Abstract
Smoking is one of the most important leading death cause worldwide. From a toxicological perspective, cigarette smoke serves hazards especially for the human being exposed to passive smoke. Over the last decades, the effects of natural compounds on smoking-mediated respiratory diseases such as COPD, asthma, and lung cancer have been under investigation, as well as the mechanistic aspects of disease progression. In the present study, the protective mechanism of eucalyptol (EUC), curcumin (CUR), and their combination on BEAS-2B cells were investigated in vitro to understand their impact on cell death, oxidative cell injury, and inflammatory response induced by 3R4F reference cigarette extract (CSE). According to the present findings, EUC, CUR, and their combination improved cell viability, attenuated CSE-induced apoptosis, and LC3B expression. Further, CSE-induced oxidative damage and inflammatory response in human bronchial epithelial cells were remarkably reduced by the combination treatment through modification of enzymatic antioxidant activity, GSH, MDA, and intracellular ROS levels as well as nitrite and IL-6 levels. In addition, nuclear translocation of Nrf2, a regulatory protein involved in the indirect antioxidant response, was remarkably up-regulated with the combination pre-treatment. In conclusion, EUC and CUR in combination might be a potential therapeutic against smoking-induced lung diseases through antioxidant and inflammatory pathways and results represent valuable background for future in vivo pulmonary toxicity studies.
Introduction
Tobacco is the death cause of over 8 million people each year worldwide and its consumption as cigarettes is considered as the most important health problem. 1 Lipophilic substances, mutagens, heavy metals, and carcinogens in cigarette smoke (CS) provide a basis for fatal outcomes.2,3 Chronic cigarette consumption decreases life quality as a progressive factor for diseases such as chronic obstructive pulmonary disease (COPD), pulmonary heart disease, and lung cancer.4–6 Hence, it is important to enlighten the underlying pathogenesis of cigarette-related lung diseases and develop a strategy to protect human health against CS as a part of environmental risk assessment. In recent years, the effects of natural chemopreventive agents, vitamins, and antioxidants on the human respiratory system are investigated to enlighten their activity on CS-induced toxic outcomes, such as oxidative and inflammatory response in the respiratory system.5,7–13 It is suggested that pathological consequences of smoking on lung tissue are mainly based on oxidative cell injury and inflammation resulted from the reactive oxygen species (ROS) in CS. 14 Moreover, ROS levels are augmented in patients with COPD even though ROS can not easily pass through airway epithelial cells. Therefore, antioxidant therapy might represent an important tool for the treatment and prevention of cigarette smoking-related diseases.

Natural components derived from medicinal plants such as flavonoids, alkaloids, and terpenoids have antioxidant, anti-inflammatory, or anticancer properties. 15 Among natural compounds, eucalyptol (EUC), mainly extracted from Eucalyptus species, has important mucolytic and spasmolytic acts on the respiratory tract. Moreover, EUC has clinically been proved the therapeutic potential for respiratory diseases, such as asthma and COPD. 16 Previous reports with EUC were mainly focused on its repair capacity on pulmonary tissue by reducing oxidative stress and lipid peroxidation, preventing/reducing activation of inflammatory cytokines with limited in vivo or in vitro studies.17–19 On the other hand, curcumin (CUR), is a well-known dietary and polyphenolic compound related to significantly enhanced pulmonary function due to its antioxidant and anti-inflammatory capacity.15,20 However, the efficacy of EUC and CUR combination on CS-induced bronchial epithelial toxicity has not been studied yet. Hence, the present study is aimed to investigate the effects of EUC, CUR and their combination on cigarette smoke extract (CSE)-induced toxicity in human bronchial epithelium cells, BEAS-2B.
Materials and methods
Preparation of CSE
Research grade cigarettes (3R4F containing 9.4 mg tar and 0.73 mg nicotine per cigarette) from Kentucky Tobacco Research and Development Center at University of Kentucky (Lexington, KY) were used for extraction and CSE was prepared with slight modifications.21,22 The extract was prepared by pumping smoke using 50 ml syringe. Ten of 3R4F cigarettes were puffed into 40 ml of culture medium with 2 s puff duration, 35 ml puff volume and 60 s puff cycle as represented in Figure 1. These procedures were performed in the fume hood.

Schematic diagram of CSE preparation.
Following, CSE was filtered by 0.22-µm filter, supplemented with 5% fetal bovine serum and 100 IU/ml penicillin-100 μg/ml streptomycin and vortexed. This preparation was considered as 100% CSE and aliquots were kept at −80°C for further studies. For each experiment, CSE was diluted freshly with complete cell culture medium to provide required experimental concentration of CSE.
Cell culture and cell viability
Human bronchial epithelial cell line BEAS-2B (ATCC, USA) were cultured in DMEM high-glucose medium (supplemented with 100 IU/ml penicillin, 100 μg/ml streptomycin and 5% fetal bovine serum) and incubated at 37°C with 5% CO2. BEAS-2B cells were seeded to a 96 well culture plate and incubated until they form a semi-confluent layer. To evaluate CSE cytotoxicity, CSE was applied at 1–50% (v/v) for 4 hours. BEAS-2B cells were exposed to various concentrations of EUC (6.25–200 μM) and CUR (5–20 μM) to determine their impact on cell viability. In addition, a well-known antioxidant and mucolytic agent N-acetylcysteine (NAC) (250–2000 μM) was used as positive control according to previous studies.23,24 After 2 hours of pre-treatment with EUC, CUR and NAC, cells were exposed to CSE and incubated for an additional 4 hours. After the incubation, MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) solution (0.5 mg/mL) was added to each well as 100 µL and incubated for an additional 2 hours at 37°C with 5% CO2. Next, formazan formation for each dilution was determined spectrophotometrically by dissolving formazan salt with isopropanol and compared to the values obtained from control group in triplicates. Cell viability is directly related to the absorbance of blue-violet-colored formazan read at 570 nm as optical density by plate reader (Thermo Scientific, Finland) and the decrease in viability as compared to the medium control (medium-treated cells) was calculated using the following equation:
Annexin V-PI staining
To determine the amount of cell death type induced by CSE, Annexin-V FLUOS staining kit (Roche, Germany) was employed following the method by Sur et al. with slight modifications. 25 Briefly, the cells (3 × 105) were seeded in six-well plates and incubated for 24 h. An anticancer drug, doxorubicin (2 μM), and DMSO (10%; v/v) were used as positive controls 26 ; while medium-treated cells were included in experiments as the negative control. After 2 h of pre-treatment with EUC, CUR, EUC+CUR, or NAC, cells were exposed to CSE for 4 h. Then, cells were collected and resuspended in solution containing 100 µl of incubation buffer and 3 µl of Annexin-V-Fluos labeling reagent. Following the incubation for 10 min at 37°C in the dark, 1 µl of propidium iodide was added and the Guava® easyCyte Single Sample Flow Cytometer system (Merck KGaA, Darmstadt, Germany) was employed to quantify the number of viable, apoptotic and necrotic cells. For each sample, 20 000 cells were counted and the data were analyzed by Guava InCyte (Merck KGaA, Darmstadt, Germany) software. Each experiment were performed in triplicates.
