Abstract
Although surgery with or without (neo)adjuvant chemo/radiotherapy, as the standard treatments, can be suitable therapeutic strategies for gastric cancer, side effects and drug resistance are two main treatment obstacles. It has been discovered that pomegranate and its natural derivatives, especially ellagic acid (EA), offer significant anti-cancer effects while causing trivial side effects. In this study, we aimed to explore the anti-cancer effects of EA on a human gastric adenocarcinoma cell line (AGS) as well as in immunocompromised mice bearing human gastric tumors, for the first time. HPLC was used for determining EA in samples. MTT assay, apoptosis and scratch assay, gelatin zymography, and quantitative RT-PCR were used to determine the anti-cancer properties of different concentrations of pomegranate fruit juice, pomegranate peel extract, and EA. Furthermore, the effects of these compounds were investigated on immunosuppressed C57BL/6 mice carrying human gastric cancer tumors. EA could inhibit the proliferation and migration of gastric cancer cells. It also had significant effects on reducing both expression and activity of MMP-2 and MMP-9. Further, it was demonstrated that with alterations in the expression of genes involved in apoptosis and inflammation including P53, BAX, APAF1, BCL2, iNOS, NF-κB, IL-8, and TNF-α, EA treatment led to increased cancer cell death and reduced inflammation. Furthermore, its use in mice bearing gastric tumors resulted in a significant reduction in tumor volume without any obvious side effects. Ellagic acid exhibited anti-cancer effects on gastric adenocarcinoma, and can be considered as a safe anti-cancer agent for further preclinical studies on this cancer.

Introduction
Due to high incidence and mortality rates of different types of malignancies, cancer is still one of the main challenges in all human societies. 1 Gastric cancer (GC) is one of the common malignancies in digestive system, the fifth most commonly diagnosed cancer, which remains the fourth leading cause of cancer-related death worldwide.2,3 Despite advances in the treatment of GC, which are mainly based on chemotherapy and radiotherapy methods, drug resistance and high rate of side effects are still major challenges in improving this deadly disease in the clinic.3,4 Thus, many researchers are striving to improve treatment strategies by finding better pharmacological agents with significant anti-cancer effects as well as minimal side effects. Accordingly, in recent years, it has been reported that various compounds derived from plants can be introduced as agents with anti-cancer properties in order to prevent and/or treat various malignancies.5,6
Pomegranate (Punica granatum L.), as a well-known fruit-bearing deciduous shrub, has originated from Iran though it has been cultivated in various climatic conditions in many parts of the world.7–10 The whole pomegranate fruit is a major source of dietary pomegranate phytochemicals and it is rich in polyphenols, anthocyanins, flavonoids, hydrolysable tannins, and different organic acids.11,12 Meanwhile, pomegranate and its related compounds are known as suitable chemopreventive and chemotherapeutic agents in many types of human cancers such as prostate cancer, breast cancer, colorectal cancer, and lung cancer.13,14
Polyphenols are very important herbal metabolites that can have various effects on different cancer related processes.15,16 It has been shown that different pomegranate fruit components such as juice and peel have a significant amount of polyphenols including ellagic acid (EA), punicalagin, etc..
17
EA, as a member of ellagitannins (ETs), has a polyphenolic structure (Figure 1). It has been proven that in addition to inducing apoptosis, this compound can also inhibit angiogenesis, migration, and metastasis in various malignancies.14,18–20 Furthermore, it is discovered that EA can boost the effects of chemotherapy and radiotherapy.
21
Our recent systems-pharmacology study revealed that the anti-cancer effects of EA in human malignancies are probably exerted through several main signaling pathways such as apoptosis, P53, and WNT signaling pathways. It was also found that EA has significant effects on the expression of several important genes and it can have effective interactions with several cancer-related proteins.
14
Chemical structure of EA.
However, despite numerous studies on the effects of pomegranate fruit and EA, as one of its polyphenolic derivatives, on various cancers, no study has been conducted on the effects of these compounds on gastric cancer so far. Thus, the aim of this study was to investigate the anti-tumor activities of pomegranate fruit juice (PFJ), hydroalcoholic extract of pomegranate peel (PPE), and EA on human gastric cancer cells (AGS) both in vitro and in vivo.
