Abstract
The paraoxonase (PON) family is composed of 3 proteins (PON1, PON2, and PON3), each of which plays a crucial role in the body, displaying antioxidant, anti-inflammatory, and antiatherosclerotic properties. The activities and properties of PON proteins can be modulated by a number of environmental factors, including cigarette smoke. In the present article, a review of existing literature is employed to analyze both the direct and the indirect impact of cigarette smoking on the activity of members of the PON family. Cigarette smoking leads to direct inhibition of the hydrolytic activity of PON enzymes by modification of thiol groups, by the reactions of free radicals, or by inhibiting enzyme-active regions with heavy metals. It has been shown that cigarette smoking correlates with a decrease in high-density lipoprotein (HDL) concentration as well as with an increase in other components of the lipid profile (low-density lipoprotein (LDL), triglycerides, and total cholesterol). By decreasing HDL levels, cigarette smoking likely acts indirectly to induce a decline in PON1 activity. Inhibition of PON1 activity by smoking is a reversible process after cessation of exposure to the xenobiotics in tobacco smoke.
Introduction
Cigarette smoking constitutes a major social problem due to its prevalence and its harmful effects on society. Worldwide approximately 40% of men and 9% of women smoke. 1 Each year, smoking causes more than 5 000 000 deaths, including the deaths of 1 500 000 women and 3 500 000 men. 1
Cigarette smoke contains about 6000 toxic carcinogens and mutagenic compounds. 2 It is a source of free radicals, which disrupts the pro/antioxidant balance in the body and cause changes in inter alia lipid profile compounds. The resultant peroxidation products are considered to be highly important in the pathogenesis of atherosclerosis. 3
Paraoxonase (PON) enzymes, a 3-member group composed of PON1, PON2, and PON3, are regarded as crucial in antiatherosclerotic processes. The PON enzymes display antioxidant and antiatherogenic activities—particularly enzymes are involved in inhibiting lipoprotein oxidation, inactivating toxic peroxidation products, and preventing their accumulation. Prooxidant factors, including cigarette smoke, are capable of modifying the activity and antioxidant properties of all PON forms. 4,5
Harmfulness of Cigarette Smoking
Tobacco smoke is a mixture of substances whose toxicity leads to the development of a range of diseases, particularly of the respiratory and circulatory systems. It contains a high volume of toxic, irritant, carcinogenic, and mutagenic substances that block the activity of the respiratory system. 2 It is composed of both gas and particulate phases. The gas phase includes over 500 gaseous substances such as carbon monoxide, carbon dioxide, nitrogen oxide, ammonia, pyridine, formaldehyde, hydrogen cyanide, and acrolein. The particulate phase contains over 3500 chemical compounds, mainly tar products such as benzene, benzopyrene, phenols, alkaloid nicotine, arsenic, polonium, radium, nickel, and cadmium. 6 Furthermore, the presence of free radicals, which lead to oxidation of the thiol groups of proteins and an increase in low-density lipoprotein (LDL) oxidative modification, has been observed in both the gas and the particulate phases. 6 Circulating products of lipid peroxidation and lipid profile alteration are considered to be important in the pathogenesis of atherosclerosis, which leads to consequences involving further changes in the cardiovascular system such as ischemic limbs, heart attack, and stroke. 3
Furthermore, tobacco smoke potentiates pro-inflammatory cytokine production and reduces levels of anti-inflammatory cytokines. It also leads to increased levels of immunoglobulin E antibodies and the subsequent development of atopic diseases and asthma. It has a deleterious effect on the immune system, causing relative immunodeficiency and increased susceptibility to infections. 7 Changes in coagulation have also been observed: platelets isolated from smokers showed spontaneous aggregation and a correlation has been noted between higher fibrinogen levels in smokers and the number of cigarettes smoked per day. 8
Cigarette smoking is both a direct and an indirect cause of many diseases. A strong correlation between cigarette smoking and the development of lung, larynx, oral cavity, lip, nasal cavity, sinus, throat, esophagus, stomach, pancreas, kidney, bladder, uterus, cervix ovaries, colon and rectal cancers, along with acute myeloid leukemia, has been observed. 9
A very large amount of free radicals are produced during cigarette smoking. 4 Studies demonstrate that the gas phase of tobacco smoke contains 1015 free radicals/puff, while the particulate phase contains 1017 free radicals/g. 8 Forms present in the gas phase are unstable and remain reactive for only a few seconds, while forms located in the particulate phase are stable with a life span ranging from several hours to several months. 8 They are highly reactive, engaging with every type of biomolecule present in the body: proteins (aggregation and denaturation), lipids (peroxidation), carbohydrates, and nucleic acids (changes in the structure of DNA), leading to damage to cell structures. 10,11 In addition, they destroy the body’s antioxidant defense system, disrupting the pro/antioxidant balance, and consequently leading to oxidative stress. 12
In order to prevent damage caused by oxidative stress and pro/antioxidant imbalance, the human body has developed a protective mechanism in the form of an antioxidant defense system whose task is to inhibit the generation of free radicals and participate in their transformation into inactive derivatives. 13 An example of one such protective mechanism is the behavior of antioxidants referred to as PON.