Determination of lipid peroxidation
BEAS-2B cells were harvested in T-25 flasks cell pellets were homogenized with a tissue homogenizer. Completely lysed cells were centrifuged at 30,678 g, 4°C for 15 min and the supernatants were stored at −80°C for the determination of, malondialdeyhde (MDA) level, protein content, and oxidative stress parameters. Thiobarbituric acid (TBA), a chromogenic compound, was used for lipid peroxidation assay by MDA analysis with slight modifications of the methods of Buege and Aust and Daraie et al. Overall, the method is based on the pink coloration of TBA by reacting with MDA in an acidic medium at the appropriate temperature.27,28 Briefly, TBA mixture containing trichloroacetic acid, HCl, and butylhydroxytoluene were vortexed with cell lysate or standard solution (tetramethoxypropane) in eppendorf tubes (2:1 v/v) and incubated in the hot water bath for 30 min at 95°C. After cooling, absorbance of pink chromophore for each group was measured at 540 nm spectrophotometrically. Protein content for each group was assessed by the method of Lowry et al. 29 Results were expressed as nmol/g protein and the experiment was performed in triplicates.
Determination of oxidative stress
Determination of GSH level
GSH level was measured from cell lysates with a slightly modified method by Sedlak and Lindsay. 30 Briefly, 50 µl of cell lysate was mixed with 10 µl DTNB (Ellman’s reagent, 5 5′-dithiobis-(2-nitrobenzoic acid)) solution, and then 150 µl of EDTA buffer solution (pH 8.2) was added in 96-well plate. After incubation at 37°C under dark for 30 min, the absorbance of yellow-colored chromophore was measured at 412 nm wavelength by using a UV spectrophotometer (Thermo Scientific, Finland). Results were expressed as μmol/g protein and performed in triplicates.
Determination of CAT activity
CAT activity was measured by using the correlation between carbonato-cobaltate (III) complex and catalase enzyme from a previously generated method by Hadwan. 31 Briefly, 50 µl of cell lysate is mixed with twice the amount of 10 mM H2O2 in tubes and incubated at 37°C for 2 min. Following, 600 µl of the working solution containing phosphate buffer (pH 7.4), sodium bicarbonate, and cobalt (II) nitrate hexahydrate was added and mixed with vortex immediately. Reaction tubes were kept under dark for 10 min and 250 µl of reaction for each group were transferred to 96-well plates. Corresponding absorbances were recorded at 440 nm spectrophotometrically and results were expressed as U/mg protein and performed in triplicates.
Determination of SOD activity
SOD activity, which is based on the ability of intracellular SOD to inhibit autooxidation of pyrogallol to pyrogallol-ortho-quinone in the reaction medium, was determined according to the method of Marklund and Marklund with modifications. 32 Briefly, 10 µl of cell lysate prepared from confluent T-25 cell culture flasks were incubated with 150 µl of reaction mixture containing Tris-HCl (pH 8.2) and EDTA solution in 96-well plate. Following, 2 mM pyrogallol was added to each reaction mixture and changes in absorbance were recorded immediately for 450 seconds at 420 nm. Each measurement was recorded for 30 seconds intervals for a total of 15 recordings. The first measurement after the addition of pyrogallol to reaction medium was accepted as time 0. The amount needed for the inhibition of 50% of pyrogallol autooxidation was accepted as 1 U of SOD activity. Results were expressed as U/ml and performed in triplicates.
Intracellular ROS
The protective effect of the compounds against CSE-induced intracellular ROS generation was determined by Cellular ROS Assay Kit (Abcam, Germany) employing flow cytometry. Under stress conditions, 2′,7′-dichlorofluorescin diacetate (DCFDA), a fluorescence sensitive dye deacetylated by cellular esterases and form a non-fluorescent compound, is oxidized into 2′,7′-dichlorofluorescein (DCF) by ROS produced within cells. DCF is a fluorescent compound which is detected by fluorescence spectroscopy with excitation/emission at 495 nm/529 nm, correspondingly. BEAS-2B cells were pre-treated with NAC, EUC, CUR, or EUC+CUR for 2 hours, which was followed by CSE exposure for 4 hours. Cells were harvested in 400 µl of phosphate-buffered saline (PBS) solution and then 100 µl of DCFDA (25 µM) was added to each flow cytometer tube and incubated under dark for 30 min at 37°C with 5% CO2. Cells pre-treated with cell culture medium were designated as negative control; while, the ones exposed to 100 µM of tert-butyl hydroperoxide (THBP) for 4 h were used as the positive control. Data were analyzed by CellQuest Pro Software (Becton Dickinson, USA) and performed in triplicates.