Experimental procedures
Pomegranate fruit juice preparation
After collecting pomegranate fruit from Bajestan region in Razavi Khorasan Province of Iran, the pomegranate arils were placed on a steel sieve and slowly put under physical pressure. For filtration, the extracted PFJ was placed into 50 mL falcon tubes and centrifuged at 1000 g, then filtered using a 0.45 μm nylon syringe filter, and stored at −80°C.
Preparation of pomegranate peel hydroalcoholic extract
Fresh pomegranate fruits were peeled manually with the peels dried at room temperature in the shade. The dried peels were powdered and macerated in 70% methanol, under shakeup for 72 h at room temperature and darkness. The solvent was evaporated and at the time of use, PPE was dissolved in fluids such as culture medium or phosphate-buffered saline (PBS).
Chromatographic conditions
High-performance liquid chromatography (HPLC) was carried out using a KNAUER liquid chromatograph system consisting of a quaternary pump (Smartline Pump 1000). Detection was performed on diode array detector (Smartline DAD 2800), and data were processed using EZ Chrom Elite software. The pomegranate juice and methanolic extract were subjected to reversed-phase HPLC using a gradient method of 10–100% methanol. The separation was done on ACE 5 C18 (Advanced Chromatography Technologies Limited, Aberdeen, Scotland; 10 × 4.6 mm) at a flow rate of 0.8 mL/min. The injection volume was 20 µL with the compounds monitored at 254 nm. A standard solution containing ellagic acid was injected into the HPLC device in order to validate the reliability of the system.
Cell culture
Human gastric cancer cells (AGS) were purchased from Pasteur Institute (Tehran, Iran), and grown in Roswell Park Memorial Institute (RPMI 1640) medium (Gibco, Paisley, Scotland), supplemented with 10% fetal bovine serum (FBS; Biosera, UK). A normal human dermal fibroblast cell line, HDF, was obtained from Academic Center for Education, Culture and Research (ACECR), Mashhad, Iran. HDF cells were kept in high glucose Dulbecco’s modified Eagle’s medium, DMEM-HG (Gibco, Paisley, Scotland), with 10% FBS and penicillin/streptomycin. Both cell lines were maintained at 37°C under 5 and 10% CO2, respectively, and subcultured as required.
Cell viability assay
The 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide assay (MTT; Sigma) was used to measure the cell viability. To this aim, AGS and HDF cells were seeded in 96-well plates at densities of 8000 and 6000 cells/well, respectively. The cells were treated for 24, 48, and 72 h with different serial dilutions of PFJ (100-3.125 µL/mL), PPE (200-3.125 µg/mL) dissolved in culture media, and EA (Sigma, Saint Louis, MO, USA) (100-3.125 µg/mL) dissolved in dimethyl sulfoxide (DMSO; Sigma, Saint Louis, MO, USA). Further, different concentrations of cisplatin (Mylan, France) (100-1.563 µg/mL), prepared in culture media, were used as a positive control. Following treatments at three-time intervals, 20 µL MTT (5 mg/mL) was added to each well where the plates were incubated for 3–4 h at 37°C in the dark. Then, the media were replaced with 150 µL DMSO for each well to dissolve formazan crystals. Finally, a microplate reader (Awareness Technology, USA) at 545 nm wavelength was employed to measure the absorbance. Experiments were performed in triplicate for each tested compound. 22
Apoptosis assay by flow cytometry
“FITC Annexin V Apoptosis Detection Kit with PI” (Biolegend, San Diego, CA) was used for apoptosis evaluation through flow cytometry according to the manufacturer’s instructions. Briefly, gastric cancer cells were seeded in 6-well plates at a density of 3 × 105 cells/well, incubated with EA at concentrations equal to its IC50 and IC50/2 values. After 24 h, the cells were collected, washed with PBS, and centrifuged at 130 g for 5 min. The cells were then resuspended in cell staining buffer at 4°C, centrifuged, and resuspended in binding buffer. Finally, 5 μL of FITC Annexin V and 10 μL of 20 mg/mL propidium iodide (PI) were added to flow cytometry tubes and incubated at room temperature and dark conditions for 15 min. The number of apoptotic cells was measured using a flow cytometer (BD Accuri C6), and data were analyzed by FlowJo software (version 7.6.1).