Paraoxonase
In 1946, the characteristics of a plasma enzyme capable of inducing hydrolysis in organophosphorus compounds was elaborated, 14 which was later given the name paraoxonase. Its name derives from the most commonly used substrate in in vitro reactions, paraoxon. 15 Paraoxonase (EC 3.1.8.1) is a multigene family composed of 3 members (PON1, PON2, and PON3) encoded by 3 genes, PON1, PON2, and PON3, respectively. 3 Each of these genes is located on the long arm of chromosome 7q 21.3-22.1, probably originating from the tandem duplication of a common evolutionary precursor. 16 Studies conducted on human tissues show 60% identicalness at the amino acid level and 70% identicalness at the nucleotide level within the PON family, which makes it a conservative group of enzymes and determines its high substrate specificity. 17
PON family members exhibit hydrolytic activity with a large number of different substrates,
17
including: lactonase activity, for example, against homocysteine thiolactone, arylesterase activity, for example, against phenyl acetate, and organophosphatase activity, for example, against paraoxon.
18
PON1 has been proven to exhibit all of the above-mentioned activities. PON2 and PON3 exhibit only lactonase activity. 18,19,20 Moreover, according to some studies, PON3 displays limited arylesterase activity. 19
PON1 is a calcium-dependent enzyme associated with high-density lipoprotein (HDL). Initial studies of PON1 properties showed only the ability to hydrolyze an organophosphate substrate. More recent research focuses primarily on its antioxidant and anti-inflammatory capacities as well as the potential pathophysiological effects of activity changes. 21
PON1 is a glycosylated protein of 355 amino acids with a molecular mass of 43 to 45 kDa. 22 It is composed of a 6-bladed β-propeller 22 ; each blade consists of 4 β-sheets linked to one another by a disulfide bond stabilizing the structure. 23 PON1 also contains 3 cysteines at positions 42, 284, and 353. Cysteine-284 is free, 24 and scientists have assigned it the protective role of an enzyme. 23
Initial studies on the role of PON1 show its hydrolytic activity with organophosphates and nerve gases (eg, paraoxon, diazoxon, and sarin), resulting from arylesterase and organophosphatase activities. 4,25 PON1 is also able to inhibit lipid peroxidation. The process of LDL oxidation is an important initial step in the development of atherosclerosis. Oxidized products are removed by macrophages, which transform into foam cells. Foam cells are filled with cholesteryl ester, forming atherosclerotic plaques. The role of PON1 is to inactivate the toxic products of LDL oxidation (ox-LDL). 26 Moreover, it prevents the accumulation of ox-LDL and stimulates cholesteryl efflux from macrophages via HDL. 5 PON1 also suppresses monocytes-macrophages transformation, thereby reducing the formation process of foam cells and limiting the creation of atherosclerosis plaques. 27 In addition, the presence of apolipoprotein A-I (apo A-I) in an HDL molecule stimulates lactonase activity of PON1 and the aforementioned antiatherosclerotic functions. 18 Research has frequently shown that serum PON1 activity is inversely proportional to the risk of atherosclerosis and related diseases. 18 In pathological condition, PON1 is separated from HDL and converted to a less-active form, which explains the greater probability of cardiovascular disease in patients with conditions such as diabetes. 18
Studies conducted on mice show the presence of messenger RNA PON1 in tissues such as the liver, lungs, heart, brain, intestine, and kidney. 16 Human PON1 is produced mainly in the liver, after which it is secreted into the blood, where it occurs in conjunction with HDL. 22
More than 160 polymorphisms of the PON1 gene have been elaborated; they may be present in exons (coding sequences), in introns (noncoding sequences), and in regulatory parts of the gene. 28 Most of them are the single-nucleotide polymorphisms. 25 The most important mutations are 2 of them found in the coding region: the missense mutation at position 192 (Q192R), which concerns the substitution of glutamine (Q) by arginine (R), and the amino acids substitution at position 55 (L55M), which concerns replacement of leucine (L) by methionine (M). The polymorphism Q192R has been investigated more extensively than L55M. The differences between them refer to affinity and catalytic activity with respect to numerous substrates. 25