Immunoblotting analysis
LC3B expression
To determine autophagy-dependent cell death, autophagosome marker LC3B expression was assessed by Western blotting. After 2 h pre-treatment with NAC, EUC, CUR, or EUC+CUR, CSE 20% (v/v) was added in T-25 flasks for 4 h. Cells were harvested and washed twice with PBS. Collected cell pellets were lysed in RIPA lysis buffer (10 mM Tris-HCl, pH 8, 0.32 mM sucrose, 5 mM EDTA, 2 mM dithiothreitol, 1 mM phenylmethyl sulfonylfluoride, and 1% Triton X-100). After incubation on ice for 15 min, extracts were clarified by centrifugation at 30678 g for 10 min at 4°C. The protein concentration of whole cell lysate was calculated by using Pierce bicinchoninic acid (BCA) protein assay kit (Thermo Scientific, USA).The total cell proteins were separated on SDS-polyacrylamide gel (14%) and then transferred to polyvinylidene fluoride membranes, which were blocked with 5% non-fat dry milk powder in TBST (10 mM Tris-HCl (pH 8.0), 137 mM NaCl, and 0.05% Tween-20) at room temperature for 1 h They were probed with primary antibodies LC3B 1:3000 (Abcam, USA) in TBST containing milk powder (5% w/v) β-actin (SantaCruz Biotechnology, CA, USA) as a loading control (1:5000 in TBST, v/v) overnight at 4°C. The following day, the membranes were incubated with horseradish peroxidase-conjugated secondary antibodies (1:5000 in TBST; SantaCruz Biotechnology, CA, USA). The blots were visualized using Bio-Rad Clarity Western ECL substrate (Bio-Rad Laboratory, USA) within Bio-Rad Universal Hood II- ChemiDoc™ XRS system (Bio-Rad Laboratory, USA) and all data were analyzed with ImageLab Software 6.0.1 (Bio-Rad Laboratory, USA). Total protein load and gel running were performed in triplicates.
Nrf2 expression
Cytosolic (cNrf2) and nuclear (nNrf2) Nrf2 expression were assessed to determine its translocation rate during treatments. After 2 h pre-treatment with NAC, EUC, CUR, or EUC+CUR, cells were exposed to CSE 20% (v/v) for 4 h. At the end of incubation, collected cell pellets were lysed in nuclear and cytosolic lysis buffer separately to isolate proteins in both nuclear and cytosolic fractions. Briefly, trypsinized cell pellets were centrifuged 1,000 g for 5 min at 4°C. After centrifugation, collected pellets were lysed in 100 µl of cytosolic lysis buffer containing 10 mM HEPES (pH 7.9), 10 mM KCl, 1.5 mM MgCl2, 0.5 mM dithiothreitol, 0.2 mM PMSF and 0.6% nonidet P-40, and protease inhibitor (PI) cocktail for 15 min in ice. Following, each pellet is vortexed and centrifuged at 3,000 × g for 15 min at 4°C. Supernatants were collected as cytoplasmic protein fraction. The remaining pellet was washed with cytosolic lysis buffer (without nonidet P-40). For each washing step, the pellet was resuspended in wash buffer and centrifuged at 3000 × g for 15 min at 4°C. After washing, remaining pellets were lysed in nuclear lysis buffer containing 25% glycerol (v/v), 20 mM HEPES (pH 7.9), 1.5 mM MgCl2, 2 mM KCl, 400 mM NaCl, 0.2 mM EDTA, 0.5 mM DTT, 0.2 mM PMSF and protease inhibitor cocktail. Pellets were resuspended and incubated in ice for 30 min with intermittent mixing and vortexing. Following, suspension was sonicated for 10 s and centrifuged at 16,000 g for 20 min at 4°C. Supernatants were collected as nuclear protein fraction. Following the same steps as stated in section 2.7.1, we probed the membrane with Nrf2 antibody (1:3000 in TBST v/v; Abcam USA) overnight at 4°C. For cytoplasmic protein extracts, β-actin (SantaCruz Biotechnology, CA, USA) was used as a loading control (1:5000 in TBST, v/v) while rabbit polyclonal anti-Lamin B Ab (1:5000 in TBST, v/v; Abcam, USA) was used as the loading control for nuclear protein extracts. Both nuclear and cytoplasmic protein load and gel run were performed in triplicates.
Determination of CSE-induced inflammation
Nitrite induction
Nitrite, a stable metabolite of nitric oxide and indirect marker of inflammation, was examined by using the Griess method in cell medium. 33 Briefly, cell supernatants were mixed with an equal amount of Griess reagent composed of sulfanilamide (1%, w/v), o-phosphoric acid and N-(1-naphtyl)ethylenediamine (0.1%, w/v) at 25°C and incubated for 5 min. Absorbances correlated with standard sodium nitrite concentration were detected at 540 nm using a microplate spectrophotometer (Thermo Scientific™ Multiskan, Finland). For each group, experiment was performed in triplicates.
IL-6 level
Cell supernatants of MDA assay were collected for IL-6 analysis. IL-6 level induced by CSE was measured by the human IL-6 ELISA kit (eBioscience; USA) according to manufacturer’s instruction. 50 µl of cell supernatant was used and each group was applied in duplicates.
Statistical analysis
All the data were generated as triplicates and presented as mean± standard deviation (SD) in results. Data were analyzed using GraphPad Prism 6.0 statistical Software (San Diego, California, USA) with one-way ANOVA followed multiple comparisons by Dunnett’s test. The value of p < 0.05 was regarded as the statistical significance.