Wound healing scratch assay
AGS cells were cultured in 6-well plates (1 × 105 cells/well); after 24 h when the cells were at 95–100% confluence, a wound was created with a pipette tip. The cells were washed with PBS twice, and then treated with predetermined compounds, including PFJ (100 µL/mL), PPE (100 µg/mL), EA (15 and 30 µg/mL), and serum-free media (as a control), and incubated at 37°C and 5% CO2. Next, photographs of the wounded areas were taken periodically (at 0, 12, 24, and 48 h time intervals) under a phase-contrast microscope. 23 Experiments were performed in triplicate. The percentage of cell‐covered areas was quantified using the Wimasis image analysis Wimscratch tool (Wimasis, Munich, Germany).
Gelatin zymography
Gelatin zymography was applied to investigate the possible effects of the desired compounds on the suppression of matrix metalloproteinase-2/-9 (MMP-2/-9) activity. 24 In brief, the AGS cells were treated with PFJ (100 µL/mL), PPE (100 µg/mL), EA (10 µg/mL), EA (20 µg/mL), and EA (30 µg/mL) for 48 h, in fresh serum-free media (SFM). Next, the cultured media were centrifuged at 4°C for 15 min at 25000 g, and the conditioned media were electrophoresed in a 7.5% polyacrylamide gel with 0.1% sodium dodecyl sulfate (SDS) containing 0.1% gelatin as a substrate. The gel was then washed three times with washing buffer (2.5% triton X-100) for 15 min under agitation to remove the SDS. Thereafter, the gel was incubated with developing buffer (150 mM NaCl, 50 mM Tris-HCl, 1 μM ZnCl2 and 5 mM CaCl2; pH 7.5) at 37°C for 4 h. Subsequently, gel staining was performed using Coomassie Brilliant Blue R-250 for 30 min followed by de-staining process with a solution containing 10% acetic acid plus 20% methanol in dH2O. Finally, the gel was imaged and regions of gelatin degradation as the gelatinolytic zones were analyzed by ImageJ 1.52a software (National Institute of Health, USA).
Quantitative reverse transcription-polymerase chain reaction (qRT-PCR)
Primer sequences used in this study for qRT-PCR.

In vivo studies
Animals
C57BL/6 male and female mice were purchased from Pasteur Institute (Amol, Iran), kept at 25 ± 2°C, with a relative humidity of 60 ± 5% and a 12 h light–dark cycle, and reproduced in an animal house at Ferdowsi University of Mashhad. The animals had free access to standard chow diet. All experimental protocols were approved by the Animal Ethics Committee of Ferdowsi University of Mashhad.
Immunosuppression
Immunosuppression of C57BL/6 mice was carried out according to the protocol presented by Jivrajani et al., with some modification. 26 C57BL/6 male mice (4-6 weeks old) were selected for immunosuppression. The animals were administered with 10 mg/kg ketoconazole (Amin pharmaceutical Co, Isfahan, Iran) orally and 30 mg/kg cyclosporine through intraperitoneal injection every day for 7 days. All animals were given co-amoxiclav (Farabi Pharmaceutical Co, Isfahan, Iran) (0.1 μg/mL) by drinking water throughout the study. Furthermore, on days 3 and 1 prior to tumor cell injection, cyclophosphamide (Roche chemical co, Grenzach, Germany) was injected subcutaneously at a dose of 60 mg/kg. Note that the animals were fed with autoclaved food and water during the study.