The polymorphism PON1Q192R has been extensively described. Numerous studies of genotypes—PON1192QQ, PON1192QR, and PON1192RR—have been conducted to determine the differences in the polymorphic forms of the hydrolytic activity of the enzyme and resulting susceptibility to the development of certain diseases. One of the initial studies on the functions on PON1 concerned hydrolysis of organophosphate compounds. It demonstrated that the protective function of PON1 depends not only on the level of the enzyme in the blood and tissues but also on the type of PON1 polymorphism. 4 For instance, Billecke et al 29 analyzed the efficiency of hydrolysis of more than 30 substances, including toxic ones, depending on the enzymatic activity of various genotypes of PON1Q192R. They found that the presence of the R allele leads to a 6-fold increase in the speed of hydrolysis of paraoxon and some lactones (eg, thiolactone and γ-butyrylolactone), whereas other lactones (eg, 2-benzofuran and δ-valerolactone) were much more efficiently hydrolyzed by the Q allozyme. The polymorphism of ATG to TTG has also been identified at the nucleotide level within the PON1 gene. At the amino acid level, this results in the presence of L or M at position 55, conditioning the phenotype PON155L and PON155M, respectively (polymorphism L55M). 19,30 Studies have identified positive associations between the alloform PON155M and low activity of PON1 15 and confirmed greater hydrolytic enzyme activity in the presence of leucine at position 55. 25
In terms of structure, roles, and regulation, PON 2 is far less understood than PON1. PON2 is involved in the body’s defenses against inflammatory reactions. In addition, it is also counted among the antioxidant defenses that may prevent the development of atherosclerosis. 21,31 Similar to PON1, PON2 occurs in 2 major polymorphic forms that have been identified in the coding region. Amino acid substitution at position 148 concerns the transformation of alanine into glycine, and the transformation of cysteine into serine at position 311. 25,32 Cysteine at position 311 may presumably be a potential active center of hydrolysis of oxidized lipids, and substitutions of cysteine may block catalytic activity. 33
PON2 is not a plasma enzyme, nor it is related to the HDL or LDL molecule, but it remains intracellularly bound to the cell membrane. The enzyme has been found in practically all human tissues, 21 mainly in the liver, where it is produced, as well as in the lung, placenta, testis, and heart. 31
Cells demonstrating overexpression of PON2 are able to reverse the minimal oxidation changes formed in LDL particles. This suggests that PON2 plays an anti-inflammatory role and also acts as a “cellular antioxidant.” 34 Studies indicate that PON2 in macrophages significantly reduces lipid peroxide (lipid free radicals) and inhibits the oxidation of LDL. 35 Expression of PON2 in these cells increases during oxidative stress, 34 and Shiner observed that this is dependent on the dose of oxidizing agents. 36 In addition, some of the most recent studies suggest that PON2 also possesses neuroprotective properties. 37
PON3 is a calcium-dependent glycoprotein with a molecular mass of 40 kDa, which catalyzes the hydrolysis of several substrates including pharmacological agents. 6 Human PON3 consists of 354 amino acids. 38 PON3 is detected in small amounts in the kidney and the liver, where it is synthesized. It is secreted into the bloodstream, where it is associated with HDL lipoprotein fraction. 20 There is also a hypothesis that PON3 primarily acts intracellularly and is not secreted physiologically into the blood, but its presence in serum is rather a consequence of pathological processes. 39 PON3, like PON1, has 3 conservative cysteine residues at positions 41, 283, and 351, and exhibits similar properties in both structure and activity. There are differences between forms resulting from hydrolytic activity: PON3 displays very limited arylesterase activity but shows significant lactonase activity. 19
Previous studies conducted on cell lines and transgenic mice have shown that PON3 has antioxidant and anti-inflammatory properties, and it is also able to inhibit LDL oxidative modification, which may also facilitate monocyte activation. 19,20,38 Unlike PON1 and PON2, PON3 expression is constant and does not depend on the intensity of oxidative agents. 20 In addition, Draganov 19 has shown that PON3 is 100 times more effective than PON1 in the protection of LDL against oxidative modification induced by copper ions.