Results
Cytotoxicity by MTT assay
Cytotoxic effect of CSE on BEAS-2B cell line has been investigated to determine the critical limits for applicable amounts. According to the results, 15–50% (v/v) of CSE have shown a significant reduction in cell viability. Among the tested doses of CSE, 20% (v/v) CSE with cell viability of 70.45 ± 2.28% compared to the control group has been selected for further experiments (Figure 2(a)). In the preliminary experiments, EUC was tested between 12.5–500 µM, while CUR was tested between 5–50 µM without CSE exposure. The doses exhibited cell viability above 70% were suggested as non-cytotoxic in BEAS-2B cells an thus, selected for further experiments (data were not shown). Two hours pre-treatment with EUC (12.5–200 μM), CUR (2.5–20 μM) and NAC (0.5–2 mM) followed by 4 hours CSE (20%, v/v) have been tested to assess cytotoxicity in BEAS-2B cells by MTT assay. According to our findings, EUC and CUR pre-treatments demonstrated cytotoxicity at higher doses, while NAC did not exhibit cytotoxicity between 0.5–2 mM (data was not shown). With the highest cell viability, NAC at 1 mM was selected as positive control for further experiments. On the other hand, 2 h pre-treatment with EUC (50–200 μM) and CUR (5–20 μM) and following 4 h CSE exposure led to dose-dependent response in cell viability (Figure 2(b)). Even though the selected doses of EUC and CUR were also tested as a combination (EUC+CUR), it showed no significant effect on cell viability compared to control and treatments alone, (Figure 2(c)). Among these, NAC pre-treatment displayed the most noteworthy effect on cell viability of CSE-exposed cells, according to MTT assay (Figure 2).

Dose-dependent cytotoxicity profile of CSE and treatments in BEAS-2B cell line. (a) Dose-dependent cytotoxicity of CSE in BEAS-2B. Statistical differences were indicated for control vs different doses of CSE as ap < 0.001, bp < 0.0001. (b) Cell viability of pre-treated and CSE (20%, v/v) exposed BEAS-2B cell. Statistical differences were indicated for CSE vs treatment groups as *p < 0.05, **p < 0.01, ***p < 0.001 and control vs CSE as ap < 0.0001. (c) Protective effect of NAC (1 mM), EUC (50 μM), CUR(5 μM) and EUC+CUR against CSE. Statistical differences were indicated for CSE (20%, v/v) vs treatment groups as *p < 0.05, **p < 0.01 and control vs CSE as ap < 0.001. CSE: cigarette smoke extract; CUR: curcumin; Ctrl: Control; EUC: eucalyptol; NAC: N-acetylcysteine (1 mM). All data were shown as mean ± SD.
Annexin V-PI staining
After the corresponding treatments determined in the previous section, BEAS-2B cells were examined with Annexin V-PI staining for cell death profile (Figure 3). According to our results, CSE exposure of BEAS-2B cells led to remarkable increase in early apoptosis (26.64%) while late apoptosis was recorded as 11.09% (Figure 3(b)). As similar to MTT assay results, live-cell ratio of BEAS-2B was decreased prominently (61.73%) upon CSE exposure. However, pre-treatments attenuated the CSE-induced early apoptosis (Figure 3(c) to (f)). The most effective impact on CSE-induced early apoptosis was seen with 1 mM NAC pre-treatment (Figure 3(c)). On the other hand, with increased doses of CUR (5–20 μM), late apoptosis rate increased (5.16% to 13.79%) in a dose-dependent manner (data was not shown).

CSE-induced apoptosis and the effects of pre-treatments on BEAS-2B cells. BEAS-2B cells were pre-treated with groups + 20% CSE for 4 h and stained with Annexin V-PI kit. (a) Control, (b) 20% CSE, (c) 20% CSE + 1 mM NAC, (d) 20% CSE + 50 μM EUC, (e) 20% CSE + 5 μM CUR, (f) 20% CSE + EUC+CUR. CSE: Cigarette smoke extract; CUR: Curcumin; Ctrl: Control; EUC: Eucalyptol; NAC: N-acetylcysteine.
CSE exposure significantly induced both apoptotic and necrotic cell death (Figure 4(a)). Pre-treatment with NAC notably reduced the early apoptosis rate, while the EUC+CUR combination was the most effective in lowering CSE-induced late apoptosis ratio (Figure 4(b)). Also, the lowest total apoptotic cell rate was seen with NAC and combination pre-treatment, respectively (Figure 4(b)).

CSE-induced cell death rate in BEAS-2B cells after Annexin V-PI labeling. BEAS-2B cells were pre-treated with groups followed with 20% of CSE exposure for 4 h. (a) Total apoptotic rate of BEAS-2B cells was quantified with flow cytometer after CSE exposure. DMSO (10%) and DOX (2 µM) were used as positive controls for apoptosis and necrosis, respectively. (b) CSE-induced total apoptosis rate was measured in BEAS-2B cells. aStatistical difference between control vs CSE p < 0.0001; *CSE vs treatment groups, p < 0.0001; **CSE vs 10% DMSO, p < 0.001; ***CSE vs 2 µM DOX; bNAC vs EUC, p < 0.05; cNAC vs CUR, p < 0.01. BEAS-2B: human bronchial epithelial cell line; CSE: cigarette smoke extract; CUR: curcumin (5 µM); Ctrl: Control; DMSO: dimethylsulfoxide; DOX: doxorubicin; EUC: eucalyptol (50 µM); NAC: N-acetylcysteine (1 mM); PI: propidium iodide.
Autophagy-dependent cell death
Autophagy-dependent cell death was evaluated with changes in LC3B protein expression level and the overall rate was calculated by using the ratio of LC3B-II over LC3B-I. 34 All pre-treatments down-regulated the expression of LC3B and lowered the ratio of LC3B-II/LC3B. Cells exposed to CSE showed important autophagosome formation through enhanced LC3B expression compared to the control group. The most attenuating effect on CSE-induced autophagy rate was seen with NAC pre-treatment, which was followed by EUC+CUR and EUC pre-treatment, respectively (Figure 5(b)). CUR pre-treatment has also declined CSE-induced autophagy rate, but not statistically significantly (Figure 5).

