QT (QTc) segment, and ST height were assessed using the PowerLab system (PowerLab 4/35 Data Acquisition Systems, AD Instruments, Australia) for 180 min. Thirty minutes after AlP gavage and anesthesia, rats were connected to the PowerLab system using a non-invasive cuff and electrodes. The systolic BP was measured by placing the cuff of PowerLab on the rat’s tail. The rats were anesthetized with ketamine/xylazine (50/5 mg/kg), and anesthesia was maintained with repeat of ketamine doses (25–40 mg/kg) during monitoring. Exenatide was injected at the beginning of the rat’s connection to the PowerLab system.
Measurement of blood glucose level
In order to assess the glucose level in the study groups, non-fasting blood glucose was measured from rats’ tail vein 30 min following exenatide administration using a glucometer (Accu-Chek Active, Roche Company made in Germany).
Collection of heart tissue samples
Following 12 and 24 h of AlP administration, the rats underwent surgery for collecting the heart tissue samples. The isolated hearts were washed with ice-cold saline solution and then divided into several parts. For the mitochondria analysis, about 100 mg of fresh heart tissue was used. Moreover, to determine the adenosine diphosphate (ADP)/ATP ratio, a portion of the heart tissue (approximately 100 mg) was collected into tubes containing trichloroacetic acid. The rest of the heart was immediately frozen at −80°C for other biochemical tests.
Assessment of tissue protein level
The Bradford method was used to determine the total concentration of protein in the heart samples. 23 Bovine serum albumin was used as the standard of protein concentration. Protein concentration in the samples was determined using a spectrophotometer at 595 nm as already set up in our laboratory. 24
Activity of complex I
The evaluation of complex I activity is based on the measurement of NADH consumption. Complex I (NADH dehydrogenase) activity was evaluated by the method of Sherwood et al. 25 as previously set up in our laboratory. 6 The complex I activity is reported as nmol/min/mg protein.
Activity assessment of complex II
The absorbance of dichlorophenolindophenol (DCPIP), as an indicator of complex II (succinate dehydrogenase) activity, was measured according to the method of Tompkins et al. 26 as previously set up in our laboratory. 3 The results represented nmol of DCIP/min/mg of mitochondrial protein.
Activity assessment of complex IV
The determination of complex IV (cytochrome C oxidase) activity in heart samples was performed based on the described method by Cooperstein et al. 27 as already set up in our laboratory. 7 The results were stated as the unit of k (first-order rate constant)/min/mg of mitochondrial protein.
Assessment of ADP/ATP ratio
The ADP/ATP ratio was evaluated based on the previously reported method by Hosseini et al. 28 The ATP and ADP concentration were measured using an HPLC system (Waters Chromatography Division, Milford, MA, USA) containing a solvent delivery system, 510-pump, Waters 486 UV-vis detector, and column (SUPELCOSIL™ LC-18-T). To this end, isocratic elution (4 tetrabutylammonium hydrogen sulfate in 0.1 M potassium phosphate buffer (pH = 5.5) and methanol (85:15 v/v)) in a flow rate of 1 mL per minute at 254 nm were used.
Lipid peroxidation assessment
The malondialdehyde (MDA) level was measured as the end product of lipid peroxidation (LPO), reflecting the levels of lipids peroxidation. 29 The MDA level was determined by Armstrong et al. 30 as already set up in our laboratory with some modifications. 31
Assessment of apoptosis using flow cytometry
The percentage of live, apoptotic, and necrotic cells in heart samples was determined after 24 h based on the method of Schlüter et al. 32 as already set up in our laboratory. 7 According to the kit instruction, the single cardiomyocytes were stained using annexin V-FITC and propidium iodide (PI). The cell death analysis was performed using flow cytometry (Shenzhen, Mindray, China).