Tumor model and treatments
In order to provide mice models bearing human tumors, AGS cells (6× 106 cells per mouse) were implanted subcutaneously in the right flank of mice. Nearly 12 days post-injection, when the tumor volume reached 100-200 mm3, C57BL/6 male mice were divided into nine groups (n = 6) and treated with PBS, DMSO, cisplatin, PFJ (10 mL/kg), PFJ (50 mL/kg), PPE (10 mg/kg), PPE (50 mg/kg), EA (10 mg/kg), and EA (50 mg/kg) groups.
21
EA was dissolved in DMSO and freshly diluted in culture medium before the treatment. The tumor-bearing mice were treated with desired compounds through gavage for 14 days, and the tumor volumes measured on even days of the week, using a digital caliper (Mitutoyo, Japan) with 0.01 mm precision, and further evaluated using the following formula
26

Histopathological analyses
After completion of the study, the animals were sacrificed where several organs such as liver, kidney, spleen, stomach, small intestine and colon were excised from the body and kept in 4% paraformaldehyde (PFA). For histopathological studies, samples were cut into 4 μm sections and stained with hematoxylin and eosin (HE).
Statistical analyses
The statistical analyses were performed using GraphPad Prism 5.0 (GraphPad Software INC., CA, USA) by one-way ANOVA and t-test. Further, FlowJo 7.6.1 (Tree Star, Ashland, OR, USA) and ImageJ 1.52a (NIH, Bethesda, Rockville, MD, USA) software were employed to analyze apoptosis and migration data, respectively. In this study, data have been presented as mean ± SD and considered significantly different when p-value < 0.05.
Results
Chromatographic results to investigate the presence of EA in desired samples
As displayed in Figure 2, the analysis of the HPLC chromatograms revealed that unlike PFJ lacking EA, there was a relatively significant amount of this compound in the PPE (1.62% per gram). Typical chromatograms of (a) EA, (b) PFJ, and (c) PPE.
Anticancer effects of ellagic acid on AGS cells