Pre-treatment with antioxidants alleviated CSE-induced autophagosis. BEAS-2B cells were pre-treated with related compounds and then exposed to 20% CSE for 4 h. Statistical significance between Ctrl vs. CSE ap < 0.0001; CSE vs. groups *p < 0.05; **p < 0.01. Ctrl: Control; CSE: Cigarette smoke extract; CUR: Curcumin; EUC: Eucalyptol; NAC: N-acetylcysteine; EUC+CUR: Eucalyptol and curcumin. The data were displayed as mean ± SD.
CSE induced oxidative stress
GSH level
Next, we decided to investigate the changes in oxidative stress conditions of BEAS-2B cells exposed to CSE alone or after pre-treatments to conclude the antioxidant capacity of our treatments. According to the results, CSE led to a significant increase in oxidative stress by reducing GSH levels significantly (Figure 6(a)). However, pre-treatment with NAC, also a synthetic precursor of glutathione, restored GSH level and even further than basal level (Figure 6(a)). A similar increase was also observed with pre-treatment with EUC (50 μM) and EUC+CUR combination. Also, pre-treatment with CUR (5 μM), showed a significant effect on GSH restoration by increasing its level nearly to the basal conditions (Figure 6(a)).

Effect of treatments on CSE-induced oxidative stress parameters. Antioxidant properties of applied compounds were evaluated in BEAS-2B cells exposed to 20% CSE for 4 h. (a) GSH level measurement in CSE-exposed BEAS-2B cells. (b) CAT activity analysis CSE-exposed BEAS-2B cells. (c) SOD activity analysis in CSE-exposed BEAS-2B cells. aStatistical difference between control vs CSE p < 0.001; *CSE vs groups, p < 0.05; **CSE vs groups, p < 0.01 and ***p < 0.0001.
CAT activity
Catalase (CAT) is an endogenous antioxidant enzyme responsible for detoxification of reactive hydrogen peroxide molecule into molecular oxygen and water 35 and its diminished activity was associated to pulmonary fibrosis. 36 According to our results, CSE diminished CAT activity significantly, which was blocked by pre-treatment with NAC, EUC and EUC+CUR compared to CSE group (Figure 6(b)). Besides, even the effect of CUR pre-treatment was significantly altering the effect of CSE exposure on BEAS-2B cells, it provided a limited change in restoring CAT activity relative to other treatments (Figure 6(b)).
SOD activity
As shown in Figure 6(c), CSE exposure slightly declined SOD activity in CSE group; however, pre-treatment with NAC, EUC or EUC+CUR combination enhanced SOD activity. NAC was the most promising treatment among all. On the other hand, CUR pre-treatment was not effective and resulted in pretty much similar SOD activity as in the CSE sample, which was not statistically different from the control group.
Lipid peroxidation
As shown in Figure 7, cytoprotective doses of NAC, CUR, EUC and EUC+CUR are effective against CSE-induced lipid peroxidation on BEAS-2B cells. According to our results, NAC pre-treatment again resulted in the most efficient application in restoring MDA levels, which was followed by EUC (Figure 7). Its combination with CUR showed a similar decline in MDA level, as well. Pre-treatment with CUR alone has led to a slight decline in the MDA level, albeit it was statistically insignificant (Figure 7).

CSE induced lipid peroxidation in BEAS-2B cells. Cytoprotectivity of applied compounds against lipid peroxidation rate was measured with MDA analysis in BEAS-2B cells exposed to 20% CSE for 4 h. aStatistical difference between control vs CSE; bCSE vs groups, p < 0.05; cCSE vs groups, p < 0.01. CSE: cigarette smoke extract; CUR: curcumin; EUC: eucalyptol; NAC: N-acetylcysteine. The data were shown as mean ± SD.
Nrf2 expression
Nrf2 is the master regulatory protein of specific antioxidant response maintained at a low basal level and it was reported to be associated with defects in the lung antioxidant defense. 21 Protein expression of Nrf2 in two different cellular fractions, cytosol and nuclei, were investigated in BEAS-2B cells. According to our western blot results, pre-treatment with NAC with/without CSE exposure, led to a significant cytosolic accumulation of Nrf2 as shown in Figure 8. However, pre-treatment with EUC, CUR and their combination limited cytosolic Nrf2 expression with/without CSE exposure (Figure 8(a)).