Determination of lactate level
The lactate level of heart samples was measured according to the instructions of the lactate assay kit. Briefly, homogenized heart tissue in 8% perchloric acid was used. Lactate level was assessed using a standard curve, and the results were demonstrated as a mmol/mg protein. 6
Determination of the troponin I level
The troponin I level in homogenized heart tissues was measured using the commercial kit of ZellBio GmbH from Germany. The absorbance of homogenized samples was checked using a plate reader (Synergy, BioTech Instruments Inc, Germany). 4
Assessment of brain natriuretic peptide expression level by real-time PCR method
The real-time PCR technique was used to assess the relative expression level of the brain natriuretic peptide (BNP) gene as a cardiac biomarker. To do this, total RNA was extracted from heart tissue samples using the TRIZol® reagent kit in accordance with the manufacturers’ protocol. The RNA concentration was determined by a UV-vis spectrophotometer (Thermo Fisher Scientific, CA) at 260 and 280 nm. RNAs were reversed transcribed to cDNA using the iScript cDNA synthesis kit (BioFACT, Korea). The specific primer for the BNP gene was purchased from Eurofins Genomics of Germany, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was chosen as the housekeeping gene. The real-time PCR assessment in a light cycler 96 system (Roche, Germany) was carried out by SYBER Green Master Mix (BioFACT, Korea). The relative expression of the gene was measured using the 2−ΔΔCt formula, and results were normalized with GAPDH. 33 The following primers were used in this research:
Forward primer: CGGGCTGAGGTTGTTTTAGG
Reverse primer: ATCACTTGAGAGGTGGTCCC
Histopathological assay
The animals were sacrificed 24 h post-treatment. The animals’ heart tissues were harvested and fixed in the 10% neutral buffered formalin (NBF, pH = 7.26) for 48 h, then processed and embedded in paraffin. The 5 µm thick sections were prepared and stained with hematoxylin and eosin (H&E). 33 The independent reviewer evaluated the histological slides using light microscopy. Any changes, including necrosis, hemorrhage or hyperemia, etc., were assessed in different groups.
Statistical analysis
In this study, results were reported as mean ± standard deviation (SD). Survival was evaluated using the log-rank of the Kaplan–Meier test (Mantel-Cox). A two-way ANOVA analysis was used for cardiac hemodynamic parameters and flow cytometry results. One-way ANOVA analysis was performed for other tests. Tukey’s multi-comparison tests were used as the post hoc test. Differences with p < 0.05 are considered statistically significant.
Results
Survival time
The animal mortality was recorded in AlP, AE 0.05, AE 0.1, and AE 0.2 groups (n = 10) for a maximum of 48 h. In the AlP group (LD50), the median survival (MD) was 24 h, and the survival percentage was 30%. This indicates that after 24 h, 50% of the rats were alive and at the end of 48 h, three rats out of 10 were alive. In treated groups with exenatide at doses 0.05, 0.1, and 0.2 mg/kg, MD was increased to 30, 36, and 42 h, respectively, and the survival percentage of animals in these groups was 30%, 40%, and 50%, respectively. However, the Kaplan–Meier analysis showed that the improvement in poisoned animals’ survival following the administration of exenatide doses was not statistically significant.
HR, BP, and ECG parameters
The evaluation of hemodynamic parameters suggested that HR and BP started to decrease significantly during 60–90 min after AlP administration in poisoned animals (p < 0.05). Co-treatment with exenatide resulted in significant improvement in HR reduction at 60–90 min and 90–120 min (0.1 and 0.2 m/kg) and other time intervals, including 120–150 min and 150–180 min with all three doses of exenatide (p < 0.05). Noteworthy, progressive reduction of BP by AlP could be improved at the time intervals of 120–150 min with doses of 0.1 and 0.2 mg/kg exenatide and at 150–180 min with three doses of exenatide (p < 0.05). However, the values in the exenatide co-treatment groups were not statistically similar to those in the control group for each time interval.
Changes in heart rate in various groups.

Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1 and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg).
AlP: aluminum phosphide; CON: control.
a Significantly different from the control group at p < 0.05.
b Significantly different from the AlP group at p < 0.05.
Changes in blood pressure in various groups.

Data are mean ±standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1 and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg).