Similar to PON1 and PON2, PON3 is also polymorphic, and some PON3 gene polymorphisms are able to modify activity levels of PON1 in serum. 40
Cigarette Smoking and PON Enzymatic Activity
The activity and antioxidant properties of PON proteins can be modulated by a number of environmental factors. One of them is the process of cigarette smoking, which generates prooxidant changes in the human body and disrupts lipid metabolism (Figure 1). Numerous studies have been conducted to determine the direct effects of cigarette smoking on the properties of PON family members. At the same time, smoking may exert indirect effects on PON enzymes’ activities by modifying the components of lipid profile directly related to these proteins. To verify the above-mentioned assumptions, the relationship between PON enzymes and lipid profile was examined.
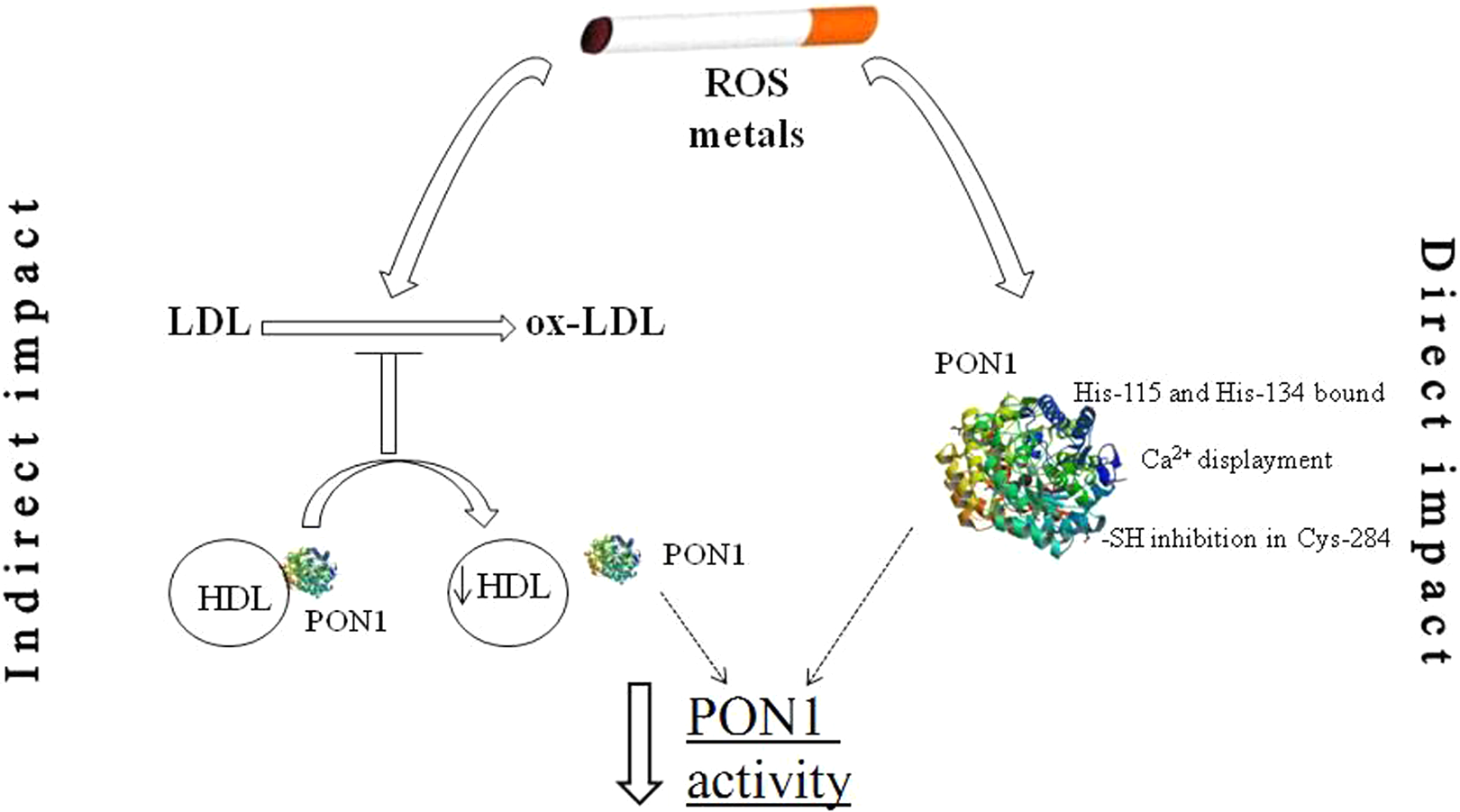
Mechanism of interaction of tobacco smoke’s xenobiotics on the PON1 activity. Cigarette smoking leads to direct and indirect inhibition of PON enzymes activity. Direct impact: metals present in tobacco smoke can interact with histidines (His-115 and His-134), display calcium ions, and modify free thiol groups. Indirect impact: cigarette smoking declines HDL concentration, leading to decrease PON1 activity, which is conjugated with HDL. PON indicates paraoxonase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; ox-LDL, oxidized low-density lipoprotein; ROS, radical oxygen species.
Inhibition of PON—The Direct Impact
Cigarette smoking is related to a decrease in HDL and increase in ox-LDL levels, which may cause pathophysiological changes in the antioxidant system. The role of the PON family is to protect HDL and LDL from oxidation. These reports led to some researchers advancing the hypothesis that cigarette smoking may be negatively correlated with their activities.
Senti et al 5 conducted research to determine the relationship between cigarette smoking and PON1 activity. Volunteers in the study were divided into smokers and nonsmokers. In both groups, the effects of cigarette smoking on the antioxidant system were determined by assaying the enzymatic activity of PON1, lipid profile, and degree of lipid oxidation. As expected, smokers had lower levels of PON1 activity, higher levels of triglyceride, and lower levels of HDL. Cigarette smoke contains a large number of substances with oxidizing properties, which generate prooxidant changes not only in cells but also in enzymatically active biomolecules. However, the exact mechanism of this