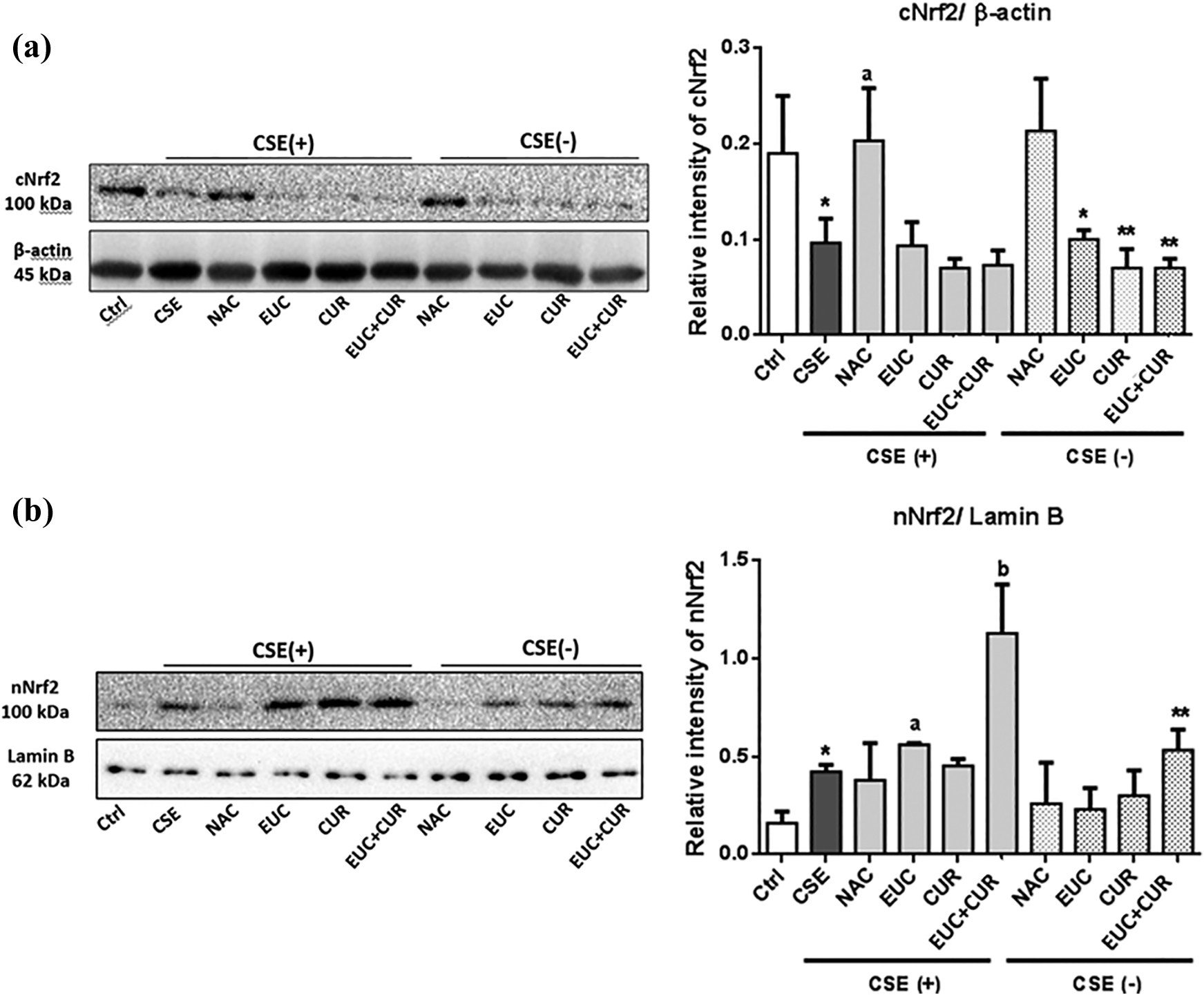
Translocation of Nrf2 in BEAS-2B cells. The effect of the compounds on the translocation of Nrf2 was examined in BEAS-2B cells pre-treated with compounds and 20% CSE for 4 h. (a) Cytosolic expression of Nrf2 (cNrf2); aStatistical difference between CSE vs NAC p < 0.01; Ctrl vs groups *p < 0.05 and **p < 0.01. (b) Nuclear translocation of Nrf2 (nNrf2); aStatistical difference between CSE vs EUC p < 0.05; bCSE vs EUC+CUR p < 0.0001; differences between Ctrl vs groups, *p < 0.01, **p < 0.0001. Ctrl: Control; CSE: Cigarette smoke extract; CUR: Curcumin; EUC: Eucalyptol; NAC: N-acetylcysteine, EUC+CUR: Eucalyptol and curcumin. The data were shown as mean ± SD.
On the other hand, pre-treatment with EUC and EUC+CUR have significantly increased the nuclear translocation of Nrf2 (Figure 8(b)). Moreover, all of the treatments increased nuclear expression of Nrf2 without CSE, but the most promising effect was observed with combination treatment, acted as Nrf2 activator itself. Especially EUC and combination pre-treatment followed by CSE exposure led to a significant nuclear translocation of Nrf2 in BEAS-2B cells (Figure 8(b)).
Intracellular ROS production
CSE exposure significantly up-regulated the intracellular ROS level based on the detected DCF fluorescence signal in each sample group (Figure 9). CSE exposure for 4 hours increased relative ROS level up to 40%. EUC and CUR combination showed more potent ROS scavenging potential compared to each treatment alone. According to the results, pre-treatment with NAC displayed a greater decline in intracellular ROS production compared to other pre-treatments (Figure 9).

Intracellular ROS level induced by CSE. The effect of the compounds on ROS generation was examined in BEAS-2B cells pre-treated with 20% CSE for 4 h. (a) Representative histograms for percentage of increase of ROS accumulation in CSE-exposed groups. Enhancement of intracellular ROS level observed via the shift of the signal curve obtained for the treated cells to the right compared with that of the control cells. (b) Bar graph presenting the mean relative ROS percentage of three independent experiments. Statistical significance between CSE vs groups *p < 0.05; **p < 0.01; ***p < 0.0001; Ctrl vs groups ap < 0.001; bp < 0.0001. CSE: cigarette smoke extract; CUR: curcumin (5 μM); DCF: Dichlorofluoresceine; EUC: eucalyptol (50 μM); NAC: N-acetylcysteine (1 mM); NC: negative control; PC: positive control THBP (100 μM). The data were shown as mean ± SD.
CSE-induced inflammatory response
Nitrite level
Nitric oxide (NO) is a critical signaling molecule that is involved in inflammatory signaling and oxidative damage as well through nitrate-nitrite-NO axis. 37 Under physiological conditions, NO itself is difficult to measure directly due to its short half-life in biological fluids, thus its inactive and stable end-product nitrite can be measured as inflammation parameter. 38 In our study, CSE significantly induced nitrite production in BEAS-2B cells compared to the non-treated control group. According to our results, 2 h pre-treatment with NAC lacks the reducing effect for nitrite in BEAS-2B cells (Figure 10). However, EUC and CUR slightly reduced the effect of nitrite in BEAS-2B cells at experimental doses. Besides, the combination of EUC+CUR significantly attenuated CSE-induced nitrite production (p < 0.05) (Figure 10).

CSE induced nitrite level in BEAS-2B cells. The effect of the compounds on nitrite production was recorded in BEAS-2B cells pre-treated with 20% CSE for 4 h. Statistical significance between CSE vs groups *p < 0.05; **p < 0.0001. CSE: cigarette smoke extract; CUR: curcumin; EUC: eucalyptol; NAC: N-acetylcysteine. The data were shown as mean ± SD.