AlP: aluminum phosphide; CON: control.
a Significantly different from the control group at p < 0.05.
b Significantly different from the AlP group at p < 0.05.
Changes in ECG parameters of various groups.

Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1 and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg).
AlP: aluminum phosphide; QTc: corrected QT; ECG: electrocardiographic; CON: control.
a Significantly different from the control group at p < 0.05.
b Significantly different from the AlP group at p < 0.05.
Blood glucose level
Changes in blood glucose level (mg/dl) of various groups.

Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1 and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg).
AlP: aluminum phosphide; CON: control.
a Significantly different from the control group at p < 0.05.
b Significantly different from the AlP group at p < 0.05.
The activity of complex I
Effects of various treatments on the activity of mitochondrial complexes and ADP/ATP ratio.
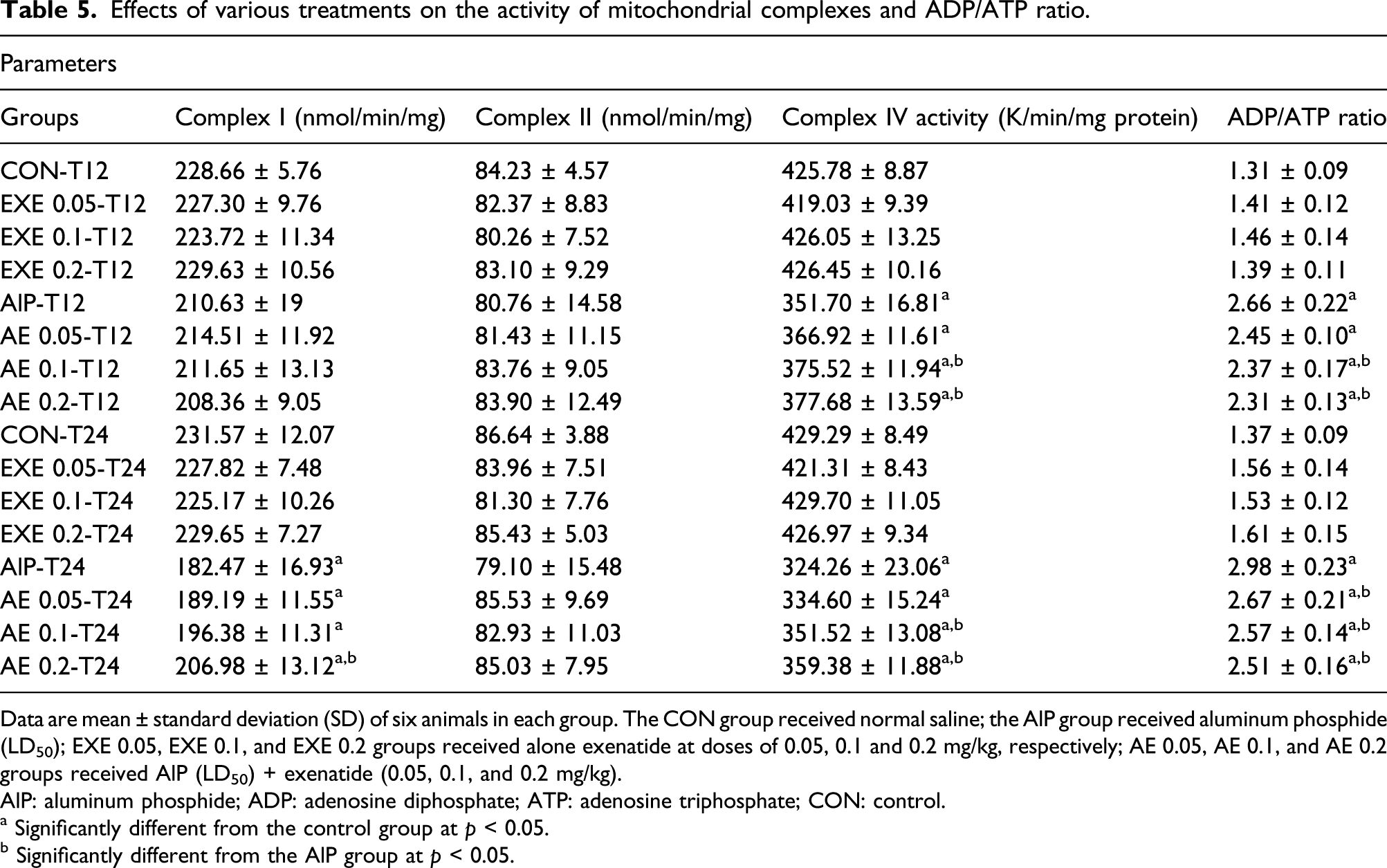
Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1 and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg).