inhibition has yet to be elucidated. Mouhamed et al 41 obtained similar results and conclusions showing a statistically significant association between cigarette smoking and decreased PON1 activity. In addition, his research took the intensity of smoking into account. Volunteers were divided into 4 groups: nonsmokers, people smoking ≤10 cigarettes per day, people smoking 11 to 20 cigarettes per day, and people smoking more than 20 cigarettes per day. In each group, regardless of the number of cigarettes smoked, there was statistically significant inhibition of PON1, which increased together with the number of cigarettes smoked per day. Additionally, a correlation was demonstrated between smoking and increased lipid peroxidation, which contributes to the weakening of the body’s antioxidant defense system. 42 These results suggest that even small amounts of harmful substances introduced into the circulatory system during smoking exceed the body’s capabilities, causing pathophysiological changes.
Studies have also been performed which were intended to facilitate a more accurate assessment of the impact of cigarette smoke on PON1 activity by standardizing exposure to tobacco smoke. To minimize the impact of various diseases and variables such as the number and type of cigarettes, a group of healthy nonsmoking volunteers was examined. The plasma of participants was exposed to cigarette smoke extract (CSE) obtained from a single cigarette. The impact of the concentration of substances contained in the CSE and the time of its incubation with plasma on the activity of PON1 were determined. The activity of PON1 decreased in proportion to increasing values of these 2 parameters (time and concentration). 43 The obtained results unequivocally indicate the presence of inhibitors of PON1 activity in cigarette smoke.
Cigarette smoke generates the formation of highly reactive oxygen species which are capable of modifying biomolecules and, consequently, would presumably inhibit PON family members. Therefore, the influence of endogenous antioxidants such as superoxide dismutase (SOD) and catalase (CAT) on the activity of PON1 in the presence of CSE has been analyzed. 43 None of the above-mentioned enzymes showed a protective effect and thus they do not impact the extent of PON1 activity inhibition. On the other hand, PON1 incubated with reduced glutathione retained a higher percentage of its initial activity. Glutathione, as distinct from SOD and CAT, has a free thiol group, which may be involved in the described protective effect.