AGS cells were treated with different concentrations of PFJ, PPE, and EA for 24, 48, and 72 h whereby a dose-dependent anti-proliferative effect was observed to various degrees in all of them. The half-maximal inhibitory concentration (IC50) values were determined as 265, 214, and 169 µg/mL for PPE as well as 60, 41, and 27 µg/mL (198, 136, 90 µM) for EA, at 24, 48, and 72 h, respectively. Furthermore, the IC50 values for cisplatin, as a control drug, were calculated as 7, 4, and 3 µg/mL (26, 15, 10 µM) at 24, 48, and 72 h, respectively (Figure 3). The compounds did not have a significant cytotoxic effect on human dermal fibroblasts (HDF) as normal cells (data not shown). The cytotoxic effects of (a) PFJ, (b) PPE, (c) EA, and (d) cisplatin on AGS gastric cancer cells, as determined by MTT assay after 24, 48, and 72 h.
EA induced apoptosis in AGS cells
The pro-apoptotic activity of EA was investigated using the “FITC Annexin V Apoptosis Detection Kit with PI” via flow cytometry. The results revealed that at 24 h, EA and cisplatin could induce apoptosis in gastric cancer cells (Figure 4). EA could induce apoptosis in AGS gastric cancer cells. Apoptotic cells were detected with FITC-Annexin V and PI staining at 24 h after treatment with (a) PBS, (b) DMSO, (c) Cisplatin, (d) 20 µg/mL EA (68 µM), and (e) 40 µg/mL EA (136 µM). Data are presented as percentage of cells placed in each quadrant.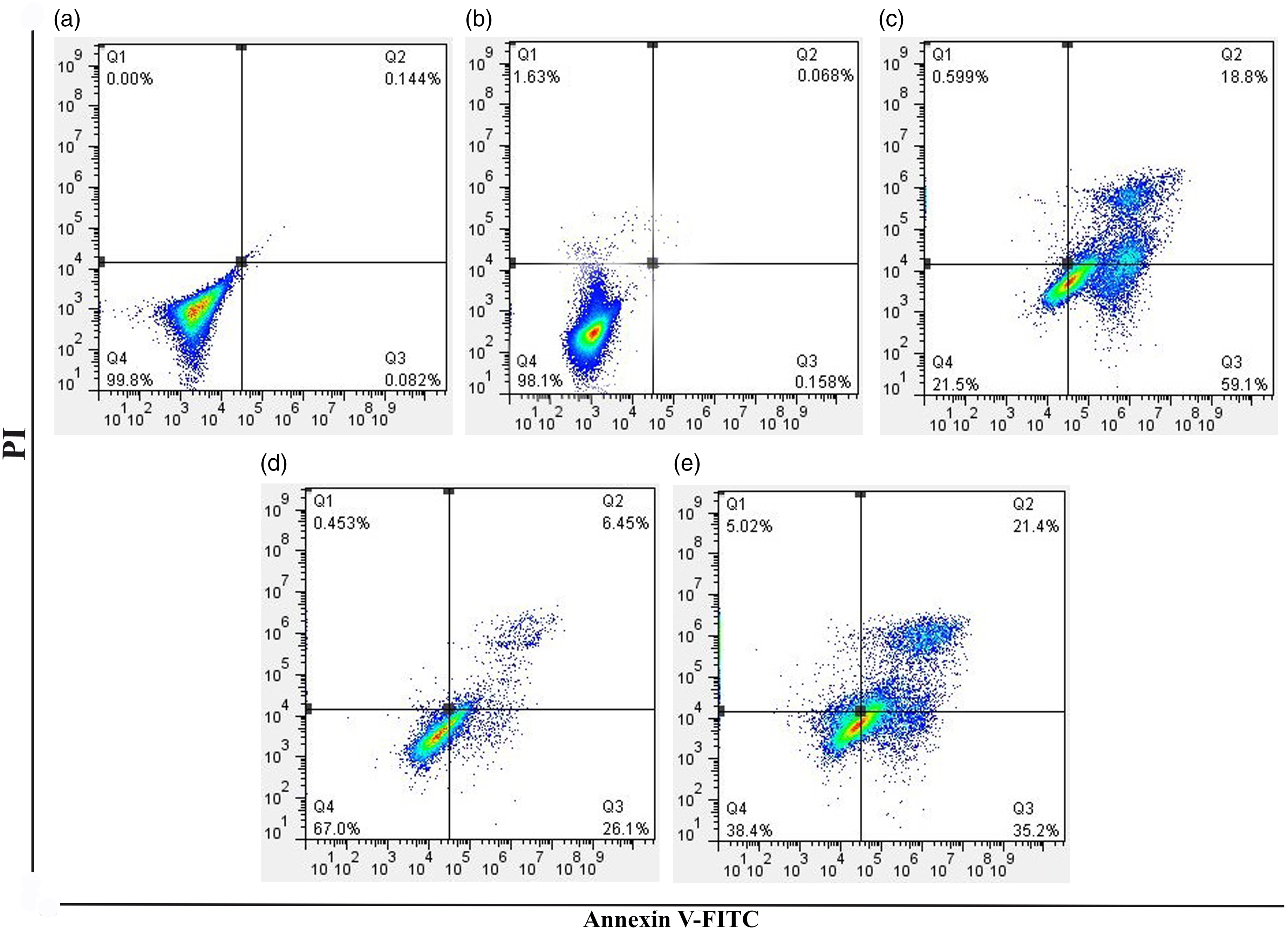
Inhibition of AGS cells migration by EA
Wound healing scratch assay was performed for investigating the effects of desired compounds on the migration ability of gastric cancer cells after treatment for 12, 24, and 48 h. The results indicated that 48 h treatment with EA (30 µg/mL) had an inhibitory effect on cell migration, whereby the size of the scratch area was not significantly reduced as compared with the control group (serum-free media) and other compounds (Figure 5). Investigating the effects of various treatments on cellular migration. AGS cancer cells were incubated with desired compounds including PFJ (100 µL/mL), PPE (100 µg/mL), and EA (15, 30 µg/mL) for 12, 24, and 48 h. (a) Scratch areas as observed under the microscope (40 x). (b) Comparing the effects of PFJ, PPE and EA on AGS cell migration in three different time periods (***p < 0.001, and ****p < 0.0001).