AlP: aluminum phosphide; ADP: adenosine diphosphate; ATP: adenosine triphosphate; CON: control.
a Significantly different from the control group at p < 0.05.
b Significantly different from the AlP group at p < 0.05.
The activity of complex II
According to the results presented in Table 5, none of the treated groups showed a statistically significant change in complex II activity.
The activity of complex IV
The activity of complex IV in the AlP group was lower compared to the control group at both times (p < 0.05). Co-treatment with exenatide doses (0.1 and 0.2 mg/kg) significantly increased this complex activity during both times. However, the activity level did not reach the control group (p < 0.05). No significant effects on complex IV activity were seen in animals that received alone exenatide doses (Table 5).
The assessment of ADP/ATP ratio
We found a significant elevation in ADP/ATP ratio in the AlP group versus the control group during both times (p < 0.05). Co-treatment with 0.1 and 0.2 mg/kg exenatide caused a significant reduction in ADP/ATP ratio compared to the AlP group after 12 h. This ratio was also lessened in the AE 0.05, AE 0.1, and AE 0.2 groups compared to the AlP-T24 group (p < 0.05). Moreover, the effect of exenatide alone on the ADP/ATP ratio was not significant (Table 5).
The evaluation of LPO
We measured the MDA level as a well-known marker for LPO assessment of heart tissue. In the rats treated with AlP, the MDA level was significantly higher than the control group in both times (p < 0.05). However, the animals under co-treatment with exenatide doses (0.05, 0.1, and 0.2 mg/kg) had lower MDA levels than those in the AlP-T12 and AlP-T24 groups. These reductions were not statistically the same as the control groups (p < 0.05). Moreover, MDA levels in groups that received exenatide alone were statistically similar to the control group (Figure 1). Effects of treatments on LPO in rat heart tissue. Data are mean ± SD of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1, and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg). All groups treated during 12 and 24 h. a: Significantly different from the control group at p < 0.05. b: Significantly different from the AlP group at p < 0.05. LPO: lipid peroxidation; SD: standard deviation; AlP: aluminum phosphide; CON: control.
Apoptosis using flow cytometry
As shown in Figures 2 and 3, in the AlP-T24 group, the percentage of live cells was lower while the percentage of apoptotic cells was higher than the control group (p < 0.05). Co-treatment with exenatide in a dose-dependent way caused a significant elevation in live cells and a significant reduction in apoptotic cells percentage. Noteworthy, these changes were different from the control values (p < 0.05). Further, the values of live and apoptotic cells were not statistically significant in the groups that received exenatide alone compared to the control group. Flow cytometry analysis of the cardiac cells in different groups. The cell death was evaluated under annexin-V/PI staining and using flow cytometry analysis. The CON-T24 group received normal saline; the AlP-T24 group received only aluminum phosphide (LD50); EXE 0.05-T24, EXE 0.1-T24, and EXE 0.2-T24 groups received only exenatide at doses of 0.05, 0.1, and 0.2 mg/kg, respectively; AE 0.05-T24, AE 0.1-T24, and AE 0.2-T24 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg). All groups treated in 24 h time. AlP: aluminum phosphide. Effects of treatments on live and apoptotic cells of rat heart tissue. Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1, and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg). All groups treated in 24 h time. a: Significantly different from the control group at p < 0.05. b: Significantly different from the AlP group at p < 0.05. AlP: aluminum phosphide; CON: control.