IL-6 level
CSE exposure of BEAS-2B cells significantly increased IL-6 level compared to the non-treated control group. Similar to the results of nitrite level analysis, IL-6 level was not significantly changed by NAC pre-treatment compared to the CSE group (Figure 11). However, EUC+CUR combination led to a drastic decline in CSE-induced IL-6 level. This result demonstrates the efficacy of EUC and CUR combination at the molecular level compared to the NAC application.

Relative IL-6 level of CSE-exposed BEAS-2B cells. The effect of the compounds on IL-6 level was measured in BEAS-2B cells pre-treated with 20% CSE for 4 h. aStatistical difference between Ctrl vs CSE p < 0.0001; bCSE vs groups p < 0.0001. Differences between two groups: *p < 0.001 and **p < 0.0001. CSE: cigarette smoke extract; Ctrl: control; CUR: curcumin; EUC: eucalyptol; NAC: N-acetylcysteine. The data are shown as mean ± SD.
Discussion
Bronchial epithelia are one of the most important primary targets of CS exposure. In this study, EUC, CUR, and their combination acted as cytoprotective and ameliorative in the oxidative and inflammatory response induced by CSE in the BEAS-2B toxicity in vitro. CSE used in our study is also representing passive smoke exposure for non-smokers, which are mainly exposed to acute secondhand smoke during their daily life. Therefore, in vitro models representing chronic exposure (a total of 24–72 or more hours of exposure) have not been followed in the present study. According to limited data in the literature, respiratory mucolytic and spasmolytic EUC is shown to be cytoprotective in BEAS-2B cell line up to 10 µg/mL (64.82 μM). 39 On the other hand, CUR, a well-known traditional medicine, which has been widely used as protective against pulmonary diseases,40,41 displayed the least effect on cell viability (p < 0.05) among all treatments in our study (Figure 2). In a report by Vanella et al., it was indicated that 5 μM CUR pre-treatment had no proliferative action on BEAS-2B cells exposed to CSE. 24 However, Zhu et al. suggested that CUR pre-treatment reversed BaP-induced cytotoxicity up to 5 μM in BEAS-2B cells. 42 Similarly, several reports indicated that CUR between 2.5–7.5 μM has been found to be ameliorative in human bronchial epithelial cells,43,44 which was supportive of our observations as well. In addition, results of Zhu and colleagues has showed that the increased dose of CUR led to a prominent decline in cell viability, which was similar to the represented findings in our study. On the other hand, positive control NAC (1 mM), a potent radical scavenger and mucolytic agent, demonstrated the most promising protective effect in the cell viability against CSE (p < 0.01) (Figure 2). In addition to its antioxidant and anti-mucolytic activity, NAC might be suggested to have a broad therapeutic range up to 10 mM. Therefore, it differs from the natural substances investigated in the present study and might be suggested as a suitable positive control in pulmonary toxicity studies based on the previous findings.11,24,45 In this study, CSE exposure led to an increase in early apoptotic cell rate significantly by Annexin V-PI staining (p < 0.0001) (Figure 4). CSE-induced apoptosis is reported to be related to activation of cleaved caspase-9 and caspase-8, 46 or anti-apoptotic Bcl-xL decline and apoptotic caspase-3 activation in a time and dose-dependent manner. 47 In the present study, pre-treatment with EUC, CUR, and combination attenuated CSE-induced early apoptotic cell death ratio (Figure 4(a)). EUC pre-treatment significantly reduced CSE-induced early apoptotic cell death ratio in BEAS-2B cells (p < 0.0001) (Figure 4(b)). Moreover, CUR, also reduced the apoptotic cell ratio induced by CSE. According to a study by Zhu et al., it was indicated that lower doses of CUR (5 μM) acted as cytoprotective while higher doses acted as pleiotropic and induce apoptosis in human bronchial epithelial cells 42 . which correlates the findings of the present study. Therefore, it might be suggested as CUR has dual effects on cell survival, depending on the dose. On the other hand, NAC led to the most prominent decline in CSE-induced apoptosis, which was correlated with our cell viability results. It was stated that apoptotic cell death might be triggered by excessive ROS production, which can be reversed by scavenging of radicals, mainly H2O2 and O2•−11. In the present study, CSE exposure significantly elevated ROS level, which was attenuated by treatments (Figure 9). The most promising ROS scavenging activity was seen with NAC, which was followed by combination pre-treatment in parallel with cell death results (Figures 2 and 9). Hence, it is plausible to suggest that preventive action of treatments, particularly NAC, acted as protective on CSE-induced ROS production and oxidative injury. Aside from apoptosis, autophagy-dependent cell death initiated by autophagosome formation has been reported to have a pivotal role in lung disease progression such as COPD. 46 During autophagy-dependent cell death, cytoplasmic free form LC3B-I is converted to autophagosome formation indicator LC3B-II in the mammalian system. 48 According to our findings, autophagy rate (LC3B-II/LC3B-I) through autophagosome formation was significantly increased with CSE exposure (Figure 5). Similar to our results, Zhou et al. reported that CSE may lead to apoptosis in parallel with autophagy-dependent cell death through LC3B expression. 49 In the present study, pre-treatment with EUC reduced CSE-induced autophagy rate significantly by diminishing LC3B-II/LC3B-I ratio (p < 0.05) (Figure 5). Previously it was reported that EUC has induced LC3B-II accumulation in mouse endothelia in vitro, thus increased autophagy rate. 50 However, the data on the effects of EUC on autophagy is insufficient. According to our findings, EUC diminished CSE-induced autophagy through LC3B-II/LC3B-I rate in BEAS-2B cells. On the other hand, CUR pre-treatment has led to a slight decrease in autophagy rate, which was insignificant compared to the CSE group. In parallel to the results of apoptotic cell death, combination treatment significantly reduced CSE-mediated autophagy through LC3B expression. However, LC3B-II/LC3B-I rate was not statistically different than the one recorded with CUR or EUC alone. Thus, no additive effect was observed on CSE-induced autophagosome formation. Overall, it might be reasonable to suggest that not only mitochondrial dysfunction but also apoptosis and autophagy-dependent cell death are involved in CSE-induced toxicity in BEAS-2B as target adverse outcomes. In addition to LC3B expression, p62 and Atg5 expression involved in autophagy have been reported to be impaired in human bronchial epithelial cells isolated from smoker COPD patients. 51 Therefore, autophagy has an important role in COPD progression and other smoking-related pulmonary diseases,12,46 thus representing an important therapeutic target.