The effects of desired compounds on MMP-2/-9 activity
In order to explore the possible inhibitory effects of the desired compounds on MMP-2/-9 activity, gelatin zymography assay was carried out. The data revealed that, apart from PFJ (100 µL/mL), the other compounds i.e. PPE (100 µg/mL) and EA (10, 20, and 30 µg/mL) had significant effects on reducing the activity of both enzymes compared to the control (no treatment) group. In addition, as outlined in Figure 6, a significant decrease was observed in the levels of MMP-2/-9 with elevating the EA concentrations. The effects of various treatments on MMP-2/-9 activity of AGS cells. The proteolytic activity of MMP-2/-9 is determined by gelatin zymography in supernatants collected 24 h after treatment with PFJ (100 µL/mL), PPE (100 µg/mL), and EA (10, 20, and 30 µg/mL). (a) Active MMP-2 (72 kDa) and MMP-9 (92 kDa) proteins are shown. (b) Statistical analysis of MMP-2/-9 enzymatic activities of three independent tests is shown. *p < 0.05, ***p < 0.001, and ****p < 0.0001 compared to the control group (no treatment).
Effects of PFJ, PPE, and EA on gene expression pattern of AGS cells
To better understand the effects of PFJ (100 µL/mL), PPE (100 µg/mL), and EA (15, 30 µg/mL) on different cancer-related processes, the expression of particular genes involved in (i) apoptosis including P53,
27
BAX,
27
APAF1,
28
and BCL2,
27
(ii) migration, such as MMP-2, and MMP-9,
29
and (iii) cancer related inflammation including iNOS,
30
NF-κB,
31
IL-8,
31
and TNF-α
32
were verified. The data showed that some of these treatments significantly altered the expression of related genes. For example, PFJ (100 µL/mL) had a significant effect on only two genes (BAX, and APAF1), while EA (30 µg/mL) significantly altered the expression of all investigated genes (Figure 7). Changes in mRNA expression of desired genes in human AGS gastric cancer cells treated with (a) PFJ (100 µL/mL), (b) PPE (100 µg/mL), and (c, d) EA (15 and 30 µg/mL). (e) Classification of desired genes and comparing the changes in their expression after treatment with the considered compounds. *p < 0.05, **p < 0.01, and ***p < 0.001 compared to the control group (no treatment).
Tumor growth inhibition by EA in a mouse model with gastric cancer
As depicted in Figure 8, by measuring the changes in tumor volumes, it was found that the mice treated with PPE (50 mg/kg), EA (at both concentrations), and cisplatin had a lower growth rate, as compared to the control groups (PBS and DMSO), both during the study and on the last day. EA inhibits the growth of AGS related tumors in immunocompromised C57BL/6 mice. (a) Relative variations of tumor volumes (mm3) after treatment with PBS, DMSO, cisplatin, PFJ (10 mL/kg), PFJ (50 mL/kg), PPE (10 mg/kg), PPE (50 mg/kg), EA (10 mg/kg), and EA (50 mg/kg). (b) Comparison between tumor volumes in different groups on the final day. (c) Representative tumors related to each treatment. (d) Histopathological images of liver, kidney, spleen, stomach, small intestine, and colon tissues after different treatments (HE staining). All data are presented as mean ± SD.
Analysing blood cells and histopathological data
According to the data obtained, the mean number of neutrophils, lymphocytes, and red blood cells in different groups of treated mice at the end of the study did not change significantly. Furthermore, in order to check whether the desired compounds had any side effects on normal tissues, after hematoxylin and eosin staining, we examined the tissues from several organs including liver, kidney, spleen, stomach, small intestine, and colon under a light microscope. As observed in Figure 8(d), the data revealed that none of the studied compounds had any significant side effects on these tissues.