Lactate level
Results revealed an increased lactate level in the AlP group versus the control group in both times (p < 0.05). Exenatide in groups of AE 0.1-T12 and AE 0.2-T12 significantly decreased the lactate levels in comparison with the AlP-T12 group. Moreover, lactate levels reduced considerably in poisoned animals after co-treatment with exenatide doses (0.05, 0.1, and 0.2 mg/kg) during 24 h. However, the values were not same as the control groups (p < 0.05). As well, there were no significant differences between alone exenatide groups and control groups at both times (Figure 4). Effects of treatments on lactate level in rat heart tissue. Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1, and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg). All groups treated during 12 and 24 h. a: Significantly different from the control group at p < 0.05. b: Significantly different from the AlP group at p < 0.05. AlP: aluminum phosphide; CON: control.
The assessment of troponin I level
As shown in Figure 5, AlP administration increased troponin I levels compared to the control group in both times (p < 0.05). The co-administration with all three doses of exenatide (0.05, 0.1, and 0.2 mg/kg) decreased this biomarker level compared to the AlP group during 12 and 24 h. However, the values were not reversed back to the control levels (p < 0.05). As well, no significant difference was observed between the groups that received exenatide alone and the control group. Effects of treatments on troponin I level in rat heart tissue. Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1, and 0.2 mg/kg, respectively; AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg). All groups treated during 12 and 24 h. a: Significantly different from the control group at p < 0.05. b: Significantly different from the AlP group at p < 0.05. AlP: aluminum phosphide; CON: control.
Relative gene expression of BNP
Real-time PCR results indicated that the BNP gene expression in the AlP-treated rats was significantly more than those in the control group (p < 0.05). The BNP gene expression was noticeably down-regulated with exenatide co-treatment at 0.1 and 0.2 mg/kg doses compared to the AlP-T24 group. Of note, this down-regulation was different from the control group (p < 0.05). As well, there was no significant difference for this biomarker in different doses of exenatide alone compared to the control group (Figure 6). Effects of treatments on mRNA expression of BNP in rat heart tissue. Data are mean ± standard deviation (SD) of six animals in each group. The CON group received normal saline; the AlP group received aluminum phosphide (LD50); EXE 0.05, EXE 0.1, and EXE 0.2 groups received alone exenatide at doses of 0.05, 0.1, and 0.2 mg/kg, respectively; and AE 0.05, AE 0.1, and AE 0.2 groups received AlP (LD50) + exenatide (0.05, 0.1, and 0.2 mg/kg). All groups treated in 24 h time. a Significantly different from control group at p < 0.05. b Significantly different from AlP group at p < 0.05. BNP: brain natriuretic peptide; SD: standard deviation; A1P: aluminum phosphide; CON: control.
Histopathological results
All H&E-stained heart sections from different experimental groups were evaluated histologically (Figure 7). Micrographs of heart tissue in normal saline and treated groups with different exenatide doses (0.05, 0.1, and 0.2 mg/kg) were normal without any histopathological changes. Histopathological evaluation of the AlP-intoxicated heart tissue showed myocardial cell necrosis and severe congestion. Micrographs of cardiac tissue sections in AlP +EXE 0.05 treated group had close similarities to the AlP-intoxicated group with the presence of myocardial necrosis (Zenker’s necrosis). In poisoned animals treated with 0.1 and 0.2 mg/kg exenatide, less cardiomyocyte necrosis was seen compared to those co-treated with dose of 0.05 kg/mg (Figure 7). Histopathologic sections of heart in different experimental groups. A: normal saline, B: EXE 0.05, C: EXE 0.1, D: EXE 0.2, E: AlP, F: AlP +EXE 0.05, G: AlP + EXE 0.1, H: AlP + EXE 0.2, black arrows: myocardial necrosis, red arrows: congestion, H&E × 400 stain. Bar: 50 µm. AlP: aluminum phosphide.