This suggests that the inhibition of PON1 activity caused by smoking occurs via the modification of free thiol groups. To verify this hypothesis, an analogous experiment was conducted using compounds containing rich sulfhydryl (thiol) groups, such as N-acetyl-cysteine, dithiothreitol, 2-mercaptoethanol, and
Substances in Cigarette Smoke Influencing the Activity of PON
Cigarette smoke contains thousands of chemically active substances. In order to identify compounds which inhibit the hydrolytic activity of PON1, a number of substances were analyzed in both the gas phase and the particulate phase of tobacco smoke. 43 These include nicotine, palmitic acid, acrolein, acetonitrile, and acetaldehyde. Human plasma was incubated with these chemicals and with CSE. The concentrations of substances used in the study were 20 times higher than those normally present in a single cigarette. After 6 hours of incubation at 37°C, total PON hydrolytic activity was measured. The results obtained after incubation of plasma with CSE confirmed that acetaldehyde inhibits the hydrolytic activity of PON1. However, other substances, such as nicotine, palmitic acid, acetonitrile, and acrolein, do not show statistically significant inhibition of PON1 activity.
Other inhibitors of PON enzyme activities may also be heavy metals. Cigarette smoke contains nickel, cadmium, lead, and mercury, which are characterized by high toxicity to humans. The presence of these metals in the human body causes a significant increase in the presence of free radicals. An excess of free radicals leads to pro/antioxidant imbalance and in consequence oxidative stress. This can be prevented by compounds with antioxidant properties, including PON proteins, which inactivate the toxic products resulting from LDL oxidation as well as preventing accumulation. PON1 is inactivated during this process, in particular by the displacement of calcium ions and the reduction of chemically active free thiol groups of cysteine at position 284. 23 This demonstrates that heavy metals derived from cigarette smoke can directly inhibit the activity of PON enzymes.
The modification of activity of PON enzymes may also be a result of direct interaction with protein. Nickel present in tobacco smoke may be associated with histidines (His-115 and His-134), which are important for maintaining the arylesterase activity of PON1. Furthermore, nickel together with other metals, such as mercury, zinc, and cadmium, through the affinity of the cysteine residues, can influence the activity of PON1. Two of these, combined by a disulfide-bridge, are responsible for hydrolytic activity while one from the free sulfhydryl group is essential in maintaining the antioxidant properties of the enzyme. 28 This information suggests that the decrease in PON proteins activities caused by cigarette smoke may be due to inhibition of the enzyme active regions by heavy metals.
Polymorphisms of PON and Cigarette Smoke
Cigarette smoking is negatively correlated with general hydrolytic activity of PON enzymes, which is variable depending on the member and polymorphism of the enzyme. 5,42,43 It is possible that the process of PON proteins inhibition caused by cigarette smoking occurs in varying degrees depending on the genotype. Numerous studies have been conducted in order to accurately determine the relation between smoking and the inhibition of various forms of PON.
As previously described, the PON family possesses antioxidant properties and is likely one of the crucial enzymes in preventing lipoproteins oxidation. This description of the properties of PON enzymes is the result of many studies in which patients having diseases associated with lipid metabolism disorders were investigated.
Turban et al 45 studied the relation between the polymorphism PON1Q192R and the development of coronary atherosclerosis among smokers. The genotype in almost 400 patients was determined by polymerase chain reaction. Participants were divided according to polymorphism types into 3 groups: QQ, QR, and RR. The lipid profile, body mass index (BMI), and systolic/diastolic blood pressure were determined in each of these groups. Among these groups, association correlations between cigarette smoking and polymorphism PON1Q192R showed no statistically significant differences. This demonstrates the lack of a link between PON1 and atherogenic changes caused by cigarette smoking.