Discussion
Gastric cancer is still one of the human malignancies with high mortality rates, especially in developing countries.2,3 Unfortunately, common therapeutic strategies including chemotherapy impose serious side effects on patients. Thus, many researchers globally are looking for anti-cancer effects of natural compounds with minimal side effects. In this study, the anti-cancer properties of three pomegranate phytoconstituents including PFJ, PPE, and EA were investigated on human gastric cancer cells.
So far, many studies have shown the anti-cancer effects of pomegranate and its derived compounds in various types of cancers, but no study has examined these effects on gastric cancer. 33 In this context, it has been shown that PFJ, thanks to its rich polyphenolic composition, can have anti-cancer activity against several human malignancies such as prostate, colon, liver, and breast cancer.34,35 Further, other studies have reported that PPE also has a high content of polyphenolic compounds and therefore, it can induce cell death in various cancers including breast, melanoma, and colon malignancies.36–38 It has also been approved that EA, as one of the main polyphenols in PFJ and PPE, can have significant anti-tumor effects on different human cancers including bladder, blood, breast, cervical, colorectal, liver, pancreas, and prostate cancer.10,14,39
Cancer cell migration is one of the important processes involved in cancer progression and metastasis. It has been found that MMPs, in particular MMP-2 and MMP-9, as enzymes playing a key role in regulating cell-matrix interactions, are essential in migration-related processes.40,41 Our study revealed that EA has a significant effect on gastric adenocarcinoma cell migration in a dose-dependent manner. It was also shown that in addition to effectively reducing enzymatic activity of MMP-2/-9, EA could reduce the expression of genes associated with these enzymes.42,43
Meanwhile, numerous studies have demonstrated that natural polyphenols, such as EA, can play different inhibitory roles in various pathways involved in human cancers. 44 Based on the present results, EA caused more drastic changes in the expression of desired genes in a dose-dependent manner, compared to PFJ and PPE. EA exhibited cancer inhibitory effects and led to a reduction in the expression of inflammatory related genes. So far, many studies have explored the effects of this natural compound on the expression of specific genes in various cancer cells.45,46,47 Qiu et al. indicated that EA could induce P53 expression in human bladder cancer cells and thus it can be effective for increased programmed cell death. 46 Furthermore, Madhukar et al. revealed that EA alone or in combination with radiation can promote the accumulation of P53 in HeLa cells. 48 Several studies have shown that up-regulation of BCL2, as an important antiapoptotic gene, has been observed in various types of human cancers, making it a good target for cancer treatment. 49 Similar to our results, a number of studies by examining the effects of EA on cancer cells have shown that this compound has a reducing effect on the expression of BCL2 in different types of human malignancies such as colorectal, pancreas, and prostate cancer.43,50–54 On the other hand, it has been shown that following cellular stress, BAX protein induces apoptosis in cancer cells through activating caspase molecules. 55 In this regard, various studies have shown that EA has significant effects on increasing the amounts of BAX protein in various types of human malignancies such as bladder, colorectal, pancreas, and prostate cancers.43,51,53,54,56,57 Further, APAF1, similar to BAX, via activating different caspases such as caspase 3 and 9, can induce programmed cell death in cancerous cells. 28 In this context, Ho et al. suggested that EA can cause apoptosis by promoting the levels of APAF1 in human bladder cancer TSGH-8301 cells. 56
It has also been demonstrated that inflammatory conditions in different organs and the presence of inflammatory microenvironment around the cells are important in the initiation, development, and progression of various cancers such as gastric cancer. Studies have also reported that in general, cancer and inflammation have reciprocal effects on each other.31,58 In this regard, the anti-inflammatory effects of different parts of pomegranate fruit and its derivatives have been proven.59,60 Thus, in this study, for the first time the effects of PFJ, PPE, and EA were investigated on altering the expression of some important inflammatory genes (NF-κB, iNOS, IL-8, TNF-α) in gastric cancer cells. Our data revealed that PPE only affected the expression of some of the desired genes (NF-κB, iNOS, TNF-α) and PFJ had no effect in this regard, while both concentrations of EA significantly altered the expression of all investigated inflammatory genes (Figure 7).