Oxidative injury and lipid peroxidation are the other important key events involved in CS-induced target toxicity.21,34,52 According to results, CSE exposure reduced CAT activity (p < 0.001), intracellular GSH level (p < 0.001) and up-regulated MDA level (p < 0.05) compared to control (Figure 7). However, in contrast to in vitro findings,13,53 SOD activity was slightly decreased, which might be due to the low relative O2•− content of CSE or instability of O2•− (Figure 6). Previously, O2•− anion was indicated as an unstable ROS present in the gas phase of CSE. 54 Therefore, the observed result is suggested to be due to the unstable nature of O2•− in used CSE. On the other hand, EUC pre-treatment attenuated CSE-induced oxidative stress by enhancing the activity of both SOD and CAT in BEAS-2B (p < 0.01). Also, intracellular GSH has been replenished significantly by EUC compared to both CSE and control groups (p < 0.01), which might represent an important source for GSH deposition in the biological system (Figure 6). On the other hand, CUR treatment significantly increased intracellular GSH level and CAT activity compared to the CSE group (p < 0.01) (Figure 6). However, no significant effect was seen on SOD activity and MDA level. Therefore, it can be suggested that CUR at lower doses did not act as potent as EUC on SOD activity and lipid peroxidation induced by CSE. Moreover, the combination treatment has showed similar results observed by EUC treatment alone. Therefore, EUC+CUR combination did not act as additive or synergistic on quenching CSE-mediated oxidative stress. Aside from its effects on enzymatic antioxidant activity, CSE is well-known to enhance electrophilic attack and activates nuclear translocation of Nrf2, thus up-regulates antioxidant defense as a cellular response.5,21 Therefore, targeting Nrf2 is suggested as an important therapeutical strategy in the treatment of chronic airway diseases.55,56 CSE exposure significantly induced Nrf2 nuclear translocation due to increased oxidative stimuli (Figure 8) and pre-treatment with EUC, CUR and their combination followed by CSE exposure increased expression of nNrf2 and acting as Nrf2 activators itself. Therefore, it might be suggested that EUC, CUR, and their combination attenuated CSE-induced oxidative injury through Nrf2 activation. Previously, EUC has been suggested to act as a Nrf2 activator. 57 Besides, Jiang et al. revealed that EUC activated nuclear translocation of Nrf2 and acted as Keap-1 inhibitor, thus decreased cytoplasmic accumulation of Nrf2 in rat liver. 58 Similar to the mentioned studies, our data indicate that Nrf2 activation was significantly enhanced by EUC, which might promote cell survival in CSE-exposed BEAS-2B cells (Figure 8). On the other hand, CUR pre-treatment activated nuclear translocation of Nrf2 as well, as suggested in the literature.8,59,60 Similar to findings of Shen et al. CUR acted as a weak Nrf2 inducer in our study as well, possibly due to its low bioavailability. 60 On the other hand, combination of EUC and CUR showed the most promising effect on Nrf2 activation (Figure 8). However, Nrf2-dependent antioxidant gene expression such as HO-1, glutamate-cysteine ligase, and NAD(P)H-quinone oxidoreductase-1 might be investigated to elucidate the adaptive response driven by EUC and CUR. Also, NAC treatment was found to interfere with nuclear translocation of Nrf2 which might be due to cysteine residues on Nrf2 inhibitor Keap-1 and consequent interference with the ubiquitination of Nrf2, which resulted in its proteasomal degradation. 61 Therefore, cysteine and Keap-1 interaction might be the reason for Nrf2 inactivation by NAC treatment. Another contributing factor for the progression of CS-induced pulmonary diseases is inflammation.62–64 In the present study, CSE exposure elevated the levels of both stable metabolite of NO, nitrite, (Figure 10) and pro-inflammatory cytokine IL-6 (Figure 11) in BEAS-2B cells (p < 0.0001). Although EUC and CUR treatments did not show a noteworthy decrease in nitrite level alone, combination treatment diminished the CSE-induced nitrite level significantly (p < 0.05) (Figure 10). Moreover, the pro-inflammatory IL-6 level was decreased with each treatment, particularly with combination treatment as well (p < 0.0001) (Figure 11). Since Nrf2 activation is involved in IL-6 release by up-regulating anti-inflammatory gene expression and mediating the pro-inflammatory cytokine release, 65 EUC and EUC+CUR combination induced Nrf2 activation might be involved in the reduction of CSE-induced IL-6 level.
Conclusion
In conclusion, the present study demonstrated that the EUC and CUR combination reduced CSE-induced oxidative stress and inflammation, thus promoting cell survival in vitro as valuable background for further in vivo studies. Moreover, the Nrf2-ARE pathway might represent an important tool for interpretation of pulmonary toxicity owing to its regulatory effect on cytoprotection through variable mechanisms. Therefore, the present study might represent a valuable background for further studies on the toxicity assessment of tobacco products.
Footnotes
Acknowledgment
The authors would like to thank Prof. Dr. Mustafa Çulha for equipment support during flow cytometer studies and Dr. Mohammed Ali Selo for technical help for nuclear protein isolation.
Author contributions
Conceptualization: RR, HS, HC; Data curation: RR, DO, DY; Formal analysis: RR, HS; Investigation: RR; Methodology: RR, HS, HC; Project administration: HS, RR; Resources; Software: HS, HC, DY; Supervision: HS, HC; Visualization: RR; Roles/Writing—original draft: RR; Writing—review and editing: RR, HS, HC, DY, DO.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