Different results were obtained in the case of patients diagnosed with acute myocardial infarction. The study group consisted of 156 postinfarction patients and a control group of 310 healthy volunteers. Participants were divided into the following groups: never smoked, past smoker, and current smoker. Within these groups, the genotype PON1 was determined. A statistically significant relationship was demonstrated between PON1192QQ, smoking, and myocardial infarction, while in the case of other genotypes, this relation was not obtained. 46 In contrast, a subsequent study by the same author showed no relationship between cigarette smoking and the polymorphism PON1Q192R. 5 The lack of coherence of these results is probably due to differences in the disease entities of the studied groups. Another group of researchers also found no relationship between PON1 polymorphisms and cigarette smoking. 41 This suggests that cigarette smoking inhibits PON1 hydrolytic activity regardless of the polymorphism.
However, other studies show a correlation between PON1, smoking, and HDL. In a study conducted on a group of 799 healthy volunteers, Frances et al 47 analyzed the relationship between the polymorphic forms of the PON1 gene (Q192R and L55M), serum lipids, and cigarette smoking. In the case of the Q192R polymorphism, a statistically significant difference between the genotypes RR and RQ and HDL concentrations was observed. The R allele carriers displayed reduced HDL levels, and the decline in HDL levels was stronger in smokers (with the same genotype).
Malin et al 48 evaluated the effectiveness of PON1L55M in smokers and nonsmokers by measuring carotid artery intima–media thickness (CIMT) via ultrasound. Participants were divided, depending on the genotype into 2 groups: homozygote LL and M allele carriers (MM and ML). The parameters of lipid profile, BMI, systolic, and diastolic blood pressure were also determined. Nonsmokers with the genotype LL exhibited a roughly 9.9% higher value of CIMT than carriers of the M alleles. Opposite results were obtained from the group of smokers, in which the genotypes MM and ML indicated a predisposition to higher CIMT performance. These results suggest that cigarette smoking modifies the properties of PON1, and the effectiveness of this process probably depends on the PON1L55M polymorphism. Other studies also confirm the relationship between polymorphism L55M and PON activity. In a volunteer group of 138 nonsmokers and 162 smokers, lipid profile, genotype, and PON1 activity as well as concentrations of cotinine—a metabolite of nicotine—were estimated. Among smokers, the results showed a significant relationship between the activity of PON1 and polymorphism L55M. The lowest enzyme activity was observed among homozygotes MM, while the highest among LL. In addition, smokers with polymorphism L55M were more exposed to lower values of PON1 activity. This suggests once again that the L55M polymorphism predisposes smokers to lower values of PON1. 41
The dependence concerning the polymorphism of PON2 was also analyzed. Some studies have shown an increased risk of coronary heart disease associated with the occurrence of polymorphism PON2C311S (among nonsmokers), 49 while other authors have only observed it in smokers. 50 The results of analyses are inconsistent and lead to ambiguous conclusions. Some researchers have claimed the existence of a minor connection between PON polymorphisms and cigarette smoking, while others have completely denied one exists. The resulting discrepancy may be the result of differences in the pathophysiological states of the study group or in some cases of its low abundance.
Inhibition of PON—The Indirect Impact
HDL is probably required to maintain the hydrolytic activity of PON, and decreased HDL concentration may impact that activity. 44 Thus, hypothetically, cigarette smoking may indirectly inhibit the PON family by reducing HDL levels. To verify this possibility, the relation between HDL-deficit states and degree of PON proteins activity should be analyzed.
James et al 51 examined PON1 activity among groups of patients with deficiencies of HDL and apo A-I, closely associated with HDL. The study group consisted of patients with diagnosed Tangier disease (familial HDL deficiency) and patients with apo A-I mutation divided into 3 groups according to place of residence: A-I-Pisa, A-I-Helsinki, and A-I-Milano. In the plasma of patients with Tangier disease from the group A-I-Pisa, a total absence of HDL was noted (value 0 mmol/L) along with decreased levels of apo A-I to less than 5% of the reference values. However, the A-I-Helsinki and A-I-Milan groups retained 50% to 70% of the apo A-I concentration as well as 59% and 37% of HDL reference values, respectively. A complete absence of or a significant reduction in HDL levels may suggest that the activity of PON1 oscillates around the minimum value. However, patients with complete deficiency of HDL (Tangier disease, A-I-Pisa) retained around 50% of the activity of PON1 marked within the control group. The reduction of PON1 in the A-I-Helsinki group was less significant, and the values obtained in the A-I-Milan group did not differ statistically from the control group. This research demonstrates that in states of genetic HDL deficiency, PON1 is still present in plasma and exhibits hydrolytic activity, which is itself limited but still present in significant quantities. The results of this study seem to be of particular clinical relevance. An antisclerotic role is presently attributed to PON proteins, resulting from the protection of LDL against oxidation. Maintenance of this function of PON family members in a state of HDL deficiency may explain why patients with genetically determined lipoprotein deficiency are not exposed to vascular diseases to the extent that could be expected. The study demonstrates that, despite the complete absence of circulating HDL, PON1 activity was not entirely inhibited but only reduced. This suggests that HDL plays an important role in maintaining PON activity but it is probably not indispensable in this process. Analyzing the results obtained, it can be assumed that the reduction in HDL levels caused by cigarette smoking may have a small indirect effect on the activities of PON proteins associated with this lipoprotein.