Discussion
The present in-vivo study was conducted to assess the role of exenatide on AlP-induced cardiac injuries in rats. The mechanisms involved in AlP-induced cardiotoxicity are summarized in Figure 8. Considerable evidence has shown that cardiovascular dysfunction is a leading cause of death during the initial 24 h of AlP poisoning.
3
Based on previous studies, the main mechanism of phosphine toxicity is the dysfunction of the mitochondrial electron transfer chain (ETC) and subsequently induction of oxidative stress and apoptosis.
34
A wide range of studies has demonstrated the positive effects of exenatide on cardiac injuries.11,13,14 To the best of our knowledge, this is the first report on the cardioprotective effects of exenatide following AlP poisoning. In our study, the effects of exenatide on cardiac hemodynamic and ECG parameters and molecular changes were investigated following AlP poisoning. The mechanisms involved of AlP cardiotoxicity. AlP inhibited the function of mitochondrial complexes I and IV, reduced ATP synthesis, induced LPO and apoptosis, and increased lactate levels. Moreover, AlP led to an alteration in ECG and hemodynamic parameters. AlP: aluminum phosphide; LPO: Lipid peroxidation; ROS: reactive oxygen species; ATP: adenosine triphosphate; ECG: electrocardiography; HR: heart rate; BP: blood pressure; QTc: corrected QT.
The present findings showed that AlP led to severe hypotension, decreased HR, and ECG abnormalities, such as ST alterations, QTc prolongation, and QRS widening.3–6 In this study, BP and HR started to decrease in animals around an hour after exposure to AlP at LD50. 3 Co-treatment with exenatide doses in poisoned rats partially prevented hypotension, but this improvement was not similar to non-poisoned rats. In addition, the use of exenatide in a dose-dependent manner improved significantly AlP-induced bradycardia. The QRS complex in ECG represents a change in left and right ventricular depolarization. 35 In our study, the QRS widening occurred following AlP poisoning, and treatment with three doses of exenatide could reverse significantly QRS widening in poisoned rats. The QTc interval is another parameter demonstrating impaired left ventricular function and fatal arrhythmia. 36 In our study, the administration of exenatide doses could significantly shorten AlP-induced QTc prolongation. The improvement effects of this parameter started at 120 min after co-treatment with 0.1 and 0.2 mg/kg exenatide. The ST segment representing the end of ventricular depolarization and the restart of ventricular repolarization, and any alteration in the ST height indicates myocardial and pericardial damages. According to previous studies, administration of AlP can induce either depression or elevation in ST height.7,37 We found that the ST segment elevation occurred after AlP poisoning, and exenatide co-administration in a dose-dependent manner could ameliorate the ST alterations at the end of 3 h. It is also worth noting that exenatide alone did not show any significant changes in the cardiac parameters described above. Various reports have confirmed the protective effect of exenatide on heart dysfunction.11,13,22 Accordingly, exenatide can enhance cardiac contractility through improvement of cellular metabolism efficiency (shifting metabolism from fatty acids to carbohydrates) in myocardial disorders. Furthermore, it can reduce ventricular arrhythmias through interacting with mitochondrial ATP-sensitive potassium (KATP) channels.14,38 Our results are in line with a previous study that showed the healing effects of exenatide on doxorubicin-induced ECG abnormalities. 39 In various studies, the cardioprotective effects of exenatide have been attributed to the improvement of cellular metabolism, cardiac contractility, modulation of mitochondrial function, reduction of oxidative stress, and inhibition of apoptosis.40–43 These mechanisms may play a role in the ameliorative effects of exenatide on hemodynamic and ECG parameters in the current study.
It has been reported that hyperglycemia is a risk factor for mortality due to AlP poisoning. Hyperglycemia management can play a beneficial role in treating these patients by increasing glucose uptake into cells and reducing oxygen consumption.44,45 Our findings showed that the mean blood glucose levels of animals significantly increased following AlP poisoning compared to the control group. This is in accordance with previous studies that AlP could cause hyperglycemia. Another key finding of our study was that exenatide at doses (0.05, 0.1, and 0.2 mg/kg) had the ability to lower the blood sugar in poisoned animals without significantly changing the blood sugar in normal animals. It indicates that exenatide’s anti-diabetic function depends on blood glucose levels, and this medication does not cause hypoglycemia.