Several studies have suggested that in addition to initiation and progression of various human malignancies including breast, bladder, gastric, and colorectal cancers, iNOS, IL-8, and TNF-α, as key mediators of inflammation, are also involved in different cancer related processes such as metastasis, angiogenesis, and resistance to chemotherapy.61–66 In line with our results, several studies have shown that pomegranate derivatives, and especially EA, can reduce inflammatory conditions by significantly reducing the expression of genes involved in this process. In this regard, some studies have shown that EA and EA-enriched pomegranate extract can inhibit NF-κB as well as down-regulate iNOS, TNF-α, and COX-2 expression in colon carcinoma.67,68 Furthermore, Kim et al. demonstrated that EA and pomegranate ellagitannins could reduce the expression at both mRNA and protein levels of several inflammatory-related factors such as iNOS, TNF-α, COX-2, and NF-κB in colon inflammation and ulceration. 69 In another study, Bishayee et al. reported that pomegranate extract remarkably moderated the hepatic expression of iNOS and NF-κB in rats with hepatocarcinoma. 70 Khan et al. revealed that pomegranate fruit extract reduced the levels of NF-κB, along with the expression of iNOS, CD31, AKT, and phosphatidylinositol 3-kinase (PI3K) in the lung tumors. 71 In addition, according to Umesalma et al., EA could exhibit an anti-inflammatory effect through down-regulating iNOS, TNF-α, and IL-6 via NF-κB inhibition. 67 Another study discovered that in pancreatic adenocarcinoma, EA can suppress cancer cell proliferation and reduce the activity of NF-κB. 72 Furthermore, Zhao et al. discovered that EA can significantly reduce the cellular level of IL-8 in pancreatic cancer cells. 43
In addition to in vitro experiments, the effects of PFJ, PPE, and EA were also investigated in immunocompromised C57BL/6 mice, as an animal model bearing human gastric tumor. Our data revealed that EA suppressed tumor growth and reduced final tumor volume in a dose-dependent manner. It was also found that PPE significantly but slightly inhibited the growth of human cancer cell mass. Notably, the studied compounds did not have any side effects on the liver, kidney, spleen, stomach, small intestine, and colon tissues. In this regard, many studies have reported the anti-cancer effects of EA on different human cancers such as pancreas, 73 breast,74,75 bladder, 76 colon, 77 and prostate 54 in vivo, and almost all of them have considered this polyphenol as a safe natural compound for treatment of cancer.
Nevertheless, it should be noted that, as shown in the results section, HPLC data revealed that, unlike the PFJ, there were high levels of EA in the PPE. Thus, more anti-cancer effects of the extract as compared to fruit juice can be clearly attributed to the presence of high amounts of EA in its composition.
Conclusion
Our results revealed that in the case of gastric cancer, EA in comparison to PFJ and PPE, had far more significant anti-cancer effects in several aspects such as apoptosis induction, reducing cell migration, altered expression of genes associated with programmed cell death, reduced expression of inflammatory genes, as well as inhibition of tumor growth in immunocompromised mice. These properties offer a rationale for nominating this natural polyphenol as a key chemotherapeutic agent against gastric cancer. Nevertheless, further studies are required before EA can be introduced as a drug for gastric cancer in the clinic.
Footnotes
Acknowledgments
We greatly acknowledge the contribution of Ferdowsi University of Mashhad to provide this great chance and high-quality lab services to carry out this investigation. Moreover, the authors would like to thank Dr. Norouzpour Laboratory, Mr. Mahdi Mirahmadi (ACECR) and Mr. Mostafa Ganjouri for their great help and technical advices.
Author contributions
The authors’ responsibilities were as follows: H. Cheshomi and M. M. Matin designed the study; M. M. Matin and H. Cheshomi analyzed data and performed statistical analysis; H. Cheshomi, A. R. Bahrami, H. Rafatpanah, and M. M. Matin contributed to sample measurements and data collection; M. M. Matin and H. Cheshomi wrote the article. All authors contributed to the manuscript review and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ferdowsi University of Mashhad [grant number 50828].