Reversibility of the PON Inhibition Caused by Cigarette Smoke
As shown earlier, cigarette smoking results in a reduction in activities of PON proteins. Research has been conducted to determine whether these changes are reversible. James et al 44 divided a group of 597 people into current smokers, former smokers, and never smokers. In each group, the concentration of PON1 was measured, as well as its arylesterase and organophosphatase activities. Both activities recorded the lowest value in current smokers and the highest in nonsmokers. Among former smokers, intermediate values were demonstrated between nonsmokers and smokers. The differences in hydrolytic activity of former and current smokers were statistically significant. This suggests that smoking cessation reverses existing changes. Furthermore, in order to complete the analysis, the concentration and activity of PON1 were identified depending on the time elapsed since smoking cessation. 44 The 75 exsmokers who had given up smoking before the beginning of the study demonstrated a positive correlation between the length of time following the cessation of smoking and PON1 activity. There were significant differences across all groups.
This study indicates that cigarette smoking is an independent factor associated with a significant decrease in the activity and concentration of PON. However, shortly after cessation of smoking there is an increase in PON1 enzyme activity. 44
As Senti et al 5 reported, the process of PON1 inhibition is reversible. Their study was conducted on a group of smokers and nonsmokers who were further divided into physically active and inactive participants. The criterion for the division was the limit of daily calories consumed during exercise: >300 kcal for active people and <300 kcal for inactive. Physically active people have significantly higher levels of PON1 activity compared to inactive people. In addition, no statistically significant difference was demonstrated in PON1 activity between smokers and nonsmokers who were active physically.
These studies clearly show that the inhibition of the enzymatic activity of PON1 caused by cigarette smoking is a reversible process. The changes that occur are not permanent and can be completely eliminated by physical activity. This demonstrates the important role of physical activity in the regulation of PON1 activity, while also encouraging further research to identify the mechanism underlying this process.
Conclusion
Cigarette smoking is a leading prooxidant factor. An enzyme that prevents oxidative stress is PON. Smoking upsets the pro/antioxidant balance and thereby reduces the hydrolytic activity of PON family members by modification of free thiol groups and/or by direct inhibition of the enzyme active areas by heavy metals. The process of inhibition of PON enzymes caused by cigarette smoking is a reversible process. This effect increases together with the time elapsing from smoking cessation. It should also be noted that PON is functionally and structurally related to HDL; however, a total absence of HDL in blood does not completely inhibit the hydrolytic activity of PON.
It is also worth mentioning that PON1 activity and hydrolytic properties have been carefully examined in relation to different treatment groups and experimental conditions, while PON2 and PON3 have not been described in sufficient detail. It is known that PON2 is located in practically all tissues of the human body, which likely facilitates the holistic action of this enzyme, and PON3 is more effective in protecting LDL antioxidants than PON1. However, it is necessary to continue analysis of these enzymes, particularly considering that they may be important in preventing a number of diseases as well as due to their participation in antioxidant processes. In addition, the interactions between the PON family’s members should be examined in order to accurately understand the mechanism of the protective action of the PON family against the xenobiotics in tobacco smoke.
Footnotes
Author Contribution
Milnerowicz, H contributed to conception and design, critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Kowalska, K contributed to analysis and interpretation, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Socha, E contributed to analysis and interpretation, drafted the manuscript, gave final approval, and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant ST-852 from Wrocław Medical University and by Wrocław Medical University Foundation.