Electron transfer chain is a series of protein complexes that synthesize cellular ATP using electron carriers’ energy from redox reactions. Phosphine gas interferes with the ETC through non-specific cytochrome C oxidase inhibition and impairs oxidative phosphorylation, leading to reduced ATP synthesis. Aluminum phosphide through inhibiting cellular respiration and the energy production process activates hydroxyl ions, causing widespread toxicity and cell death. Therefore, mitochondrial dysfunction is the main contributor to the toxicity caused by AlP.3,5,46 In the present research, the effect of phosphine on the activities of mitochondrial complexes I, II, and IV were investigated in cardiac tissue. There was no significant change in the activity of complex I at the 12 h time and complex II in the two times. However, we reached a significant reduction in the activity of complex I at the time of 24 h and complex IV in the two times following AlP poisoning. These results were in line with previous studies reporting that AlP reduced complex I and IV activities, whereas it had no effect on complex II activity.4,6 Albeit, some studies showed that AlP has no effect on complex I activity.3,5 Additionally, in line with earlier studies, AlP increased the ADP/ATP ratio compared to the control group during 12 and 24 h.3,4,6 A possible explanation for this might be due to the inhibition of ETC complexes by AlP. In addition, it might be mediated through elevation of ATP consumption during repairing heart damage following AlP poisoning. The present study revealed a rise in complex I activity through administration of 0.2 mg/kg exenatide at 24 h. Another finding was the improvement of complex IV activity through the doses of 0.1 and 0.2 mg/kg exenatide at both time points. Also, co-administration of exenatide could significantly reduce the ATP/ADP ratio after 12 h by the doses of 0.1 and 0.2 mg/kg and 24 h by the doses of 0.05, 0.1, and 0.2 mg/kg. These results are consistent with others noted that exenatide ameliorates mitochondrial respiration. It seems that exenatide improves oxidative phosphorylation and mitochondrial respiration along with increasing ATP production through elevation of synthesis and activity of mitochondrial enzymes, particularly cytochrome C oxidase (complex IV).20,22,36 Studies have also shown that treatment with exenatide increases mitochondrial mass and stimulates mitochondrial biogenesis. 47 These findings suggest that exenatide can protect mitochondria as an important target of AlP-induced toxicity in the heart.
Oxidative stress occurs due to an imbalance between the free radical and anti-oxidant defense system.48,49 It has been demonstrated that AlP can make a considerable change in oxidative stress biomarkers such as reactive oxygen species (ROS) and LPO. 50 In this research, we found that AlP significantly increased LPO levels that is in line with previous reports. According to the literature, AlP induces oxidative stress following ETC impairment. Inhibition of ETC by AlP leads to the excessive formation of free radicals, including peroxide and superoxide radicals. Free radicals induce malfunction of macromolecules, alteration of membrane permeability, LPO, cellular damages, and cell death.51–54 Our results showed that co-treatment with various doses of exenatide (0.05, 0.1, and 0.2 mg/kg) reduced LPO levels in rats poisoned with AlP during both times. This finding was in accordance with others mentioning anti-oxidant effect of exenatide.16,18 Based on the evidence, exenatide can modulate the production of ROS in cardiomyocytes through its ability to protect the respiratory chain and improvement of the endogenous anti-oxidant capacity, including catalase, glutathione peroxidase 1, and manganese superoxide dismutase.13,16,18,40 As a result, it can prevent oxidative damages caused by AlP.
Apoptosis is a physiological process of cell death and has a key role in cellular homeostasis. Physiological and pathological stimuli cause apoptosis,55–57 which can further bring about various illnesses such as autoimmune diseases and cancer, if dysregulated.58–60 AlP poisoning can enhance the process of apoptosis by releasing cytochrome C into the cytosol and activating caspases. Possible reasons behind this event are altering the mitochondrial membrane integrity, reducing the MMP, and opening the mPTP.6,33 In the current study, the flow cytometry analyses demonstrated the lower percentage of live cells and upper percentage of apoptotic cells in the AlP group versus the control group. Considering this subject, other studies also reported the same findings.3,5,6 It should be noted that co-treatment with exenatide in a dose-dependent manner increased live cells while decreased apoptotic cells in heart tissue. It seems that exenatide is able to prevent AlP-induced apoptosis through modulating mitochondrial function and reducing oxidative stress. Based on previous studies, exenatide involves in the up-regulation of anti-apoptotic genes including Bcl-2, down-regulation of pro-apoptotic genes such as Bax, and the reduction of cytochrome C release. Moreover, exenatide reduces caspases activity, including caspases 3, 8, and 9.16,18
Lactate is generated by lactate dehydrogenase enzyme under anaerobic conditions such as hypoxia or disorder of the aerobic pathway. An increase in the body’s lactate level leads to lactic acidosis. 6 In the current study, an elevation of lactate levels induced by AlP was reported in both times, reduced by co-administration of exenatide at doses of 0.1 and 0.2 mg/kg after 12 h and at all three doses after 24 h. This result may be explained by the fact that exenatide during AlP poisoning can boost aerobic pathway efficiency by improving mitochondrial ETC function.
Troponin I is a protein released from myocytes into the bloodstream due to myocardial damages. This protein is a specific biomarker with high sensitivity in cardiomyocyte injury detection.61,62 In this context, our study indicated that AlP poisoning increased troponin I level in the heart tissue during both times. In addition, the level of this biomarker significantly decreased as a result of co-treatment with exenatide doses (0.05, 0.1, and 0.2 mg/kg). As well, BNP (a polypeptide with 32 amino acids) is the most important biomarker in heart failure diagnosis. The release of this biomarker from heart cells is strongly associated with left ventricular wall pressure 63 Based on our findings, an elevation in BNP gene expression was observed in cardiac tissue after AlP poisoning. This is while exenatide co-treatment at 0.1 and 0.2 mg/kg doses was noticeably able to down-regulate the BNP gene expression. Reduced troponin I and BNP levels by exenatide indicated cardioprotective effects of exenatide in AlP-poisoned animals through its anti-oxidant properties. Since AlP poisoning is followed by increased LPO and membrane degradation, exenatide can prevent the leakage of these markers into the bloodstream through reducing the level of LPO.
In our study, histopathological findings such as myocardial cell necrosis and severe congestion confirmed the induction of heart injury by AlP. These changes were all mitigated by administration of exenatide carious doses, particularly the dose 0.2. Besides, our study confirms that exenatide alleviates heart injuries in an almost dose-dependent manner. Based on findings, this is meditated through improving mitochondrial function, reducing oxidative stress, and inhibiting apoptotic pathways.
Conclusion
In general, AlP exposure induced severe damage in rats’ heart tissue through oxidative stress, apoptosis, and an imbalance in the redox status. These changes were associated with histopathological damages and altered cardiac physiological functions, including BP, HR, and ECG. Co-treatment with exenatide doses showed a notable improvement in AlP-induced histopathological and physiological damages in the heart. Based on our results, exenatide has a potential protective role against AlP-induced cardiac disorders. Therefore, exenatide is a good candidate as an adjunct choice along with other supportive treatments of AlP poisoning. The present conclusions remain to be confirmed in clinic by further studies.
Footnotes
Author contributions
MA gave the idea. MB, MR, SH, MA, and MS collaborated to conduct some experimental investigations. MG, MA, and MRHS participated in the study of animals. BB and HHA prepared the manuscript. MB, MN, and SNO were advisors. BB did the work as her PhD thesis. MA supervised the whole study. The final version was read and approved by all authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study coded 97-03-33-40308 was supported by a grant from TUMS.
